Microstructure and properties of AZ31 magnesium alloy with rapid solidification
ZHAO Hong-liang(�Ժ���), GUAN Shao-kang(���ܿ�),ZHENG Fei-yan(֣����), LI Qing-kui(�����), WANG Li-guo(������)
(College of Materials Engineering, Zhengzhou University, Zhengzhou 450002, China)
Abstract: Rapidly solidified (RS) AZ31 magnesium alloy ribbons were made using melt spinning technique. The results show that its microhardness increases with the wheel speed, and after heat treatment, the microhardness of the ribbons produced at 1600r/min also increases. Rapid solidification leads to reduction of grain size. When the wheel speed reaches 1600r/min, no Mg17Al12 phase precipitates, while heat treatment at 200�� leads to precipitation of Mg17Al12 phase. Al-Mn intermetallic compounds with size no larger than 10nm appear in as-spun ribbons. The corrosion potential of the as-cast ingots is lower than that of the as-spun ribbons.
Key words: rapid solidification; magnesium alloy; microstructure; properties CLC number: TG0146.22
Document code: A
1 INTRODUCTION
In recent years, magnesium alloys have been used in automotive, aerospace and electronic industries where mass reduction is an important requirement. Magnesium alloys show light mass, high stiffness, high specific strength, good dimensional stability and damping capacity[1-5]. Magnesium is the lightest space structural metal, but its low mechanical properties and poor corrosion resistance limit its applications[6-8]. Refinement of microstructure through rapid solidification processing can result in stronger, easily workable and more corrosion resistant magnesium alloys[9, 10]. However, rapid solidification processing of magnesium alloys poses critical challenges due to the very high chemical reactivity of magnesium metal itself and a number of its important alloying elements. The available literature on rapidly solidified magnesium alloys is very sparse[11-13] and no significant work has been undertaken so far. In the present study, the technology of processing of rapidly solidified (RS) ribbons in an alloy based on Mg-Al-Zn system has been established. The ribbons are characterized through metallographic and X-ray diffraction techniques. The effect of the wheel speed on thickness and microhardness is also studied. The corrosion resistance of the ribbons is measured.
2 EXPERIMENTAL
Alloy of Mg-3%Al-1%Zn-0.2%Mn was prepared in steel crucible in an electric resistance furnace. Magnesium, aluminium and zinc metal ingots of 99.9% purity were used. Manganese was added as Al-10%Mn hardener. The melt was poured into 30mm-diameter steel moulds and ingots were cast under the protective cover of SF6 gas. These ingots were proof machined. RS ribbon making experiments were carried out using melt spinning technique. The 25mm-diamater specimens, cut from test bars, were charged into the cylindrical quartz glass tube of the melt spinning unit and the chamber with the tube was evacuated to a vacuum level of 133Pa. Subsequently, argon was purged and vacuum level was brought to 13300Pa. The alloy was melted through induction heating, in the tube under argon atmosphere. The molten alloy was then pressurized to 0.7MPa with argon gas to force the metal through 0.3mm��10mm orifice at the bottom of the tube. The speed of the copper wheel (350mm-diameter), was varied between 500 (9.16m/s) to 2200r/min (40.30m/s) and RS ribbons were produced. The ribbons produced at a wheel speed of 1600r/min were heated to 200�� for 2h. The ingots and ribbons were mounted, polished and etched with 3% nital for metallographic analysis. The ribbons in as-spun and heat treatment conditions were twin-jet electropolished for TEM analysis. Microhardness of ribbons, measured using Vickers pyramid indentor at a load of 0.98N. X-ray diffraction study was carried out using Co K�� radiation. Corrosion resistance of the ribbons and the ingots were compared through measuring their corrosion potential.
3 RESULTS AND DISCUSSION
3.1 Effect of wheel speed on thickness and microhardness
Chemical compositions of the magnesium alloy are listed in Table 1. Cooling rate during liquid to solid transformation increases with the increase of wheel speed. In general, increased cooling rate leads to refinement of microstructure, enhancement of solubility of solutes, etc, and thus improves the properties[10]. The effect of wheel speed on thickness of ribbons is given in Table 2, which shows that as the wheel speed is increased, the ribbon thickness is inherently decreased. It is also clear from Table 2 that an increase in wheel speed
Table 1 Chemical composition of magnesium alloy(mass fraction, %)

Table 2 Effect of wheel speed on ribbon thickness and microhardness

results in an increase of microhardness.
3.2 Effect of wheel speed and heat treatment on microstructures and phase transformations
X-ray diffraction results(Fig.1(a), Fig.1(b)) reveal the presence of Mg17Al12 phase under the as-cast and the rapidly solidified at the wheel speed of 800r/min, but no Mg17Al12 phase precipitated when the wheel speed reached 1600r/min(Fig.1

