Trans. Nonferrous Met. Soc. China 23(2013) 2079-2084
Folate-conjugated Fe3O4 nanoparticles for in vivo tumor labeling
Chun-jiao ZHOU1, Shao-hua WANG2, Yu ZHOU1, Peng-fei RONG2, Zi-zi CHEN2, Jin-yan LIU2, Jian-da ZHOU2
1. College of Science, Hunan Agricultural University, Changsha 410128, China;
2. The Third Xiangya Hospital, Central South University, Changsha 410013, China
Received 8 March 2012; accepted 10 October 2012
Abstract: Highly biocompatible superparamagnetic Fe3O4 nanoparticles were synthesized by amide of folic acid (FA) ligands and the NH2-group onto the surface of Fe3O4 nanoparticles. The as-synthesized folate-conjugated Fe3O4 nanoparticles were characterized by X-ray diffraction diffractometer, transmission electron microscope, FT-IR spectrometer, vibrating sample magnetometer, and dynamic light scattering instrument. The in vivo labeling effect of folate-conjugated Fe3O4 nanoparticles on the hepatoma cells was investigated in tumor-bearing rat. The results demonstrate that the as-prepared nanoparticles have cubic structure of Fe3O4 with a particle size of about 8 nm and hydrated diameter of 25.7 nm at a saturation magnetization of 51 A・m2/kg. These nanoparticles possess good physiological stability, low cytotoxicity on human skin fibroblasts and negligible effect on Wistar rats at the concentration as high as 3 mg/kg body mass. The folate-conjugated Fe3O4 nanoparticles could be effectively mediated into the human hepatoma Bel 7402 cells through the binding of folate and folic acid receptor, enhancing the signal contrast of tumor tissue and surrounding normal tissue in MRI imaging. It is in favor of the tumor cells labeling, tracing, magnetic resonance imaging (MRI) target detection and magnetic hyperthermia.
Key words: Fe3O4 nanoparticles; superparamagnetic property; folic acid; tumor; conjugation; magnetic resonance imaging (MRI); cell labeling
1 Introduction
Tumor cell membranes and Fe3O4 nanoparticles are both negatively charged, which affects tumor cell uptake of Fe3O4 nanoparticles. The naked Fe3O4 nanoparticles cannot effectively label tumor cells. To achieve an effective labeling of magnetic nanoparticles on tumor cells, the optimal method is to couple magnetic nanoparticles to specific targeting molecules which have high affinity with the target tumor cells [1,2]. Many specific targeting molecular markers for the tumor cells were mentioned in many reports [3-7]. However, some agents are expensive and anti-body molecules are too big and hinder the crossing of the coupled magnetic nanoparticles through biological barriers [8]. Folic acid is a small-molecule vitamin that is essential for the human body, especially single carbon metabolism of eukaryotic cells and nucleoside synthesis. It is an important coenzyme in DNA synthesis and serves as a co-factor in the synthesis of several enzyme systems. Its molecular structure is shown in Fig. 1. Compared with protein-targeting molecules such as antibodies, folic acid is stable, low cost and non-immunogenic. In addition, folic acid binds strongly to folic acid receptors through ligand-receptor binding. Therefore, folic acid can be efficiently mediated into tumor cells and may be useful in targeting these cells [9].
The expression of the folate receptor is highly conserved in normal animal cells. This may be due to the lack of a key enzyme for folate biosynthesis in normal animal cells, so folate receptors on the cell surface actively absorb exogenous folate to maintain normal life activities. However, folate receptors are over-expressed in many malignant tumors, such as ovarian cancer, nasopharyngeal cancer, kidney cancer, breast cancer, endometrial cancer, testicular cancer, and liver cancer. Because folate receptors are over-expressed on the surface of malignant tumor cells, and folic acid has a high affinity and specificity with acid analogs, we can mediate folic acid endocytosis into tumor cells through specific binding with folic acid substances. Therefore, folic acid is of high potential as a tumor-targeting molecule [10-14].
