Trans. Nonferrous Met. Soc. China 25(2015) 3560-3568
Microstructural evolution during homogenization of DC cast 7085 aluminum alloy
Yun-jia SHI1, Qing-lin PAN1, Meng-jia LI1, Zhi-ming LIU2, Zhi-qi HUANG2
1. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
2. Guangdong Fenglu Aluminum Co., Ltd., Foshan 528133, China
Received 22 December 2014; accepted 13 March 2015
Abstract: The microstructural evolution of a DC cast 7085 alloy during homogenization treatment was investigated by optical microscopy, scanning electron microscopy, energy dispersive X-ray spectrometry (EDS), differential scanning calorimeter (DSC) and X-ray diffraction (XRD). The results showed that serious dendritic segregation existed in the as-cast 7085 alloy. Numerous eutectic microstructures and phases were observed at the grain boundary. During homogenization process, eutectic ��(Al)+T(AlZnMgCu) microstructure gradually was dissolved into matrix. Intermetallic S(Al2CuMg) phase formed and grew along the eutectic microstructure and disappeared into the matrix completely when it was homogenized at 460 ��C for 24 h. It could be found that the evolution of primary eutectic structure of 7085 alloy consisted of three processes, dissolution of eutectic ��+T microstructure, phase transformation from T phase to S phase and the dissolution of S phase. The optimum homogenization parameter was at 470 ��C for 24 h.
Key words: 7085 aluminum alloy; homogenization treatment; microstructural evolution; intermetallic phase
1 Introduction
7085 aluminum alloy was introduced by Alcoa a few years ago as the new generation of high strength thick plate alloy. It is widely used in aerospace industry owing to its high strength, high toughness and low quenching sensitivity [1,2]. As is well known, the microsegregation of main elements is inevitable for 7xxx aluminum alloy during casting, and massive undissolved secondary phases occur in the ingot. The constituent phases, such as ��(MgZn2), T(AlZnMgCu), S(Al2CuMg) and ��(Al2Cu), are commonly observed in Al-Zn-Mg- Cu alloys [2-7]. However, the coarse intermetallic phases (above 1 ��m) usually deteriorate the stress corrosion resistance, fracture toughness and fatigue strength of these alloys [5,8,9]. Therefore, in order to get good workability and service performance of alloys, homogenization treatment is needed for Al-Zn-Mg-Cu alloy ingots before plastic processing. It plays a key role in dissolving large soluble non-equilibrium intermetallic phases and eliminating the microsegregation in the matrix [10-12].
Intermetallic phases and their evolution during homogenization of Al-Zn-Mg-Cu alloy have been extensively investigated. Usually, phase dissolution and transition will differ in different alloying elements and homogenized processes [13,14]. CHEN et al [15] revealed that after pretreatment at 450 ��C for 35 h, ��(MgZn2) phase dissolved completely, while T((AlCuZn)49Mg32) and S(Al2CuMg) phases were still observed in 7055 alloy after being treated at 470 ��C for 50 h. DENG et al [5] reported that intermetallic S(Al2CuMg) phase nucleated and grew along the grain boundaries during homogenization at 400 ��C for 10 h, and disappeared completely after treatment under conditions of (400 ��C, 10 h) + (470 ��C, 24 h) + (485 ��C, 4 h). However, HOU et al [6] represented that during the high-temperature homogenization heat treatment at 485 ��C for 5-20 min, ��(MgZn2) phase was transformed to S(Al2CuMg) phase. LIU et al [3] reported that the phase transition from Mg(Zn,Al,Cu)2 to S(Al2CuMg) phase was very difficult when Zn content was higher than 8% (mass fraction). To date, the evolution behavior of the intermetallic phases during different homogeniza-tion temperatures of 7085 alloy is not well known. The purpose of the present work was to investigate the effects of homogenization temperature on the microstructure and composition distribution of 7085 aluminum alloy, and to study the evolution of intermetallic phases during homogenization. Besides, the homogenization kinetic equation of the experimental 7085 alloy was also established.
