Trans. Nonferrous Met. Soc. China 23(2013) 2650-2656
Influence of Ga and In on microstructure and electrochemical properties of Mg anodes
Yan FENG, Ri-chu WANG, Chao-qun PENG
School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 20 August 2012; accepted 28 December 2012
Abstract: The influence of Ga and In on the electrochemical properties of Mg anode materials were investigated by the polarization and galvanostatic curve tests. The microstructure and the corroded surface of the Mg-In-Ga alloys were observed by scanning electron microscopy (SEM). The corrosion product of the Mg-0.8%In (mass fraction) and Mg-0.8%Ga-0.3%In alloy were determined by X-ray diffraction. The results show that no second phase exists in the Mg-xIn (x=0-0.8%) allloys. Intergranular compounds containing Ga and In elements occur in the Mg-0.8%In-xGa (x=0-0.8%) alloys. The addition of In into Mg as well as the addition of 0.05%-0.5%Ga into Mg-In alloy promotes the corrosion resistance. The addition of Ga into Mg-In alloys also promotes the electrochemical activity. The Mg-0.8%In-0.8%Ga alloy has the most negative mean potential, -1.682 V, which is more negative than -1.406 V in AZ91D. The corrosion type of the Mg-In-Ga alloys is general corrosion and the corrosion product is Mg(OH)2.
Key words: Mg anode; alloying element; Ga; In; galvanostatic curve; corrosion resistance; electrochemical activity
1 Introduction
Mg alloys are widely used in high energy density batteries and sacrificial anodes due to many advantages, such as rapid activation, low specific mass, low electrode potential and high current capacity [1-5]. Since 1960 s, Mg alloy batteries have been the subject of comprehensive interest and have been developed for some military and commercial applications, such as torpedo, electromotive and unmanned underwater vehicle power source.
The developed Mg anode materials with high cell voltage are AP65 (Mg-6%Al-5%Pb) [6,7], Mg-7%Tl- 5%Al [1] and Mg-Hg-Ga alloys [8]. The specific energy of the seawater battery using Mg-Hg-Ga alloys as anode can reach 150 W・h/kg [9,10], compared with 30 W・h/kg of lead acid battery. The open potential of Mg-Hg alloys in a 3.5% NaCl solution is -2.0 V (vs SCE) [8], compared with -1.8 V (vs SCE) of Mg-6%Al-5%Pb alloys [6]. However, the good results of the Mg anodes obtained in high power seawater battery still meet with problems such as bad deformability, large self-corrosion velocity and low current efficiency [9,10]. Especially the Cl- in seawater produces acid environment for the Mg anodes and increases corrosion of the Mg anodes [11-14]. In order to solve these problems, some alloying elements with high over-potential of hydrogen revolution and large electrochemical activity, such as In, Zn, Mg and Re elements, were added into Mg anodes in recent years [15-18].
According to Refs. [8,13,19-24], Ga and Hg can greatly activate Mg anodes due to the dissolution- redeposition mechanism and Ga can also enhance the corrosion resistance of the amalgams [25]. Due to the toxicity of Hg, In is chosen to replace Hg. In this work, the effect of Ga and In on electrochemical corrosion behavior of Mg anode materials was studied.
2 Experimental
The Mg-In and Mg-In-Ga alloys, with the given chemical compositions shown in Table 1, were sealed in Fe flask under Ar atmosphere and melted in muffle. The chemical composition of the specimens was determined by emission spectrum analysis (ESA) and atomic absorption spectrochemical analysis. The small amounts of impurities such as Fe and Ca were confirmed. The alloys were prepared three times and the deviation of the main composition is under 0.5%.
Microstructure and corroded surface of the specimens were observed using SEM (KYKY-2800). The corrosion product was determined by XRD with a Rigaku D/Max 2500 V diffractometer with Cu Kα radiation.
Potentiodynamic and galvanostatic experiments were performed with a Potentiostat-Galvanostat (Model 263A) in a 3.5% NaCl solution. The scanning rate of potentiodynamic measurement is 5 mV/s and the anodic current density in the galvanostatic test is 180 mA/cm2. The specimens were polished with emery paper and buffed to a mirror finish. Each of them was sealed with epoxy resin except for an exposed surface of 10 mm×10 mm submitted to the electrochemical tests in a three-electrode cell. A platinum sheet was used as the auxiliary electrode and a SCE with a standard electrode potential of 0.2412 V(SHE) was used as the reference electrode.
