Trans. Nonferrous Met. Soc. China 26(2016) 2479-2484
Enhancement of bio-oxidation of refractory arsenopyritic gold ore by adding pyrolusite in bioleaching system
Xu ZHANG1,2, Ya-li FENG1, Hao-ran LI2
1. School of Civil and Environmental Engineering, University of Science and Technology Beijing, Beijing 100083, China;
2. State Key Laboratory of Biochemical Engineering, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, China
Received 28 August 2015; accepted 14 June 2016
Abstract: Pyrolusite was added in the bioleaching process to enhance the bio-oxidation process. Bioleaching tests at different dosages of pyrolusite ore, pH and inoculation amounts of Acidithiobacillus ferrooxidans were studied. The results showed that the time of the bio-oxidation process was decreased obviously and the arsenic leaching rate reached 94.4% after the bioleaching. The bio-oxidation of arsenopyrite and the effective extraction of manganese from pyrolusite were achieved by the bioleaching process. After bioleaching, the leaching rate of gold from the reaction residues reached 95.8% by cyanide leaching. In the bio-oxidation process, pyrolusite increased the redox potential of the solution to accelerate the bioleaching rate. The experiment showed that there were two reaction modes in the bioleaching process.
Key words: arsenopyritic gold ore; pyrolusite; bio-oxidation; Acidithiobacillus ferrooxidans
1 Introduction
Many sulfide gold ores contain various amounts of arsenic, often in the form of arsenopyrite, FeAsS[1]. For refractory, arsenic-bearing sulfide gold ores, lots of the fine disseminated gold is encapsulated in the sulfide matrix of FeAsS and the gold cannot be recovered by conventional leaching techniques. In recent years, the advances of biomining technology have focused on achieving effective recovery of precious metals by improving the efficiency of bioleaching [2,3]. One of the major advances in bioleaching is the development of the stirred-tank process for bio-oxidation of refractory arsenopyritic gold ores [4-6]. Bio-oxidation makes use of iron and sulfur oxidizing bacteria to catalyze the oxidation of sulfides, liberating gold for subsequent cyanidation [7,8]. Acidithiobacillus ferrooxidan is one of the primary micro-organisms in the commercial bio-oxidation industries. The bacteria obtain energy by oxidizing ferrous iron and elemental sulfur that generate ferric ions and sulfuric acid [9]. Bio-oxidation of refractory gold is a mature technology and is used to treat a refractory arsenopyritic gold resource in several countries.
However, bio-oxidation process needs a long time for pretreatment (often 6 d are required to dissolve arsenopyrite) and stringent process control is required to maintain bacterial activity [6,10,11]. So, the technology of enhancing the bio-oxidation of a refractory arsenopyritic gold ore is necessary to be developed. Pyrolusite is an important manganese mineral in manganese ores, which is distributed widely in the world and can be used as an oxidizing agent for the oxidation of sulphide ore in sulfuric acid solution [12]. In this work, pyrolusite was added in the bioleaching process to enhance the bio-oxidation of a refractory arsenopyritic gold ore. The bio-oxidation of arsenopyrite and the high extraction of manganese from pyrolusite were achieved during the bioleaching process. The impacts of pyrolusite ore dosage, pH and inoculation amount on the bio-oxidtion were studied. The kinetics of the bioleaching system was well described. The bio-oxidation of arsenopyrite is based on arsenic dissolved into the solution and the extraction of manganese from pyrolusite is based on manganese leaching efficiency.
2 Experimental
The refractory arsenopyritic gold ore was obtained from Zhuzhou Smelting Plant in Hunan Province, China. The ore was ground to the required powder with the particle size (d90) of 0.045 mm. Table 1 shows the element composition of the arsenopyritic gold ore used for the experiments. The ore samples by XRD (Fig. 1) were mainly composed of quartz, arsenopyrite and pyrite. The pyrolusite samples were obtained from a manganese ore mine in Yunnan Province, China. The ore was crushed and ground into powder with the particle size (d90) of 0.074 mm. The chemical multi-elemental analysis results are shown in Table 2. The pyrolusite ore samples were mainly pyrolusite, and the main gangue minerals were quartz and feldspar.