2 Experimental
The 7085 alloy with dimensions of d260 mm �� 800 mm was produced by semi-continuous casting method. The chemical composition of this alloy is Al-7.24Zn-1.42Mg-1.50Cu-0.092Zr-0.046Fe-0.041Si (mass fraction, %). Slices with dimensions of 10 mm �� 10 mm �� 3 mm were cut from the half position between the center and the surface of as-cast ingot. The specimens were homogenized for 24 h at 440, 450, 460, 470 and 480 ��C, respectively. The homogenization was performed in a salt bath furnace with a heating rate of 5 ��C/min. The temperature variation was controlled within ��2 ��C. These homogenized specimens were all air-cooled to room temperature.
The samples for microstructural observations were examined by optical microscopy (OM, Leica) and scanning electron microscopy (SEM, FEI Sirion 200). The specimens for OM observation were chemically etched by anode coating solution (11 g H3BO3, 20 mL HF and 1000 mL H2O). SEM observation was carried out in the backscattered electron imaging mode, operated at 20 kV. The map and line scanning analyses were conducted on an energy dispersive X-ray spectrometer (EDS, Genesis 60S) operated at 20 kV. Differential scanning calorimetry (DSC) analyses were conducted in a purified argon atmosphere using a NETZSCH STA-449C instrument with a heating rate of 10 ��C/min from room temperature to 700 ��C. X-ray diffraction (XRD) studies were undertaken on a D/max 2500 PC diffractometer with Cu K��1 radiation.
3 Results and discussion
3.1 Characterization of as-cast microstructure
The microstructures of as-cast alloy are shown in Fig. 1. It was a typical as-cast eutectic structure showing serious dendritic segregation (Fig. 1(a)). And lots of intermetallic phases existed at the grain boundaries. The average grain size of as-cast alloy was about 140 ��m. Table 1 lists the EDS results of the intermetallic phases in as-cast alloy. Only Al, Zn, Mg and Cu elements were detected by EDS at Point A, suggesting that it may be the mixture of ��(Al) and quaternary phase AlZnMgCu. The grey phase at Point B was close to ��(Al2Cu) with solute of elements Zn and Mg. EDS analysis revealed that the lamellar eutectic phase at Point C was Al7Cu2Fe phase. MONDAL and MUKHOPADHYAY [16] studied the nature of intermetallic alloy phases in 7055 aluminum alloy, and found that ��, T and S phases were solid solutions with extended composition ranges. It can be inferred that the quaternary phase AlZnMgCu at Point A belonged to T phase.
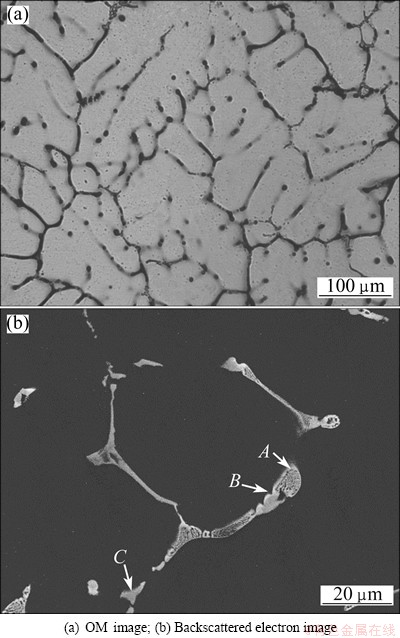
Fig. 1 Microstructures of as-cast 7085 alloy
Table 1 Chemical compositions of intermetallic phases in as-cast 7085 alloy shown in Fig. 1(b)
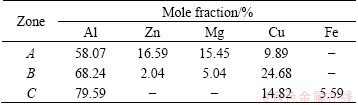
Figure 2 shows the microstructure and the distribution of main elements Al, Cu, Zn and Mg in as-cast 7085 alloy. The segregation of elements Cu, Zn and Mg is obvious at the grain boundaries, and the concentration of the elements decreases from grain boundary to inside. It is obvious that the segregation of element Cu is greater than that of Zn and Mg. It is well known that the segregation and component inhomogeneity of as-cast alloy usually have hereditary influence on semi-products and finished products. Therefore, the homogenization is necessary to eliminate severe microsegregation in as-cast 7085 alloy. Generally, an equation is used to describe the relationship between diffusion coefficient and temperature [17,18], and the mathematical formula is shown as follows:
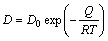 (1)
(1)
where D0 is the diffusion coefficient, Q is the diffusion activation energy, R is the mole gas constant and T is the thermodynamic temperature. From Eq. (1), it can be seen that thermodynamic temperature is proportional to diffusion coefficient, in other words, the higher the temperature is, the faster the diffusion speed is. As a result, it is easy to eliminate the microsegregation in the as-cast alloy. However, it should be noted that the overburnt phenomenon will occur in samples which are treated at high temperatures. So, an optimum parameter of homogenization is very important to explore for practical application.

