J. Cent. South Univ. Technol. (2009) 16: 0594-0598
DOI: 10.1007/s11771-009-0099-y
Inhibitory effect of Cr(Ⅵ) on activities of soil enzymes
PENG Bing(彭 兵), HUANG Shun-hong(黄顺红), YANG Zhi-hui(杨志辉),
CHAI Li-yuan(柴立元), XU You-ze(许友泽), SU Chang-qing(苏长青)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: To evaluate the influence of various Cr(Ⅵ) concentrations (0.05, 0.25, 0.50, 1.00 and 2.00 g/kg) on the activity of soil enzymes, the activities of catalase, polyphenol oxidase, dehydrogenase, alkaline phosphatase in soils were investigated in the incubation experiment with a period of 35 d. The results indicate that all the tested Cr(Ⅵ) concentrations significantly inhibit dehydrogenase activity by over 70% after 35 d. The activity of alkaline phosphatase is slightly inhibited during the whole experiment except for on the day 7. Cr(Ⅵ) has no obvious effect on the activity of catalase in soil. On the contrary, Cr(Ⅵ) stimulates the activity of polyphenol oxidase. The results suggest that dehydrogenase activity can be used as an indicator for assessing the severity of chromium pollution.
Key words: Cr(Ⅵ); soil; pollution; enzyme activity; inhibition
1 Introduction
Industrial waste residues are deposited on soils and the heavy metals in residues are easily released into soil environment by leaching. Heavy metal ions are recognized to be highly toxic because of not being biodegradable. Such metals can accumulate in the environment, which can cause acute and chronic toxicity in soil ecosystems and exhibit the impact on human health even at very low concentration through their action on enzymes of the metabolic chain [1-2]. Chromate manufacturing, ore refining, production of steel and alloys, pigment manufacturing, corrosion inhibition, leather tanning, wood preservation, and combustion of coal and oil are mostly responsible for chromium (Cr) discharge into the environment. Cr- containing slag is one of the most hazardous solid wastes because of its high content of dissolvable Cr(Ⅵ), which is known to be toxic to plants and animals because of its strong oxidizability and potential carcinogenicity [3-4]. Therefore, monitoring of this metal is important due to the potential health and ecological hazard. Soil enzymes play an important role in soil metabolism, monitoring and activating biochemical processes of decomposition and the synthesis of mineral and organic matter occurring all the time, which is crucial for the nutrient cycles and transformation. Heavy metals above a certain level in soils may affect the biochemical processes by influencing enzyme activities [5]. It has been reported that heavy metals inhibit enzyme activities by denaturing proteins, masking catalytically active groups, or competing with the metal ions involved in the formation of complexes between enzyme and substrate [6-7]. Furthermore, the interaction of a single molecule of inhibitor with an enzyme can result in a noticeable reduction of the enzyme activity [8]. The enzyme activity in soils is proposed to be an early and sensitive indicator to measure the degree of soil degradation in both natural and agro-ecosystems, and thus it is suitable for assessing the effect of contaminants on soil quality [9-10]. The aim of this study is to investigate the effect of Cr(Ⅵ) on the activities of soil enzymes including catalase, dehydrogenase, polyphenol oxidase and alkaline phosphatase catalyze.
2 Experimental
2.1 Soil sampling and preparation
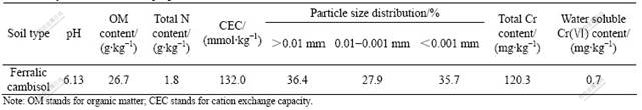
Soil was taken from the surface layer (0-20 cm) of the agricultural field in Xiangxiang County, Hunan Province, China. The soil was classified as ferralic cambisol according to FAO/UNESCO soil classification system. Its main physical and chemical properties are presented in Table 1. The sampled soil was air-dried and then sieved through a 2-mm mesh prior to use.
2.2 Incubation experiment
250 g of the sieved soil (dry mass) was transferred into a 500 mL plastic beaker. The soil moisture was
Table 1 Physical and chemical properties of soil
adjusted to 60% of the soil water holding capacity (WHC) by adding deionized water and then pre-incubated at 25 ℃ for 3 d (conditioning period). After conditioning, the soil were spiked with 0 (control), 0.05, 0.25, 0.50, 1.00 and 2.00 g/kg Cr(Ⅵ) to yield the polluted soils. K2Cr2O7 was used as Cr(Ⅵ) source. The polluted soil was incubated at 25 ℃ for 35 d. During the incubation process, water content of soils was adjusted to 60% of WHC by adding deionized water equivalent to the loss of water every 2 d. Each treatment of Cr(Ⅵ) concentration had three replications. On the day 0, 7, 14, 21, 28 and 35, the incubated soil was used for the determination of the activities of catalase, dehydrogenase, polyphenol oxidase and alkaline phosphatase.