Table 1 Chemical compositions of Mg-In-Ga alloys
3 Results and discussion
3.1 Effect of Ga and In on microstructure
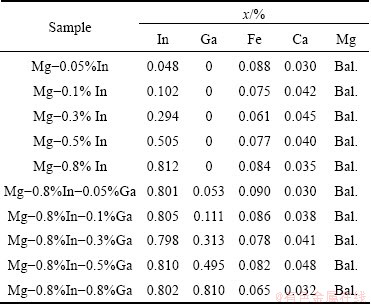
Figure 1 shows the SEM images of the cast Mg-In and Mg-In-Ga alloys. From Figs. 1(a) and (b), it can be seen that no second phase exists in the Mg-In specimens. Figures 1(c) and (d) show that intergranular compounds containing Ga and In elements exist in the Mg-In-Ga alloys. The amount of the intergranular compounds increases and the grain size decreases when the Ga content increases from 0.3% to 0.8%. According to ESA measurements in Fig. 2, the intergranular compound in Mg-0.8%In-0.3%Ga alloy contains 2.09% In, 1.51% Ga and the intergranular compound in Mg-0.8%In-0.8%Ga alloy contains 2.52% In, 2.32% Ga. The addition of Ga in Mg-In alloys inspires the formation of the intergranular compounds containing Ga and In.

Fig. 1 SEM images of Mg-Ga-In alloys
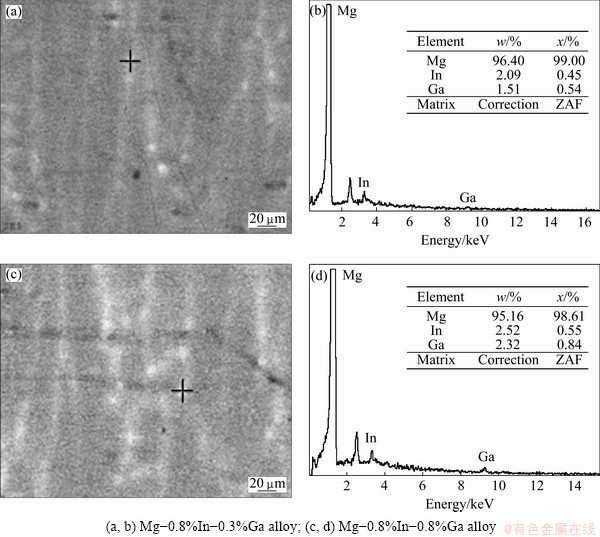
Fig. 2 ESA of grain boundary of Mg-In-Ga alloys
3.2 Effect of Ga and In on electrochemical corrosion behavior
Figure 3 shows the polarization curves of the Mg-In alloys and Mg-In-Ga alloys in the 3.5% NaCl solution. Tafel curves were fitted using CHI660A software. During the potentiodynamic scanning, a stead state current is attained for each applied potential step. Started from the rest potential, the anode polarization shows a remarkable increase in the anodic current density. When the potential rises near the rest potential, the anodic dissolution rate was accelerated and increased linearly with the increase of potential. Whole reaction is controlled by activation polarization. The black corrosion products flake off continuously in the process of polarization, which is consistent with the electrochemical reaction of an amalgam with moisture [22].