Fig. 2 SEM image (a) and main elements distribution (b-e) of as-cast 7085 alloy
3.2 DSC analysis of as-cast and homogenized alloys
Figure 3 shows the DSC curves of as-cast ingot and homogenized alloys at different temperatures for 24 h. Two endothermic peaks were observed in the as-cast 7085 alloy, which were sited at 475.1 and 637.4 ��C, respectively. The endothermic peak at 475.1 ��C is reduced gradually with the increase of homogenization temperature, which may correspond to the dissolution of some non-equilibrium phases during homogenization. The endothermic peak at 637.4 ��C corresponds to the melting point of 7085 alloy. Figure 4 represents the optical microstructures of 7085 alloy homogenized at 470 and 480 ��C for 24 h, respectively. Upon homogenization at 470 ��C for 24 h, most of the residual phases are dissolved and the dendritic network structure is reduced. However, when the temperature increases to 480 ��C, the melting spherical compounds can be observed both at the grain boundaries and in triple conjunctions (Fig. 4(b)), indicating that the specimen was seriously over-burnt. In conclusion, the suitable temperature for homogenization is 470 ��C.
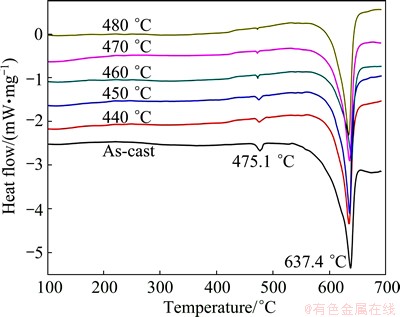
Fig. 3 DSC curves of as-cast ingot and homogenized alloys at different temperatures for 24 h
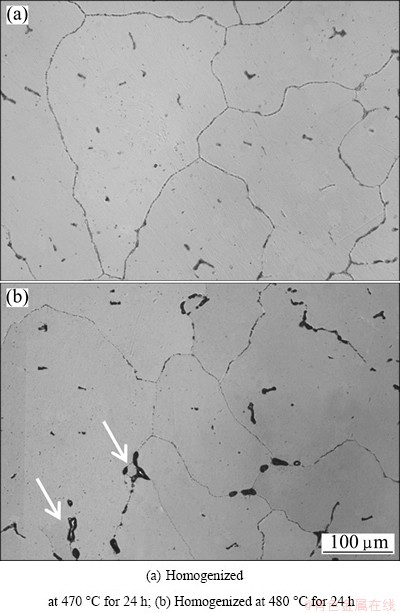
Fig. 4 Optical microstructures of specimens
3.3 XRD analysis
XRD results of 7085 alloy are shown in Fig. 5. The microstructure of as-cast 7085 alloy contained ��(MgZn2) phase and Al7Cu2Fe phase. It should be noted that ��(MgZn2) phases were not detected by EDS in Section 3.1, because during the semi-continuous casting process, high cooling speed caused the non-equilibrium solid solution of elements Al and Cu in ��(MgZn2) phase [2,19]. Consequently, plenty of eutectic ��(Al)+T(AlZnMgCu) microstructures instead of ��(MgZn2) phases were observed in as-cast microstructure of 7085 alloy. S(Al2CuMg) phase was not identified from XRD pattern in as-cast 7085 alloy. When the alloy was homogenized at 440 ��C for 24 h, the peak of S(Al2CuMg) phase appeared. And with the increase of homogenization temperature, diffraction peak of S(Al2CuMg) decreased and then disappeared at 460 ��C for 24 h. When temperature reached 470 ��C, there was no obvious diffraction peak in XRD patterns except for ��(Al) and Al7Cu2Fe. In this work, ��(Al2Cu) phases were not detected by XRD, which may be due to too low content of this phase in the matrix.
