Trans. Nonferrous Met. Soc. China 24(2014) 3008-3013
Separation of macro amounts of tungsten and molybdenum by ion exchange with D309 resin
Xiao-ying LU, Guang-sheng HUO, Chun-hua LIAO
School of Metallurgy and Environment, Central South University, Changsha 410083, China
Received 25 May 2014; accepted 2 September 2014
Abstract: Based on the difference in tendency to polymerize between tungsten and molybdenum, a new method using D309 resin was propounded. The batch tests indicate that the optimum pH value and contact time for the separation are 7.0 and 4 h respectively, the maxium separation factor of W and Mo is 9.29. And the experimental resules show that isothermal absorbing tungsten and molybdenum belongs to Langmuir model and Freundlich model respectively, and the absorbing kinetics for tungsten is controlled by intra-particle diffusion. With a solution containing 70 g/L WO3 and 28.97 g/L Mo, the effluent with a mass ratio of Mo to WO3 of 76 and the eluate with a mass ratio of WO3 to Mo of 53.33 are obtained after column test.
Key words: high concentrated tungstate and molybdenum solution; ion exchange; batch tests; column tests
1 Introduction
Nowsday, tungsten and molybdenum are widely applied to many hi-tech industrial products. However, there is a great challenge for separating tungsten and molybdenum effectively because they have many similar properties [1,2]. There are many methods for separating tungsten and molybdenum, such as precipitation [3], solvent extraction [4], and ion exchange [5,6]. Most of these methods are based on the different affinities of tungsten and molybdenum towards sulfur [7], which more easily makes MoO4- transform into MoS42- by sulfurizing agents, while tungsten still exists in the form of WO42-. For example, the current technology for deep removal of molybdenum from tungstate solution is based on adsorbing MoS42- by copper compounds, which has been widely applied in China. However, when the Mo to W molar ratio is more than 1/20, a worse result will be obtained [8]. The scheelite (CaWO4) gradually became the main resource because of great consumption of wolframite (FeWO4, MnWO4), and the scheelite includes more Mo than wolframite, which results in feed solutions containing plenty of tungsten and molybdenum, and can obviously add the difficulty for separating tungsten and molybdenum [9,10].
Except for the different affinities of tungsten and molybdenum towards sulfur, the different tendencies to polymerize between tungsten and molybdenum are also used to separate tungsten and molybdenum [11]. According to Refs. [12,13], WO42- and MoO42- can form isopolytungstate and isopolymolybdate respectively: 7H++6MO42-=HM6O215-+3H2O, where M represents W or Mo. ZHANG et al [14] studied the thermodynamic analysis for separation of tungsten and molybdenum in W-Mo-H2O system and concluded that the polymeric degree of tungsten is significantly higher than that of molybdenum in the pH range of 6.5-7.5, which indicates that the polymeric ability of monotungstate is stronger. Compared with the monomeric anions, heteropoly anions have higher affinity to some anion adsorbents. Taking the large-size heteropoly anions with slow diffusion rate in the adsorbent interior into account, macroporous resins are considered to be more suitable.
Among these methods, it has been illustrated that ion exchange has a great potention because this method has many advantages, such as, shorter flow process, higher recovery and less pollution. According to reports, an extractant of N1923 with primary amine can get better result in selective extraction of tungsten from sodium molybdate solution [15]. So, D309 resin with primary amine is selected in the present experiment.
This study mainly focuses on the effective separation from a solution with high concentration of tungsten and molybdenum by ion exchange with D309 resin, and the adsorption behavior and the rate controlling steps were also studed.
2 Experimental
Initial solutions were synthesized by Na2WO4 (AR) and Na2MoO4 (AR). The D309 resin was used as adsorbent with the main characteristics listed in Table 1.
Prior to testing, the D309 resin was firstly soaked in water for 24 h, and then soaked in 4% NaOH (0.04 g/mL) for 12 h. After rinsing with distilled water, the resin was activated with 4% HCl (v/v) for 12 h.
Batch tests were carried out in the constant temperature oscillator using a 100 mL conical flask. Flask containing 50 mL solution (70 g/L WO3 and 28.97 g/L) was shaken for 4 h after putting 10 mL resin into. Then the loaded resin was rinsed with distilled water.
