Speciation of chromium in soil inoculated with Cr(Ⅵ)-reducing strain, Bacillus sp. XW-4
来源期刊:中南大学学报(英文版)2009年第2期
论文作者:徐卫华 刘云国 曾光明 周鸣 樊霆 王欣 夏文斌
文章页码:253 - 253
Key words:Bacillus sp.; Cr(Ⅵ) reduction; available chromium; Cr fraction
Abstract: Cr(Ⅵ)-amended soil was inoculated with Cr(Ⅵ)-reducing strain, Bacillus sp. XW-4 and incubated at 28 ℃ in an incubator. Cr(Ⅵ) reduction, available Cr and Cr fraction in soils were studied. The results show that addition of Bacillus sp. XW-4 can promote Cr(Ⅵ) reduction, but inoculation of this strain has a negative effect on the decrease of available Cr content in soil. In controls (without this strain) amended with 100 and 200 mg/kg of Cr(Ⅵ), Cr(Ⅵ) contents decrease to about 41 and 92 mg/kg respectively after incubation of 4 d, while in soil inoculated with XW-4, Cr(Ⅵ) contents decrease to about 18 and 60 mg/kg, respectively. The content of available Cr in soils with inoculation of XW-4 is higher than that in controls. Chromium is partitioned into water soluble Cr, exchangeable Cr, precipitated Cr, Cr bound to organics and residual Cr. The highest content of Cr is observed in residual form and water soluble Cr is not detected for all treatments after 42 d, but the soils inoculated with Bacillus sp. XW-4 contain higher content of exchangeable Cr and lower content of precipitated Cr than the soil without the inoculum. Inoculation of Bacillus sp. XW-4 can increase chromium activity in soils.
基金信息:the Doctoral Foundation of Ministry of Education of China
J. Cent. South Univ. Technol. (2009) 16: 0253-0257
DOI: 10.1007/s11771-009-0043-1
![]()
XU Wei-hua(徐卫华), LIU Yun-guo(刘云国), ZENG Guang-ming(曾光明),
ZHOU Ming(周 鸣), FAN Ting(樊 霆), WANG Xin(王 欣), XIA Wen-bin(夏文斌)
(College of Environmental Science and Engineering, Hunan University, Changsha 410082, China)
Abstract: Cr(Ⅵ)-amended soil was inoculated with Cr(Ⅵ)-reducing strain, Bacillus sp. XW-4 and incubated at 28 ℃ in an incubator. Cr(Ⅵ) reduction, available Cr and Cr fraction in soils were studied. The results show that addition of Bacillus sp. XW-4 can promote Cr(Ⅵ) reduction, but inoculation of this strain has a negative effect on the decrease of available Cr content in soil. In controls (without this strain) amended with 100 and 200 mg/kg of Cr(Ⅵ), Cr(Ⅵ) contents decrease to about 41 and 92 mg/kg respectively after incubation of 4 d, while in soil inoculated with XW-4, Cr(Ⅵ) contents decrease to about 18 and 60 mg/kg, respectively. The content of available Cr in soils with inoculation of XW-4 is higher than that in controls. Chromium is partitioned into water soluble Cr, exchangeable Cr, precipitated Cr, Cr bound to organics and residual Cr. The highest content of Cr is observed in residual form and water soluble Cr is not detected for all treatments after 42 d, but the soils inoculated with Bacillus sp. XW-4 contain higher content of exchangeable Cr and lower content of precipitated Cr than the soil without the inoculum. Inoculation of Bacillus sp. XW-4 can increase chromium activity in soils.