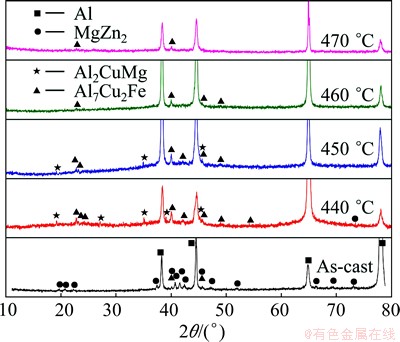
Fig. 5 XRD patterns of 7085 alloy heat-treated at different temperatures for 24 h

Fig. 6 Backscattered electron images of 7085 alloy homogenized at different temperatures for 24 h
3.4 Microstructural evolution of as-cast and homogenized alloys
Figure 6 shows the distribution of intermetallic phases of as-cast ingot and specimens homogenized at different temperatures for 24 h. Serious segregation existed at the grain boundaries of as-cast 7085 alloy, and the intermetallic phases were coarse and continuously distributed. After the alloy was homogenized at 440 ��C for 24 h, the volume of interdendritic phases decreased significantly, the continued intermetallic phases along grain boundaries became discontinuous, and the residual phases became smaller and sparser. It should be noted that a grey intermetallic phase was observed in the matrix (Figs. 7(a) and (b)). According to the EDS analysis, it contained 21.71% Mg, 58.09% Al and 20.20% Cu (mole fraction), the mole ratio of Mg to Cu was close to 1:1. Therefore, it could be considered that the grey intermetallic phase was S(Al2CuMg) phase. S(Al2CuMg) was not detected in as-cast microstructure by using SEM, EDS and XRD measurements, which agreed with the results from Refs. [2,5,20]. During homogenization, with the diffusion of alloying elements from eutectic structures to the matrix, ��+T eutectic microstructure gradually dissolved into the matrix, while S(Al2CuMg) phase formed and grew along the eutectic microstructure, which indicated that the transformation from T to S(Al2CuMg) phase occurred [2]. The diffusion velocity of Cu was slower than that of Mg and Zn, which resulted in higher concentration of Cu in the eutectic structure regions. FAN et al [20] pointed out that during the transformation from T to S(Al2CuMg), the driving force for the phase transformation from T to S(Al2CuMg) should be supersaturation of Cu in the regions of eutectic structures, which made S(Al2CuMg) a stable phase. However, when the specimen was homogenized at 460 ��C for 24 h, S(Al2CuMg) phase could not be detected both in X-ray diffraction and EDS analysis, which showed that S(Al2CuMg) phase gradually disappeared and finally dissolved into the matrix completely with increasing homogenization temperature. This was because the diffusion of alloying elements from the interdendritic regions to the matrix led to the decrease of the supersaturation extent of alloying elements in eutectic regions [2]. After homogenization at 470 ��C for 24 h, the least amount of residual phases was obtained (Fig. 6(e)), and the residual phases were magnified in Fig. 7(c). According to the results of EDS analysis, the residual phase contained 84.94% Al, 4.67% Fe and 10.39% Cu (mole fraction), there were no Mg and other element in the phase, which indicated that the residual phase belonged to Al7Cu2Fe phase. Al7Cu2Fe phase was an indissolvable impurity phase in the 7xxx aluminum alloys, and hard to be completely eliminated by homogenization treatment [21]. According to the results of XRD, SEM and EDS analyses, it can be concluded that the evolution of primary eutectic structure during homogenization consists of three processes as follows: dissolution of eutectic ��+T microstructure, phase transformation from T to S phase and the dissolution of S phase.