Table 1 Chemical multi-elemental analysis of arsenopyritic gold ore (mass fraction, %)
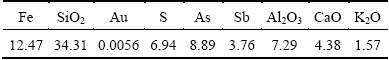

Fig. 1 XRD pattern of arsenopyritic gold ore
Table 2 Chemical multi-elemental analysis of pyrolusite ore (mass fraction, %)

Acidithiobacillus ferrooxidans were isolated from the acid mine water of a copper mine in Gansu Province, China, and was obtained through isolation, purification and various stages of adaptation to sulfide minerals. The culture medium was 9K medium. The composition of 9K medium was as follows: 3.0 g/L (NH4)2SO4, 0.1 g/L KC1, 0.5 g/L K2HPO4, 0.5 g/L MgSO4・7H2O, 0.01 g/L Ca(NO3)2, 44.1 g/L FeSO4・7H2O, and pH 1.8 at first by H2SO4. All systems were incubated for at least 48 h.
Bioleaching experiments were carried out in 350 mL Erlenmeyer flasks with 150 mL solution. The flasks were placed on constant temperature and humidity table with 30 °C and 160 r/min. The pyrolusite ore dosage, pH and inoculation amount of Acidithiobacillus ferrooxidans were studied in the experiments and 5 g of the refractory arsenopyritic gold ore was added in every experiment. In the experiments, H2SO4 was uninterruptedly supplied to control the solution pH. The reaction residues were analyzed for the determination of manganese and arsenic by atomic fluorescence spectrometry (AFS) and inductively coupled plasma atomic emission spectrometry (ICP-AES). The reaction residues were decomposed by mixed acid (H2SO4 and H3PO4). Prior to atomic fluorescence spectrometry (AFC), the details of the technique were performed with the determination of manganese in ferrophosphorus as previous described in Refs. [12,13]. Some reaction residues were examined to extract gold from the reaction residues by cyanide leaching. The cyanide leaching was performed using 20 g of reaction residues in 200 mL of 0.1% NaCN solution at pH 10.5 for 24 h.
3 Results and discussion
3.1 Effect of pyrolusite ore dosage on bioleaching
The effect of pyrolusite ore dosage on the bioleaching was investigated at the pyrolusite ore dosage of 0, 8, 10, 12, 14 and 16 g, with inoculation amount of 20 mL (1×107 cell/mL) and pH 1.8 (maintained constant in bioleaching). The results are shown in Fig. 2. As shown in Fig. 2(a), increasing pyrolusite ore dosage increased the leaching rate of arsenic. The bio-oxidation of the refractory arsenopyritic gold ore was analyzed by the leaching rate of arsenic.
It was shown that bio-oxidation of a refractory arsenopyritic gold ore was enhanced with the increase of pyrolusite ore dosage added to the bioleaching system. It was also seen that when the pyrolusite ore dosage was less than 12 g, increasing pyrolusite ore dosage had a significant improvement effect on the bio-oxidation of the refractory arsenopyritic gold ore. But when the pyrolusite ore dosage was more than 12 g, there was a slight improvement on the bio-oxidation. It was shown in Fig. 2(b) that manganese was extracted from pyrolusite with the bio-oxidation of the refractory arsenopyritic gold ore. And it was seen in Fig. 2 that when the pyrolusite ore dosage was 12 g, the leaching rate of arsenic attained 91.85% and the leaching rate of manganese reached 92.26% after 56 h. This suggested that it was suitable for the bioleaching system at the pyrolusite ore dosage of 12 g.