In adsorption isotherm tests, 4.0 mL of D309 resin was mixed with 50 mL solutions containing different initial concentrations, in which the concentrations of WO3 ranged from 10 to 80 g/L and the increment was 10 g/L, accordingly the concentrations of Mo ranged from 4.14 to 33.10 g/L and the increment was 4.14 g/L. In all cases, the pH of the solution was maintained at 7.0 and all samples were stirred for 4 h at ambient temperature.
For adsorption kinetics tests, 2.0 mL of D309 resin (particle size 0.6-1.0 mm) was stirred in 500 mL solution (pH=7.0) containing 70 g/L WO3 and 28.97 g/L Mo at 30, 45 and 60 ��C respectively. The WO3 and Mo concentrations could be regarded as constants during the reaction process because the adsorbed metals contents were less than 1.5%. After the desired contact time, the resin was rapidly separated from the solution and then washed with distilled water. The adsorption rate of WO42- ions was calculated according to the following equation:
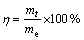 (1)
(1)
where mt is adsorption amount at time t; me is the equilibrium adsorption amount.
In column tests, 287 mL of D309 resin was wet-packed into the glass column (20 mm in diameter, 900 mm in height) with solution containing 70 g/L WO3 and 28.97 g/L Mo, keeping a flow rate of 66.88 mL/h by upstream flow, which is much closer to plugflow [16]. Then the loaded resin was rinsed with distilled water, and desorbed using 1 mol/L NaOH solution with a linear flow rate of 10.45 mm/min.
All samples were analyzed by inductively coupled plasma atomic emission spectroscopy (ICP-OES). The adsorption rate and separation factor of tungsten and molybdenum were calculated as follows:
 (2)
(2)
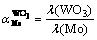 (3)
(3)
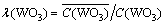 (4)
(4)
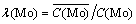 (5)
(5)
where C0 is initial concentration; C is instantaneous concentration; V is solution volume; 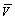 is resin volume;
is resin volume; 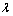 is distribution ratio;
is distribution ratio; 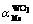 is separation coefficient of WO3 and Mo;
is separation coefficient of WO3 and Mo; 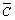 is concentrations in resin; C is concentration in solution.
is concentrations in resin; C is concentration in solution.
3 Results and discussion
3.1 Batch tests
3.1.1 Effect of pH value
pH directly affectes the existing forms of tungsten and molybdenum. The experimental results are shown in Fig. 1. From Fig. 1, as pH of solution decreases from 10 to 8.0, the absorption quantity of W exceeds a little that of Mo. So the separation factor is low. The reason is that W and Mo do not polymerize within this pH range [14]. While as pH decreases to 7.0, the adsorbed amount of WO3 increases rapidly, meanwhile the adsorption quantity of Mo changes slightly. The reason is that tungsten is transformed into HW6O215-, while dominant form of molybdenum is still monoions. D309 resin prefers to absorb the isopolytungstate than to absorb molybdate, so the separation factor significantly increases to 9.29. Continuing to lower pH value, tungsten and molybdenum are both present in the form of anionic polymerizations. The comparable adsorption capacities result in the small separation factor. As a conclusion, pH=7.0 should be selected in the following experiments.
Table 1 Characteristics of D309 ion exchange resin (obtained from manufacturer)

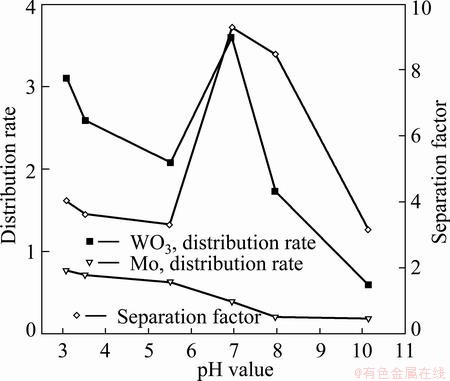
Fig. 1 Effect of pH value of solution on separation of tumgsten and molybdenum
3.1.2 Effect of contact time
The influence of contact time on adsorption rate was studied and the results are shown in Fig. 2.
It can be seen from Fig. 2, the adsorption rate of molybdenum rises rapidly within 1 h. The shorter equilibrium time attributes to the fast diffusion rate of the small-size MoO42- and a large number of available vacant adsorptive sites. Whereas, the equilibrium time for adsorbing tungsten on D309 resin is 4 h, due to the slow diffusion rate of the large-size heteropoly anions. So, the optimum contact time for the separation of tungsten and molybdenum is 4 h.