Fig. 3 Polarization curves of Mg-In alloys (a) and Mg-In-Ga alloys (b) in 3.5% NaCl solution at 298 K
Table 2 Electrochemical parameters of Mg-In-Ga alloys after aging treatments calculated from tests
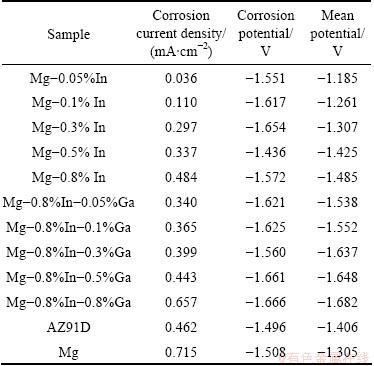
Table 2 shows the corrosion potential and corrosion current densities calculated from the polarization curves in Fig. 3. It can be seen that the Mg-In and Mg-In-Ga alloys both have lower corrosion current density than pure Mg. The additions of In and Ga into Mg both decline the corrosion current density and promote the corrosion resistance. Moreover, the Mg-In and Mg-In-Ga alloys have lower corrosion current density than AZ91D except Mg-0.8%In and Mg-0.8%In- 0.8%Ga alloys. In Mg-In alloys, the corrosion current density increases with the increasing In content. The smallest corrosion current density of 0.036 mA/cm2 exists in the Mg-0.05%In alloy. The largest corrosion current density of 0.484 mA/cm2 exists in the Mg-0.8%In alloy. The addition of 0.05%Ga into the Mg-0.8%In alloy reduces the corrosion current density to 0.340 mA/cm2. While as the content of Ga increases to 0.8%, the corrosion current density of the Mg-0.8%In-0.8%Ga alloy increases to 0.657 mA/cm2.
The alloying elements and intergranular compounds influence the corrosion resistance of the Mg-In-Ga alloys. As seen in Fig. 2, no second phase exists in the Mg-In alloys while intergranular compounds containing Ga and In elements exist in the Mg-In-Ga alloys. The standard electrode potentials of In and Ga are -0.14 V and -0.56 V, respectively, which both are more positive than standard electrode potential of Mg, -2.7 V. The solid-solution of In and Ga in а-Mg moves the electrode potential of the Mg-In and Mg-In-Ga alloys positive. The electro-negativity discrepancy as well as the driving force of corrosion galvanic cell is reduced. The corrosion resistance of Mg-In and Mg-In-Ga alloys is promoted. Due to the solution of Ga in the Mg-In-Ga alloys, whose corrosion resistance is better than the corrosion resistance of the Mg-In alloys. Meanwhile, the intergranular compounds containing Ga and In elements have more positive standard electrode potential due to the high content of Ga and In in the Mg-In-Ga alloys. The intergranular compounds work as cathode in the corrosion galvanic cell. When the amount and size of the intergranular compounds increase, the driving force of corrosion galvanic cell increases. As a result, the largest corrosion current density exists in Mg-0.8%In-0.8%Ga alloy.
Figure 4 shows the galvanostatic curves of Mg-In and Mg-In-Ga alloys at the 180 mA/cm2 anodic current density. Serious polarization behavior occurs in pure Mg with the test time prolonging, while the Mg-In and Mg-In-Ga alloys show no polarization behavior. Only galvanostatic curves display some oscillations in the Mg-0.3%In alloys and Mg-0.8%In-0.5%Ga alloy. The mean potentials of the alloys were measured from the galvanostatic curves in Table 2. It can be seen that the mean potentials of the alloys move negative when the addition of In and Ga contents increases. In Mg-In alloys, the largest mean potentials of -1.485 V exists in the Mg-0.8%In alloy. The addition of In improves the electrochemical activity of the pure Mg. The addition of 0.8%Ga into the Mg-0.8%In alloy moves the mean potential to -1.682 V, which is more negative than -1.406 V in AZ91D. The addition of Ga promotes the electrochemical activity of the Mg-In alloy.