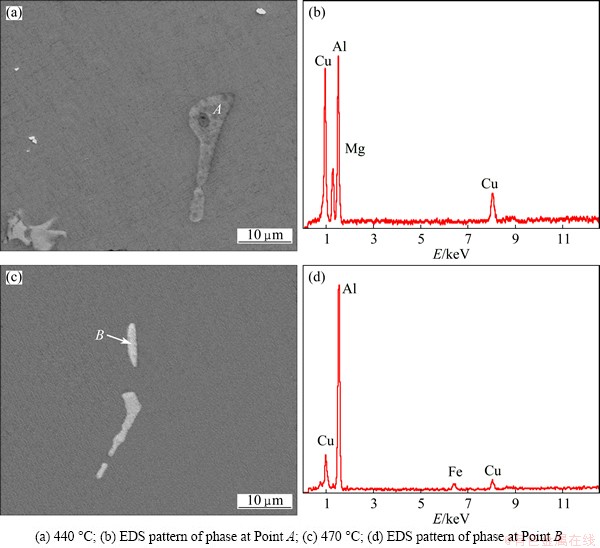
Fig. 7 Backscattered electron images (a, c) and EDS results (b, d) of alloys homogenized at different temperatures for 24 h

Fig. 8 Line scanning analysis of as-cast 7085 alloy
3.5 Line scanning and homogenization kinetic analysis
The line scanning analysis of as-cast 7085 alloy is shown in Fig. 8. It can be seen that the distribution of Zn, Mg and Cu elements along interdendritic region varied periodically. The diffusion law along interdendritic region is an important and suitable method to evaluate the element distribution during homogenization [5,17]. According to Refs. [22,23], the initial concentration of the elements along the interdendritic region could be approached by Fourier series components in a cosine function:
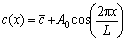 (2)
(2)
where 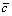 is the average concentration of the element, A0 is the initial amplitude of the composition segregation, and L is the interdendritic spacing, which can be shown as
is the average concentration of the element, A0 is the initial amplitude of the composition segregation, and L is the interdendritic spacing, which can be shown as
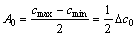 (3)
(3)
According to the second Fick��s law and the boundary conditions [24], A(t) can be expressed as
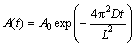 (4)
(4)
From Eq. (1) to Eq. (4), the equation can be rewritten as
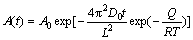 (5)
(5)
LIU et al [25] assumed that the element distribution was homogeneous when the composition segregation amplitude was reduced to 1%, that is
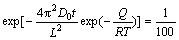 (6)
(6)
By taking natural logarithms of both sides of Eq. (6), we can obtain
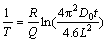 (7)
(7)
Equation (7) is the homogenization kinetic equation. For a given alloy, the values of diffusion coefficient D0 and diffusion activation energy Q can be obtained. Therefore, the corresponding homogenization kinetic curve can be simulated. It was reported that the diffusion coefficient of Cu element in aluminum alloy was smaller than that of Mg and Zn at the same temperature [26]. And WANG et al [19] represented that the homogenization process was mainly controlled by the diffusion of Cu. Substituting D0(Cu) (0.084 cm2/s) and Q(Cu) (136.8 kJ/mol) into Eq. (7), the homogenization kinetic curves of 7085 alloy for different interdendritic spacings can be obtained, as shown in Fig. 9. It can be seen that if the interdendritic spacing L is larger, higher temperature and longer time are needed for full homogenization. And the homogenization time is reduced obviously with the increase of the homogenization temperature. According to Eq. (7), suitable homogenizing parameters were obtained by substituting the average interdendritic spacing L into Eq. (7). Figure 10 shows the relationship between homogenization time and interdendritic spacing of aluminum alloy at 470 ��C. The average interdendritic spacing L of as-cast 7085 alloy is 38.5 ��m, which is based on quantitative metallographic analysis. As seen in Fig. 10, the corresponding homogenization time is 25 h. The calculated results agree well with the experimental results in the present work. Hence, the homogenization time is suitable at 24 h.