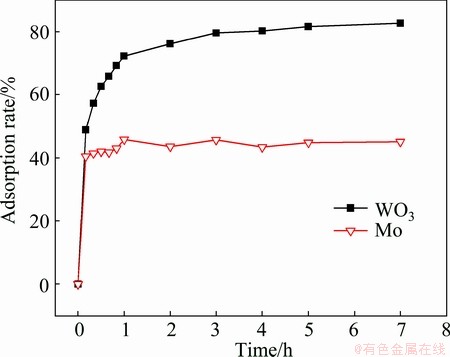
Fig. 2 Effect of contact time on separation of tungsten and molybdenum
3.2 Adsorption isotherm of D309 resin for absorbing tungsten and molybdenum
In the ion exchange process, the adsorption isotherm may reflect the ions equilibrium distribution between liquid and resin at a ceitain temperature. The adsorption data of binary system were simulated with multi-component models in the present study, which are all extended or modified from mono-component models mentioned earlier. For the sake of simplicity, in this research, Henry model, Langmuir model and Freundlich model were used to describe the equilibrium isotherm data. The mathematical expressions of the three models are described as follows:
Henry model:
Qe=KHCe (6)
Freundlic model:
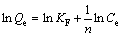 (7)
(7)
Langmuir model:
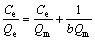 (8)
(8)
where KH is Henry model constant; Qe is equilibrium loading capacity of resin; Ce represents equilibrium concentration of solution; KF and n are Freundlich model constants relative to the sorption capacity and sorption intensity, respectively; Qm is saturated loading capacity; b is affinity parameter or Langmuir sorption constant.
These experiments were carried out as explained above and the result is shown in Fig. 3. Fitting the experimental results by above models respectively, the model with the largest correlation coefficient is the corresponding adsorption isotherm model.
As shown in Fig. 4, the adsorption of molybdenum belongs to Freundlic model: lnQ=1.08lnC+0.58. The Freundlich constant, KF, can be taken as a relative indicator of the adsorption capacity [16]. The 1/n value indicates the relative distribution of energy sites and depends on the nature and intensity of the adsorption process [17]. On the other hand, in view of R2 values, the Langmuir model exhibits a good fit to the experimental data of tungsten, indicating that the adsorption of tungsten may belong to Langmuir model.

Fig. 3 Relationship between equilibrium concentration of Mo (a), WO3 (b) and equilibrium loading capacity

Fig. 4 Fitting curves by Freundlich model for Mo (a) and Langmuir model for WO3 (b)
3.3 Adsorption kinetics of D309 resin
Macroporous ion exchange resins have porous structure having many micro-pores and macro-pores [18]. Hence, the ion exchange process occurs on the surface of the resin particles and the internal micro-particles simultaneously. So the control step in ion exchange always includes film diffusion (FDC), intra-particle diffusion (PDC) and chemical reaction. The mathematical expressions of the three models are described as follows:
Film diffusion: F=kt
Intra-particle diffusion: 1-3(1-F)2/3+2(1-F)=kt
Chemical reaction: 1-(1-F)1/3=kt
where F is adsorption rate; t is time; k is the apparent rate constant.
It can be seen from Fig. 5 that the reaction rate increases with the increase of reaction temperature. The experimental data were fitted by the each model introduced obove. Only PDC model is suitable and the fitting curves are shown in Fig. 6.
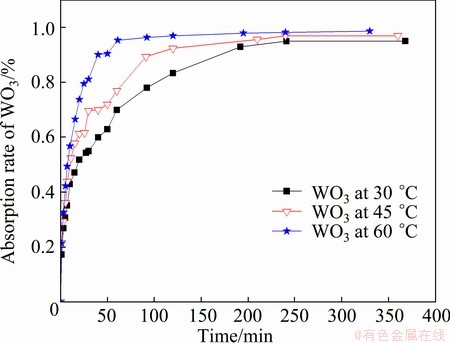
Fig. 5 Adsorption curves under different temperatures

Fig. 6 Fitting curves by PDC model
As shown in Fig. 6, 1-3(1-F)2/3+2(1-F) and t have a good linear relationship at different temperatures. Adsorption of tungsten onto D309 resin belongs to intra-particle diffusion control model. On the other hand, adsorption of molybdenum onto D309 resin reaches equilibrium quickly, so the useful experimental data are not obtained, but it can be concluded that D309 resin adsorbs molybdenum more easily than tungsten.