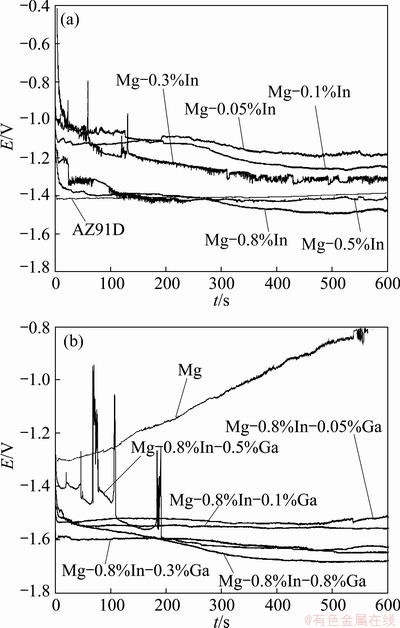
Fig. 4 Galvanostatic curves of Mg-In alloys (a) and Mg-In-Ga alloys (b) at 180 mA/cm2 current density
The electrochemical activities of the alloys are also relevant to the amount and size of the intergranular compounds. In pure Mg, it is difficult for the corrosion products to strip from the surface. The thick coverage of the corrosion products leads to serious polarization behavior in pure Mg. In the Mg-In-Ga alloys, the intergranular compounds containing Ga and In work as cathode in the electrochemical reaction. The activation reaction of the surface begins from the interfacial zone between а-Mg and compounds. The corrosion of the interfacial zone leads to the dissolution of the anodes, which agrees with the previous work [21,22]. The largest amount and size of the compounds in the Mg-0.8%In-0.8%Ga leads to homogeneous generalized dissolution of matrix, which enlarges the area of the activation reaction and enhances the electrochemical activity.
Considering the comprehensive property of the Mg alloys as anode materials, the best alloy is Mg-0.8%In-0.3%Ga alloy with a corrosion current density of 0.399 mA/cm2 and a mean potential of -1.637 V. Comparing with other Mg-In-Ga alloys, Mg-0.8%In-0.3%Ga alloy has relative larger electrochemical activity and lower corrosion current density.
3.3 Activation mechanism
Figure 5 shows SEM images of the corrosion surface of the Mg-0.8%In and Mg-0.8%In-0.3%Ga alloys after galvanostatic tests for 600 s. From Fig. 5(a), it can be seen that the alloy is mainly attacked by general corrosion in the а-Mg solid solution. Visible corrosion product accumulated on the surface of the Mg-0.8%In alloy and is difficult to be stripped from the Mg matrix. This thick corrosion product layer seriously impedes the electrochemical reaction of Mg with NaCl solution as well as the electrochemical activity of the Mg-In alloys. In the Mg-0.8%In-0.3%Ga alloy, a thin layer of loose and porous corrosion product can be seen in Fig. 5(b). Due to the reaction 2H++2e→H2, some bubble corrosion product can be seen. Some bubbles crack and make the activated а-Mg exposed. This demonstrates that the corrosion product can strip easily from the surface of the alloy and maintain the electrochemical reaction between Mg and NaCl solution. The Mg-0.8%In-0.3%Ga alloy has more negative mean potential and better electrochemical activity than Mg-0.8%In alloy. Figure 5(c) shows the corrosion surface of the Mg-0.8%In-0.3%Ga alloy after removing the corrosion product. It can be seen that the general corrosion occurs in the Mg-0.8%In-0.3%Ga alloy. It leads to the homogenous corrosion and better corrosion resistance without pitting in-depth. The Mg-0.8%In-0.3%Ga alloy has more negative mean potential and better electrochemical activity than Mg-0.8%In alloy.
Figure 6 shows that the corrosion products of the Mg-0.8%In alloy and Mg-0.8%In-0.3%Ga alloy are Mg(OH)2 and no alloying elements Ga and In exist. It can be detected that the main electrochemical reaction is Mg2++2H2O→Mg(OH)2+2H+. The alloying elements Ga and In react with water and dissolve in the solution. The electrochemical reactions are Ga→Ga3++3e, In→In++e.

Fig. 5 SEM images of surface of Mg-0.8%In alloys (a) and Mg-0.8%In-0.3%Ga alloys (b, c) after galvanostatic test in 3.5% NaCl solution at 298 K for 600 s
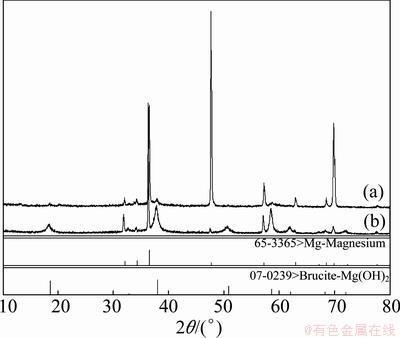
Fig. 6 X-ray analysis of corroded products of Mg-0.8%In- 0.3%Ga (a) and Mg-0.8%In (b) alloys after galvanostatic test
4 Conclusions
1) No second phase exists in the Mg-xIn (x=0-0.8%) alloys. Intergranular compounds containing Ga and In elements occur in the Mg-0.8%In-xGa (x=0-0.8%) alloys.
