
Preparation and electrochemical performance of
nanosized Co3O4 via hydrothermal method
YANG You-ping(杨幼平), LIU Ren-sheng(刘人生), HUANG Ke-long(黄可龙),
WANG Li-ping(王丽平), LIU Su-qin(刘素琴), ZENG Wen-wen(曾雯雯)
School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China
Received 15 July 2007; accepted 10 September 2007
Abstract: The hydrotalcite-type cobalt compounds were prepared through oxidation of Co(OH)2 gel using NH4OH as precipitating agent and H2O2 as oxidant. These hydrotalcite-type cobalt compounds were transformed into Co3O4 through hydrothermal decomposition with nanostructural deformation. The precursor and product were characterized by Fourier-transform infrared(FT-IR) spectrum, X-ray diffractometry(XRD) and transmission electron microscopy(TEM). The electrochemical performances of as-prepared nanosized Co3O4 as anode materials in lithium-ion batteries were tested by charge-discharge test in the voltage range of 0-3.0 V. The influence of morphology of Co3O4 particle on the capacity and cycling performance was studied. The results show that the shape and size of the final product can be controlled by altering cobalt sources. The irregular cubic Co3O4 with the average particle size of about 10 nm shows the best electrochemical performance. After 10 charge-discharge cycles, the specific charge capacity retains 555 mA?h/g.
Key words: hydrotalcite-type cobalt compounds; nanosized Co3O4; hydrothermal method; anode materials
1 Introduction
In recent years, Co3O4 has attracted increasingly attention due to its wide applications in catalysts, gas sensors, magnetic semiconductors, negative electrodes in rechargeable lithium-ion batteries[1-6]. The performance of Co3O4 in its applications such as catalysts and magnetic materials can be promising owing to the high surface area and narrow size distribution. Therefore, the synthesis of uniform Co3O4 nanocrystal has been the target of material chemists. In this field, various synthesis methods have been reported, such as thermal decomposition of solid phase[7], chemical spray pyrolysis[8], and sol-gel method[9]. However, the size distribution of Co3O4 nanocrystal made by the above methods is wide. Hydrothermal method[10] is an efficient technique for preparing fine oxide powers by reaction with high temperature and high pressure in solution.
In this work, the hydrotalcite-type cobalt compounds were prepared using NH4OH as precipitating agent and H2O2 as oxidant, and then were transformed to Co3O4 under hydrothermal condition. The electro- chemical performances of as-prepared nanosized Co3O4 as anode materials in lithium-ion batteries were tested.
2 Experimental
2.1 Samples preparation
All of the starting materials were in analytical grade. 15 mmol cobalt salt (Co(CH3COO)2・4H2O, CoSO4・7H2O, or Co(NO3)2・6H2O) was dissolved into deionized water containing dispersant polyethylene glycol(PEG) with relative molecular mass of about 20 000, then excessive amount NH4OH was added with electromagnetic stirring at 30 ℃ to form Co(OH)2 gel. The pH value was monitored to 8-9. A certain volume of 30% (mass fraction) H2O2 was dropped into the above suspension. Finally, all of them were transferred into a Teflon-lined stainless steel autoclave with the volume of 100 mL, and the autoclave was filled with deionized water up to 70% of the total capacity. The sealed autoclave was heated to 160 ℃ in 1 h and maintained for 16 h, then cooled to room temperature in air naturally. The black products were centrifuged and washed with deionized water and absolute ethanol for three times, respectively, then dried in a vacuum oven at 80 ℃ for 6 h.
2.2 Samples characterization
The crystal structures of samples were identified by X-ray diffraction(XRD) in the 2θ range of 10?-80? using a Japan Rigaku D/Max-2500 X-ray diffractometer with graphite monochromatic Cu Kα radiation (λ=0.154 056 nm). Fourier transform infrared(FT-IR) spectra were measured with Avatar 360 FT-IR spectrophotometer within a wavelength range from 4 000 cm-1 to 400 cm-1. The morphology and size of the as-prepared Co3O4 were observed with Model JEM-1230 transmission electron microscope(TEM).
2.3 Electrode preparation and electrochemical per- formance test
The electrode was prepared by coating the mixture of Co3O4 (80%, mass fraction), acetylene black (10%) and polytetrafluoroethylene(PTFE) binder (10%) onto a 1 cm2 stainless steel current collector. The electrode was dried in a vacuum oven at 80 ℃ and then pressed at 20 MPa. The electrochemical cell consisted of a Co3O4 working electrode and a lithium foil counter electrode. They were assembled in a MBRAUN glove box under argon atmosphere with H2O and O2, in which the volume fraction of H2O and O2 was less than 10-6. The electrolyte was 1 mol/L LiPF6 dissolved in a mixture of ethylene carbonate(EC), dimethyl carbonate(DMC) and methyl-ethyl carbonate(EMC) with a volume ratio of 1?1?1. The cell was discharged and charged between 0.01 and 3.0 V versus Li+/Li at a rate of 0.1C.