Based on the fitting line slope obtained from Fig. 6, the apparent rate constants k for the resin adsorption for tungsten under various temperatures are calulated. At 30, 45, and 60 ��C, the apparent rate constants are 5.44��10-3, 6.93��10-3 and 1.14��10-2 respectively. According to the Arrhenius formula: ln k=-E/(RT)+B, apparent activation energy can be calculated to be 20.56 kJ/mol.
3.4 Column tests
3.4.1 Loading test
In loading tests, the breakthrough point takes place when the concentrations of tungsten and molybdenum in effluent begin to increase predominantly.
Figure 7 illustrates the loading curves of tungsten and molybdenum on D309 resin. It can be seen that the breakthrough volume of molybdenum is 500 mL, while almost no tungsten presents in the effluent until the volume of liquid reaches 1070 mL. The solution mainly containing molybdenum and trace tungsten can be obtained from the effluent from 500 mL to 1070 mL, whose mass ratio of Mo to WO3 reaches 76 in contrast with the initial ratio of 0.414. The removal rate of tungsten reaches 95.14%. It can be concluded that D309 resin is suitable for the high-concentration solution.

Fig. 7 Loading curves
3.4.2 Stripping test
The D309 resin is weak base anion exchange resin with primary amine group (��CH2NH2), which implies that the loaded resin can be desorbed by NaOH solution easily.
As shown in Fig. 8, the eluate from 683 mL to 1273 mL has a mass ratio of WO3 to Mo 53.33 while the ratio of 2.41 in the initial solution. The saturated adsorption capacities of WO3 and Mo for the collected effluents are 289.40 mg/mL and 41.68 mg/mL respectively. The removal rate of molybdenum is 85.60%.
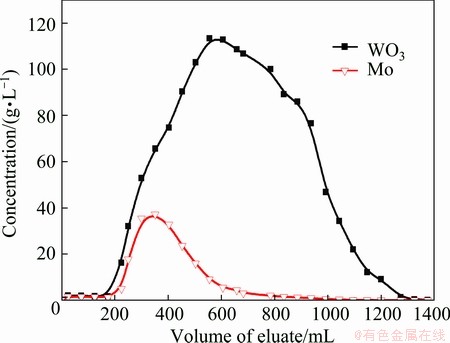
Fig. 8 Stripping curves for loaded D309 resin
4 Conclusions
1) Batch tests indicate that the optimum pH value for the separation is 7.0 and the optimum contact time is 4 h. In this case, the separation factor of W and Mo reachs 9.29.
2) The D309 resin absorbing tungsten and molybdenum belongs to Langmuir model and Freundlich model respectively, and the absorbing process for tungsten is controlled by intra-particle diffusion.
3) The column tests with a solution containing 70 g/L WO3 and 28.97 g/L Mo show that the effluent with a mass ratio of Mo to WO3 of 76 and the eluate with a mass ratio of WO3 to Mo of 53.33 can be obtained.
References
[1] WIBERG E, WIBERG N, HOLLEMAN A F. Inorganic chemistry [M]. San Diego: Academic Press, 2001: 1385-1386.
[2] HUO Guang-sheng, PENG Chao, SONG Qiong, LU Xiao-ying. Tungsten removal from molybdate solutions using ion exchange [J]. Hydrometallurgy, 2014, 147-148: 217-222.
[3] ZHAO Zhong-wei, CAO Cai-fang, CHEN Xing-yu. Separation of macro amounts of tungsten and molybdenum by precipitation with ferrous salt [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(12): 2758-2763.
[4] ZHAO Z W, XIAO L P. Study on removing Mo from tungstate solution by activatedcarbon loaded with copper [J]. Refract Metal and Hard Mater, 2010, 28: 503-507.
[5] HU Zhao-rui. The generation and development of tungsten ion exchange meltingprocess [J]. China Tungsten Industry, 1997(7): 41�C43. (in Chinese)
[6] KLIMENKO G L, BLOKHIN A A, GLEBOVSKIJ V G, ERMOLOV S N, MAJOROV D Y, KOPYRIN A A. Use of ion exchange method in production of high purity tungsten and molybdenum [J]. Metally, 2001(3): 49�C51.
