J. Cent. South Univ. Technol. (2008) 15: 674-678
DOI: 10.1007/s11771-008-0125-5

Comparison of capacitive behavior of activated carbons with different pore structures in aqueous and nonaqueous systems
ZHOU Shao-yun(周邵云), LI Xin-hai(李新海), WANG Zhi-xing(王志兴),
GUO Hua-jun(郭华军), PENG Wen-jie(彭文杰)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: The pore structures of two activated carbons from sawdust with KOH activation and coconut-shell with steam activation for supercapacitor were analyzed by N2 adsorption method. The electrochemical properties of both activated carbons in 6 mol/L KOH solution and 1 mol/L Et4NPF4/PC were compared, and the effect of pore structure on the capacitance was investigated by cyclic voltammetry, AC impedance and charge-discharge measurements. The results indicate that the capacitance mainly depends on effective surface area, but the power property mainly depends on mesoporosity. At low specific current (1 A/g), the maximum specific capacitances of 276.3 F/g in aqueous system and 123.9 F/g in nonaqueous system can be obtained from sawdust activated carbon with a larger surface area of 1 808 m2/g, but at a high specific current, the specific capacitance of coconut-shell activated carbon with a higher mesoporosity of 75.1% is more excellent. Activated carbon by KOH activation is fitter for aqueous system and that by steam activation is fitter for nonaqueous system.
Key words: activated carbon; supercapacitor; aqueous system; nonaqueous system; capacitance
1 Introduction
Supercapacitor is a kind of new energy storage device between secondary battery and physical capacitor. As a new device with great application prospect, it has become a research hotspot in recent years for its friendliness to environment, high safety, good power property and cycling performance. At present, because of its stable electrochemical behavior, good cycling performance and low cost, activated carbon has become a mainstream electrode material for commercial supercapacitors. The storage of energy exists in the double layer on the electrode-electrolyte interface[1]. Specific surface area and pore structure are key physical property parameters. The pore structure of activated carbon depends on the solvated ionic radius of electrolytes[2]. The system with larger ions chooses microporous carbon, which will lead to low utility ratio of material and inappeasable power property; and mesoporous carbon is used for the system with smaller ions, which will be difficult to bring a high specific capacitance. SOFFER and KORESH et al[3-4] proposed that the pores with size larger than 0.5 nm can be utilized in KOH aqueous system. LIN et al[5] reported that the smallest pore size which can form double layer in H2SO4 system is 0.8 nm. ZULETA et al[6] detected the ionic diameters of Et4N+ and BF4- to be 0.68 and 0.45 nm, respectively. The popular conclusion is that if there are more mesopores wider than 2 nm, there will be more advantageous for forming double layer and high power property[7-8]. So it is very important to choose activated carbon with appropriate pore structure in different systems. In this work, the capacitive behaviors of two activated carbons with different pore structures by KOH and steam activation in KOH aqueous system and Et4NPF4/PC nonaqueous system were compared, so as to supply some references for choosing appropriate activated carbon materials in both systems.
2 Experimental
2.1 Test on physical properties of activated carbon
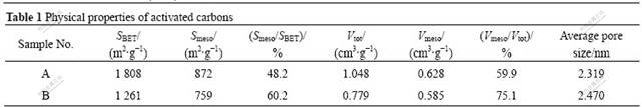
Activated carbons were provided by Huaxian Company (China, sawdust by KOH activation, marked as “sample A”) and Kuraray Chemistry Company (Japan, coconut shell by steam activation, marked as “sample B”). Their physical properties are listed in Table 1. Specific surface area and pore size distribution were tested by a N2 isothermal adsorption tester and calculated based on BET and BJH methods.
2.2 Preparation of electrode and assembly of coin- type capacitor
85% (mass fraction) activated carbon, 10% acetylene black, 5% polytetrafluoroethylene(PTFE),
Table 1 Physical properties of activated carbons
appropriate ethanol were mixed and stirred in the agate mortar until homogeneous black slurry was obtained, then the blended slurries were pressed onto foamed nickel (used in aqueous system) or stainless steel net (used in nonaqueous system) current collector and cut it out to prepare electrode with diameter of 14 mm. In the vacuum condition, electrode was dried at 120 ℃ for 6 h. The test capacitor consisted of two identical electrodes separated by a porous membrane, and 6 mol/L KOH so- lution or 1 mol/L Et4NPF4/PC as electrolyte, respectively. The assembly of the coin-type capacitors was carried out in atmosphere for aqueous system and in a dry Ar-filled glove box for nonaqueous system.
