J. Cent. South Univ. Technol. (2007)05-0601-06
DOI: 10.1007/s11771-007-0115-z 
Preparation and properties of pitch carbon based supercapacitor
LIU Ye-xiang(��ҵ��), LI Jing(�� ��), LAI Yan-qing(������),
SONG Hai-sheng(�κ���), ZHANG Zhi-an(���ΰ�)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: Using the mesophase pitch as precursor, KOH and CO2 as activated agents, the activated carbon electrode material was fabricated by physical-chemical combined activated technique for supercapacitor. The influence of activated process on the pore structure of activated carbon was analyzed and 14 F supercapacitor with working voltage of 2.5 V was prepared. The charge and discharge behaviors, the properties of cyclic voltammetry, specific capacitance, equivalent serials resistance (ESR), cycle properties, and temperature properties of prepared supercapacitor were examined. The cyclic voltammetry curve results indicate that the carbon based supercapacitor using the self-made activated carbon as electrode materials shows the desired capacitance properties. In 1 mol/L Et4NBF4/AN electrolyte, the capacitance and ESR of the supercapacitor are 14.7 F and 60 m��, respectively. The specific capacitance of activated carbon electrode materials is 99.6 F/g; its energy density can reach 2.96 W?h/kg under the large current discharge condition. There is no obvious capacitance decay that can be observed after 5000 cycles. The leakage current is below 0.2 mA after keeping the voltage at 2.5 V for 1 h. Meanwhile, the supercapacitor shows desired temperature property; it can be operated normally in the temperature ranging from -40 �� to 70 ��.
Key words: carbon based supercapacitor; physical-chemical combined activation; specific capacitance; equivalent serials resistance; temperature property
1 Introduction
Supercapacitor is a kind of new energy storage device, which can fill the gap between the conventional capacitor and the battery[1-3]. Supercapacitor has many advantages, such as good pulse charge and discharge character, rapid discharge ability, long life span, no environment pollution[4-5]. Supercapacitors are now utilized in many fields, such as space industry, national defense, war industry, electrical vehicle, wireless communication, and consumptive electronics. It is estimated that the total market value would be about one billion by 2015, in the commercial, industrial and automotive applications[6].
According to the energy storage principle, supercapacitor can be categorized into carbon based supercapacitors that store energy by electric double layer (Helmholtz layer) and the metal-oxide based and the polymer based supercapacitors that store energy by pseudo capacitance[7-10]. Carbon based supercapacitor is also called electric double layer supercapacitor(EDLC).
There are many types of carbon materials that can be used for carbon based supercapacitor, such as carbon aero gel[11], carbon nanotube[12], carbon fiber[13], activated carbon powder[14]. The attributes of carbon (surface area, pore structure and specific capacitance) are the key factors to determine the properties (capacitance, equivalent serials resistance(ESR) and energy density, power density) of carbon based supercapacitor. Because activated carbon electrode materials enjoy many advantages of the stable electrochemical properties, wide electrochemical window, perfect cycle property and lower cost, activated carbon is now the main commercially utilized supercapacitor electrode material.
In this experiment, low-cost mesophase pitch was selected as raw materials and physical-chemical combined activated technique was utilized to fabricate the activated carbon powder that has both the high surface area and ideal pore structure. This activated carbon powder was used as electrode material and activated electrode was prepared. Meanwhile, 1 mol/L Et4NBF4 was used as electrolyte and cylindrical type supercapacitor was also prepared, the size of prepared supercapacitor was d12.5 mm��28 mm. Properties of prepared supercapacitor, including capacitance properties, capacitance, ESR, current leakage, cycle property, discharge at different power density, temperature properties were discussed.
2 Experimental
2.1 Preparation of activated carbon electrode materials
