
Dendrite coherency point of A357 alloys
LI Jian-feng(李建峰), KOU Hong-chao(寇宏超), WANG An-jia(王安家),
LI Jin-shan(李金山), HU Rui(胡 锐), FU Heng-zhi(傅恒志)
State Key Laboratory of Solidification Processing, Northwestern Polytechnical University,
Xi’an 710072, China
Received 28 July 2006; accepted 15 September 2006
Abstract: The dendrite coherency point was determined for A357 alloys using double-thermocouples technique. The effects of the cooling rate and different treatments such as grain refinement, modification, and superheat on the dendrite coherency point were investigated. The results show that the increase of the cooling rate and refinement significantly postpones the coherency point of A357 alloys. The morphology of silicon phase is modified, which slightly leads to lower solid fraction (fs) at the coherency point. But the fscoh value decreases after superheat treatment at 850 ℃ for 30 min. The grain size plays a decisive role in the fscoh value of A357 alloys. The smaller the grain size is, the more the fscoh value increases, which is beneficial to improving the final property of this alloy, while the secondary dendrite arm spacing and modification have almost no influence on fscoh.
Key words: A357 alloys; dendrite coherency point; cooling rate; melt treatments
1 Introduction
During solidification, the dendrite can move freely at the early stage of the solidification process. With the growth of the dendrite, the dendrite tips begin to impinge upon their neighbors (Fig.1), so that the solid in the mushy zone like a skeleton throughout the solidifying area. The dendrite coherency point(DCP) refers to the install at which the growing dendrites begin to establish a network. The volume fraction of solid at this point is called the coherency fraction solid (fscoh)[1-2].
Recently, the dendrite coherency point has been widely used as an important solidification parameter for aluminum alloys because it marks the transition from mass to interdendritic feeding to compensate for shrinkage in casting processes[1-7]. At this point the melt likes a solid, which results in defect generation, such as hot tearing, macrosegregation and porosity, and often assumed to commence after this point [5-6].
There are three techniques for determining the DCP [1-2,5]: torque technique, single-thermocouple technique and double-thermocouples technique. Compared with the torque technique and single- thermocouple technique, the double-thermocouples technique is based on the assumption that the establishment of a dendritic network at the DCP will result in a rapid decrease of the temperature difference between the wall and the center. So this is an exacter technique than others for its static measurement.
Previous work has been focused on the effects of alloying element and cooling rate on DCP of aluminum alloys[1-6, 8-10], but there is few study on the effect of melt treatment which are important methods for improving the performance of alloys.
In this paper, the dendrite coherency point using double-thermocouples technique was determined and the effects of different cooling rates and melt treatments on DCP of A357 alloys were focused on. This would provide a theory basis for investigating the solidification procedure in aluminum alloys.
2 Experimental
The A357 alloys were supplied commercially. The optical emission and inductively coupled plasma spectroscopy were used to verify the chemical compositions. An Al-5%Ti master alloy was added to the A357 alloys to refine the grain size, and an Al-10%Sr master alloy was used to modify the eutectic microstructure. Precast alloy ingot was melted in electric resistance furnace. Melt treatments processing are shown in Table 1 and the contents of refiner, modifier and other parameters are shown in Table 2. The optimized contents of titanium and strontium were 0.1% and 0.01% according to the Refs.[11-12].
Fig.1 Instant of collision in dendrite growth
The cylindrical sample crucibles were machined from a high-density graphite rod to an inner diameter of 30 mm and 80 mm in depth, with 10 mm-thick walls and base (Fig.2). The insulation board (thickness of 10 mm) was used above and below the crucibles to minimize the heat loss in the axial directions. The thermocouples were immersed in melt against the wall and in the center and the crucibles were preheat to 300 ℃ to eliminate the wall chill effect.
3 Results and discussion
3.1 Cooling curve
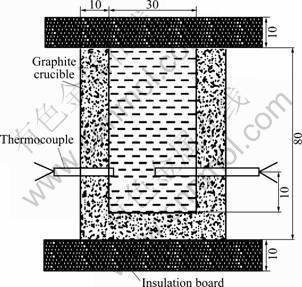
Fig.2 Schematic diagram of experimental setup for double- thermocouples (mm)
The cooling curves were measured in solidification before determining the dendrite coherency point of A357 alloys, and the temperature difference between the wall (θwall) and center (θcenter) of graphite crucible was calculated. According to Ref.[3], the fraction of solid corresponding to the maximum of temperature difference between θwall and θcenter after nucleation was taken as the dendrite coherency point. Then the temperature (θcc) and time (tcc) of the dendrite coherency point were available. Fig.3 shows the cooling curves of graphite crucible center, wall and corresponding calculated temperature difference (Δθ) under air-cooling condition.
The corresponding parameters from cooling curves by other different treatments, such as the temperature and the time of dendrite coherency are shown in Table 3.
Table 3 summarizes the coherency fraction solid results with double-thermocouples technique, where each result is the average of three measurements.