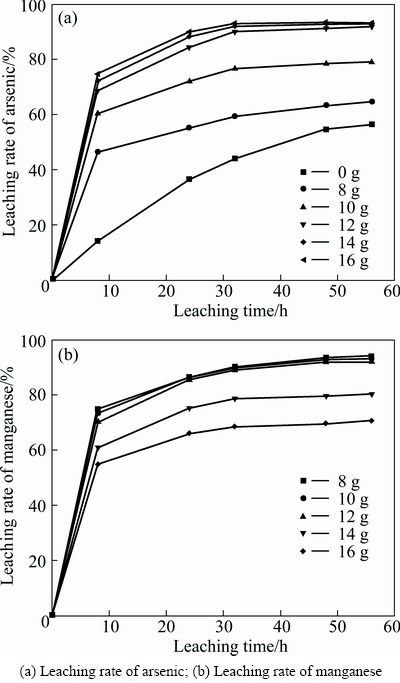
Fig. 2 Effect of pyrolusite ore dosage on bioleaching
3.2 Effect of pH on bioleaching
The bioleaching system was obviously affected by pH. The following reaction equations account for the above experimental kinetic data [14]:
FeAsS+7MnO2+6H2SO4=7MnSO4+FeAsO4+6H2O (1)
FeAsS+4MnO2+4H2SO4=4MnSO4+FeAsO4+S+4H2O (2)
As shown in Eqs. (1) and (2), the acid was consumed during the bioleaching. And the bacteria are able to be reproduced in a range of pH. In the experiments, H2SO4 was uninterruptedly supplied to control the solution pH. The effect of pH on the bioleaching was investigated at pH 1.4, 1.6, 1.8, 2.0 and 2.2(maintained constant in bioleaching), respectively, the pyrolusite ore dosage of 12 g and inoculation amount of 20 mL (1×107 cell/mL). As shown in Fig. 3(a), the leaching rate of arsenic presented very similar trend during bioleaching at pH 1.4, 1.6 and 1.8 and the arsenic extraction significantly decreased at pH 2.0 and 2.2. During the bioleaching, the leaching rate of arsenic increased obviously, and subsequently began to slow down, and then kept stable. At pH 1.4, 1.6, 1.8, 2.0 and 2.2, the leaching rates of arsenic after leaching for 56 h were 95.03%, 94.40%, 91.85%, 86.47% and 74.86%, separately. As shown in Fig. 3(b), at pH 1.4, 1.6, 1.8, 2.0 and 2.2, the leaching rates of manganese after 56 h were 95.32%, 94.60%, 92.26%, 85.93% and 70.60%. From Fig. 3, it showed that the extractions of arsenic and manganese were well achieved by the bioleaching process at pH 1.6.
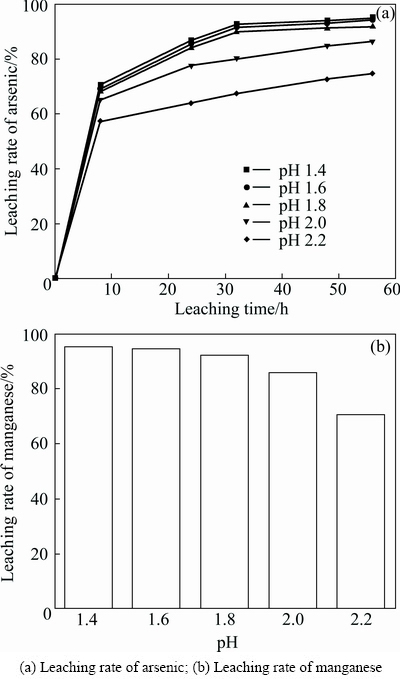
Fig. 3 Effect of pH on bioleaching
3.3 Effect of inoculation amount on bioleaching
The experiments were carried out at four different Acidithiobacillus ferrooxidans inoculation amounts of 0, 10, 20 and 30 mL (1×107 cell/mL), pH 1.6 and the pyrolusite ore dosage of 12 g. It was obvious that the bacteria attached themselves onto the surface of the sulfide ores and directly solubilized the surface through hypothesized enzymatic oxidation reactions [15,16]. Acidithiobacillus ferrooxidans catalyzed the oxidation of arsenopyrite and the arsenic was dissolved into the solution. As shown in Fig. 4, increasing inoculation amount accelerated the leaching rate of arsenic when the inoculation amount was less than 20 mL. When the inoculation amount was more than 20 mL, enough bacteria attached themselves onto the surface of the sulfide ore and the inoculation amount slightly improved the leaching rate of arsenic. The leaching rates of manganese after leaching for 56 h were 72.16%, 86.40%, 94.60% and 94.82%, respectively, when the inoculation amount was 0, 10, 20 and 30 mL (1×107 cell/mL). The reaction residues after oxidation were washed with 0.1 mol/L HCl, distilled water and dried, and then were examined by cyanide leaching and the leaching rate of gold reached 95.8%. The leaching rate of arsenic was only 54.92% after the bioleaching without pyrolusite and the leaching rate of gold was 67.2%, much less than 95.8%.
