
Synthesis and properties of bulk nanocrystalline Mg2Si through
ball-milling and reactive hot-pressing
XIONG Wei(熊 伟), QIN Xiao-ying(秦晓英), KONG Ming-guang(孔明光), CHEN Li(陈 莉)
Key Laboratory of Materials Physics, Institute of Solid State Physics, Chinese Academy of Sciences,Hefei 230031, China
Received 9 November 2005; accepted 20 June 2006
Abstract: Bulk nanocrystalline Mg2Si(n-Mg2Si) was prepared by mechanical milling and reactive hot-pressing in vacuum and its mechanical properties were characterized. The results indicate that the grain sizes of Mg and Si decrease non-monotonously with increasing ball-to-powder mass ratio(BPR). The content of Mg2Si phase increases with increasing milling time; however, pure Mg2Si phase cannot be obtained by direct ball milling. Moreover, monolithic is Mg2Si phase can only be obtained when as much as 10% of excess Mg was blended in the raw mixtures. The relative density (D) and fracture toughness as well as hardness of bulk Mg2Si samples can be raised effectively by increasing the pressure in hot-pressing. After hot-pressing at 400 ℃ for 60 min under 1.5 GPa, highly dense(D>98%) n-Mg2Si (d=60 nm) was obtained, whose fracture toughness (KIC≈1.46 MPa・m1/2) and Vickers hardness (HV≈ 4.38 Gpa) are superior to those of conventional coarse-grained Mg2Si.
Key words: Mg2Si; nanocrystalline; ball-milling; hot-pressing; mechanical propert
1 Introduction
The intermetallic compound Mg2Si is useful as strengthening phase in the metal matrix composites (MMC) due to its low density (1.99 g/cm3) and high strength-to-mass ratio[1-5]. It is also promising as a basic material for thermoelectric energy conversion[6-8]. The phase purity and microstructure of the product Mg2Si is difficult to control by conventional melting technique because of the great discrepancy of melting point between Mg and Si and the high vapor pressure of Mg at the silicon melting point (1 412 ℃). For in-situ synthesis of monolithic Mg2Si from the elemental mixtures of magnesium and silicon powder and refining the grain size of Mg2Si to nano-scale, the mechanical alloying(MA) technique is often employed in the previous decade[9-13]. However, it is usually difficult or even impossible to synthesize Mg2Si thoroughly by MA technique with an ordinary planetary ball mill, and a large quantity of impurities, such as iron, cannot be avoidable.
From the thermodynamic point of view, Mg2Si can be formed through solid-state reaction between Mg and Si because of its minus free energy. Consequently, we attempted to produce nanocrystalline Mg2Si bulk by a two-step process consisting of ball milling and reactive hot-pressing. The milling process is performed to intimately mix the elements and refine the grain sizes of Mg and Si. The subsequent low-temperature hot-pressing is utilized to accelerate the formation Mg2Si phase and to obtain Mg2Si bulk. In this paper the influences of preparation parameters on the composition, micro- structure and mechanical properties of n-Mg2Si bulk are investigated.
2 Experimental
The initial materials were elemental Mg (99%, 150 μm) and Si (99%, 150 μm) powders, which were blended in a stoichiometric proportion of Mg2Si or over- stoichiometrically with some surplus Mg. After blending, the powder mixture was sealed into a stainless-steel vial (since ball milling in vacuum is necessary for active element Mg, stainless steel instead of ceramics vial was utilized in present work) together with process controlling agent of ball-milling(PCA), argon gas (99.999%) and hardened-steel balls of 19 and 10 mm in diameter (big-to-small ball ratio 1:4). The milling was performed in a planetary-type ball mill at a speed of 250 r/min. Then, the as-milled powders were conveyed to a tungsten carbide(WC) mould and hot- pressed in vacuum. The resulting specimens have cylindrical shape with a diameter of 14 mm and a thickness of 4-5 mm.
The microstructures and morphology of the synthesized powders and bulks were characterized by X-ray diffraction(XRD) on a diffractometer (Philips- PW1700) with Co Kα radiation, field-emission scanning microscopy(FE-SEM) and energy dispersive spectro- scopy(EDS). Mean grain sizes of the obtained samples were evaluated based on Scherrer formula with the strongest peaks of Mg2Si(220), Mg(101) and Si(111), respectively. Densities of Mg2Si bulk specimens were measured by Archimedes principle.