3.2 Fraction solid model
There are various mathematical models with certain assumptions for simulation of fraction solid from experimentally obtained fundamental parameters[13-15]. A linear model has been adopted without any theoretical foundation, while lever rule model assumes a very small solidification rate in binary alloys. Scheil model presumes a homogeneous liquid phase and no solute back diffusion in solid phase. In this study, a new model was proposed to calculate the fraction solid, based on the heat balance of solid and liquid in melt (the deduction of the model will be in described another paper).
Table 1 Melt-treatment process of A357 alloys

Table 2 Treatment process and serial sample No. of A357 alloys
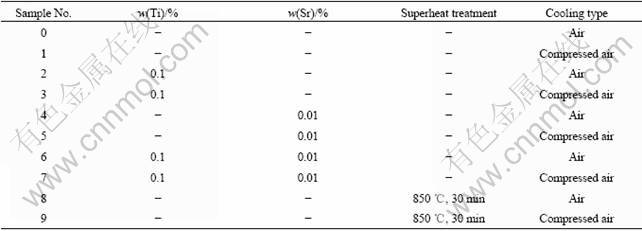
Table 3 Correlative determined dates received from cooling curves and dendrite coherency fraction solid results by different treatments
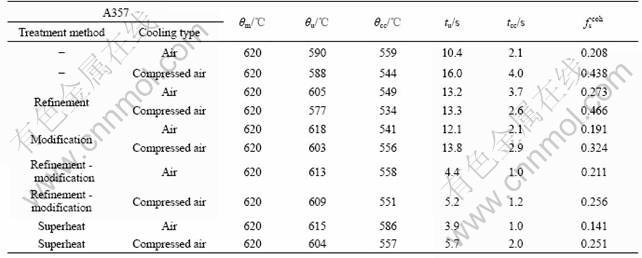
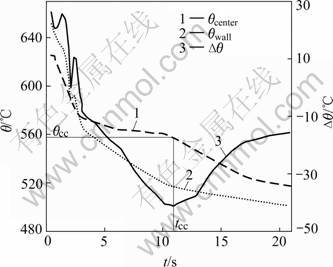
Fig.3 Cooling curves of A357 alloy measured in center and against wall of samples and corresponding calculated temperature difference curve at air-cooling
The recorded temperature and time dates were processed using the model of heat balance in solidification. The fraction solid at coherency point is determined as
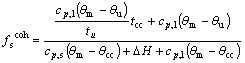 (1)
(1)
where cp,l is the special heat of liquid alloy, cp,s is the special heat of solid alloy, ΔH is the latent heat of solidification, θm is the melting point of alloy, θu is the temperature of recalescence point, θcc is the temperature of dendrite coherency point, tu is the time of recalescence point, tcc is the temperature of dendrite coherency point.
3.3 Cooling rate
Fig.4 shows the effect of cooling rate on dendrite coherency in the A357 alloys. In both alloys an increase in cooling rate gives a decrease in the grain size and a later coherency point.
When the graphite crucible was cooled with compressed air, the melt near the wall is solidified at higher cooling rate priority. The kind and number of heterogeneous nucleation are increased at higher undercooling, and the fine crystal grain can be formed. As the cooling rate increases, the growth of dendrite becomes slowly and the amount of dendrite increases, as a result, the fscoh value increases. The shrinkage that accrued in solidification will be fed when the melt has good flowability before the DCP. At the same time, the hot cracking at the end of solidification also can be prevented by liquid alloy. So the postponement of the DCP is beneficial to preventing these defects[8,14].
3.4 Melt treatment
The coherency solid fraction with different melt treatments is shown in Fig.4. The grain refinement improves the fscoh value for the increase of heterogeneous nucleation prevented by the forming and growth of dendrite.
After added modifier of Al-10%Sr, the Si phase is modified by the free Sr which is precipitated from the α-Al solid solution and from Al4Sr compound. The growth of Si shows anisotropic nature and has a marked preferred orientation of [111] on (111) surface without modification, so the Si phase grows quickly. But after added modifier, modified atoms are adsorbed in twinning saddle and hinders the growth of Si atom or atomic group, and then the silicon morphology is changed[14]. The fscoh value after modification has little change, while the grain size decreases. It is found that the fscoh value increases with decreasing grain size, but the secondary dendrite arm spacing has little effect on the fscoh value.
The morphologies of Si phase and Fe phase are changed after A357 alloy superheated at 850 ℃ for 30 min, and the fscoh value decreases with increasing grain size. Generally speaking, the mechanism of crystal melt is that the atomic group split gradually. During the melting process, atomic group splits gradually, but after the process is interrupted by external condition, some informations of original material may be reserved and transfer to the secondary solidification structure. For A357 alloy, the melt structure at general pouring temperature is located in microscopic non-uniform field which is aluminum-rich at low temperature. After the melt is superheated, the metastable colloidally particle disappears, and the melt structure is able to change from microscopic non-uniform field at low temperature to quasimicrocosmic uniform field, consequently the structure has changed. That is to say, the modified structure can be obtained without modifier addition.