In this study, the APTES-modified superparamagnetic Fe3O4 nanoparticles were conjugated to folic acid, and the folate-conjugated Fe3O4 nanoparticles were mediated into hepatoma cells in tumor-bearing rats, in order to elucidate the in vivo labeling effect of folate-conjugated Fe3O4 nanoparticles on the hematoma cells, MRI imaging and magnetic hyperthermia [15].
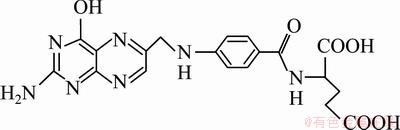
Fig. 1 Structure of folic acid
2 Experimental
Ferric chloride hexahydrate (99.99%), ferrous sulfate heptahydrate (99.9%), sodium dodecyl benzene sulfonate (DBS, analytical reagent), folic acid (analytical reagent), dimethyl sulfoxide (DMSO, analytical reagent) and toluene (analytical reagent) were provided by Beijing Chemical Reagent Company, China. ATPES (analytical reagent), 1,3-dicyclohexylcarbodiimide (DCC, analytical reagent), N-hydroxysuccinimide (NHS, analytical reagent) and pentobarbital sodium imported packaging were all provided by Shanghai Chemical Reagent Company, China. RPMl1640 medium (Gibco, USA), human skin fibroblast cell line (from Dr. Yong Chen, Hunan Province Population and Family Planning Commission, China), SRB (Sigma, USA), and male Wistar rats of 180 g were provided by the Experimental Animal Center, Central South University, China.
Folic acid (0.132 g) was dissolved in 10 mL DMSO, subjected to ultrasound for 10 min, mixed with 35 mg NHS, 62 mg DCC and 5 mL ATPES-modified Fe3O4 nanoparticles (containing 5 mg Fe3O4 nanoparticles), subjected to ultrasound for 20 min, and stirred overnight. The mixed solution was separated with an external magnetic field, alternately rinsed with ultra-pure water and ethanol 4-5 times, then put through dialysis and dispersed in ultrapure water, to obtain folate-conjugated Fe3O4 nanoparticles.
The structure of the sample was determined with a Bruker D8 X-ray diffractometer. Scanning voltage was 40 kV. Scanning current was 40 mA at 0.25 (°)/min with sampling interval of 0.02°. TEM was done using JEM 3010 microscope. FTIR spectra were obtained using a WQF 410 FTIR spectrometer with a resolution of 4 cm-1 and scans of 32. A small amount of nano-composite powders were milled with KBr, and the mixture was pressed into a disc for analysis. The magnetic measurement was measured using a Lake Shore 7310 VSM at 27 °C. The dynamic light scattering characterization was measured with dynamic light scattering instrument HPPS-ET 5002 (Malvern Instruments, UK) using the He-Ne laser (λ=632.8 nm). The data were collected using back-scattering mode at (25±0.1) °C.
Human skin fibroblast cell lines at logarithmic growth phase were cultured and added to the first line of wells in a 24-well plate, with 0.2 mL in each well. Cells were respectively incubated with folate-conjugated superparamagnetic Fe3O4 nanoparticles (100, 200, 300, 400 μg/mL Fe) and saline. After cells were adhered to 24-well plates and culture fluid was disposed, 50 μL of 500 mL/L trichloroacetic acid was added to each well, and the plate was kept at 4 °C for 1 h. Cells were washed 5 times. After air drying, cells were stained with 4 g/L SRB for 0.5 h, washed 3 times with acetic acid and dried, then dissolved in tris solution. The absorbance at 515 nm was determined with enzyme-linked immunosorbent assay.
Folate-conjugated Fe3O4 nanoparticles were injected via the tail vein of Wistar rats, which were observed for 72 h for activities, death and damage to major organs after death. The acute side effects were also observed to make a preliminary evaluation of its safety.