2) The additions of In into Mg promote the corrosion resistance and the addition of 0.05%-0.5%Ga into Mg-In alloys promotes the corrosion resistance and electrochemical activity. The addition of 0.8% Ga into the Mg-0.8%In alloy moves mean potential to -1.682 V, which is more negative than -1.406 V in AZ91D.
3) The corrosion types of the Mg-In and Mg-In-Ga alloys are general corrosion. The corrosion products of the Mg-In and Mg-In-Ga alloys are Mg(OH)2.
4) Considering the combinational property of the Mg alloys as anode materials, the best alloy is Mg-0.8%In-0.3%Ga alloy with a corrosion current density of 0.399 mA/cm2 and a mean potential of -1.637 V.
References
[1] DORON A, GURUKAR S S, ELENA L, ARIEL M, OREN M, ORIT C. Progress in rechargeable magnesium battery technology [J]. Advanced Materials, 2007, 19(23): 4260-4267.
[2] RENUKA R. Influence of allotropic modifications of surphur on the cell voltage in Mg-CuI(S) seawater activated battery [J]. Materials Chemistry and Physics, 1999, 59(1): 42-48.
[3] JEROME G, DANIEL C, KATIA G, MARC D S, FRANCIS M, ANDRE H. Magnesium batteries: Towards a first use of graphite fluorides [J]. Journal of Power Sources, 2007, 173(1): 592-598.
[4] RENUKA R. AgCl and Ag2S as additives to CuI in Mg-CuI seawater activatied batteries [J]. Journal of Applied Electrochemistry, 1997, 27(12): 1394-1397.
[5] ZHAO Hong-yang, BIAN Pei, JU Dong-ying. Electrochemical performance of magnesium alloy and its application on the sea water battery [J]. Journal of Environmental Sciences Supplement, 2009, 21(1): S88-S91.
[6] UDHAYAN R, MUNIYANDI N, MATHUR P B. Studies on magnesium and its alloys in battery electrolytes [J]. Corrosion, 1992, 27(1): 68-71.
[7] UDHAYAN R, BHATT D P. On the corrosion behaviour of magnesium and its alloys using electrochemical techniques [J]. Journal of Power Sources, 1996, 63(1): 103-107.
[8] FENG Yan, WANG Ri-chu, YU Kun, PENG Chao-qun, JIN Zhan-peng, ZHANG Chun. Activation of Mg-Hg anodes by Ga in NaCl solution [J]. Journal of Alloys and Compounds, 2009, 473(1-2): 215-219.
[9] RAO K V. Performance evaluation of Mg-AgCl batteries for under water propulsion [J]. Defence Science Journal, 2001, 5(2): 161-170.
[10] SONG Guang-lin, ATRENS A. Corrosion mechanisms of magnesium alloys [J]. Advanced Engineering Materials, 1999, 1(1): 11-33.
[11] FENG Yan, WANG Ri-chu, YU Kun, PENG Chao-qun, LI Wen-xian. Influence of Ga and Hg on microstructrue and electrochemical corrosion behavior of Mg alloy anode materials [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(6): 1363-1366.
[12] WANG Lei, ZHANG Bo-ping, TADASHI S. Corrosion behavior of AZ91 magnesium alloy in dilute NaCl solutions [J]. Materials & Design, 2010, 31(2): 857-863.
[13] UHLENHAUT D I, FURRER A, UGGOWITZER P J, LOFFLER L F. Corrosion properties of glassy Mg70Al15Ga15 in 0.1 M NaCl solution [J]. Intermetallics, 2009, 17(10): 811-817.
[14] MA Zhen-qing, PANG XU, ZUO Lie, CAO lin, ZENG Su-min. Mechanism of activation of anode of mg alloy seawater battery [J]. Surface Technology, 2008, 37(1): 5-7.