2.3 Test on electrochemical performance of super- capacitor
AC impedance (10 kHz-1 mHz), cyclic voltammetry (5 mV/s) and charge-discharge tests of supercapacitors were performed on a CHI660A electrochemical workstation.
3 Results and discussion
3.1 Pore structure analysis
Fig.1 shows N2 adsorption/desorption isotherms of samples A and B at 77.4 K. Both of the activated carbons are found to give typical type-Ⅰ nitrogen isotherms with an approximate smooth beeline at high relative pressure and rise very sharply to a high adsorptive volume at low relative pressure, which indicates that they both possess lots of micropores[9]. The desorption/desorption isotherms of sample B have a clear hysteresis loop, but those of

Fig.1 N2 adsorption/desorption isotherms of activated carbons
sample A have no at high relative pressure, which indicates sample B has more mesopores than sample A[10]; besides, the same conclusion can also be drawn according to Smeso/SBET and Vmeso/Vtot in Table 1. Judging from adsorptive volume and cumulative pore volume, the specific surface area of sample A is larger than that of sample B. Therefore, larger specific surface area of sample A is based on the precondition that it has more micropores.
Fig.2 shows the pore size distribution curves. For sample A, the pore size with maximal probability is 2.25 nm, and mesopores from 2 to 6 nm mainly distribute; and for sample B, besides the pores which are smaller than 1.95 nm, it has a focalization of pores at 3.75 nm. The average pore size of sample A is smaller than that of sample B. In Table 1, judging from Smeso/SBET and Vmeso/Vtot of samples A and B, the same result can also be achieved. Hence, sample B is more appropriate than sample A for the system with larger electrolyte ions, that is to say, activated carbon by KOH activation is fitter for aqueous system and that by steam activation is fitter for nonaqueous system.

Fig.2 Pore size distribution of activated carbons
3.2 Comparison of cyclic voltammetry behavior
Cyclic voltammetry curves of both activated carbons in KOH aqueous system and in Et4NPF4/PC nonaqueous system are shown in Fig.3. All of the curves do not show any major Faradic reactions within scan voltage window and have a typical capacitive character. According to the principle that specific capacitance keeps direct ratio with specific current at the same scan rate[11], sample A with larger specific surface area has bigger specific capacitance gained from the plateau current of rectangle pattern; however, the same relationship between specific capacitance and BET specific surface area does not exist and specific surface area increased does not bring proportionate specific capacitance. This result ascribes that micropores with ultrafine pore size are difficult to be utilized by electrolyte ions and are null surface areas. The ratio of specific capacitance of both activated carbons in KOH aqueous system is more approximate to the ratio of specific surface area than that in Et4NPF4/PC nonaqueous system because the specific surface area is utilized more adequately in KOH aqueous system which has smaller ions. Hence, the decreasing extent of plateau current of sample A from KOH aqueous system to Et4NPF4/PC nonaqueous system is larger than that of sample B. At the moment the scan direction changes, the more quickly the current responds, the better capacitance and power properties of activated carbon can be gained. Hysteresis current of sample B is smaller than that of sample A because electrolyte ions in micropores are difficult to reverse quickly owing to small diffusion path and large surface tension, which causes the current expressed hysterically when the scan direction changes[12].

Fig.3 Cyclic voltammetry curves of activated carbons
3.3 Comparison of AC impedance
For an ideal capacitor, the Nyquist plot would show a vertical line only coinciding with the vertical axis with the real part of Z being 0. For an ideal capacitor connected in series with a resistor, the vertical line in the Nyquist plot is shifted on the vertical axis by the amount of R. In practice, for a typical porous activated carbon electrode, generally, the Nyquist plot can be divided into three distinct frequency zones: zoneⅠis a semicircle in high frequency region, and the first intercept of semicircle shows the resistance of electrolyte, and the diameter of semicircle represents the resistance due to all the connections which mainly include the connections between the activated carbon and the current collector as well as the electrolyte; zone Ⅱ is medium frequency region, in this frequency range, an oblique line with approximate 45? slope expresses the diffused resistance and ascribes that the electrolyte ions reach electrode area deeper in the activated carbon pores with a longer path; zone Ⅲ is vertical line which is perpendicular to horizontal axis and shows pure capacitance behavior in the low frequency region[13]. Fig.4 shows AC impedance curves of activated carbons in both systems. In KOH aqueous system, there is no obvious semicircle which almost degenerates to a beeline, showing that the contact resistance is small. The difference is that plot of sample A extends relatively wide and shows a bigger diffused resistance than that of sample B in medium frequency region. In the low frequency region, plot of sample B is approximately perpendicular to the real axis, while sample A is difficult to express pure capacitance behavior, so double layer capacitance is also under the influence of frequency. Thus it can be concluded that if mesoporosity is larger, the capacitive behavior will be better. In Et4NPF4/PC nonaqueous system, both activated carbons have similar Nyquist plots with a clear semicircle in high frequency region. Compared with aqueous system, solution resistance and contact resistance increase remarkably as the consequence of the differences of ionic conductivity and ionic diameter of both electrolytes, the diffused resistance is larger in medium frequency region and the plots cannot show pure capacitance behavior in low frequency region. The experiments indicate the electrolytes with different ionic diameters require strict pore structure of activated carbon.