Fig. 4 Effect of inoculation amount on bioleaching
3.4 Chemical reaction and kinetics in bioleaching
The bioleaching was based on the redox reaction and aqueous electrochemical system. The kinetics of the gold leaching process was related to the potential (φ) and pH value in aqueous solutions (φ-pH diagram) [17]. φ-pH diagram could describe the chemical reactions in the As-Fe-Mn-S-H2O system (Fig. 5). The following equilibrium equations were used for describing the φ-pH diagram of the As-Fe-Mn-S-H2O system at 30 °C [18,19].
FeAsS+6H2O-11e=Fe2++ +
+ +12H+
+12H+
φ=0.282-0.064pH+0.00536lg[Fe2+]+0.00536lg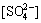 +0.00536lg
+0.00536lg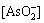 (3)
(3)
 +2H2O-2e=
+2H2O-2e= +2H+
+2H+
φ=0.665-0.059pH+0.0295lg -0.0295lg
-0.0295lg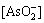 (4)
(4)
Fe2+-e=Fe3+φ=0.771+0.059[Fe3+]-0.059lg[Fe2+] (5)
Fe3++H2O=Fe(OH)3+3H+3pH=4.82-lg[Fe3+] (6)
Fe2++3H2O-e=Fe(OH)3+3H+φ=1.050-0.177pH+0.059lg[Fe2+] (7)
MnO2+4H++2e=Mn2++2H2Oφ=0.974-0.118pH (8)
The main form of pyrolusite is MnO2 and pyrolusite is used as an oxidizing agent during the bioleaching. When the pyrolusite was added in the bioleaching solution, the φ of the solution varied from 0.90 to 0.85 V at pH 1.2-2.0; whereas the φ of the arsenopyrite dissolved varied from 0.21 to 0.16 V at pH 1.2-2.0. The analysis of the diagram demonstrated that arsenopyrite was composed of dissolved compound with the highest valences of the elements, such as 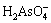 , Fe3+ and
, Fe3+ and 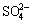 , during the bioleaching. The φ of the solution was increased to above 0.85 V with adding pyrolusite in the bioleaching system, whereas the φ of the solution was only 0.67 V (or less than 0.67 V) without adding pyrolusite. Increasing the solution φ, the oxidation of arsenopyrite was accelerated during the bioleaching process.
, during the bioleaching. The φ of the solution was increased to above 0.85 V with adding pyrolusite in the bioleaching system, whereas the φ of the solution was only 0.67 V (or less than 0.67 V) without adding pyrolusite. Increasing the solution φ, the oxidation of arsenopyrite was accelerated during the bioleaching process.

Fig. 5 φ-pH diagram of As-Fe-Mn-S-H2O system at 30 °C
The application of pyrolusite in bioleaching system to enhance the bio-oxidation of a refractory arsenopyritic gold ore was a complex process. From the experimental data, it was concluded that there were two reaction modes in the bioleaching process, which are shown in Fig. 6. In Mode I, arsenopyrite and pyrolusite particles contacted with each other to have reactions in the solution, and in Mode II, Acidithiobacillus ferrooxidans were adsorbed on the surface of arsenopyrite to release Fe3+ by improving the bio-oxidation.