[15] XI Bei-hua, XIA Tian. Survey of power battery for torpedo propulsion [J]. Torpedo Technology, 2005, 13(2): 7-12.
[16] FONT S, DESCROIX J P, SARRE G, CHERRY H. Advanced reserve batteries for torpedoes propulsion [C]//Penniton: Proceedings of the 31st Power Sources Symposium, 1984: 362-368.
[17] KIM J G, JOO J H, KOO S J. Development of high-driving potential and high-efficiency Mg-based sacrificial anodes for cathodic protection [J]. Journal of Materials Science Letters, 2000, 19(6): 477-479.
[18] MA Zhen-qing, LI Wen-xian, YU Kun. Electrochemical characteristics of magnesium alloys in synthetic seawater [J]. Materials Protection, 2002, 35(12): 16-18.
[19] FLAMINI D O, SAIDMAN S B, BESSONE J B. Aluminium activation produced by gallium [J]. Corrosion Science, 2006, 48(6): 1413-1425.
[20] DENG Shu-hao, YI Dan-qing, ZHAO Li-hong, ZHOU Ling-ling, WANG Bin, JI Cheng-nian, LAN Bo. Study on Mg alloy anode material for seawater battery [J]. Bettery Technology, 2007, 131(5): 402-405.
[21] SHAYEB H A, WAHAB F M, ABEDIN S Z. Effect of gallium ions on the electrochemical behaviour of Al, Al-Sn, Al-Zn and Al-Zn-Sn alloys in chloride solutions [J]. Corrosion Science, 2001, 43(4): 643-645.
[22] NESTORIDI M, PLETCHER D, WOOD R J K, WANG S C, JONES R L, STOKES K R. The study of aluminium anodes for high power density Al/air batteries with brine electrolytes [J]. Journal of Power Sources, 2008, 178(1): 445-455.
[23] FENG Yan, WANG Ri-chu, YU Kun, PENG Chao-qun, LI Wen-xian. Influence of Ga content on electrochemical behavior of Mg-5at%Hg anode materials [J]. Materials Transaction, JIM, 2008, 49(5): 1077-1080.
[24] BESSONE J B. The activation of aluminium by mercury ions in non-aggressive media [J]. Corrosion Science, 2006, 48(12): 4243-4256.
[25] PINSSCO M R, ANGELINI E, CORDANO E, PRSALBINO F. Structural characterisation and corrosion resistance of Ga-precious metal alloys formed by liquid-solid reaction at room temperature [J]. Journal of Alloys and Compounds, 2001, 317-318: 411-418.
合金元素Ga和In对Mg阳极组织和电化学性能的影响
冯 艳,王日初,彭超群
中南大学 材料科学与工程学院,长沙 410083
摘 要:采用动电位极化和恒电流曲线测试合金元素Ga和In对Mg阳极材料电化学性能的影响。采用扫描电镜法分析Mg-In-Ga合金的显微组织和腐蚀表面,并用X射线衍射法检测Mg-0.8%In合金和Mg-0.8%Ga-0.3%In 合金的腐蚀产物。结果表明:Mg-xIn (x=0-0.8%) 合金中没有第二相出现,Mg-0.8%In-xGa (x=0-0.8%)合金中存在富含Ga和In元素的晶间化合物。合金元素In和0.05%-0.5%Ga的添加提高了镁阳极的耐腐蚀性能,Ga元素的添加更促进了Mg-In合金的电化学活性。Mg-0.8%In-0.8%Ga合金的平均电位最负,为-1.682 V,此电位比AZ91D合金的-1.406 V更负。Mg-In-Ga合金的腐蚀类型是全面腐蚀,其腐蚀产物是Mg(OH)2。
关键词:Mg阳极;合金元素;Ga;In;恒电流曲线;抗腐蚀性;电化学活性
(Edited by Chao WANG)
Foundation item: Project (20110162120051) supported by Specialized Research Fund for the Doctor Program of Higher Education, China; Project (51101171) supported by the National Natural Science Foundation of China
Corresponding author: Ri-chu WANG; Tel: +86-13755199741; E-mail: fengyanmse@yahoo.com.cn
DOI: 10.1016/S1003-6326(13)62781-8