3 Results and discussion
Fig.1 shows the XRD patterns for the precursors, hydrotalcite-type cobalt compounds, obtained using different cobalt sources without hydrothermal treatment. All peaks shown in Fig.1 can be indexed to hydrotalcite-type cobalt compound, which is consistent with the reported data[11-14], d(003)= 2d(006)=3d(009). When excessive amount NH4OH is added to the solution of cobalt salt, there forms Co(OH)2 gel. After H2O2 is dropped into the above suspension, some divalent cobalt cations are substituted by trivalent cobalt cations, and anion species in suspension are intercalated into the interlayer space to compensate the extra position charge possessed by the trivalent cobalt cations. This structural alternation leads to the formation of hydrotalcite-like structures.
Fig.2 shows FT-IR spectra of the precursors prepared using different cobalt sources. The broad peaks
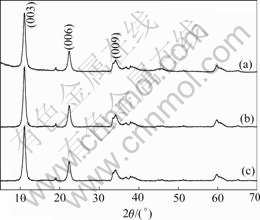
Fig.1 XRD patterns of hydrotalcite-type cobalt compounds using different cobalt sources: (a) Co(CH3COO)2; (b) CoSO4; (c) Co(NO3)2
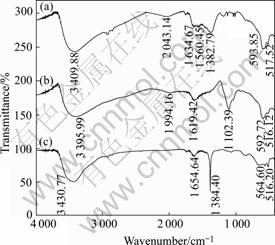
Fig.2 FT-IR spectra of precursors obtained using different cobalt sources: (a) Co(CH3COO)2; (b) CoSO4; (c) Co(NO3)2
at 3 395.99-3 430.77 cm-1 are assigned to the stretching vibration of the O―H bond, υOH, which indicates the presence of hydroxyl ions due to the metal-OH layer and/or water in the crystal, and the peaks at 1 619.42- 1 654.64 cm-1 are assigned to the bending modes of water. The peaks at 516.12-517.52 cm-1 and 564.60- 593.85 cm-1 can correspond to the presence of Co―O species.
In Fig.2(a), the peaks, at around 1 560.45 cm-1 and 1 382.79 cm-1, result from the asymmetric stretching and symmetric stretching mode of CH3COO-, respectively. In Fig.2(b), the peak at 1 102.39 cm-1 is assigned to the asymmetric stretching mode of  in the precursor. In Fig.2(c), the peak, at around 1 384.40 cm-1, belongs to the υ3 vibrational mode of
in the precursor. In Fig.2(c), the peak, at around 1 384.40 cm-1, belongs to the υ3 vibrational mode of  . Based on FT-IR spectra, the hydrotalcite-type cobalt compounds formed are indicated as
. Based on FT-IR spectra, the hydrotalcite-type cobalt compounds formed are indicated as 
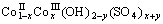 and
and (NO3)x+y using Co(CH3COO)2, CoSO4 and Co(NO3)2 as cobalt sources, respectively.
(NO3)x+y using Co(CH3COO)2, CoSO4 and Co(NO3)2 as cobalt sources, respectively.
Generally speaking, divalent metal cation hydroxides have a layered structure, while metal hydroxides having a formula of [M(OH)]x+(An-)x/n・yH2O are known as layered double hydroxides (LDH)[15]. When adding oxidant H2O2 to the LDH suspension, the oxidation of Co2+ into Co3+ yields positively charged hydroxide layers 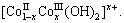 According to charge equilibrium, the anions such as CH3COO-,
According to charge equilibrium, the anions such as CH3COO-,  and
and  are then intercalated into the interlamellar spaces. Then the hydrotalcite-type cobalt compounds are formed. Upon the hydrothermal treatment of the hydrotalcite-type hydroxide at 160 ℃ in the autoclave for 16 h, all the hydrotalcite-type cobalt compounds are converted into Co3O4 nanoparticles.
are then intercalated into the interlamellar spaces. Then the hydrotalcite-type cobalt compounds are formed. Upon the hydrothermal treatment of the hydrotalcite-type hydroxide at 160 ℃ in the autoclave for 16 h, all the hydrotalcite-type cobalt compounds are converted into Co3O4 nanoparticles.