4 Conclusions
1) Bioleaching tests at different dosages of pyrolusite ore, pH and inoculation amount of Acidithiobacillus ferrooxidans were performed. Pyrolusite ore was used as an oxidation agent and the fine gold was released from the sulfide matrix with the bio-oxidation. The time of the bio-oxidation process decreased obviously after pyrolusite was added into the bioleaching system.
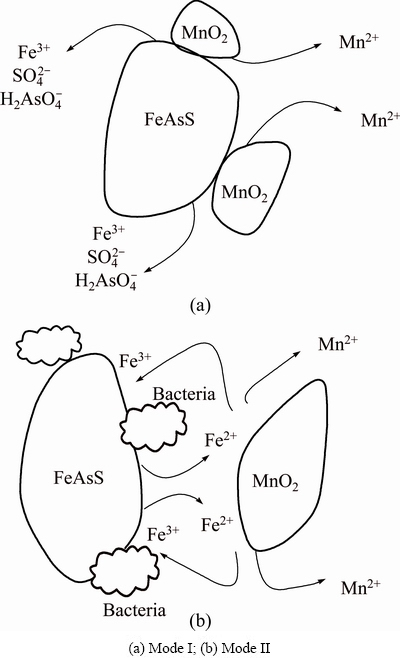
Fig. 6 Reaction modes in bioleaching
2) In the bio-oxidation process, pyrolusite increased the redox potential of the solution to accelerate the bioleaching rate. The leaching rate of arsenic reached 94.4% and the leaching rate of manganese reached 94.6% after bioleaching at pH 1.6, inoculation amount of 20 mL and the pyrolusite ores dosage of 12 g. The leaching rate of gold from the reaction residues reached 95.8% by cyanide leaching.
3) The application of pyrolusite in bioleaching system to enhance the bio-oxidation of a refractory arsenopyritic gold ore was a complex process. In the bio-oxidation process, pyrolusite increased the redox potential of the solution to accelerate the bioleaching rate. There were two reaction modes in the bioleaching process.
References
[1] JIANG Tao, LI Qian, YANG Yong-bin, LI Guang-hui, QIU Guan-zhou. Bio-oxidation of arsenopyrite [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1433-1438.
[2] DAS A P, SUKLA L B, PRADHAN N, NAYAK S. Manganese biomining: A review [J]. Bioresource Technology, 2011, 102(16): 7381-7387.
[3] GONZALEZ R, GENTINA J C, ACEVEDO F. Continuous biooxidation of a refractory gold concentrate [J]. Process Metallurgy, 1999, 9(9): 309-317.
[4] ARSHADI M, MOUSAVI S M. Enhancement of simultaneous gold and copper extraction from computer printed circuit boards using Bacillus megaterium [J]. Bioresource Technology, 2014, 175: 315-324.
[5] CLIMO M, WATLING H R, BRONSWIJK W V. Biooxidation as pre-treatment for a telluride-rich refractory gold concentrate [J]. Minerals Engineering, 2000, 13(12): 1219-1229.
[6] GONZALEZ R, GENTINA J C, ACEVEDO F. Biooxidation of a gold concentrate in a continuous stirred tank reactor: Mathematical model and optimal configuration [J]. Biochemical Engineering Journal, 2004, 19(1): 33-42.
[7] AHMANN D, KRUMHOLZ L R, HEMOND H F, LOVLEY D R, MOREL F M M. Microbial mobilization of arsenic from sediments of the aberjona watershed [J]. Environmental Science & Technology, 1997, 31(10): 2923-2930.
[8] CASSITY W D, PESIC B. Interactions of Thiobacillus ferrooxidans with arsenite, arsenate and arsenopyrite [J]. Process Metallurgy, 1999, 9(9): 521-532.
[9] SUN Li-xin, ZHAND Xu, TAN Wen-song, ZHU Ming-hong. Effects of dissolved oxygen on the biooxidation process of refractory gold ores [J]. Journal of Bioscience & Bioengineering, 2012, 114(5): 531-536.