A human hepatocellular carcinoma subcutaneous tumor xenograft model was established in tumor-bearing rats, which were housed in a standard superclean environment for 1-2 weeks and used for in vivo cell labeling experiments when the subcutaneous tumor grew to 5 mm3. After tumor-bearing rats were anesthetized with 3% sodium pentobarbital, an MRI scan of their legs was performed to obtain plain scan unenhanced T2WI images. MRI was executed using a 1.5 T imager (Avanto 1.5 T, Simenze, Germany). The scanning protocol included a T2-weighted fast spin-echo sequence with a repetition time of 2000 ms, an echo time of 68 ms, a field-of-view of 120 mm, a matrix of 512×512 and a slice thickness of 3 mm. Following this protocol, 9 axial liver slices were taken with 4 acquisitions. Then the same tumor-bearing rat was injected with folate-conjugated Fe3O4 nanoparticles via the tail vein with 0.4 mg/kg body mass. 45 min later, the same position was scanned with the same parameters to obtain an enhanced T2WI image.
After MRI examination was completed, rats were killed quickly and the tumor was removed. The tumor growth and invasion situation were observed. Samples were fixed with 10% formalin, embedded in paraffin, and cut into serial sections. One section was selected from every 5 sections with 7 μm in thickness. Two sets of slices for hematoxylin-eosin staining and Prussian blue staining were collected from each sample. The tumor morphology and Fe content were determined under a light microscope.
3 Results and discussion
3.1 Folate-conjugated superparamagnetic Fe3O4 nanoparticles characterization
The folate-conjugated superparamagnetic Fe3O4 nanoparticles were characterized with X-ray diffraction analysis, as shown in Fig. 2. There are seven diffraction peaks at 2θ values of 30.1°, 35.4°, 43.1°, 53.2°, 57.0°, 62.7° and 74.0°, corresponding to the cubic phase of Fe3O4 (220), (311), (400), (422), (511), (440) and (533) planes. The diffraction peak position and relative intensity are well matched with the standard card (JCSPD No. 82―1533) [16]. There are no other impurity peaks observed, indicating that folate-conjugated Fe3O4 nanoparticles have a cubic structure. The particle size estimated by the Scherrer formula is about 8 nm.
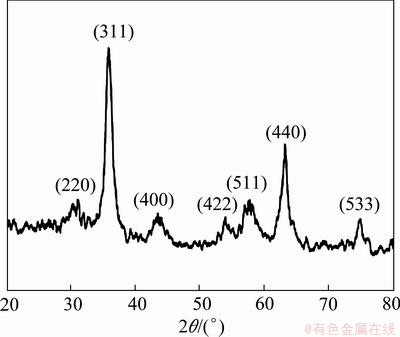
Fig. 2 XRD pattern of folate-conjugated Fe3O4 nanoparticles
TEM image of folate-conjugated superparamagnetic Fe3O4 nanoparticles is shown in Fig. 3. Most of the nanoparticles are spherical and the average size of particles is initially estimated as 8 nm, which is consistent with the X-ray diffraction analysis.
The surface groups of folate-conjugated super- paramagnetic Fe3O4 nanoparticles were characterized by FTIR (Fig. 4). The absorption peaks at 596 cm-1 and 445 cm-1 are attributed to Fe―O bond absorption. Absorption at 1643 cm-1 can be attributed to N―H bending vibration, which is also favorable evidence for the existence of primary amine in molecules. Absorption at 1604 cm-1 corresponded to the aromatic ring stretching vibration in the folic acid molecule, absorption at 1396 cm-1 corresponded to the benzoic vibrations in folic acid molecule [14], and absorption at 1533 cm-1 can be attributed to the characteristic absorption of amide [17]. Infrared results proved that folic acid molecules have successfully conjugated onto the surface of Fe3O4 nanoparticles.