2.3 Chemical analysis
Catalase activity in soils was measured using the KMnO4 titrimetric method. A 100-mL measuring flask was added with 5.0 g of fresh soil and 0.5 mL of toluene, shaken thoroughly, and then placed in refrigerator (4 ℃) for 30 min. Thereafter, 25 mL of 3% H2O2 solution was added into the above flask that was placed in refrigerator (4 ℃) for another 30 min. 25 mL of 1 mol/L H2SO4 was mixed and the suspension was filtered. 2 mL of filtrate was taken and added with 4 mL of 0.5 mol/L H2SO4. The residual H2O2 in filtrate was titrated with 0.1 mol/L of KMnO4 in the presence of H2SO4. A control test without soil was carried out identically at the same time. Catalase activity is expressed in mL/(g?h).
Dehydrogenase activity was determined as follows: 2 g of fresh soil was placed in an 18 mm×20 mm glass tube containing 2 mL of 3% 2, 3, 5 tri-phenyl tetrazolium chloride (TTC) and incubated at 30 ℃ for 24 h. After incubation, 10 mL of acetone was added, and the suspension was homogenized via intermittent agitation for 2 h (once every 0.5 h) and then filtered in darkness. Reactive products were measured at 485 nm using a spectrophotometer (VIS-7220). A sample without soil containing 2 mL of buffer solution instead of TTC was used as a control. Dehydrogenase activity (vs triphenyl formazon) is expressed in mg/(kg?d).
Polyphenol oxidase activity was measured by a UV-Vis spectrophotometer. Briefly, 1 g of fresh soil was transferred into an 18 mm×20 mm glass tube and mixed with 10 mL of 1% pyrogallol solution. After incubation at 30 ℃ for 1 h, 12.5 mL of 0.5 mol/L HCl was added into the mixture and shaken manually. The gallic acid produced was extracted with 10 mL of ether and measured spectrophotometrically at 430 nm. Activity (vs gallic acid) was quantified by reference to a calibration curve constructed using gallic acid standard and expressed in mg/(g?h).
Alkaline phosphatase activity was determined by the following procedure. 1.0 g of fresh soil was mixed with 0.2 mL of toluene, 4.0 mL of modified universal buffer solution (pH 11.0) and 1.0 mL of 0.025 mol/L p-nitrophenol phosphate, and then incubated at 37 ℃ for 1 h. After incubation, the mixture was mixed with 1.0 mL of 0.5 mol/L CaCl2 and 4.0 mL of 0.5 mol/L NaOH, and then filtered through a filter paper. The concentration of the product (p-nitrophenol) in the filtrate was determined colorimetrically at 400 nm. Activity (vs phenol) was quantified by reference to a calibration curve constructed using p-nitrophenol standard under the same condition and expressed in mg/(g?h).
Soil pH was determined in a 1:2.5 (ratio of mass of soil to volume of distilled water, mg/mL) suspension using a pH meter (PHS-3C). Soil organic matter was determined by oxidation with potassium dichromate and titration of excessive dichromate with ammonium ferrosulphate. Total N content was measured by digestion with the concentrated H2SO4 followed by micro-kjeldahl procedure. Cation exchange capacity (CEC) was determined with the ammonium acetate method. Soil particle size distribution was determined using the pipette method. Total Cr content was determined by digestion with a mixture of 10 mL of nitric acid, 5 mL of 1?1 sulfuric acid, and 5 mL of hydrofluoric acid, followed by ICP-AES analysis (Perkin Elmer 3300). Water soluble Cr(Ⅵ) content was determined using a 1, 5-diphenylcarbohydrazide spectro- photometric method.
2.4 Statistical analysis
An analysis of variance was carried out by the general linear model procedure of the SAS software package. Significant differences in enzymatic activities between means for different treatments were compared by using the least significant difference test at P<0.05.