[10] GAO Guo-long, LI Deng-xin, ZHOU Yong, SUN Xian-hao, SUN Wen. Kinetics of high-sulphur and high-arsenic refractory gold concentrate oxidation by dilute nitric acid under mild conditions [J]. Minerals Engineering, 2009, 22(10): 111-115.
[11] LIU Xin-xing, WANG Guo-hua, HUO Qiang, XIE Jian-ping, LI Shou-peng, WU Hai-yan, GUO Yu-jie. Novel two-step process to improve efficiency of bio-oxidation of Axi high-sulfur refractory gold concentrates [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(12): 4119-4125.
[12] CAI Zhen-lei, FENG Ya-li, LI Hao-ran, DU Zhu-wei, LIU Xin-wei. Co-recovery of manganese from low-grade pyrolusite and vanadium from stone coal using fluidized roasting coupling technology [J]. Hydrometallurgy, 2013, 131(1): 40-45.
[13] WANG Zhen-kun, CHEN Yan, LI Yi, WANG Jian. Determination of manganese in ferrophosphorus by periodate spectrophotometry [J]. Metallurgical Analysis, 2010, 30(12): 53-55.
[14] CORKHILL C L, VAUGHAN D J. Arsenopyrite oxidation―A review [J]. Applied Geochemistry, 2009, 24(12): 2342-2361.
[15] BOON M. The mechanism of ‘direct’ and ‘indirect’ bacterial oxidation of sulphide minerals [J]. Hydrometallurgy, 2001, 62: 67-70.
[16] LI Qing-cui, LI Deng-xin, CHEN Quan-yuan. Prediction of pre-oxidation efficiency of refractory gold concentrate by ozone in ferric sulfate solution using artificial neural networks [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(2): 413-422.
[17] KONYRATBEKOVA S S, BAIKONUROVA A, USSOLTSEVA G A, ERUST C, AKCIL A. Thermodynamic and kinetic of iodine- iodide leaching in gold hydrometallurgy [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(11): 3774-3783.
[18] LI Hong-xu, LI Chao, ZHANG Zhi-qian. Decomposition mechanism of pentlandite during electrochemical bio-oxidation process [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(3): 731-739.
[19] RAWLINGS D E. High level arsenic resistance in bacteria present in biooxidation tanks used to treat gold-bearing arsenopyrite concentrates―A review [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1311-1318.
添加软锰矿强化复杂难选冶含砷金矿的生物氧化过程
张 旭1,2,冯雅丽1,李浩然2
1. 北京科技大学 土木与环境工程学院,北京 100083;
2. 中国科学院 过程工程研究所 生化工程国家重点实验室,北京 100190
摘 要:通过控制生物浸出试验中软锰矿用量、过程pH和细菌接菌量等条件,对添加软锰矿强化复杂难选冶含砷金矿的生物氧化过程进行研究。结果表明,添加软锰矿可以缩短含砷金矿的生物氧化时间,砷的浸出率达到94.4%。反应过程中实现了含砷金矿中砷黄铁矿氧化的同时,软锰矿中锰元素高效浸出。生物浸出渣的氰化浸出实验结果表明,经软锰矿强化生物氧化后,生物浸出渣中金的氰化浸出率达到95.8%。生物浸出过程中,添加软锰矿能提高生物浸出溶液的氧化还原电位,从而促进生物氧化过程,且添加软锰矿后生物浸出过程中存在两种不同的反应方式。
关键词:含砷金矿;软锰矿;生物氧化;氧化亚铁硫杆菌
(Edited by Wei-ping CHEN)
Foundation item: Project (2015ZX07205-003) supported by the National Water Pollution Control and Treatment Science, China; Project (DY125-15-T-08) supported by China Ocean Mineral Resource R & D Association; Project (2012BAB07B05) supported by the National Key Technology R & D Program of China; Project (2012AA062401) supported by the National High-tech Research and Development Program of China
Corresponding author: Ya-li FENG; Tel: +86-10-62332467; E-mail: ylfeng126@126.com
DOI: 10.1016/S1003-6326(16)64339-X