When coal tar-pitch (soft point 83 ��, provided by Wuhan Ferrous Company) was carbonized at 500 ��, the easily emitted compound with small molecule was removed and the mesophase pitch was obtained. The mesophase pitch was ground and sieved in order to collect the grains whose average size was in the range of 100-200 ��m. Sieved mesophase pitch was mixed with KOH (analytical grade, provided by Shanghai Rreagent Factory ) in nickel crucible, and the mass ratio of alkali to carbon was 3?1. In the activation process, the samples were first heated at 800 �� in argon atmosphere��and argon flow was kept at 60 mL/min, the heating rate was 5 ��/min, and heating time was 1 h. After this process, CO2 was led into and the time of combined activated process was 2 h. After activated process, the activated carbon was washed first by l mol/L HCl, then distilled water was used to wash it until its pH value reached 6.5. After rinsing process, the activated carbon was dried in an oven at 110 �� for 10 h. The characterization of the porous texture of the activated carbons was conducted using physical adsorption of N2 at 77 K in the Autosorb-6 apparatus. The morphologies of active carbon powder and electrode were examined by JSM-5600LV scanning electron microscope.
2.2 Preparation of activated carbon electrode and cylindrical type supercapacitor
80%(mass fraction) self-made active carbon, 10 % acetylene black, 10 % adherent reagent were mixed and stirred in the agate mortar until homogeneous black slurry was achieved, which then was spread out onto a aluminum film of 30 ��m. In the vacuum condition, electrode film was dried at 100 �� for 10 h; the roller was then used to roll the electrode film until its thickness was less than 190 ��m; and the lead wire was dot welted on the electrode films. Two electrode films were separated by a separator, and the three films were wrapped in the certain wrapping machine. The wrapped electrode films were put into electrolyte in the inert atmosphere and were put onto rubber packing. At last the whole was put into aluminum case with the size of d12.5 mm��28 mm, and sealed in the self-made sealer.
2.3 Examination of pitch carbon based super- capacitor
The specific capacitance, ESR, the galvanostatic measurement and the cyclic voltammeter(CV) measurements were performed in the 273A EG&G Princeton Applied Research Potentiosat. As shown in Eqns.(1) and (2), the ESR was calculated according to voltage drop of the charge and discharge curve. Specific capacitance of activated carbon was calculated by E(t) slope[15]:
R����E/I (1)
C��(2It)/(m��E) (2)
where I is the discharge current, t is the discharge time, m is the mass of carbon on a electrode, and ��E is the voltage drop in discharge, excluding the portion of IR drop; the factor of ��2�� comes from the fact that the total capacitance measured from the test cells is the addition of two equivalent single-electrode capacitances.
3 Results and discussion
3.1 Influence of activated technique on physical property of activated carbon
The activated methods can be described as physical activation (CO2, H2O used as activated regent) and chemical activation (KOH, NaOH, ZnCl2 used as activated regent). The activated effect was determined by the method of activation, the attributes of precursor and the concrete activated techniques, such as activated temperature, the rate of heating, ratio of activated regent, and so on. Chemical activation, especially KOH used as activated regent, has the advantage of low activated temperature and high activated efficiency[16-17]. But when KOH was used as activated regent, the pore structure was formed by the corrosion of micro-graphic layer, and the amount of microspore of activated carbon was high and it was difficult to control the pore structure. When the physical-chemical activated technique was used at first, the graphic layer in carbon precursor was permeated into by melted KOH and a great amount of microspores were formed. When CO2 was led in the activated process, the microspore on the surface may have the priority to be activated and widened, because in the CO2 activated process, the pore structure was formed by the oxidation of active point of precursor, in the pore widening process, the mesopore may be formed, and the tendency of microspore formation may be wakened. Meanwhile, KOH on the surface of carbon might prevent the pore structure from collapsing. Compared with the KOH activated process, the physical-chemical combined technique might effectively enhance the amount of mesopores. The physical parameters of activated carbon are listed in Table 1, where the surface area of activated carbon is 1 959 m2/g, pore volume is 1.059 cm3/g, and volume fraction of mesopore is 19.96%, average pore diameter is 2.224 nm.
Fig.1 shows the N2 adsorption isotherms of the activated carbons prepared by physical-chemical combined technique. It can be seen from Fig.1 that the N2 adsorption isotherms of prepared activated carbon are almost type I according to BET classification, which indicates that microspores occupy most proportion of the activated carbon pore structure. Meanwhile, it can also be seen that the plateau has the shape of a small gradual increase along the pressure axis, which indicates that this activated carbon has a significant mesopore volume.
Table 1 Physical parameters of activated carbon
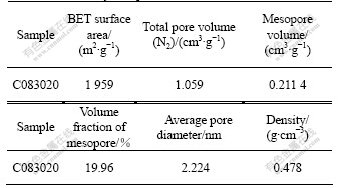