Key words: Bacillus sp.; Cr(Ⅵ) reduction; available chromium; Cr fraction
1 Introduction
Chromium is one of the most hazardous pollutants. The wide applications of chromium have resulted in its occurrence as a common contaminant in soils. Chromium commonly exists in two oxidation states, Cr(Ⅵ) and Cr(Ⅲ), under natural environmental conditions. These two states are different in toxicity, solubility and mobility [1-2]. Cr(Ⅵ) is well known to be highly toxic and a human mutagen. Cr(Ⅵ) is highly soluble, mobile, and consequently, bioavailable in the environment as a potential soil contaminant. Cr(Ⅲ) is, however, much less toxic than Cr(Ⅵ). Cr(Ⅲ) is rather immobile due to its low solubility, high adsorption and complexation. Due to the vastly differing toxicity of these two oxidation states, the approaches to facilitate transformation of Cr(Ⅵ) to Cr(Ⅲ) are of great interest in remediation of Cr(Ⅵ)-contaminated soils.
![]()
Previous studies have demonstrated that a variety of microorganisms such as Pseudomonas sp. [3-4], Bacillus sp. [5-6], Escherichia sp. [7-8] and Enterobacter sp. [9-10] have the ability to reduce Cr(Ⅵ). Bioremediation using microorganisms offers an attractive option for detoxification of Cr(Ⅵ) pollution [4,11]. But, most of these Cr(Ⅵ)-reducing strains are used in the treatment of Cr(Ⅵ)-contaminated water, and limited information is available on application of these strains to Cr(Ⅵ)-contaminated soil. In this study, the exogenous Cr(Ⅵ)-reducing bacterium, Bacillus sp. XW-4 was inoculated into Cr(Ⅵ)-amended soil to determine the effects of this strain on Cr(Ⅵ) reduction, available chromium and chromium fraction, and evaluate the potential of this Cr(Ⅵ)-reducing strain to remediate Cr(Ⅵ)-contaminated soil.
2 Materials and methods
2.1 Soil sample
Soil samples were collected from Yuelu Mountain located in Changsha, Hunan Province, China. Soils were sampled from 0-20 cm layer. The visible plant debris, rock fragments, and soil fauna in soil samples were removed by hand. The collected soil was air-dried, and then passed through a 1 mm sieve. Some physico- chemical properties of this treated soil sample were measured. The results are listed in Table 1.
2.2 Microorganisms and cultivation conditions
2.2.1 Preparation of nutrient media
The liquid nutrient medium consisted of beef extract (0.5 g), peptone (1 g), NaCl (0.5 g) in 100 mL distilled water. The pH value of the medium was adjusted to 7.2 with 10% (mass fraction) NaOH and 10% (mass fraction) HCl. Nutrient media were autoclaved at 120 ℃ for 20 min before they are used for cultivation.
Table 1 Properties of soil sample

2.2.2 Cells cultivation
Cr(Ⅵ)-reducing strain, Bacillus sp. XW-4 was used in this study. Bacillus sp. XW-4 was isolated from chromium landfill and its ability to reduce Cr(Ⅵ) was reported in Ref.[12]. This strain was enriched by transferring one loop of cells from the agar slants to 250 mL flasks containing 100 mL of previously sterilized liquid nutrient medium, and then the flasks were incubated at 37 ℃ for 24 h by shaking at 150 r/min in an orbital incubator. The cells were then centrifuged at 4 000 r/min for 20 min. The supernatant was removed and cells pellet was washed three times with sterile distilled water. The cells suspension with the concentration of 1.0×107 /mL was prepared in sterile distilled water.
2.3 Experimental design
Soil sample of 400 g was added into 500 mL plastic container. Several K2Cr2O7 solutions with different Cr(Ⅵ) concentrations were prepared and 100 mL of each concentration was homogeneously sprayed in individual soil with a sparger. Two of Cr(Ⅵ) contents, 100 and 200 mg/kg, were prepared in soil treatments. The cells suspension (20 mL) of Bacillus sp. XW-4 was then inoculated to soil. The control (without the strain of XW-4) was added with 20 mL sterile distilled water.
All of samples were sealed up and incubated at 28 ℃ in an incubator. Soil samples were drawn at incubation of 1, 2, 3, 4, 5, 6 and 7 d for determining Cr(Ⅵ) content. At incubation of 7, 14, 21, 28 and 35 d, the content of available chromium was determined. Chromium fraction was analyzed at incubation of 42 d. Samples (1 g) were drawn at three different sites and the mixture was used for analysis. The experiments were conducted in replicate and the results were presented as the mean values of replicates.