Fig. 3 TEM image of folate-conjugated Fe3O4 nanoparticles
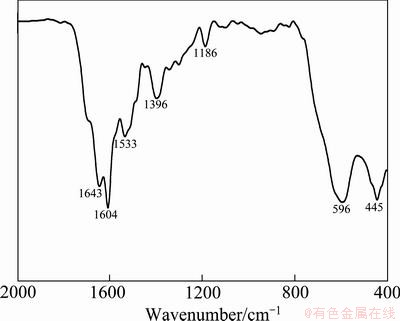
Fig. 4 FTIR image of folate-conjugated Fe3O4 nanoparticles
Magnetic hysteresis loop of folate-conjugated superparamagnetic Fe3O4 nanoparticles is shown in Fig. 5. The hysteresis loop passes through the original point, and the coercivity and remanence are close to zero, indicating that folate-conjugated Fe3O4 nanoparticles are superparamagnetic. Saturation magnetization is 51 A・m2/kg.
The particle size distribution of folate-conjugated Fe3O4 nanoparticles is shown in Fig. 6. Results show that the particle size distribution is relatively narrow, with an average of 25.7 nm.
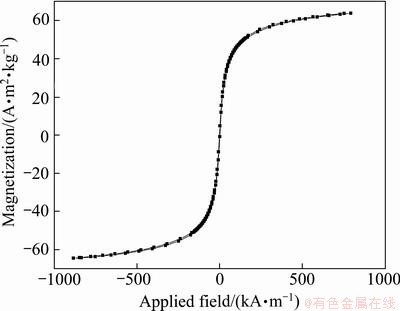
Fig. 5 Magnetic hysteresis loop of folate-conjugated Fe3O4 nanoparticles
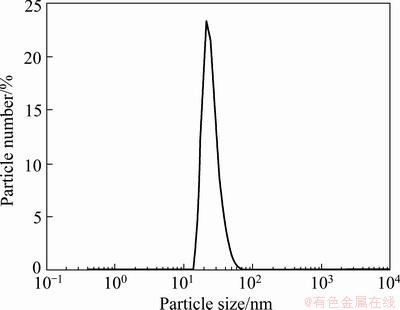
Fig. 6 Particle size distribution of folate-conjugated Fe3O4 nanoparticles
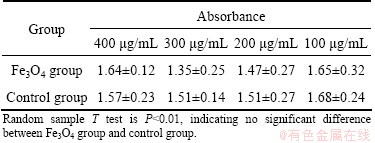
Table 1 Effect of folate-conjugated superparamagnetic Fe3O4 nanoparticles on growth of human skin fibroblasts by SRB assay
3.2 Cell safety test of folate-conjugated superparamagnetic Fe3O4 nanoparticles
Cell safety test of folate-conjugated super-paramagnetic Fe3O4 nanoparticles by SRB assay (Table 1) shows that, the folate-conjugated super- paramagnetic Fe3O4 nanoparticles have the similar influence on the growth of human skin fibroblasts. As normal saline, they cannot significantly inhibit cell growth compared with normal saline. Statistical analyses show no significant difference between the two groups. This is evidence that folate-conjugated superparamagnetic Fe3O4 nano- particles have no significant toxicity on human skin fibroblasts.
Folate-conjugated Fe3O4 nanoparticles with the concentration of 1 mg/kg body mass were injected into tail vein of Wistar rats. After 72 h, there is no abnormal activity, no rat death, or no marked changes of major organs after death. There is no significant difference between two groups. When the concentrations reach 3 mg/kg body mass, the heart rate, pulse rate and breathing of part of the Wistar rat speed up with no rat death, or no marked changes of major organs after death. When the concentrations reach 30 mg/kg body mass, part of the Wistar rats were killed. The results of in vivo acute toxicity test of Wistar rats show that folate-conjugated superparamagnetic Fe3O4 nanoparticles have negligible effect on Wistar rats at the concentration as high as 3 mg/kg body mass.