Fig.1 Isotherms of activated carbon
Fig.2 shows the pore size variation of microspores for the prepared activated carbon. It can be seen from Fig.2 that there is a pore diameter concentration plateau between 1.250 and 1.559 nm, which indicates the activated carbons with larger proportion of large microspore.

Fig.2 Microspore distribution of activated carbon
Fig.3(a) and Fig.3(b) show SEM images of the activated carbon powder and the electrode of activated carbon, respectively. It can be found that the activated carbon has the irregular shape; the diameter of activated carbon ranges from several ��m to several tens ��m.

Fig.3 SEM images of activated carbon powder(a) and carbon electrode(b)
There is no obvious pore structure to be found on the surface of activated carbon, which indicates that there is large proportion of micropore in the activated carbon. It can also be seen from SEM image of activated carbon electrode that the activated carbon powder is homogeneously mixed with acetylene black to form the net-work structure, and there are obvious pores between carbon particles, which may contribute to the accessibility of electrolyte.
3.2 Cyclic voltammograms examination of pitch carbon based supercapacitor
Fig.4 shows the cyclic voltammograms(CV) property of pitch carbon supercapacitor at different scan rates ranging from 2 to 20 mV/s, and the voltage window range of CV curve is from 0 to 2.5 V. According to the CV curve, the reversibility of the supercapacitor electrode can be analyzed. For ideal supercapacitor, its CV curve may show rectangle shape, but in the real examination process, the electrolyte ion may be prevented by the migration force and the polarized resistance is produced to make the CV curve of supercapacitor a little difference from the ideal rectangle. It can be seen from Fig.4(a) that the CV curves of supercapacitor show the regular rectangle-like shape, and there is no obvious oxidation or reduction peak in these curves, indicating that the energy is stored on the basis of the electric double layer. Fig.4(b) shows the relationship of peak current and scan rate, from which it can be seen that on the stable electrochemical window of 0-2.5 V, with the increase of scan rate, the peak current increases linearly, which indicates that the scan rate has no influence on the capacitance of carbon electrode, and the supercapacitor shows the ideal reversibility and the capacitance behavior.

Fig.4 CV properties of pitch carbon supercapacitor at different scan rates
(a) CV curves at different scan rates; (b) Relationship between peak current(Ip) and scan rate
3.3 Charge and discharge process of pitch carbon based supercapacitor
Fig.5 shows the charge and discharge curve of pitch carbon supercapacitor at the current of 0.6 A, and the maximum voltage is 2.5 V. It can be seen from Fig.5 that the voltage may change linearly with the change of charging time on the condition of keeping the current stable, which indicates that the reaction on the surface is mainly reaction of the movement of ion. The mirror-like curve indicates the ideal capacitance property of prepared supercapacitor, which is in accordance with the result observed from the CV curve, and the capacitance of carbon-based supercapacitor calculated by the slope of the curve is 14.7 F, and the ESR is 60 m��, the specific capacitance of activated carbon is 99.6 F/g.

Fig.5 Charge and discharge curve of pitch carbon supercapacitor
Fig.6 shows the discharge property of pitch carbon supercapacitor at different current densities. It can be seen from Fig.6(a) that the curves of the discharge still show the obvious linear shape when the current increases by eight times, which indicates that the increase of current dose not cause the polarized phenomenon of electrode. Meanwhile, as shown in Fig.6(b), when the discharge current increases from 0.2 to 1.6 A, there is no obvious capacitance decay. The energy density of this supercapacitor is 2.96 W?h/kg at the current of 1.6 A, which indicates that the supercapacitor has an ideal power property and is suitable for the large current discharge process.