The Vickers hardness of Mg2Si bulk was determined by indentation method. The loading mass is 100 g and the loading time is 20 s. The corresponding indentation fracture toughness of Mg2Si bulk was estimated roughly from[14]
KIC=0.016(E/H)1/2P/c3/2 (1)
where c is the half-length of the resultant indentation crack, and E and H are the elastic modulus (120 GPa[9]) and the Vickers hardness of Mg2Si bulk, respectively. Each datum point was calculated based on six indentation measurements.
3 Results and discussion
3.1 Influence of ball-to-powder mass ratio(BPR) on grain sizes of Mg-Si powders
It is well known that the ball-to-powder mass ratio (BPR) plays very important roles in controlling the microstructures of the milled powders, such as the particle size distribution, the degree of disorder and the final stoichiometry. Fig.1 shows the dependence of mean grain sizes of Mg and Si on BPR after milling for 25 h. It can be seen that the mean grain sizes of Mg and Si decrease non-monotonously with increasing BPR. Instead, the plots of mean grain sizes of Mg and Si versus BPR have wavy shape (periodicity-like structure) with two valleys located at BPR 20 and 50 when BPR increases from 10 to 60. The mean grain sizes of Mg and Si at the first valley are 58 and 70 nm respectively, and 26 and 43 nm respectively at the second valley, which indicates that BPR 20 and BPR 50 are preferable in the present system.
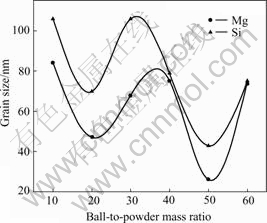
Fig.1 Dependence of mean grain sizes of Mg and Si on ball-to-powder mass ratio(BPR)
Usually at a high BPR, because of increase in the mass proportion of the balls, the number of collisions per unit time increases and consequently more energy is transferred to the powder particles leading to fast refinement microstructure[15]. However, the present result shows the grain size of Mg (or Si) dose not decrease monotonously with increasing BPR. This phenomenon may reflect that the effective collision number (the collision of milling balls with powders) would decrease corresponding to some specific BPR (here BPR=30 or BPR>50), leading to the appearance of non-monotonous behavior.
3.2 Influence of milling time on formation of Mg2Si phase
Fig.2 shows the XRD patterns of Mg-Si mixtures ball-milled for 7, 18, 25, 40 and 50 h. After 7 h and 18 h milling, as shown in Figs.2(a) and (b), the diffraction peaks of Mg and Si are still sharp and the diffraction peaks of Mg2Si are invisible, which implies that ball-milling in these durations only cause a mechanical mixing and cracking of Mg-Si powders. With the increase of milling time, as shown in Figs.2(c), (d) and (e), the diffraction peaks of Mg ((101) peak at 42?) and Si ((111) peak at 33?) are broadened and decreased evidently due to mechanically-induced lattice strain and crystallite size reduction. At the same time, the Mg2Si phase ((220) peak at 46?) appears and becomes much sharper, indicating the solid-state reaction of Mg to Si and formation of Mg2Si phase.
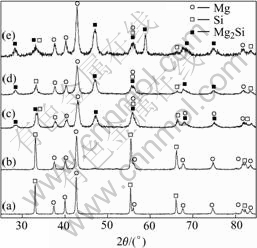
Fig.2 XRD patterns of Mg-Si powders ball-milled for different times: (a) 7 h; (b) 18 h; (c) 25 h; (d) 40 h; (e) 50 h
However, prolonging the milling time leads the reduction of grain size to become slow and results in the monotonous increase of iron impurity. As shown in Fig.3, iron (Fe) concentration increases from 0.63% to 4.84% (mass fraction) when milling time is prolonged from 7 h to 50 h. From the viewpoint of refining grain sizes and reducing the impurity content, the milling time of 25 h is an appropriate choice. Under this condition the obtained mean grain sizes of Mg2Si, Mg and Si are approximately 11 nm, 26 nm and 43 nm, respectively.
