
Effect of nonlinear liquidus and solidus on dendrite growth in bulk undercooled melts
CHEN Zheng(陈 正), WANG Hai-feng(王海丰), LIU Feng(刘 峰), YANG Wei(杨 伟)
State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, Xi’an 710072, China
Received 26 November 2008; accepted 29 June 2009
Abstract: On the base of nonlinear liquidus and solidus, an extended model for dendrite growth in bulk undercooled melts was developed under local non-equilibrium conditions both at the interface and in the bulk liquid. In terms of thermodynamic calculations of the phase diagram, the model predictions are relatively realistic physically, since few fitting parameters are used in the model predictions. Adopting three characteristic velocities, i.e. the critical velocity of absolute solute stability (VC*), the velocity of maximal tip radius (VRm), and the velocity of bulk liquid diffusion (VD), a quantitative agreement is obtained between the model predictions and the experimental results in undercooled Ni-0.7%B and Ni-1%Zr (molar fraction) alloys, and the overall solidification process can be categorized.
Key words: dendrite growth; undercooled melts; Ni-based alloys
1 Introduction
The growth of unconstrained dendrites in undercooled melts has been a major subject in the theoretical arena of solidification research. Model developed by BOETTINGER et al[1], referred to as BCT model, was well accepted for dendrite growth in rapid solidification process. Applying the thermodynamic driving force[2] and the model of interfacial solute-trapping presented by AZIZ[3-4], BCT model allows deviations of interface composition from the local equilibrium value with the advancement of S/L interface. On this basis, the linear kinetic model proposed by TURNBULL[5] was adopted to relate the interfacial driving force to the tip velocity, V, while the marginal stability criterion of TRIVEDI and KURZ[6] was used to predict the tip radius.
So far, a lot of experiments[7-11] have shown that, above a critical undercooling DT*, dendrite growth deviates from power law to linear growth. Unfortunately, a physically realistic interpretation for these phenomena cannot be obtained by BCT model. This infers that the BCT model should be revised or modified. Introducing a relaxation effect, i.e. non-equilibrium liquid diffusion, GALENKO and DANILOV[12-13] developed a model, where the bulk liquid diffusion speed, VD, has a finite value and the solute transport is described by a partial differential equation of hyperbolic type[14-16]. Applying this model, a better prediction for the transition from power law to linear growth was achieved.
However, all the above models were developed assuming linear liquidus and solidus, which is generally valid for small DT[17]. Subjected to large DT, analysis of dendrite growth should refer to non-linear liquidus and solidus. In the present work, the non-equilibrium interface kinetics and the non-equilibrium liquid diffusion have been combined with non-linear liquidus and solidus, and then a steady-state dendrite growth model, as an extension of GALENKO an DANILOV’s model[12-13], is proposed, including a marginal stability criterion in highlight of the kinetic effect. The present model is applied to rapid solidification of undercooled Ni-0.7%B and Ni-1%Zr (molar fraction) alloys.
2 Model formulation
2.1 Interfacial driving force and interface response function
For dendrite growth to occur, a net decrease of the free energy, DG, is needed as the driving force for the S/L interface migration. Combined with the relaxation effect, the expression of DG for a dilute alloy is given as[18]
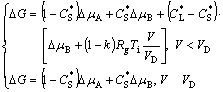 (1)
(1)
where Dμ is the chemical potential difference between solid and liquid; CL* and CS* are the non-equilibrium concentrations in liquid and solid, respectively, at the interface, corresponding to Ti, for a curved interface; and CLeq and CSeq, the equilibrium concentrations in liquid and solid, respectively, at the interface, subjected to the curvature correction, are respectively replaced by CLeq′ and CSeq′[17]:
 (2)
(2)
where DTR is the curvature undercooling. The interface response function can be deduced by combining Eqs.(1) and (2) with the linear kinetic law in Refs.[5, 19]. A detailed derivation is available in Ref.[20]:

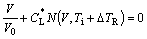 (3)
(3)
where
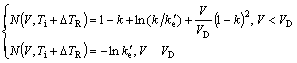 (4)
(4)
where V0 is the upper limit of the interface advancement, k=CS*/CL* and ke′=CSeq′/CLeq′ are the non-equilibrium and equilibrium partition coefficients subjected to the curvature correction, respectively. Eq.(3) is the interface response function related to the interfacial temperature Ti and the liquid composition CL* for a curved interface. In contrast with BCT model[1] and GALENKO and DANILOV’s model[12-13], in which the slope of liquidus mL and the equilibrium partition coefficient ke are assumed to be constant, the current model adopts composition- dependent mL and ke, which are directly obtained from thermodynamic calculations of the phase diagram (i.e. with non-linear liquidus and solidus).