Fig.6 Discharge properties of pitch carbon supercapacitor at different current densities
(a) Discharge curve at different current densities; (b) Capacitance decay at different charge current
3.4 Examination of cycle property and current leakage of pitch carbon based supercapacitor
Fig.7 illustrates the charge and discharge curve of activated carbon electrode over 5000 cycles, and the charge and discharge current is 200 mA. In the first 500 cycles, the specific capacitance of active carbon electrode shows a little decay behavior. After 500 cycles, the decay rate of specific capacitance is very slow. Compared with the capacitance of 500th cycle, the capacitance decay rate is less than 3% because energy, different from the rechargeable lithium ion battery, is stored by electric double layer. There is not the intercalation of ion into the electrode materials that may cause the volume change, the supercapacitor has the advantage of desired cycle property.

Fig.7 Charge and discharge curve of activated carbon electrode
Leakage current is an important parameter to analyze the property of supercapacitor. There are many reasons for the current leakage: the reaction of impurity ion on the electrode, the micro-contact of the electrode, and the diffusion of electrolyte ion on the electrode surface because of the concentration gradient. Fig.8 shows the curve of leakage current at 2.5 V, from which it can be seen that the current leakage is very low and the current leakage value is less than 0.2 mA after keeping the voltage at 2.5 V for 1 h.

Fig.8 Examination of leakage current of supercapacitor
3.5 Examination of temperature property of pitch carbon based supercapacitor
Temperature property is also an important property for supercapacitor. In the operating process of supercapacitor, the environment temperature may vary greatly, which requires that the supercapacitor has the good electric property in the different temperature conditions. Fig.9 shows the change of capacitance and ESR of pitch carbon supercapacitor with the variation of temperature. It can be seen from Fig.9(a) that the capacitance keeps stable in the range from -30 �� to 50 ��. The capacitance has the tendency to decay when the temperature rise to 70 ��. This is because with the increase of temperature, the electrolyte ion of electric double layer has the tendency to move from the electric double layer. For the same reason, at the low temperature, because the heating movement becomes slow, and the electric double layer is relatively stable, the capacitance of pitch carbon supercapacitor at low temperature is relatively high.