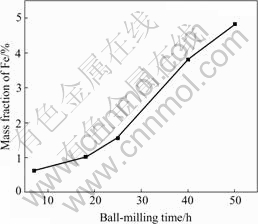
Fig.3 Dependence of concentration of impurity Fe on ball- milling time
3.3 Influence of Mg content on phase purity of obtained bulk samples
Because of the high volatility of Mg, it is difficult to obtain monolithic Mg2Si by hot-pressing the milled Mg-Si powders blended according to the stoichiometric composition of Mg2Si, as shown in Fig.4(a). It can be found that there are some weak peaks of Si in the pattern. Similarly, trivial Si phase is still present in the bulk specimen obtained by hot-pressing the ball-milled powder with excess 5% Mg (Fig.4(b)). However, as blended powders contain up to 10% excess Mg, the obtained bulk specimen is monolithic Mg2Si phase without Mg, Si or other impurity phase, as shown in Fig.4(c).
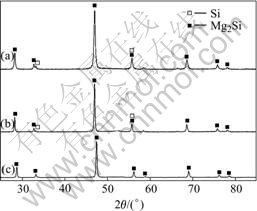
Fig.4 XRD patterns of bulk specimens for different Mg-to-Si ratio in initial Mg-Si mixed powders: (a) Stoichiometrically; (b) 5% excess Mg; (c) 10% excess Mg
To obtain monolithic Mg2Si the loss of element Mg, which mainly takes place in the ball-milling process and the heating process of hot-pressing, had to be considered. As compared with Si, Mg is plastic, easy to cold-weld, and adhesive to the vial and balls during the ball-milling process, leading to the loss of Mg. While in the heating process of hot-pressing in vacuum, some Mg can be volatilized from the interstices of the WC mould because of comparatively great vapor pressure of Mg element. It is found that the amount of Mg loss varies with the variation of the technique parameters of ball-milling and hot-pressing. Consequently, the Mg content should be adjusted according to concrete technique parameters.
3.4 Variations of relative density of Mg2Si bulk with hot-pressing parameters
Fig.5 shows the variation of the relative density of Mg2Si bulk (hot-pressed at 400 ℃ for 1 h) with the hot-pressing pressure(p). It can be seen that the change of relative density of Mg2Si bulk with hot-pressing pressure exhibits two stages: 1) low pressure fast-rise stage (125 MPa<p<300 MPa) in which the relative density of Mg2Si bulk rises steeply from 72% to 82%; 2) stably-rise stage (300 MPa<p<1500 MPa) where the relative density increases stably from 82% up to about 98.5%. According to the compaction theory of powder metallurgy, the first stage (125 MPa<p<300 MPa) can be ascribed to the elimination of holes, the sliding and displacement of Mg2Si particles, and the second stage (300 MPa<p<1 500 MPa) could be attributed to the rearrangement, fracture and deformation of brittle Mg2Si particles.
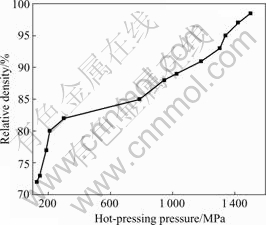
Fig.5 Variation of relative density of Mg2Si bulk with hot- pressing pressure (hot-pressed at 400 ℃ for 1 h)
At the same time, the influence of other hot-pressing parameters on the relative density Mg2Si bulk has been investigated. Fig.6 shows the variation of the relative density of Mg2Si bulk with the hot-pressing temperature (hot-pressed under 300 MPa for 1 h). One can see that the relative density of Mg2Si bulk increases monotonously from 74% to 86% as hot-pressing temperature increases from 300 to 500 ℃. It should be pointed out that the self-ignition phenomenon of Mg2Si bulk will occur when the hot-pressing temperature is lower than 300 ℃, because of the existence of Mg grains unreacted in Mg2Si bulk. While the hot-pressing temperature is over 500 ℃, the grain size of Mg2Si rises steeply[16].
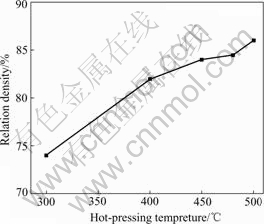
Fig.6 Variation of relative density of Mg2Si bulk with hot- pressing temperature (hot-pressed under 300 MPa for 1 h)
Similarly, Fig.7 shows the variation of the relative density of Mg2Si bulk with the hot-pressing time (hot-pressed at 400 ℃ under 300 MPa). It shows that the relative density of Mg2Si bulk increases from 78% to 87.5% when the hot-pressing time increases from 30 to 90 min. However, as hot-pressing time is prolonged further, the increase of the relative density becomes slow.