3.3 Tumor cell labeling results
Figure 7 shows the unenhanced T2WI image of the leg of tumor-bearing rats (Fig. 7(a)) and the enhanced T2WI image of the leg of tumor-bearing rats at 40 min after injection of folate-conjugated Fe3O4 nanoparticles (Fig. 7(b)). In MRI unenhanced T2WI imaging of the leg of tumor-bearing rats, the MRI signals showed no significant difference between the tumor and surrounding normal tissue and the tumors were hard to detect. However, the enhanced T2WI imaging displayed significantly more MRI signal contrast in the tumor and surrounding normal tissue and the MRI scan could detect the presence of the tumor. This enhanced contrast occurred because Fe3O4 nanoparticles modified by folic acid could bind to folate receptors that were highly expressed in tumor cells. The folate-conjugated Fe3O4 nanoparticles were then transported into the tumor cells and labeled the tumor cells, thus the MRI T2 signals of the labeled tumor were significantly negatively enhanced. Accordingly, the imaging contrast between normal tissue and tumor tissue became apparent, and the labeled tumor was clearly visible.
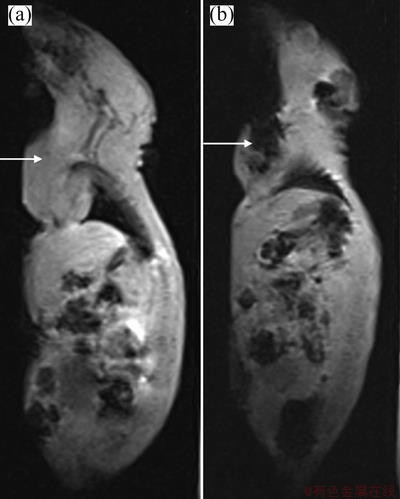
Fig. 7 Unenhanced T2WI image of leg of tumor-bearing rats (a) and enhanced T2WI image of leg of tumor-bearing rats at 40 min after injection of folic acid-modified Fe3O4 nano-magnetic fluid (b) (Arrows refer to the position of the tumor)
After MRI imaging and relevant examination, the samples from the human tumor subcutaneous xenograft model in tumor-bearing rats were subjected to histopathological detection, including conventional hematoxylin-eosin staining and Prussian blue staining. Conventional hematoxylin-eosin staining results showed that the established human tumor subcutaneous xenograft model coincided with hepatocellular carcinoma. Prussian blue staining results showed many blue-stained particles in hepatoma carcinoma cells (Fig. 8), and these particles were the folic acid-modified Fe3O4 nanoparticles. Prussian blue staining pathologically verified the presence of Fe (i.e., folic acid-modified Fe3O4 nanoparticles) in hepatoma carcinoma cells, indicating that the folate-conjugated Fe3O4 nanoparticles were available to enter the hepatoma carcinoma cells through the high affinity between folate receptor and folic acid, and these particles can label the cells in vivo. This is evidence that folate-conjugated Fe3O4 nanoparticles can be used for targeted labeling of human liver tumors in tumor bearing rat.