Fig.1 XRD patterns of ingots and ribbons
(c)). When the ribbons produced at 1600r/min were heated at 200�� for 2h, the Mg17Al12 phase precipitated again(Fig.1(d)).
The quantity of Mg17Al12 decreases with the increase of wheel speed. Because the solidification of the RS ribbons is very fast, Mg17Al12 phase has no time to precipitate. So, the faster the cooling rate, the less the Mg17Al12 phase. When the wheel speed reaches 1600r/min, no Mg17Al12 phase can be found. Heat treatment at 200�� for 2h leads to precipitation of Mg17Al12 phase and takes away the solute from the matrix.
Microstructures(Fig.2(b)) of as-spun ribbons, processed at wheel speed of 800r/min, shows fine grain size (6-15��m) compared to 100-150��m obtainable through as-cast of this alloy(Fig.2(a)). TEM morphologies of ribbons processed at speed of 1600r/min shows the grain size is 1-2��m (Fig.3(a)) and is finer than that of the 800r/min. All these indicate that the increase of the microhardness is a direct manifestation, resulting from reduction of grain size.

Fig.2 Microstructures of AZ31 magnesium alloy

Fig.3 TEM morphologies of ribbons processed at speed of 1600r/min
From the Mg-Al phase diagram, the AZ31 magnesium alloy consists of ��-Mg (hexagonal crystal structure, space group P63/mmc, a=0.32094nm and c=0.52105nm) and ��-Mg17Al12 (cubic crystal structure, a=1.056nm) phases. Fig.2 (a) shows typical microstructure of AZ31 magnesium alloy in cast state. Many Mg17Al12 eutectic phase precipitates along grain boundaries in cast state, visible as ��necklace�� type structure. The length of the ��-Mg17Al12 ranges from tens of micrometers to one hundred micrometers and the width is about 10��m. Those large Mg17-Al12 phase with ��necklace�� type structure was harmful to the workability of the AZ31 magnesium alloy[14]. The quantity of Mg17Al12 clearly decreases and is invisible in RS ribbons (Fig.2(b)). Moreover, small-sized Al-Mn intermetallic compounds were observed in as-spun ribbons(Fig.3(b)). These Al-Mn compounds distribute homogenously in the ��-Mg matrix and the size is no larger than 10nm. Al-Mn intermetallic compound is a brittle phase and it is harmful to the properties of materials in cast state. But in as-spun conditions, the homogenous distribution and small size make it become a strengthening phase.
Fig.4 shows Mg17Al12 phase precipitated after heated at 200�� for 2h, which is consistent with the XRD results in Fig.1. The precipitates are hard and contribute to the high hardness values[15]. On the other hand, the size of ��-Mg17Al12 precipitated becomes smaller clearly after heat treatment. The length of ��-Mg17Al12 is between two and four hundred nanometers and the width is about twenty five nanometers. The tiny Mg17Al12 precipitated is very beneficial for the improvement of the ribbons�� properties. So after heat treatment, the microhardness of the ribbons produced at the wheel speed of 1600r/min increases (Table 2).