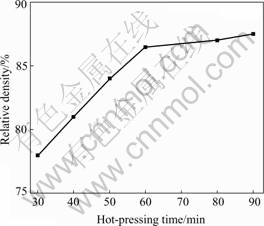
Fig.7 Variation of relative density of Mg2Si bulk with hot- pressing time (hot-pressed at 400 ℃ under 300 MPa)
Moreover, the grain sizes of Mg2Si bulk obtained are in the range of 30-60 nm, which agrees basically with the SEM results, as shown in Fig.8. It can be seen that most of the particles in Mg2Si bulk are made up of several nanocrystalline Mg2Si grains.

Fig.8 SEM fractographs of nanocrystalline Mg2Si bulk: (a) Low magnification; (b) High magnification
3.5 Vickers hardness and fracture toughness of nanocrystalline Mg2Si bulk
Fig.9 shows the dependence of fracture toughness and Vickers hardness on the relative density of Mg2Sibulk. As the relative density of Mg2Si bulk increases from about 72% to 98.5%, the fracture toughness and the Vickers hardness increase from about 0.69 to 1.46 MPa・m1/2 and from about 1.35 to 4.38 GPa, respectively. It is worthwhile to note that the values of KIC and HV of the obtained high-density Mg2Si bulk are higher than the values of conventional polycrystalline Mg2Si (KIC 0.8- 1.0 MPa・m1/2 and HV 4.1GPa)[17] produced by induction melting and casting, indicating that the mechanical properties of nanocrystalline Mg2Si are superior to those of the conventional coarse-grained Mg2Si. The great hardness can be ascribed to the fine grain size of n-Mg2Si, for one knows from Hall-Petch relation that the yield strength (or hardness) is proportional to d-1/2 (here d is the mean grain size). The increase of KIC with increasing density mainly originates from the elimination of defects (microvoids) which can act as cracking sources and degenerate toughness of the material. The enhancement of fracture toughness as compared with that of the conventional polycrystalline Mg2Si is maybe related to large volume fraction of grain boundaries Ct (Ct=5% for Mg2Si with mean grain size of 60 nm[18]). Previous work[19] indicated that the rate of grain boundary(GB) sliding through GB diffusion is proportional to d-3, and the contribution of GB sliding to deformation in n-Mg2Si is much greater than that in coarse-grained (normally grain sizes have the order of micrometers) Mg2Si.The larger strain rate of GB sliding in n-Mg2Si could release the stress concentration accumulated around crack tips and inhibit creak extension, which would result in increase of fracture toughness KIC of n-Mg2Si as compared with coarse grained Mg2Si.
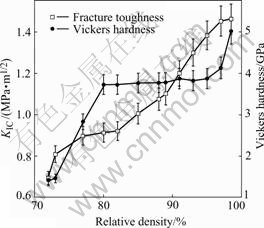
Fig.9 Dependence of fracture toughness and Vickers hardness on relative density of Mg2Si bulk
4 Conclusions
Bulk n-Mg2Si was prepared by mechanical balling and reactive hot-pressing in vacuum and its mechanical properties were characterized. The decrease of grain sizes d of Mg and Si decreases non-monotonously with increasing ball-to-powder mass ratio(BPR). The content of Mg2Si increases with increasing milling time. However, pure Mg2Si phase cannot be obtained by direct ball milling. Moreover, monolithic Mg2Si phase can only be obtained when as much as 10% excess Mg was blended in the raw mixtures. The relative density D and fracture toughness as well as hardness of bulk Mg2Si samples can be raised effectively by increasing the pressure in hot-pressing. After hot-pressing at 400 for 60 min under 1.5 GPa, highly dense (D>98%) n-Mg2Si (d=60 nm) was obtained, whose fracture toughness (about 1.46 MPa・m1/2) and Vickers hardness (about 4.38 GPa) are superior to those of conventional coarse- grained Mg2Si. The increase of the hardness can be ascribed to the refinement of grain size of Mg2Si bulk, and the enhancement of fracture toughness can be attributed to the large volume fraction of grain boundaries in n-Mg2Si.