3 Results and discussion
3.1 Catalase activity
As shown in Fig.1(a) and Fig.2, the changes of
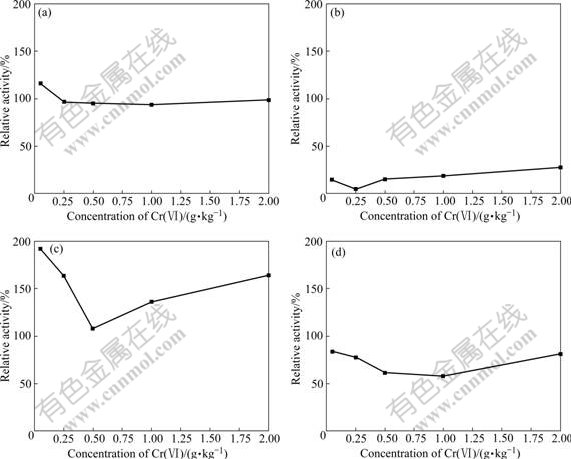
Fig.1 Effect of Cr(Ⅵ) concentration on relative activities of soil enzymes with respect to control: (a) Catalase; (b) Dehydrogenase; (c) Polyphenol oxidase; (d) Alkaline phosphatase
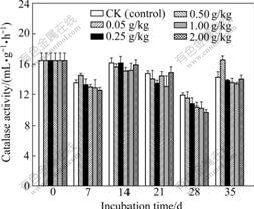
Fig.2 Changes in catalase activity of soils added with different concentrations of Cr(Ⅵ) (Each value is mean of three replicates; and bars represent standard deviations)
catalase activity caused by various Cr(Ⅵ) concentrations are not significant. The activity of this enzyme remains quite stable, and its change does not exceed 17% and is close to that of the control soil. At low concentrations of Cr(Ⅵ), such as 0.05 g/kg, the slight activation effect for catalase activity is found. Its activity is 116% of the control soil and 16% of activation is found after 35 d. In previous literatures, the effects of heavy metal on catalase activity in soil are not consistent [11-12]. It was reported that catalase activity was not markedly inhibited by heavy metals [11]. Contrast results were elucidated by KHAN et al [12] who noted that heavy metals such as Cd and Pb slightly inhibited the catalase activity. The extent of inhibition increased significantly with increasing the level of heavy metals and varied with the incubation periods.
3.2 Dehydrogenase activity
Dehydrogenase activities are presented in Fig.3. On all the sampling dates, dehydrogenase is active in the control. Moreover, the activity of dehydrogenase in the control increases with the increase of incubation time. However, a significant decrease of dehydrogenase activity for all Cr(Ⅵ) treatments is observed as early as on the day 7 and dehydrogenase activity is far lower than that of the control. Similar results are also found on other sampling dates. Dehydrogenase activity decreases with the increase of Cr(Ⅵ) concentration when Cr(Ⅵ) is lower than 0.25 g/kg and the lowest dehydrogenase activity is obtained for the treatment of 0.25 g/kg Cr(Ⅵ). However, when Cr(Ⅵ) is higher than 0.25 g/kg, dehydrogenase activity increases with the increase of the Cr(Ⅵ) concentration on the day 7. Similar trends are also obtained on the day 21, 28 and 35. Fig.1(b) also shows the total effect of Cr(Ⅵ) on dehydrogenase activity, expressed as a relative activity with respect to control. All the tested Cr(Ⅵ) concentrations cause an obvious inhibition of dehydrogenase activity by 70% after 35 d. The strongest inhibition occurs for treatment of 0.25 g/kg Cr(Ⅵ), and dehydrogenase activity for this Cr(Ⅵ) concentration declines to 5% of the control (95% inhibition). 72% inhibition effect is caused by 2.00 g/kg Cr(Ⅵ).
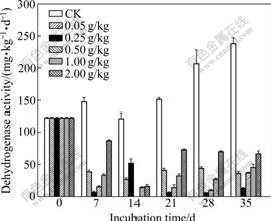
Fig.3 Changes in dehydrogenase activity of soils added with different concentrations of Cr(Ⅵ) (Each value is mean of three replicates; bars represent standard deviations)
Activities of dehydrogenase in soils added with different Cr(Ⅵ) concentrations on all sampling dates are all lower than those in the control. This result indicates that Cr(Ⅵ) has a negative influence on dehydrogenase activity. It is generally accepted that dehydrogenase activity is one of the most sensitive microbial parameters towards metal contamination because dehydrogenase only occurs within living cells. Dehydrogenase activity can reflect the ability of microorganism taking part in redox reaction in soil [13-14]. The stimulation of dehydrogenase by heavy metal in soils is principally contributed to micronutrients of microbial populations, especially at low concentrations. In contrast, the inhibition of higher heavy metal concentration on dehydrogenase might be due to the reduced microbial numbers and activity in soils because the activity is derived from the intracellular enzyme [1]. However, soil is heterogeneous system in which the reactivity of colloids may interfere with the inhibition and/or activation [15].