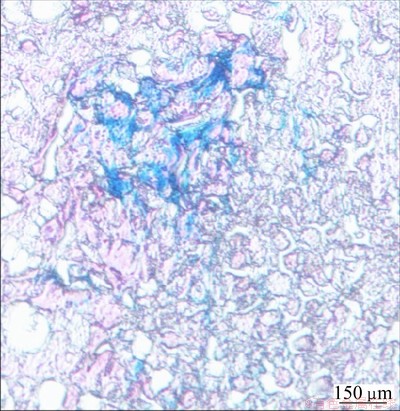
Fig. 8 Prussian blue staining of tumor tissue
4 Conclusions
The highly biocompatible folate-conjugated superparamagnetic Fe3O4 nanoparticles were synthesized through amide action using inexpensive and environmentally friendly iron salt and FA as raw materials. The prepared folate-conjugated super- paramagnetic Fe3O4 nanoparticles are cubic-phase Fe3O4 with a particle size of about 8 nm and hydrated diameter of 25.7 nm, and the shape is close to spherical. There are functional groups such as carboxyl on the surface and the nanoparticles are superparamagnetic, with a saturation magnetization of 51 A・m2/kg. And they could transfer into tumor cells and label tumor cells effectively. Moreover, they show good safety to human skin fibroblasts or no apparent acute side effects on Wistar rat and good labeling efficiency of hepatoma cells in vivo. This is convenient to use folate-conjugated super- paramagnetic Fe3O4 nanoparticles for the tumor cells labeling, tracing, MRI target detection and magnetic hyperthermia.
References
[1] WEISSLEDER R, BOGDANOV A, PAPISOV M. Drug targeting in magnetic resonance imaging [J]. Magn Reson Q, 1992, 8(1): 55-63.
[2] HUH Y M, JUN Y W, SONG H T, KIM S, CHOI J S, LEE J H, YOON S, KIM K S, SHIN J S, SUH J S, CHEON J. In vivo magnetic resonance detection of cancer by using multifunctional magnetic nanocrystals [J]. J Am Chem Soc, 2005, 127: 12387-12391.
[3] HULTMAN K L, RAFFO A J, GRZENDA A L, HARRIS P E, BROWN T R, O’BRIEN S. Magnetic resonance imaging of major histocompatibility class II: Expression in the renal medulla using immunotargeted superparamagnetic iron oxide nanoparticles [J]. Acs Nano, 2008, 2(3): 477-484.
[4] FENG B, HONG R Y, WANG L S, GUO L, LI H Z, DING J, ZHENG Y, WEI D G. Synthesis of Fe3O4/APTES/PEG diacid functionalized magnetic nanoparticles for MR imaging [J]. Colloids and Surfaces A: Physicochem Eng Aspects, 2008, 328: 52-59.
[5] CHEN W H, YI P W, ZHANG Y, ZHANG L M, DENG Z W, ZHANG Z J. Composites of inodextran-coated Fe3O4 nanoparticles and graphene oxide for cellular magnetic resonance imaging [J]. Appl Mater Interfaces, 2011, 3: 4085-4091.
[6] GARCIA I, GALLO J, GENICIO N, PADRO D, PECADES S. Magnetic glycol nanoparticles as a versatile platform for selective immunolabeling and imaging of cells [J]. Bioconjugate Chem, 2011, 22: 264-273.
[7] POSELT E, SCHMIDTKE C, FISCHER S, PELDSCHUS K, SALAMON J, KLOUST H, TRAN H, PIETSCH A, HEINE M, ADAM G, SCHUMACHER U, WAGENER C, FORSTER S, WELLER H. Tailor-made quantum dot and iron oxide based contrast agents for in vitro and in vivo tumor imaging [J]. Acs Nano, 2012, 6(4): 3346-3355.
[8] MCNEIL S E. Nanotechnology for the biologist [J]. J Leukoc Biol, 2005, 78: 585-594.
[9] ROSS J F, CHAUDHURI P K, RATNAM M. Differential regulation of folate receptor isoforms in normal and malignant tissues in vivo and in established cell lines [J]. Physiologic and Clinical Implications Cancer, 1994, 73: 2432-2443.
[10] PARK E K, LEE S B, LEE Y M. Preparation and characterization of methoxy poly (ethylene glycol)/poly (epsilon-caprolactone) amphiphilic block copolymeric nanospheres for tumor-specific folate-mediated targeting of anticancer drugs [J]. Biomaterials, 2005, 26: 1053-1061.
[11] SUDIMACK J, LEE R J. Targeted drug delivery via the folate receptor [J]. Adv Drug Deliv Rev, 2000, 41(2): 147-162.