2.4 Analytical methods
2.4.1 Analysis of Cr(Ⅵ)
Cr(Ⅵ) was extracted with 0.1 mol/L KH2PO4 [13], and the extraction was performed by shaking 3 g of soil with 15 mL of extractant for 1 h. The mixture was then centrifuged at 5 000 r/min for 20 min. The supernatant was filtered and then analyzed for the concentration of Cr(Ⅵ). Cr(Ⅵ) was measured colorimetrically by reaction with diphenylcarbazide in acid solution. The absorbance was measured at 540 nm by using a UV 754V model spectrophotometer.
2.4.2 Analysis of available chromium
Extraction of available chromium was performed with 0.05 mol/L EDTA by shaking 3 g of soil sample with 15 mL of extractant for 1 h [14]. The soil suspension was then centrifuged at 5 000 r/min for 20 min. The supernatant was filtered and analyzed for available Cr by flame atomic absorption spectrometry (FAAS, Perkin Elmer 700).
2.4.3 Chromium fraction
Chromium fraction was determined by sequential extraction scheme reported by CHEN et al [15]. Chromium in various chemical forms was extracted as follows: (1) water soluble Cr, 3 g of soil sample extracted with 15 mL of distilled water; (2) exchangeable Cr, the residue from water soluble extracted with 15 mL of 1 mol/L NH4Ac; (3) precipitated Cr, the residue from exchangeable extracted with 15 mL of 2 mol/L HCl; (4) organics-bound Cr, the residue from precipitated firstly extracted with 15 mL of 5% H2O2 and then with 15 mL of 2 mol/L HCl; (5) residual Cr, total chromium was subtracted by the content of water soluble, exchangeable, precipitated and organics-bound Cr. Total chromium was obtained by the method of H2SO4-HNO3-HF digestion, and the procedure was as follows: soil was first digested with 10 mL of nitric acid and 5 mL of sulfuric acid solution (V(sulfuric acid)?V(distilled water)=1?1), and then treated with 5 mL of hydrofluoric acid; then the remaining material was dissolved with a mixture (3 mL) of hydrochloric acid and distilled water (1?1, volume ratio). Sequential extractions were carried out in centrifuge tubes by shaking for 2 h and then remaining stationary for 2 h. Between successive extractions, separation was performed by centrifugation at 5 000 r/min for 20 min. The supernatant was filtered and then used to analyze the content of chromium, while the residue was added to next extractants. Chromium in each form was determined by flame atomic absorption spectrometry (FAAS, Perkin Elmer 700).
3 Results and discussion
3.1 Variation of Cr(Ⅵ) content
As shown in Fig.1, Cr(Ⅵ) content decreases fast, especially during the first 4 d. In controls (without XW-4) amended with 100 and 200 mg/kg of Cr(Ⅵ), the contents of Cr(Ⅵ) decrease to about 41 and 92 mg/kg respectively, after incubation of only 4 d. This indicates that Cr(Ⅵ) is reduced in soils. The organic matter probably plays an important role in Cr(Ⅵ) reduction since the content of organic matter in soil is as high as 45.98 mg/kg (Table 1). It has been reported that Cr(Ⅵ) can be reduced by organic matter directly [16-18] or through microbially mediated pathways [19-20]. In addition, soil moisture increased as soil was amended with 100 mL of K2Cr2O7 solution. Reducing conditions and the formation of Cr(Ⅲ) are favored as soil moisture content increases [21].
From Fig.1, it can be seen that Cr(Ⅵ) content is lower with inoculation of Bacillus sp. XW-4, compared with that without inoculation of this strain. In soils amended with 100 and 200 mg/kg of Cr(Ⅵ), Cr(Ⅵ) contents decrease to about 18 and 60 mg/kg, respectively, in the presence of Bacillus sp. XW-4, after incubation of only 4 d. This may be due to that the inoculated strain contributes to Cr(Ⅵ) reduction since this strain is capable of reducing Cr(Ⅵ) in Ref.[12]. SRIVASTAVA and THAKUR [22] introduced Aspergillus niger to Cr(Ⅵ)-amended soil and found that this fungus removes chromate by accumulation of chromium in fungal mycelium, but the accumulation of chromium by the inoculated strain was not determined in this study.