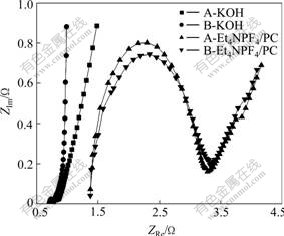
Fig.4 AC impedance curves of activated carbons
Fig.5 shows frequency―specific capacitance curves calculated by AC impedance[14]. Because of distributed capacitance effect and asymmetrical distribution of electrolyte resistance in pores, when the electrochemical system is given a high AC signal frequency, specific capacitance will be small because electrolyte ions can only form the double layer on the ektexine of electrode and do not reach the depth of activated carbon pores. Specific capacitance increases gradually along with lessening frequency, and electrolyte ions have enough time to penetrate into the micropores which are hidden deeply in the activated carbon, consequently, the utility ratio of materials is improved. When AC signal frequency falls to a fixed value called cut-off frequency, surface area will be utilized adequately and specific capacitance will become an invariable plateau. The higher the cut-off frequency is, the better the power property and the higher the capacitance retention at large specific current can be realized[13]. Relatively small mesoporosity decides that cut-off frequency of sample A is lower than that of sample B; and it is even difficult to reach the cut-off frequency in KOH aqueous system. Therefore, controlling mesoporosity is a crucial factor to balance specific capacitance and power property.

Fig.5 Frequency―specific capacitance curves of activated carbons
3.4 Comparison of cycling charge-discharge
Fig.6 shows charge-discharge curves of super- capacitor in both systems charged to 1.0 V and 2.7 V at 1 A/g, respectively. All of the charge-discharge curves are regular triangles, which indicates their typical capacitive characters. The specific capacitances of activated carbons are 276.3 and 157.8 F/g in aqueous system, 123.9 and 96.9 F/g in nonaqueous system, respectively. However, the charge time of sample A which has more micropores in KOH aqueous system is longer because sample A needs to conquer stronger infiltrating resistance and consume more energy when ions permeate into micropores. More remarkable potential saltations in nonaqueous system from charge and discharge switching show that PC system has larger resistance, and the equivalent serials resistances (ESR) of activated carbons are 2.421 and 1.486 Ω in aqueous system, 8.951 and 7.776 Ω in nonaqueous system, respectively.
Fig.7 shows specific capacitance at different specific current. Specific capacitance of sample A with larger specific surface area is larger at low specific current and falls quickly as increasing specific current because transfer velocity of ions increases and utility ratio of micropores decline[15]. In contrary, specific capacitance of sample B hardly decreases in aqueous system, mesopore structure makes K+ and OH- with small diameters transfer relatively uncontrolled at high specific current; in nonaqueous system, it also falls mildly and is even higher than that of sample A at the high specific current. Specific capacitances of both activated carbons decrease with the specific current to a greater extent in nonaqueous system. Than in aqueous system, thus it can be concluded that the system with bigger ions requires more mesopores.

Fig.6 Charge-discharge curves of supercapacitors at specific current of 1 A/g
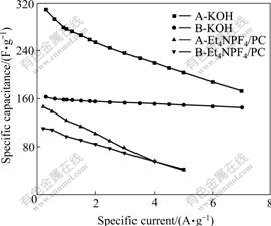
Fig.7 Relationship between specific capacitance and specific current
4 Conclusions
1) Specific capacitance mainly depends on effective surface area, but power property mainly depends on mesoporosity. The maximum specific capacitances of 276.3 F/g in aqueous system and 123.9 F/g in nonaqueous system can be obtained from sawdust activated carbon with a larger surface area of 1 808 m2/g at low specific current of  but the specific capacitance of coconut-shell activated carbon with a higher mesoporosity of 75.1% is more excellent at high specific current.
but the specific capacitance of coconut-shell activated carbon with a higher mesoporosity of 75.1% is more excellent at high specific current.