[12] ZHANG Y, SUN C, KOHLER N, ZHANG M Q. Self-assembled coatings on individual monodisperse magnetite nanoparticles for efficient intracellular uptake [J]. Biomed Microdevices, 2004, 6(1): 33-40.
[13] SUN C, SZE R, ZHANG M Q. Folic acid-PEG conjugated superparamagnetic nanoparticles for targeted cellular uptake and detection by MRI [J]. J Biomed Mater Res Part A, 2006, 45: 550-557.
[14] ZHU Y F, FANG Y, KASKEL S. Folate-conjugated Fe3O4@SiO2 hollow mesoporous spheres for targeted anticancer drug delivery [J]. J Phys Chem C, 2010, 114: 16382-16388.
[15] WANG Dong-sheng, LI Jian-guo, LI He-ping, TANG Fa-qing. Preparation and drug releasing property of magnetic chitosan-5-fluorouracil nano-particles [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(5): 1232-1236.
[16] PENG Jian, ZOU Fen, LIU Lu, TANG Liang, YU Li, CHEN Wei, LIU Hui, TANG Jing-bo, WU Li-xiang. Preparation and characterization of PEG-PEI/Fe3O4 nano-magnetic fluid by co-precipitation method [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(2): 393-398.
[17] CAO L J, HU Y P, GU L, SHEN H X, LI Q H, LAN P, FANG Y. Preparation and characterization of composite microspheres of nano zinc ferrite/poly(D, L-lactide-co-alanine) [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(2): 360-365.
叶酸偶联的Fe3O4纳米颗粒对活体肿瘤细胞的标记
周春姣1,王少华2,周 宇1,容鹏飞2,陈孜孜2,刘进言2,周建大2
1. 湖南农业大学 理学院,长沙 410128;
2. 中南大学 湘雅三医院,长沙 410013
摘 要:采用酰胺化反应方法,通过Fe3O4纳米颗粒表面氨基基团和与肿瘤细胞具有高度亲和力的叶酸分子的羧基间反应进行偶联得到目标材料,利用X射线衍射、傅立叶红外光谱仪、透射电镜、振动样品磁强计、动态光散射仪等对所合成的材料进行表征,且以瘤鼠为动物模型,考察其活体肝癌细胞标记效果和MRI成像功能。结果表明,合成的材料为立方相的Fe3O4,粒径约8 nm,水合直径约25.7 nm,呈近似球形形貌,表面分布有羧基等功能基团,呈超顺磁特性,饱和磁化强度为51 A・m2/kg,能靶向进入肝癌细胞内,提高肿瘤与周围正常组织的MRI成像信号对比。实验证实,叶酸偶联的超顺磁Fe3O4纳米颗粒,不仅细胞毒性小,而且因其表面的叶酸与叶酸受体之间的高强结合力,能通过配体-受体结合作用被高效介导进入肿瘤细胞内,实现对癌细胞的标记、示踪、MRI靶向检测及磁热疗。
关键词:Fe3O4纳米颗粒;超顺磁性;叶酸;肿瘤;偶联;核磁共振成像;细胞标记
(Edited by Xiang-qun LI)
Foundation item: Project (2011JQ028) supported by the Fundamental Research Funds for the Central Universities, China, Projects (2008SK3114, 2010SK3113) supported by Hunan Provincial Science & Technology Plan, China; Project (B2007086) supported by Science & Research Funds of Hunan Health Department, China; Project (12JJ5057) supported by Natural Science Foundation of Hunan Province, China; Projects (XCX1119, XCX12073) supported by University Students Innovative Experiment Plan Project of Hunan Agricultural University, China
Corresponding author: Jian-da ZHOU; Tel: +86-731-8861815; Fax: +86-731-88618326; E-mail: zhoujianda@hotmail.com
DOI: 10.1016/S1003-6326(13)62699-0