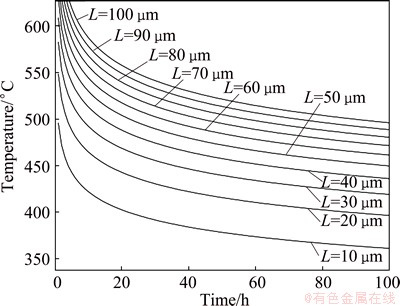
Fig. 9 Homogenization kinetics curves of 7085 alloy
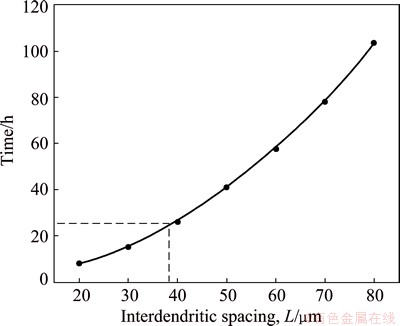
Fig. 10 Relationship between homogenization time and interdendritic spacing of 7085 aluminum alloy at 470 ��C
Figure 11 represents the line scanning traces of 7085 alloy homogenized at 470 ��C for 24 h. It can be seen from Fig. 11 that the segregation of Zn and Mg is almost eliminated, and only small segregation of Fe and Cu exists along the grain boundary. This shows that the element distribution is more homogeneous and only small numbers of irregular Al7Cu2Fe phases exist in the aluminum matrix after homogenization. The optimum single-stage homogenization process of 7085 alloy is at 470 ��C for 24 h.
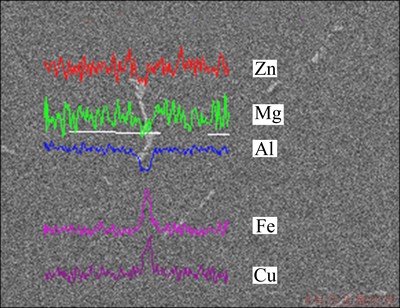
Fig. 11 Line scanning analysis of 7085 alloy homogenized at 470 ��C for 24 h
4 Conclusions
1) Serious segregation exists in as-cast 7085 alloy, the distribution of Cu, Zn and Mg elements along interdendritic region vary periodically. The as-cast microstructure consists of eutectic ��(Al)+T(AlZnMgCu), ��(Al2Cu) phase and Al7Cu2Fe phase.
2) By increasing homogenization temperature, the intermetallic phases become smaller and sparser, and the main elements are distributed more homogeneously.
3) The evolution of primary eutectic structure during homogenization consists of three processes: dissolution of eutectic ��+T microstructure, phase transformation from T to S phase and the dissolution of S phase.
4) The optimum single-stage homogenization treatment of 7085 alloy is at 470 ��C for 24 h, which well agrees with the results of homogenization kinetic analysis.
References
[1] SHUEY R T, BARLAT F, KARABIN M E, CHAKRABARTI D J. Experimental and analytical investigations on plane strain toughness for 7085 aluminum alloy [J]. Metallurgical Materials Transactions A, 2009, 40: 365-376.