2) Activated carbon by KOH activation is fitter for aqueous system because of its larger specific surface area and that by steam activation is fitter for nonaqueous system because of its high mesoporosity.
References
[1] ZHENG J P, JOW T R. A new charge storage mechanism for electrochemical capacitors [J]. Journal of Electrochemical Society, 1995, 142(1): 6.
[2] SALITRA G, SOFFER A, ELIAD L, COHEN Y, AURBACH D. Carbon electrodes for double-layer capacitors (Ⅰ): Relations between ion and pore dimensions [J]. Journal of Electrochemical Society, 2000, 147(7): 2486-2493.
[3] SOFFER A, EOLAMN M. The electrical double layer of high surface porous carbon electrode [J]. Journal of Electroanalytical Chemistry, 1972, 38: 25-43.
[4] KORESH J, SOFFER A. Stereoselectivity in ion electroadsorption and in double-layer charging of molecular sieve carbon electrodes [J]. Journal of Electroanalytical Chemistry, 1983, 147: 223-234.
[5] LIN C, RITTER J A, POPOV B N. Correlation of double-layer capacitance with the pore structure of sol-gel derived carbon xerogels [J]. Journal of Electrochemical Society, 1999, 146(10): 3639-3643.
[6] ZULETA M, BJ?RNBOM P, LUNDBLAD A, LUST E, KASUK H, NURK G. Determination of diffusion coefficients of BF4- inside carbon nanopores using the single particle microelectrode technique [J]. Journal of Electroanalytical Chemistry, 2006, 586(2): 247-259.
[7] ZHAO Jia-chang, LAI Chun-yan, DAI Yang, XIE Jing-ying. Synthesis of mesoporous carbon as electrode material for supercapacitor by modified template method [J]. Journal of Central South University of Technology, 2005, 12(6): 647-652.
[8] ZHOU Shao-yun, LI Xin-hai, WANG Zhi-xing, GUO Hua-jun, PENG Wen-jie. Effect of activated carbon and electrolyte on properties of supercapacitor [J]. Trans Nonferrous Met Soc China, 2007, 17(6): 1328-1333.
[9] LI Jing, LI Jie, LAI Yan-qing, SONG Hai-sheng, ZHANG Zhi-an, LIU Ye-xiang. Influence of KOH activation techniques on pore structure and electrochemical property of carbon electrode materials [J]. Journal of Central South University of Technology, 2006, 13(4): 360-366.
[10] GREGG S J, SING K S W. Adsorption, surface area and porosity [M]. London: Academic Press, 1982.
[11] ?LVAREZ S, BLANCO-L?PEZ M C, MIRANDA-ORDIERES A J, FUERTES A B, CENTENO T A. Electrochemical capacitor performance of mesoporous carbons obtained by templating technique [J]. Carbon, 2005, 43(4): 866-870.
[12] SHOU Zhao-hui. Preparation and EDLC application of mesoporous carbons with high specific surface [D]. Changsha: Central South University, 2004. (in Chinese)
[13] K?TZ R, CARLEN M. Principles and applications of electrochemical capacitors [J]. Electrochimica Acta, 2000, 45: 2483-2498.
[14] NIAN Y R, TENG H. Influence of surface oxides on the impedance behavior of carbon-based electrochemical capacitors [J]. Journal of Electroanalytical Chemistry, 2003, 540(2): 119-127.
[15] GAMBY J, TABETNA P L, SIMON P, FAUVARQUE J F, CHESNEAU M. Studies and characterizations of various activated carbons used for carbon/carbon supercapacitors [J]. Journal of Power Sources, 2001, 101(1): 109-116.
(Edited by YANG Hua)
Foundation item: Project(2007BAE12B01-2) supported by the National Key Project Scientific and Technical Supporting Programs Funded by Ministry of Science and Technology of China
Received date: 2008-01-25; Accepted date: 2008-04-24
Corresponding author: LI Xin-hai, Professor, PhD; Tel: +86-731-8836633; E-mail: xhli@mail.csu.edu.cn