Fig.1 Variation of Cr(Ⅵ) content in soil amended with: (a) 100 mg/kg Cr(Ⅵ); (b) 200 mg/kg Cr(Ⅵ)
3.2 Variation of available Cr content
The variation of available Cr content in soil is shown in Fig.2. It is obvious that the content of available chromium gradually decreases in all of soil treatments. But, the soils inoculated with Bacillus sp. XW-4 contain higher content of available chromium than the controls. This indicates that inoculation of Bacillus sp. XW-4 has a negative effect on the decrease of available Cr content, though it promotes Cr(Ⅵ) reduction in soil (Fig.1). The reasons for that are not quite clear. But the metabolisms of Bacillus sp. XW-4 may change the soil properties such as soil pH and organic constituents, and thereby indirectly affect the content of available Cr. It was reported that the lowering of pH reduced the sorption of heavy metals in soil [23-25]. Addition of some organic wastes could increase the extractable heavy metals content in soil [26-27]. Further explorations are needed for explaining the effects of Bacillus sp. XW-4 on available Cr content observed in this study.

Fig.2 Variation of available Cr content in soil amended with: (a) 100 mg/kg Cr(Ⅵ); (b) 200 mg/kg Cr(Ⅵ)
3.3 Chromium fraction
Chromium may have different degrees of mobility and transportation depending on its chemical forms in soil. The chemical speciation of chromium is an important facet of the chemistry of this pollutant with respect to both environment and plant uptake of metals [28]. Chromium fraction was determined after incubation of 42 d. The results are shown in Fig.3. It is observed that in all treatments, the residual Cr content is greater than other Cr forms and water soluble Cr is not detected after 42 d. Fig.3 also shows that the exchangeable Cr content in controls is lower than that in treatments inoculated with Bacillus sp. XW-4, while the precipitated Cr content is higher in controls than that in treatments inoculated with Bacillus sp.XW-4. This demonstrates that this bacterium affects the distribution of Cr in various fractions and that inoculation of this strain can probably slower the transformation of water soluble and exchangeable Cr to precipitated Cr, thus increase the activity and mobility of chromium in soil. SHI et al [29] found that the inoculation of Pseudomonas aeruginosa ZD4-3 significantly increases the activity of Cu, Pb, and Zn, and inoculation of Phanerochaete chrysosprim also increases the activity of Cu and Zn, but decreases the activity of Pb in soil contaminated by heavy metals. Therefore, besides the ability of Cr(Ⅵ) reduction, the effects of bacteria on chromium fraction should be evaluated when Cr(Ⅵ)-reducing bacteria are applied to remediation of Cr(Ⅵ)-contaminated soil.
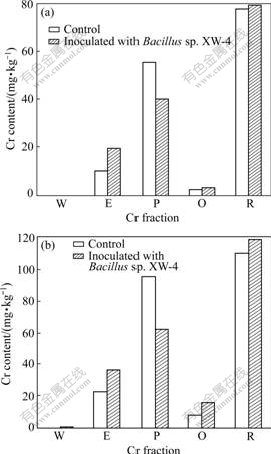
Fig.3 Chromium fraction in soil amended with: (a) 100 mg/kg Cr(Ⅵ); (b) 200 mg/kg Cr(Ⅵ) (W denotes water soluble Cr; E denotes exchangeable Cr; P denotes precipitated Cr; O denotes organics-bound Cr; R denotes residual Cr)
4 Conclusions
(1) Addition of exogenous Cr(Ⅵ)-reducing bacterium, Bacillus sp., XW-4, can promote Cr(Ⅵ) reduction in soils. In soils amended with 100 and 200 mg/kg, with inoculation of Bacillus sp.XW-4, Cr(Ⅵ) contents decrease to about 18 and 60 mg/kg respectively, after incubation of 4 d, while in controls without inoculation of XW-4, Cr(Ⅵ) contents decrease only to about 41 and 92 mg/kg.