Fig.9 Temperature properties of pitch carbon supercapacitor
(a) Specific capacitance as function of temperature;(b)Equivalent series resistance as function of temperature
It can be seen from Fig.9(b) that the ESR decreases with the increase of the temperature in the temperature range from -40 �� to 25 �� because the movement of ion may speed up at high temperature and the polarized resistance of electrode is reduced. But with the further increase of temperature, the heating movement of electrolyte ion in disorder direction may influence the effective movement of electrolyte ion under the force of the electric field, and reduce the effect of low ion polarized resistance caused by enhancing ion movement rate. Therefore, in the temperature ranging from 25 �� to 70 ��, the ESR of pitch carbon supercapacitor decreases a little with the increase of temperature. But in the temperature ranging from -40 to 70 ��, the pitch carbon supercapacitor shows obvious capacitance behavior, the shape of supercapacitor is not changed, and there is also no damage or electrolyte leakage phenomenon during the cycle process.
4 Conclusions
1) Using the mesophase pitch as precursor, KOH and CO2 as activated agent, the activated carbon electrode material was fabricated by physical-chemical combined activated technique for supercapacitor. The carbon materials have the high surface area and ideal pore structure, which is suitable for engineering preparation of supercapacitor.
2) Using the pitch carbon as electrode material, the cylinder supercapacitor was prepared, and the size of case is d12.5 mm��28 mm, the capacitance of supercapacitor is 14.7 F in the electrolyte of BF4(CN)4/AN, and ESR is 60 m��. The specific capacitance of activated carbon material is 99.6 F/g. and the prepared pitch carbon supercapacitor shows ideal capacitance behavior.
3) In the current ranging from 0.2 to 1.6 A, pitch carbon supercapacitor shows ideal power discharge behavior, and its capacitance is almost unchanged, the energy density of this supercapacitor is 2.96 W?h/kg at the current of 1.6 A. The current leakage is less than 0.2 mA after keeping stable voltage of 2.5 V for 1 h. After 5000 cycles, the capacitance decay is less than 3%, compared with the capacitance of 500th cycle.
4) The prepared supercapacitor shows ideal temperature properties, which can be operated at the temperature range of -40 to 70 ��. The capacitance of supercapacitor may increase with the increase of temperature, and the ESR is relatively high at low temperature.
References
[1] KOTZ R, CARLEN M. Principle and application of electrochemical capacitors[J]. Electrochimica Acta, 2000, 54(11): 2483-2498.
[2] BONNEFOI L, SIMON P, FAUVARQUE J F, et al. Electrode optimisation for carbon power supercapacitors[J]. Power Source, 1999, 87(2): 1113-1119.
[3] WANG Xiao-feng, RUAN Dian-bo, WANG Da-zhi. Hybrid electrochemical supercapacitors based on polyaniline and activated carbon electrodes[J]. Acta Phys Chim Sin, 2005, 21(3): 261-266.
[4] LAI Yan-qing, LI Jing, LI Jie, et al. Preparation and electrochemical characterization of C/PANI composite electrode materials[J]. Journal of Central South University of Technology, 2006, 13(4): 353-359.
[5] LI Jing, LI Jie, LAI Yan-qing, et al. Influence of KOH activation techniques on pore structure and electrochemical property of carbon electrode materials[J]. Journal of Central South University of Technology, 2006, 13(4): 360-366.
[6] VIX-GUTERL C, SAADALLAH S. supercapacitor electrodes from new ordered porous carbon materials obtained by a template procedure[J]. Materials Science and Engineering B, 2004, B108(2): 148-155.
[7] WANG Yong-gang, ZHANG Xiao-gang. Preparation and electrochemical capacitance of RuO2/TiO2 nanotubes composites[J]. Electrochemical Acta, 2004, 49(12): 1957-1962.
[8] PEDRO G R, MALGORZATA C, KARINA C G, et al. Hybrid organic�Cinorganic nanocomposite materials for application in solid-state electrochemical supercapacitors[J]. Electrochemistry Communications, 2003, 5(2): 149-153.
[9] CONWAY B F. Transition from ��supercapacitor�� to ��battery�� behavior in electrochemical energy storage[J]. Journal of Electrochemical Society, 1991, 6(1): 1439-1448.
[10] PASSERINI V, VIDAKOVIC T, DEKANSKI A, et al. The properties of carbon�Csupported hydrous ruthenium oxide obtained from RuOxHy sol[J]. Electrochimica Acta, 2003, 48(25/26): 3805-3813.
[11] LI Wen-cui, REICHENAUER G, FRACKE J, et al. Carbon aerogel derived from cresol-resorcinol-formaldehyde for supercapacitor[J]. Carbon, 2002, 40(12): 2955-2959.
[12] FRACKOWIAK E, JUREWICZ K, DELPEUX S, et al. Nanotubular materials for supercapacitor[J]. Journal of Power Source, 2001, 98(1): 822-825.
[13] KIM Y J, HORIE Y, MATSUZAWA Y, et al. Structure features necessary to obtain a high specific capacitance in electric double layer capacitor[J]. Carbon, 2004, 42(12/13): 2423-2432.
[14] QU De-yang. Studies of the activated carbon used in double-layer supercapacitors[J]. Journal of Power Source, 2002, 109(1): 403-411.
[15] PORTET C, TABERNA P L, SIMON P, et al. Modification of Al current collector surface by sol-gel deposit for carbon-carbon supercapacitor applications[J]. Electrochimica Acta, 2004, 49(9): 905-912.
[16] LOZANO-CASTELL? D, LILLO-R?DENAS M A, CAZORLA-AMOR?S D, et al. Preparation of activated carbons from Spanish anthracite ��: Activation by KOH[J]. Carbon, 2001, 39(5): 741-749.
[17] LILLO M A, JUAN-JUAN J, CAZORLA-AMOR?S D, et al. About reaction occurring during chemical activation with hydroxides[J]. Carbon, 2004, 42(7): 1365-1369.
Foundation item: Project(2007BAE12800) supported by the National Supported Plan for Science and Technology; Project(06FJ4059) supported by the Hunan Provincial Academician Foundation
Received date: 2007-03-12; Accepted date: 2007-05-20
Corresponding author: LI Jing, Doctoral candidate; Tel:+86-731-8830474; E-mail:csu_lijing@126.com
(Edited by YANG Hua)