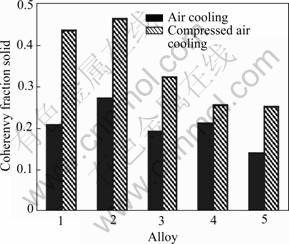
Fig.4 Coherency solid fraction with different melt treatments: 1 Without treatment; 2 Grain refinement; 3 Modification treat- ment; 4 Grain refinement and modification treatment; 5 Super- heat treatment
3.5 Grain size and secondary dendrite arm spacing
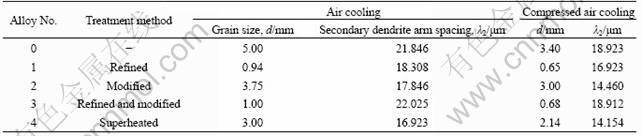
Table 4 summarizes the results of the grain size and secondary dendrite arm spacing with different treatments. The increased cooling rate gives lower grain size and secondary dendrite arm spacing. The grain size and secondary dendrite arm spacing show obvious decrease with adding refiner to melt. Modification treatment has slight effect on grain size and secondary dendrite arm spacing while combined treatments make the grain size decrease. Superheat treatment shows similar trends to modification treatment.
Table 4 Results of grain size and secondary dendrite arm spacing
4 Conclusions
1) The cooling rate and refinement significantly increase the coherency solid fraction in A357 alloys.
2) The morphology of silicon phase is changed, which slightly leads to lower fs at the coherency point. But the fscoh value decreases after superheat treatment at 850 ℃ for 30 min.
3) Grain size plays a decisive role in the fscoh value of A357 alloys. The smaller the grain size is, the more the fscoh value increases easily, which is beneficial to improving the final property of this alloy, while the secondary dendrite arm spacing and modification have almost no influence on fscoh.
References
[1] VELDMAN N L M, DAHLE A K, STJOHN D H. Dendrite coherency of Al-Si-Mg alloys [J]. Metall Mater Trans A, 2001, A32: 147-155.
[2] JIANG H, KIERKUS W T, BARDES B P. Determining dendrite coherency point characteristics of Al alloys using single-thermocouple technique [J]. ASF Transactions, 1999, 68: 169-172.
[3] B?CKERUD L, CHAI G, TAMMINEN J. Solidification characteristics of aluminium alloys [J]. Foundry Alloys Norway: ASF/SkanAluminium, 1990, 2: 128-129.
[4] DAHLE A K, STJOHN D H. Rheological behaviour of the mushy zone and its effect on the formation of casting defects during solidification [J]. Acta Metallurgica Inc, 1999, 47: 31-41.
[5] DAHLE A K, ARNBERG L. Development of strength in solidifying aluminum alloys [J]. Acta Metallurgica Inc, 1997, 45: 547-559.
[6] STANGELAND A, MO A, NIELSEN O. Development of thermal strain in the coherent mushy zone during solidification of aluminum alloys [J]. Metall Mater Trans A, 2004, A35: 2903-2915.
[7] ARNBERG L, DAHLE A K, PARADIES C J, SYVERTSEN F. Feeding mechanisms in aluminum foundry alloys [J]. ASF Transactions, 1995, 115: 753-759.
[8] MACKAY R I, DJURDJEVIC M B, SOKOLOWSKI J H. Effect of cooling rate on fraction solid of metallurgical reactions in 319 alloy [J]. ASF Transactions, 2000, 25: 521-530.
[9] CHAI G, B?CKERUD L, ARNBERG L. Relation between grain size and coherency parameters in aluminum alloys [J]. Materials Science and Technology, 1995, 11: 1099-1103.
[10] DAHLE A K, ARNBERG L. On the assumption of additive effect of solute element in dendrite growth [J]. Mater Sci Eng A, 1997, A225: 38-46.
[11] CHEN Zhong-wei. Solidification Microstructure Control of Ternary Aluminium Alloys [D]. Xi’an: Northwestern Polytechnical Univer- sity, 2003.
[12] ZHANG Jin-shan, XU Chun-xiang, HAN Fu-yin. Effect of composite modifier on structures and properties of hypereutectic Al-Si alloy [J]. The Chinese Journal of Nonferrous Metals, 2002, 12(1): 107-110. (in Chinese)
[13] DJURDJEVIC M B, KIERKUS W T, BYCZYNSKI G E. Modeling of fraction solid for 319 aluminum alloy [J]. ASF Transactions, 1999, 14: 173-179.
[14] KURZ W. Fundamentals of Solidification [M]. Switzerland: Trans tech Publications, 1984.
[15] BAKHTIYAROV S I, OVERFELT R A, TEODORESCU S G. Fraction solid measurements on solidifying melt [J]. Journal of Fluids Engineering, 2004, 126(2): 193-197.
(Edited by LI Xiang-qun)
Foundation item: Project(5131902) supported by the Major State Basic Research Development Program of China
Corresponding author: LI Jian-feng; Tel: +86-29-88493484; E-mail: lijianfeng@mail.nwpu.edu.cn