(2) In soils inoculated with Bacillus sp.XW-4, the content of available Cr is higher than that in the controls. This indicates that Bacillus sp.XW-4 can increase the content of available Cr.
(3) Inoculation of Bacillus sp.XW-4 affects Cr fraction. It is found that precipitated Cr content is lower, but exchangeable Cr content is higher with inoculation of Bacillus sp.XW-4 than those without inoculation of this strain. Inoculation of Bacillus sp.XW-4 may increase chromium activity in soil.
References
[1] FENDORF S E. Surface reactions of chromium in soils and waters [J]. Geoderma, 1995, 67(1/2): 55-71.
[2] KOTA? J, STASICKA Z. Chromium occurrence in the environment and methods of its speciation [J]. Environ Pollut, 2000, 107(3): 263-283.
[3] GANGULI A, TRIPATHI A K. Survival and chromate reducing ability of Pseudomonas aeruginosa in industrial effluents [J]. Lett Appl Microbiol, 1999, 28(1): 76-80.
[4] KONOVALOVA V V, DMYTRENKO G M, NIGMATULLIN R R, BRYK M T, GVOZDYAK P I. Chromium (Ⅵ) reduction in a membrane bioreactor with immobilized Pseudomonas cells [J]. Enzyme Microb Tech, 2003, 33(7): 899-907.
[5] GARBISU C, ALKORTA I, LLAMA M J, SERRA J L. Aerobic chromate reduction by Bacillus subtilis [J]. Biodegradation, 1998, 9(2): 133-141.
[6] PHILIP L, IYENGAR L, VENKOBACHAR C. Cr(Ⅵ) reduction by Bacillus coagulans isolated from contaminated soils [J]. J Environ Eng, 1998, 124(12): 1165-1170.
[7] SHEN H, WANG Y T. Characterization of enzymatic reduction of hexavalent chromium by Escherichia coli ATCC 33456 [J]. Appl Environ Microb, 1993, 59(11): 3771-3777.
[8] ACKERLEY D F, GONZALEZ C F, KEYHAN M, BLAKE R, MATIN A. Mechanism of chromate reduction by the Escherichia coli protein, NfsA, and the role of different chromate reductases in minimizing oxidative stress during chromate reduction [J]. Environ Microbiol, 2004, 6(8): 851-860.
[9] OHTAKE H, FUJII E, TODA K. Reduction of toxic chromate in an industrial effluent by use of a chromate-reducing strain of Enterobacter cloacae [J]. Environ Technol Lett, 1990, 11(5): 663-668.
[10] REGE M A, PETERSEN J N, JOHNSTONE D L, TURICK C E, YONGE D R, APEL W A. Bacterial reduction of hexavalent chromium by Enterobacter cloacae strain HO1 grown on sucrose [J]. Biotechnol Lett, 1997, 19(7): 691-694.
[11] SHEN Li, XIA Jin-lan, HE Huan, NIE Zhen-yuan, QIU Guan-zhou. Biosorption mechanism of Cr(Ⅵ) onto cells of Synechococcus sp. [J]. J Cent South Univ Technol, 2007, 14(2): 157-162.
[12] LIU Yun-guo, XU Wei-hua, ZENG Guang-ming, LI Xin, GAO Hui. Cr(Ⅵ) reduction by Bacillus sp. isolated from chromium landfill [J]. Process Biochem, 2006, 41(9): 1981-1986.
[13] BECQUER T, QUANTIN C, SICOT M, BOUDOT J P. Chromium availability in ultramafic soils from New Caledonia [J]. Sci Total Environ, 2003, 301(1/3): 251-261.