Fig.4 TEM morphologies of ribbons heat treatment at 200��
3.3 Corrosion resistance of ribbons and ingots
The corrosion potential of the as-spun ribbons, processed at wheel speed of 1600r/min, is about -1.550V (Fig.5(a)), but that of the as-cast ingots is about -1.60V (Fig.5(b)). The minor the corrosion potential, the more easily the material can be eroded. So the RS magnesium alloy ribbons have a better corrosion resistance than the as-cast ingots. Increased cooling rate of the RS leads to refinement of microstructure, enhancement of solubility of solutes, etc, and thus increase the corrosion potential.
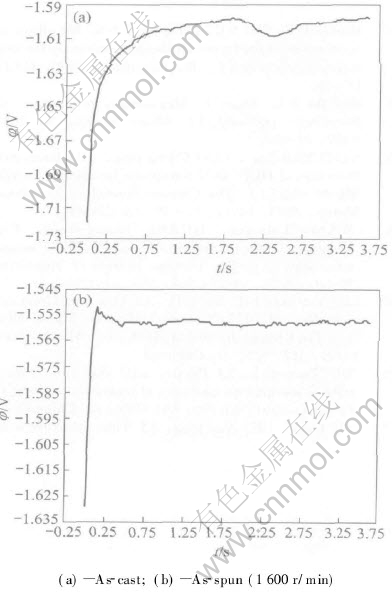
Fig.5 Corrosion potential of AZ31 magnesium alloy at different times
4 CONCLUSIONS
1) The ribbon thickness is decreased and the microhardness is increased as the wheel speed increases. After heat treatment, the microhardness of the ribbons produced at the wheel speed of 1600r/min also increases.
2) Rapidly solidification leads to small grains. As-spun ribbon processed at wheel speed of 800r/min, shows fine grain size (6-15��m). Ribbons processed at speed of 1600r/min shows the grain size finer (1-2��m).
3) When the wheel speed reaches 1600r/min, no Mg17Al12 phase precipitates, while heat treatment at 200�� leads to precipitation of the Mg17-Al12 phase. No larger than 10nm Al-Mn intermetallic compounds appeared in as-spun ribbons.
4) The corrosion potential of the as-cast ingots is lower than that of the as-spun ribbons.
ACKNOWLEDGEMENTS
The authors would like to thank Dr. Rui Zhang for helpful discussion.
REFERENCES
[1]Biswas P K, Dev S C, Krishnan C S S. Mg alloys and composites as the future challenging materials for automobile applications[J]. Ind Foundry J, 1999, 45(3): 17-26.
[2]Mordike B L, Ebert T. Magnesium, properties - applications - potential[J]. Mater Sci Eng A, 2001, A302: 37-45.
[3]XIAO Xiao-ling , LUO Cheng-ping, LIU Jiang-wen. Structure of HCP/ BCC interphase boundaries in AZ91 Mg-Al alloy[J]. The Chinese Journal of Nonferrous Metals, 2003, 13(1): 15-20. (in Chinese)
[4]WANG Ling-yun, HUANG Guang-sheng, FAN Yong-ge. Grain refinement of wrought AZ31 magnesium alloy[J]. The Chinese Journal of Nonferrous Metals, 2003, 13(3): 594-598. (in Chinese)
[5]LIU Yan-hui, LIU Xiang-fa , LI Ting-bin. Grain refining effect of Al2Ti2C master alloy on Mg-Al alloys[J]. The Chinese Journal of Nonferrous Metals, 2003, 13(3): 622-625. (in Chinese)
[6]WU Zhen-ning, LI Pei-jie, LIU Shu-xun. Present state of research on corrosion of magnesium alloys[J]. Foundry, 2001, 50(10): 583-586. (in Chinese)
[7]YU Gang, LIU Yue-long, LI Ying. Corrosion and protection of magnesium alloys[J]. The Chinese Journal of Nonferrous Metals, 2002, 12(6): 1087-1098. (in Chinese)
[8]ZHOU Hai-tao, ZENG Xiao-qin, LIU Wen-fa. Effect of Ce on microstructures and mechanical properties of AZ61 wrought magnesium[J]. The Chinese Journal of Nonferrous Metals, 2004, 14(1): 99-104. (in Chinese)
[9]SHI Fei, GUO Xue-feng, ZHANG Zhong-ming. Quasicrystal of as-cast Mg-Zn-Y alloy[J]. The Chinese Journal of Nonferrous Metals, 2004, 14(1): 112-116. (in Chinese)
[10]Govind, Nair K S, Mittal M C, et al. Development of rapidly solidified (RS) magnesium-aluminium-zinc alloy[J], Mater Sci Eng A, 2001, A304-306: 520-523.
[11]Das S K, Chang C F. Rapidly Solidified High Strength Corrosion Resistant Magnesium Base Metal Alloys[P]. US 4853035, 1989.
[12]Regazzoni G. High Strength Magnesium Alloys and Process for Obtaining These Alloys by Rapid Solidification[P]. US 4997622, 1991.
[13]CHEN Xu-hong, DING Pei-dao, YANG Chun-mei. Experimental study on twin-roll strip casting process for AZ31 magnesium alloy[J]. Light Alloy Fabrication Technology, 2003, 31(5): 19-22. (in Chinese)
[14]YU Kun, LI Wen-xian. Microstructures and properties of Mg-Al-Zn wrought alloys in as-rolling and annealing states[J]. Heat Treatment of Metals, 2002, 27(5): 8-11. (in Chinese)
[15]Luo A, Perquleryuz M O. Review-cast Mg alloys for elevated temperature applications[J]. J Mater Sci, 1994, 29: 5259-71.
(Edited by LONG Huai-zhong)
Foundation item: Project(0411051500) supported by the Natural Science Foundation of Henan Province; Project(204085) supported by the Key Project of Eduction Ministry of China
Received date: 2004-09-23; Accepted date: 2004-11-15
Correspondence: GUAN Shao-kang, PhD, Professor; Tel: +86-13523080674; E-mail: zhlwkr@zzu.edu.cn