Fig.3 shows the XRD patterns of the products obtained by the hydrothermal method. All the diffraction peaks can be indexed to a pure cubic phase of spinel Co3O4. Evaluated from Scherrer formula, the average particle sizes prepared with the hydrothermal method using Co(CH3COO)2, CoSO4, Co(NO3)2 as cobalt sources are about 18, 27 and 10 nm, respectively. No other peaks for impurities are detected.
The TEM morphologies of crystallized powders are very homogeneous with respect to particles size and shape. Fig.4(a) shows the TEM image of hydrotalcite-
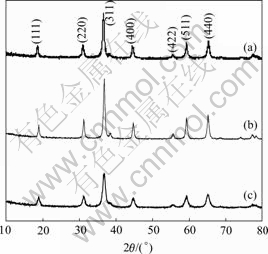
Fig.3 XRD patterns of products prepared by hydrothermal method using different cobalt sources: (a) Co(CH3COO)2; (b) CoSO4; (c) Co(NO3)2
type cobalt compound deposited using Co(CH3COO)2 as cobalt source and H2O2 as oxidant. The TEM images (Figs.4(b)-(d)) show that the converted products of hydrotalcite-type cobalt compounds by hydrothermal method, Co3O4 powders, consist of monodispersive nanoparticles using Co(CH3COO)2, CoSO4 and Co(NO3)2

Fig.4 TEM images of hydrotalcite-type cobalt compound (a) and nanocrystalline Co3O4 prepared by hydrothermal method using different cobalt sources: (b) Co(CH3COO)2; (c) CoSO4; (d) Co(NO3)2
as cobalt sources, respectively. Fig.4(b) shows the cubic grains with an average edge length of 20 nm. The nanocubes are perfect and the particle size distribution is narrow. This result suggests that the spinel cobalt oxide nanocubes may be formed through a decomposing and redepositing process under hydrothermal conditions (Figs.4(a) and (b)). Fig.4(c) shows spherical morphology, with a diameter of around 30 nm. Fig.4(d) demonstrates that the average particle size is about 10 nm. The most of the nanocubes are imperfect, and their shapes become more spherical. The size of the Co3O4 powders is generally consistent with that of the XRD analysis.
In order to obtain a discharge-charge profile of nanosized Co3O4, the cell was subjected to 10 cycles at 0.1C rate. The first discharge-charge curves for the regular cubic sample are shown in Fig.5. During the first discharge, the potential rapidly drops to a large plateau at 1.1 V, and then slowly drops to a plateau at 0.7 V. The specific discharge capacity (lithium-inserted process) in the first discharge reaches 1 380 mA・h/g and the first charge capacity(lithium-deserted process) amounts 927 mA・h/g. The irreversible capacity loss is 33% in the first cycle. The discharge-charge profiles of nanosized Co3O4 with various shapes are uniform, but their capacity and the cycling performance are different.
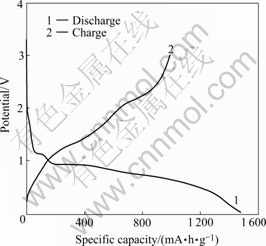
Fig.5 First discharge-charge profiles of cubic Co3O4 at 0.1C rate
The cycling performances of nanosized Co3O4 samples with different particle morphologies are shown in Fig.6. The as-prepared Co3O4 with regular cubic shape (see Fig.4(b)) has obvious decline in electrochemical capacity. After 10 discharge-charge cycles, the specific charge capacity decreases from 927 mA・h/g to 446 mA・h/g in the first charge process. The first and the tenth charge capacities of spherical Co3O4 (see Fig.4(c)) are 894 mA・h/g and 378 mA・h/g, respectively. The first charge capacity of irregular cubic Co3O4 (see Fig.4(d)) is 912 mA・h/g, and the charge capacity is retained at 555 mA・h/g after 10 cycles. It can be concluded that the electrochemical properties of Co3O4 as anode material for lithium-ion batteries are sensitive to their particle shape, and the irregular cubic Co3O4 has the best electrochemical property in all the as-prepared samples.
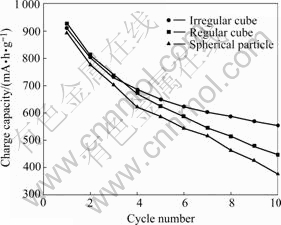
Fig.6 Cycling performance of as-prepared Co3O4 in charge-discharge process at 0.1C rate
4 Conclusions
1) The hydrotalcite-type cobalt compounds are synthesized through oxidation of Co(OH)2 gel using NH4OH as precipitating agent and H2O2 as oxidant. The process that Co2+ is partially oxidated to Co3+ yields positively charged hydroxide layers, 
 According to charge equilibrium, the anions such as CH3COO-,
According to charge equilibrium, the anions such as CH3COO-, and
and are intercalated into the interlamellar spaces.
are intercalated into the interlamellar spaces.