[14] CHEN Ying-xu, ZHU Zu-xiang, HE Zeng-yao. Cr availability in soils and its effects on pollution ecology [J]. Acta Ecol Sinica, 1995, 15(1): 79-84. (in Chinese)
[15] CHEN Ying-xu, HE Zeng-yao, WU Jian-ping. Speciation and transformation of chromium in soil [J]. Environ Sci, 1994, 15(3): 53-56. (in Chinese)
[16] DENG B, STONE A T. Surface-catalyzed chromium(Ⅵ) reduction: Reactivity comparisons of different organic reductants and different oxide surfaces [J]. Environ Sci Technol, 1996, 30(8): 2484-2494.
[17] WITTBRODT P R, PALMER C D. Reduction of Cr(Ⅵ) by soil humic acids [J]. Eur J Soil Sci, 1997, 48(1): 151-162.
[18] NAKAYASU K, FUKUSHIMA M, SASAKI K, TANAKA S, NAKAMURA H. Comparative studies of the reduction behavior of chromium(Ⅵ) by humic substances and their precursors [J]. Environ Toxicol Chem, 1999, 18(6): 1085-1090.
[19] TOKUNAGA T K, WAN J, FIRESTONE M K, HAZEN T C, OLSON K R, HERMAN D J, SUTTON S R, LANZIROTTI A. In situ reduction of chromium(Ⅵ) in heavily contaminated soils through organic carbon amendment [J]. J Environ Qual, 2003, 32(5): 1641-1649.
[20] GU B, CHEN J. Enhanced microbial reduction of Cr(Ⅵ) and U(Ⅵ) by different natural organic matter fractions [J]. Geochim Cosmochim Acta, 2003, 67(19): 3575-3582.
[21] MASSCHELEYN P H, PARDUE J H, DELAUNE R D, PATRICK W H. Chromium redox chemistry in a lower Mississippi Valley bottomland hardwood wetland [J]. Environ Sci Technol, 1992, 26(6): 1217-1225.
[22] SRIVASTAVA S, THAKUR I S. Evaluation of bioremediation and detoxification potentiality of Aspergillus niger for removal of hexavalent chromium in soil microcosm [J]. Soil Biol Biochem, 2006, 38(7): 1904-1911.
[23] YUAN G, LAVKULICH L M. Sorption behavior of Cu, Zn, and Cd in response to simulated changes in soil properties [J]. Commun Soil Sci Plant Anal, 1997, 28(4): 571-587.
[24] ARNESEN A K M, SINGH B R. Plant uptake and DTPA-extractablility of Cd, Cu, Ni and Zn in a Norwegian alum shale soil as affected by previous addition of dairy and pig manures and peat [J]. Can J Soil Sci, 1998, 78(3): 531-539.
[25] GUO Zhao-hui, LIAO Bo-han, HUANG Chang-yong. Leaching potential and changes in components of metals in two acidic ferrisols [J]. J Cent South Univ Technol, 2006, 13(6): 631-636.
[26] HE Q B, SINGH B R. Effect of organic matter on the distribution, extractability and uptake of cadmium in soils [J]. Eur J Soil Sci, 1993, 44(4): 641-650.
[27] KARACA A. Effect of organic wastes on the extractability of cadmium, copper, nickel, and zinc in soil [J]. Geoderma, 2004, 122(2/4): 297-303.
[28] OTABBONG E. Chemistry of Cr in some Swedish soils. Ⅰ: Chromium speciation in soil extracts: A comparison of different methods [J]. Acta Agric Scand, 1989, 39(2): 119-129.
[29] SHI Ji-yan, CHEN Ying-xu, LIN Qi, WANG Yuan-peng. The influence of root exudates and microbe on heavy metal activity in contaminated soil [J]. China Environ Sci, 2004, 24(3): 316-319. (in Chinese)
Foundation item: Project(20050532009) supported by the Doctoral Foundation of Ministry of Education of China; Project supported by Program for Changjiang Scholars and Innovative Research Team in University
Received date: 2008-06-28; Accepted date: 2008-08-09
Corresponding author: LIU Yun-guo, Professor, PhD; Tel: +86-731-8649208; E-mail: liuyunguo@hnu.cn
(Edited by YANG You-ping)