References
[1] LU L, LAI M O, HOE M L. Formation of nanocrystalline Mg2Si and Mg2Si dispersion strengthened Mg-Al alloy by mechanical alloying [J]. Nanostructured Materials, 1998, 10(4): 551-563.
[2] YOSHINAGA M, IIDA T, NODA M. Bulk crystal growth of Mg2Si by the vertical Bridgman method [J]. Thin Solid Films, 2004, 461: 86-89.
[3] KONDOH K, OGINUMA H, YUASA E. Solid-state synthesis of Mg2Si from Mg-Si mixture powder [J]. Materials Transactions, 2001, 42(7): 1293-1300.
[4] LI G H, KONG Q P. Processing and thermal stability of nano-Mg2Si intermetallic compound [J]. Scripta Metallurgical et Materialia, 1995, 32(9): 1435-1440.
[5] TAKEUCHI S, HASHIMOTO T, SUZUKI K. Plastic deformation of Mg2Si with the C1 structure [J]. Intermetallics, 1996, 4: 147-150.
[6] RIFFEL M, SCHILZ J. Mechanically alloyed Mg2Si1-xSnx solid solutions as thermoelectric materials [A]. 15th International Conference on Thermoelectrics [C]. Pasadena: IEEE, 1996. 133-136.
[7] RIFFEL M, SCHILZ J. Influence of production parameters on the thermoelectric properties of Mg2Si [A]. 16th International Conference on Thermoelectrics [C]. Dresden: IEEE, 1997. 283-286.
[8] TATSUOKA H, TAKAGI N, OKAYA S. Microstructures of semiconducting silicide layers grown by novel growth techniques [J]. Thin Solid Films, 2004, 461: 57-62.
[9] MU?OZ-PALOS J M, CRISTINA M C, ADEVA P. Synthesis of Mg2Si powder by mechanical alloying and its consolidation [J]. Materials Transactions, 1996, 37(10): 1602-1606.
[10] RIFFEL M, SCHILZ J. Mechanical alloying of Mg2Si [J]. Scripta Metallurgical et Materialia, 1995, 32(12): 1951-1956.
[11] LIU X P. Formation of magnesium silicide by mechanical alloying [J]. Advanced Performance Materials, 1997, 3: 275-283.
[12] LU L, LAI M O, XUE W B, NG S Y. Influence of kinetic energy on the formation of Mg2Si [J]. Z Metallkd, 2000, 91(8): 680-685.
[13] XIONG Wei, QIN Xiao-ying, WANG Li. Preparation and microstructural characterization of nanocrystalline Mg2Si intermetallic compound bulk [J]. The Chinese Journal of Nonferrous Metals, 2005, 15(3): 380-385. (in Chinese)
[14] ANSTIS G R, CHANTYIKUL P, LAWN B R. A critical evaluation of indentation technique for measuring fracture toughness (I)-direct crack measurements [J]. J Am Ceram Soc, 1981, 64: 533-538.
[15] SURYANARAYANA C. Mechanical alloying and milling [J]. Progress in Materials Science, 2001,46: 1-184.
[16] WANG L, QIN X Y. The effect of mechanical milling on the formation of nanocrystalline Mg2Si through solid-state reaction [J]. Scripta Materialia, 2003, 49: 243-248.
[17] MILEKHINE V, ONSΦIEN M I, SOLBERG J K. Mechanical properties of FeSi (ε), FeSi2 (ζα) and Mg2Si [J]. Intermetallics, 2002, 10: 743-750.
[18] M?TSCHELE T, KIRCHHEIM R. Hydrogen as a probe for the average thickness of a grain boundary [J]. Scripta Metallurgica, 1987, 21: 1101-1104.
[19] QIN X Y, ZHU X G, GAO S. Compression behavior of bulk nanocrystalline Ni-Fe [J]. J Phys Condens Matter, 2002, 14: 2605-2620.
Foundation item: Project(50371081) supported by the National Natural Science Foundation of China
Corresponding author: QIN Xiao-ying; Tel: +86-551-5592750; E-mail: xyqin@issp.ac.cn
(Edited by LONG Huai-zhong)