[2] HE L Z, LI X H, ZHU P, CAO Y H, GUO Y P, CUI J Z. Effects of high magnetic field on the evolutions of constituent phases in 7085 aluminum alloy during homogenization [J]. Materials Characterization, 2012, 71: 19-23.
[3] LIU Y, JIANG D M, XIE W L, HU J, MA B R. Solidification phases and their evolution during homogenization of a DC cast Al-8.35Zn- 2.5Mg-2.25Cu alloy [J]. Materials Characterization, 2014, 93: 173-183.
[4] GUO S, NING Z L, ZHANG M X, CAO F Y, SUN J F. Effects of gas to melt ratio on the microstructure of an Al-10.83Zn-3.39Mg- 1.22Cu alloy produced by spray atomization and deposition [J]. Materials Characterization, 2014, 87: 62-69.
[5] DENG Y, YIN Z M, CONG F G. Intermetallic phase evolution of 7050 aluminum alloy during homogenization [J]. Intermetallics, 2012, 26: 114-121.
[6] HOU W R, JI W B, ZHANG Z H, XIE J X, CHENG X L. The effect of homogenization temperature on the corrosion resistance of extruded 7050 Al-alloy bars [J]. Journal of Materials Processing Technology, 2014, 214: 635-640.
[7] DENG Y L, WAN L, WU L H, ZHANG Y Y, ZHANG X M. Microstructural evolution of Al-Zn-Mg-Cu alloy during homogenization [J]. Journal of Materials Science, 2011, 46: 875-881.
[8] LI Chun-mei, CHEN Zhi-qian, ZENG Su-min, CHENG Nan-pu, CHEN Tian-xiao. Intermetallic phase formation and evolution during homogenization and solution in Al-Zn-Mg-Cu alloys [J]. Science China Technological Sciences, 2013, 56: 2827-2838.
[9] SHARMA M M. Microstructural and mechanical characterization of various modified 7xxx series spray formed alloys [J]. Materials Characterization, 2008, 59: 91-99.
[10] DEHMAS M, WEISBECKER P, GEANDIER G, ARCHAMBAULT P, AEBY�CGAUTIER E. Experimental study of phase transformations in 3003 aluminium alloys during heating by in situ high X-ray synchrotron radiation [J]. Journal of Alloys and Compounds, 2005, 400: 116-124.
[11] WANG Guo-jun, XIONG Bai-qing, ZHANG Yong-an, LI Zhi-hui, LI Pei-yue. Microstructural characterization of as-cast and homogenized 2D70 aluminum alloy [J]. International Journal of Minerals Metallurgy and Materials, 2009, 16: 427-431.
[12] WU L M, WANG W H, HSU Y F, TRONG S. Effects of homogenization treatment on recrystallization behavior and dispersoid distribution in an Al-Zn-Mg-Sc-Zr alloy [J]. Journal of Alloys and Compounds, 2008, 456: 163-169.
[13] EIVANI A R, AHMED H, ZHOU J, DUSZCZYK J. Evolution of grain boundary phases during the homogenization of AA7020 aluminum alloy [J]. Metallurgical Materials Transactions A, 2009, 40: 717-728.
[14] LI G F, ZHANG X M, ZHU H F. Effect of minor Er and Y addition on homogenization of as-cast Al-Zn-Mg-Cu-Zr alloys [J]. Journal of Aeronautical Materials, 2010, 30: 1-6.
[15] CHEN K H, LIU H W, ZHANG Z, LI S, TODD R I. The improvement of constituent dissolution and mechanical properties of 7055 aluminum alloy by stepped heat treatments [J]. Journal of Materials Processing Technology, 2003, 142: 190-196.
[16] MONDAL C, MUKHOPADHYAY A K. On the nature of T(Al2Mg3Zn3) and S(Al2CuMg) phases present in as-cast and annealed 7055 aluminum alloy [J]. Materials Science and Engineering A, 2005, 391: 367-376.