3.3 Polyphenol oxidase activity
Polyphenol oxidase activities for different treatments and incubation time are shown in Fig.4. A pronounced increase in polyphenol oxidase activity is observed in all treatments on the day 7. Thereafter, polyphenol oxidase activities significantly decrease again. However, the change in polyphenol oxidase activities of all treatments from 14 to 35 d is not significant. When exposed to Cr(Ⅵ) for 7 d, the augmented polyphenol oxidase activity is probably caused by the action of soil microorganism’s detoxification system.
Fig.1(c) shows the total effect of Cr(Ⅵ) on polyphenol oxidase activity. All the tested Cr(Ⅵ) concentrations stimulate polyphenol oxidase activity. The strongest activation is found for the treatment of 0.05 g/kg Cr(Ⅵ), and polyphenol oxidase activity for this treatment is as much as 192% of the control soil (92% activation). Even at the highest Cr(Ⅵ) concentration (2.00 g/kg), this enzyme is activated by 60%.
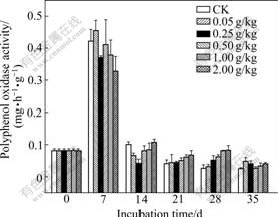
Fig.4 Changes in polyphenol oxidase activity of soils added with different concentrations of Cr(Ⅵ) (Each value is mean of three replicates, bars represent standard deviations)
3.4 Alkaline phosphatase activity
Changes of alkaline phosphatase activities in soils are shown in Fig.5. Alkaline phosphatase activities in all treatments on the day 7 are all higher than their corresponding values on the other sampling dates, indicating that alkaline phosphatase activity is influenced by the period of Cr(Ⅵ) exposure. The highest alkaline phosphatase activity is obtained for the treatments of 1.00 and 2.00 g/kg Cr(Ⅵ) on the day 7 and the lowest alkaline phosphatase activity for treatment of 2.00 g/kg Cr(Ⅵ) on the day 14. This can be explained as the suppression of some microbial populations and/or an inhibitory effect on the cellular activity by the high concentration of Cr(Ⅵ) [16]. The levels of enzymatic activities found in soil polluted by heavy metals were principally due to an indirect effect such as the inhibition of microbial growth rather than a direct inactivation of extracellular enzymes [17]. Inhibitory effect of other heavy metal on alkaline phosphatase activity was adequately reported [12]. Previous studies showed that alkaline phosphatase originated from soil bacteria, fungi and fauna [18]. Microorgainism can produce and release large amount of extracellular phosphatase due to their large combined biomass, high metabolic activity and short lifecycles [18]. The corresponding alkaline phosphatase activities for control treatment are higher than those for Cr(Ⅵ) treatments, and an inhibition of alkaline phosphatase caused by all these Cr(Ⅵ) concen- trations is less than 28%. This inhibition starts on the day 14 and maintains until the end of the experiment.
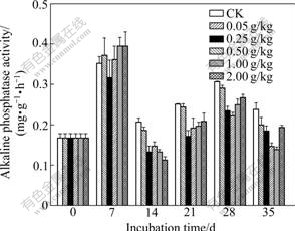
Fig.5 Changes in alkaline phosphatase activity of soils added with different concentrations of Cr(Ⅵ) (Each value is mean of three replicates, bars represent standard deviations)
Fig.1(d) shows the total effect of Cr(Ⅵ) on the activity of alkaline phosphatase. All Cr(Ⅵ) concentra- tions inhibit alkaline phosphatase activity. The strongest inhibition is found for the treatment of 1.00 g/kg Cr(Ⅵ), and alkaline phosphatase activity in this treatment is only 58% of the control soil (42% inhibition). The slightest inhibitory effect, 16% inhibition, occurs in the case of low Cr(Ⅵ) concentration (0.05 g/kg).
4 Conclusions
(1) The changes of catalase, dehydrogenase, polyphenol oxidase and alkaline phosphatase activities in soils exposed to different Cr(Ⅵ) concentrations are significantly different. Those variations depend on Cr(Ⅵ) exposure time, Cr(Ⅵ) concentration and the enzymes examined.
(2) Cr(Ⅵ) exposure shows distinctly inhibitory effect on activities of dehydrogenase and alkaline phosphatase. The effect of Cr(Ⅵ) on dehydrogenase activity is larger than that on alkaline phosphatase activity.
(3) Polyphenol oxidase activity is stimulated by Cr(Ⅵ) exposure.
(4) The changes of catalase activity caused by various Cr(Ⅵ) concentrations are minor.
References
[1] OBBARD J P. Ecotoxicological assessment of heavy metals in sewage sludge amended soils [J]. Applied Geochemistry, 2001, 16(11/12): 1405-1411.