[7] GUAN Wen-juan, ZHANG Gui-qing. Solvent extraction separation of molybdenum and tungsten from ammonium solution by H2O2-complexation [J]. Hydrometallurgy, 2012, 127�C128: 84�C90.
[8] LI Hong-gui. Production of high purity APT from scheelite and complex tungsten raw material with high Mo content [J]. Transactions of Nonferrous Metals Society of China, 2004, 14(2): 366�C369.
[9]  Ying, LI Hong-gui. Utilize tungsten concentrates of high molybdenum rationally [J]. China Tungsten Industry, 2005, 20(5): 15�C17. (in Chinese)
Ying, LI Hong-gui. Utilize tungsten concentrates of high molybdenum rationally [J]. China Tungsten Industry, 2005, 20(5): 15�C17. (in Chinese)
[10] ZHAO Zhong-wei, CAO Cai-fang, CHEN Xing-yu. Separation of macro amounts of tungsten and molybdenum by precipitation with ferrous salt [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(12): 2758�C2763.
[11] ZHAO Zhong-wei, ZHANG Jia-liang, CHEN Xin-yu. Separation of tungsten and molybdenum using macroporous resin: Equilibrium adsorption for single and binary systems [J]. Hydrometallurgy, 2013, 140: 120�C127.
[12] JIANG An-ren, JIANG Wei-zhong, PANG Zhen. Using the different ability of forming isopolyacid to separate tungsten and molybdenum [J]. Rare Metals and Cemented Carbides, 1989(2): 26�C29. (in Chinese)
[13] HUO Guang-sheng, SUN Pei-mei, JIA Gen-gui. On the possible methods for separating Mo from W [J]. Rare Metals and Cemented Carbides, 1997(1): 52�C56. (in Chinese)
[14] ZHANG Jia-liang, ZHAO Zhong-wei, CHEN Xing-yu, LIU Xu-heng. Thermodynamic analysis for separation of tungsten and molybdenum in W �CMo �CH2O system [J]. The Chinese Journal of Nonferrous Metals, 2013,23(5): 1463�C1470. (in Chinese)
[15] NING Peng-ge, CAO Hong-bin, ZHANG Yi. Selective extraction and deep removal of tungsten from sodium molybdate solution by primary amine N1923 [J]. Separation and Purification Technology, 2009, 70: 27�C33.
[16] AKSU Z. Equilibrium and kinetic modelling of cadmium (II) biosorption by C. vulgaris in a batch system: Effect of temperature [J]. Separation and Purification Technology, 2001, 21: 285-294.
[17] LAKSHMI U R, SRIVASTAVA V C, MALL I D, LATAYE D H. Rice husk ash as an effective adsorbent: evaluation of adsorptive characteristics for indigo carmine dye [J]. Journal of Environmental Management, 2009, 90: 710-720.
[18] LITTLEJOHN P, VAUGHAN J. Selectivity of commercial and novel mixed functionality cation exchange resins in mildly acidic sulfate and mixed sulfate-chloride solution [J]. Hydrometallurgy, 2012, 121-124: 90-99.
����D309��֬���ӽ�����������ٺ���
¬��ӱ�����������δ���
���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083
ժ Ҫ�������١�����ˮ��Һ�оۺ������IJ��죬���һ������D309��֬���ӽ��������١�����·�������̬ʵ������������١�������pHֵΪ7.0����ѽӴ�ʱ��Ϊ4 h���١������ϵ���ﵽ9.29���������������о�������D309��֬���١���������ֱ�����Langmuirģ�ͺ�Freundlichģ�͡���������ѧ�о�������D309��֬���ٵ�������������ɢ���ơ����ں�70 g/L WO3 �� 28.97 g/L Mo����Һ������̬���������١������������Һ��Mo��WO3 �������ȴ�76�����ý���Һ��WO3��Mo�������ȴ�53.33��
�ؼ��ʣ���Ũ��������Һ�����ӽ�������̬ʵ�飻��̬ʵ��
(Edited by Hua YANG)
Foundation item: Project (51174232) supported by the National Natural Science Foundation of China
Corresponding author: Guang-sheng HUO; Tel: +86-731-88830476; E-mail: gshuo@csu.edu.cn
DOI: 10.1016/S1003-6326(14)63438-5