[17] LI Hong-ying, SU Xiong-jie, YIN Hao, HUANG De-sheng. Microstructural evolution during homogenization of Al-Cu-Li- Mn-Zr-Ti alloy [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(9): 2543-2550.
[18] LI Bo, PAN Qing-lin, SHI Yun-jia, LI Chen, YIN Zhi-ming. Microstructural evolution of Al-Zn-Mg-Zr alloy with trace amount of Sc during homogenization treatment [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(12): 3568-3574.
[19] WANG H J, XU J, KANG Y L, TANG M G, ZHANG Z F. Study on inhomogeneous characteristics and optimize homogenization treatment parameter for large size DC ingots of Al-Zn-Mg-Cu alloys [J]. Journal of Alloys and Compounds, 2014, 585: 19-24.
[20] FAN X G, JIANG D M, MENG Q C, ZHONG L. The microstructural evolution of an alloy during homogenization [J]. Materials Letters, 2006, 60: 1475-1479.
[21] YAN Li-zhen, ZHANG Yong-an, LI Xi-wu, LI Zhi-hui, WANG Feng, LIU Hong-wei, XIONG Bai-qing. Microstructural evolution of Al-0.66Mg-0.85Si alloy during homogenization [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(4): 939-945.
[22] SHEWMAN P G. Diffusion in solids [M]. New York: McGraw-Hill, 1963: 61.
[23] JIANG Hai-chun, YE Ling-ying, ZHANG Xin-ming, GU Gang, ZHANG Pan, WU Yu-long. Intermetallic phase evolution of 5059 aluminum alloy during homogenization [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(12): 3553-3560.
[24] SAMARAS S N, HAIDEMENOPOULOS G N. Modelling of microsegregation and homogenization of 6061 extrudable Al-alloy [J]. Journal of Materials Processing Technology, 2007, 194: 63-73.
[25] LIU X Y, PAN Q L, FAN X. Microstructural evolution of Al-Cu-Mg-Ag alloy during homogenization [J]. Journal of Alloys and Compounds, 2009, 484(1-2): 790-794.
[26] LI Wen-bin, PAN Qing-lin, XIAO Yan-ping, HE Yun-bin, LIU Xiao-yan. Microstructural evolution of ultra-high strength Al-Zn- Mg-Cu-Zr alloy containing Sc during homogenization [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(10): 2127-2133.
DC����7085���Ͻ��ھ��Ȼ������е���֯�ݱ�
ʷ�˼�1��������1�����μ�1����־��2����־��2
1. ���ϴ�ѧ ���Ͽ�ѧ�빤��ѧԺ����ɳ 410083��
2. �㶫������ҵ����˾����ɽ 528133
ժ Ҫ�����ù�ѧ������ɨ��羵��EDS��������DSC������X����������ֶ��о�DC����7085���Ͻ���Ȼ�̬����֯���ݱ䡣���������7085���Ͻ����̬��֯�д������ص�֦��ƫ�������紦���ڴ����Ĺ�������֯�������ࡣ������(Al)+T(AlZnMgCu)����֯�ھ��Ȼ�����������������С���������S(Al2CuMg)�ع�����֯�γɳ�����460 ��C��24 h�ľ��Ȼ���������ȫ��������С�7085���Ͻ��ԭʼ�����ṹ���ݱ������+T��������֯���ܽ⡢T����S���ת�估S����ܽ⡣��ѵľ��Ȼ��ƶ�Ϊ470 ��C��24 h��
�ؼ��ʣ�7085���Ͻ𣻾��Ȼ�����������֯�ݱ䣻��������
(Edited by Wei-ping CHEN)
Foundation item: Project (2012CB619503) supported by the National Basic Research Program of China; Project (2013AH100055) supported by the Special Foundation for Science and Technology Innovation of Foshan, China
Corresponding author: Qing-lin PAN; Tel: +86-731-88830933; E-mail: csupql@163.com
DOI: 10.1016/S1003-6326(15)63993-0