[2] HUANG Shun-hong, YANG Zhi-hui, CHAI Li-yuan, PENG Bing, SHAO Bo-hua. Toxicity of metal chromium to microorganism activities in soil around chromium-containing slag heap [J]. Journal of Central South University: Science and Technology, 2009, 40(1): 25-30. (in Chinese)
[3] BANERJEE M, MISHRA S, CHATTERJEE J. Scavenging of nickel and chromium toxicity in Aulosira fertilissima by immobilization: Effect on nitrogen assimilating enzymes [J]. Electronic Journal of Biotechnology, 2004, 7(3): 302-309.
[4] HUANG Shun-hong, PENG Bing, YANG Zhi-hui, CHAI Li-yuan, ZHOU Li-cheng. Chromium accumulation, microorganism population and enzyme activities in soils around chromium- containing slag heap of steel alloy factory [J]. Transaction of Non-ferrous Metals Society of China, 2009, 19(1): 241-248.
[5] MEGHARAJ M, AVUDAINAYAGAM S, NAIDU R. Toxicity of hexavalent chromium and its reduction by bacteria isolated from soil contaminated with tannery waste [J]. Current Microbiology, 2003, 47(1): 51-54.
[6] GIANFREDA L. Activity of free and immobilized urease in soil: Effects of pesticides [J]. Soil Biology and Biochemistry, 1994, 26(6): 777-784.
[7] DENG S P, TABATABAI M A. Cellulase activity of soils [J]. Soil Biology and Biochemistry, 1994, 26(10): 1347-1354.
[8] RODRIGUEZ B B, BOLBOT J A, TOTHILLI E. Development of urease and glutamic dehydrogenase amperometric assay for heavy metals screening in polluted samples [J]. Biosensors and Bioelectronics, 2004, 19(10): 1157-1167.
[9] HINOJOSA M B. Soil moisture pre-treatment effects on enzyme activities as indicators of heavy metal-contaminated and reclaimed soils [J]. Soil Biology and Biochemistry, 2004, 36(10): 1559-1568.
[10] KIZILKAYA R. Dehydrogenase activity in Lumbricus terrestris casts and surrounding soil affected by addition of different organic wastes and Zn [J]. Bioresource Technology, 2008, 99(5): 946-953.
[11] BELYAEVA O N, HAYNES R J, BIRUKOVA O A. Barley yield and soil microbial and enzyme activities as affected by contamination of two soils with lead, zinc or copper [J]. Biology and Fertility of Soils, 2005, 41(2): 85-94.
[12] KHAN S, CAO Q, HESHAM A E L, XIA Y, HE J Z. Soil enzymatic activities and microbial community structure with different application rates of Cd and Pb [J]. Journal of Environmental Sciences, 2007, 19(7): 834-840.
[13] DUNGAN R S, KUKIER U, LEE B. Blending foundry sands with soil: Effect on dehydrogenase activity [J]. Science of the Total Environment, 2006, 357(1/3): 221-230.
[14] MAJER B J, TSCHERKO D, PASCHKE A, WENNRICH R, KUNDI M, KANDELER E, KNASMULLER S. Effects of heavy metal contamination of soils on micronucleus induction in Tradescantia and on microbial enzyme activities: A comparative investigation [J]. Mutation Research―Genetic Toxicology and Environmental Mutagenesis, 2002, 515(1/2): 111-124.
[15] ALEF K. Enzyme activities, in methods in applied soil microbiology and biochemistry [M]. London: Academic Press, 1995: 311-373.
[16] VITI C, MINI A, RANALLI G, LUSTRATO G, GIOVANNETTI L. Response of microbial communities to different doses of chromate in soil microcosms [J]. Applied Soil Ecology, 2006, 34(2/3): 125-139.
[17] VIG K, MEGHARAJ M, SETHUNATHAN N, NAIDU R. Bioavailability and toxicity of cadmium to microorganisms and their activities in soil: a review [J]. Advances in Environmental Research, 2003, 8(1): 121-135
[18] WANG Y P, SHI J Y, WANG H, QI L, CHEN X C, CHEN Y X. The influence of soil heavy metals pollution on soil microbial biomass, enzyme activity, and community composition near a copper smelter [J]. Ecotoxicology and Environmental Safety, 2007, 67(1): 75-81.
(Edited by CHEN Wei-ping)
Foundation item: Projects(2006AA06Z374, 2007AA021304) supported by the National High-Tech Research and Development Program of China; Project(2008SK2007) supported by the Key Program of Science and Technology of Hunan Province, China
Received date: 2008-11-06; Accepted date: 2009-03-12
Corresponding author: YANG Zhi-hui, PhD; Tel: +86-731-88830875; E-mail: yangzhihui65@yahoo.com