2) These hydrotalcite-type compounds are transformed into Co3O4 through hydrothermal decomposition with nanostructural deformation. The shape and size of the final product can be controlled by altering cobalt sources.
3) The particle shape has distinct effect on electrochemical performance of Co3O4 as anode material for lithium-ion batteries. The irregular cubic Co3O4 shows the best electrochemical performance in all the as-prepared samples. After 10 discharge-charge cycles, the specific charge capacity retains 555 mA?h/g.
References
[1] BAHLAWANE N, RIVERA E F, KOHSE-HOINGHAUS K, BRECHLING A, KLEINEBERG U. Characterization and tests of planar Co3O4 model catalysts prepared by chemical vapor deposition [J]. Appl Catal B: Environmental, 2004, 53(4): 245-255.
[2] LI Wei-yang, XU Li-na, CHEN Jun. Co3O4 nanomaterials in lithium-ion batteries and gas sensors [J]. Adv Funct Mater, 2005, 15(5): 851-857.
[3] ABE S, CHOI U S, SHIMANOE K, YAMAZOE N. Influences of ball-milling time on gas-sensing properties of Co3O4-SnO2 composites [J]. Sens Actuators B Chem, 2005, 107(2): 516-522.
[4] ICHIYANAGI Y, KIMISHIMA Y, YAMADA S. Magnetic study on Co3O4 nanoparticles [J]. J Magn Magn Mater, 2004, 272: 1245-1246.
[5] WANG G X, CHEN Y, KONSTANTINOV K, YAO J, AHN J H, LIU H K, DOU S X. Nanosize cobalt oxides as anode materials for lithium-ion batteries [J]. J Alloys Compd, 2002, 340: L5-L10.
[6] YUAN Zheng-yong, HUANG Feng, FENG Chuan-qi, SUN Ju-tang, ZHOU Yun-hong. Synthesis and electrochemical performance of nanosized Co3O4 [J]. Mater Chem Phys, 2003, 79: 1-4.
[7] YANG Hua-ming, HU Yue-hua, ZHANG Xiang-chao, QIU Guan-zhou. Mechanochemical synthesis of cobalt oxide nanoparticles [J]. Mater Lett, 2004, 58(3/4): 387-389.
[8] CHENG C, SERIZAWA M, SAKATA H, HIRAYAMA T. Electrical conductivity of Co3O4 films prepared by chemical vapour deposition [J]. Mater Chem Phys, 1998, 53(3): 255-230.
[9] CAO Jin-zhang, ZHAO Yan-chun, YANG Wu, TIAN Jian-niao, GUAN Fei, MA Yong-jun. Sol-gel preparation and characterization of Co3O4 nanocrystals [J]. Journal of University of Science and Technology Beijing, 2003, 10(1): 54-57.
[10] COTE L J, TEJA A S, WILKINSON A P, ZHANG Z J. Continuous hydrothermal synthesis and crystallization of magnetic oxide nanoparticles [J]. J Mater Res, 2002, 17(9): 2410-2416.
[11] XU Z P, ZENG H C. Interconversion of brucite-like and hydrotalcite-like phases in cobalt hydroxide compounds [J]. Chem Mater, 1999, 11(1): 67-74.
[12] XU Z P, ZENG H C. Control of surface area and porosity of Co3O4 via intercalation of oxidative or nonoxidative anions in hydrotalcite-like precursors [J]. Chem Mater, 2000, 12(11): 3459-3465.
[13] XU Z P, ZENG H C. A new approach for design and synthesis of CoII and CoII, III hydroxide materials [J]. Int J Inorg Mater, 2000, 2(2/3): 187-196.
[14] XU Rong, ZENG Hua-chun. Mechanistic investigation on self-redox decompositions of cobalt-hydroxide-nitrate compounds with different nitrate anion configurations in interlayer space [J]. Chem Mater, 2003, 15(10): 2040-2048.
[15] HOSONO E, FUJIHARA S, HONMA I, ZHOU H S. Fabrication of morphology and crystal structure controlled nanorod and nanosheet cobalt hydroxide based on the difference of oxygen-solubility between water and methanol, and conversion into Co3O4 [J]. J Mater Chem, 2005, 15: 1938-1945.
(Edited by YANG Bing)
Foundation item: Project(50542004) supported by the National Natural Science Foundation of China; Project(ZE097) supported by Creative Program of Central South University, China
Corresponding author: HUANG Ke-long; Tel: +86-731-8879850; E-mail: klhuang@mail.csu.edu.cn