2.2 Marginal stability criterion
The model of TRIVEDI and KURZ[6], as an extension of the marginal stability theory[21] to cases of high Péclet number, is valid for rapid solidification of an undercooled melt where the thermal length is much smaller, but the role of the kinetic effect is neglected. Departed from non-equilibrium liquid diffusion, a modification of T-K model was made by GALENKO and DANILOV[22], where the kinetic factor affecting the equilibrium phase diagram, i.e. the slope of the liquidus in the kinetic phase diagram, was considered, but the corresponding effect of perturbation was neglected. Based on the kinetic interface response function assuming non-linear liquidus and solidus, an approximately marginal stability criterion for a curved interface was obtained [20], and the expression of the dendrite tip radius R is
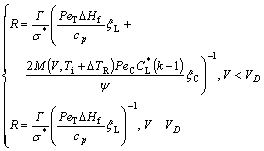 (5)
(5)
where
 (6a)
(6a)
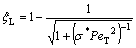 (6b)
(6b)
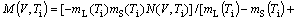
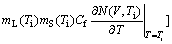 (6c)
(6c)
where Г is the Gibbs-Thompson coefficient; ?Hf is the latent heat of fusion; s*=1/4p2, the stability constant; PeC=VR/(2D), the solute Péclet number; PeT=VR/2αL, the thermal Péclet number; ψ=1-V2/VD2; D the liquid diffusion coefficient; αL the thermal diffusivity in liquid; and mL and mS the slope of liquidus and solidus, respectively. As an extension to the corresponding relations assuming linear solidus and liquidus[1, 12-13, 22], Eq.(5) implies that both dendrite tip radius R and dendrite tip velocity V are dependent on the dendrite tip temperature Ti. M(V, Ti), the slope of liquidus in the kinetic phase diagram assuming non-linear solidus and liquidus, is also dependent on both the interface temperature Ti and the interface velocity V. Based on the kinetic interface response function (Eq.(3)), the kinetic effect plays an important role in the current marginal stability analysis.
2.3 Bath undercooling component and solute trapping model
Analogously, the bath undercooling consists of four components, i.e. curvature, constitutional, kinetic and thermal undercoolings:
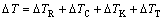 (7)
(7)
where
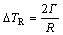 (8a)
(8a)
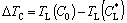 (8b)
(8b)
 (8c)
(8c)
 (8d)
(8d)
By taking into account of local non-equilibrium effects, the solute trapping model of AZIZ[3-4] is corrected by SOBOLEV[16]. In the present work, the equilibrium partition coefficient, ke, is replaced by the curvature-corrected one, ke′, and the non-equilibrium partition coefficient reads
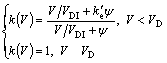 (9)
(9)
Until now, the dendrite growth model is established. Integrating Eqs.(3)-(9), for a given undercooling, the dendrite tip radius R and the dendrite growth velocity V can be uniquely solved.
3 Results and discussion
3.1 Comparison of model prediction with experimental results
The present model is used to describe rapid solidification of undercooled Ni-0.7%B and Ni-1%Zr (molar fraction) alloy melts. Since nonlinear solidus and liquidus are held for large DT, thermodynamic calculations of the phase diagram become very decisive. As shown in Fig.1, the equilibrium Ni-B and Ni-Zr phase diagrams for the nickel rich part are precisely calculated using Thermo-Calc.
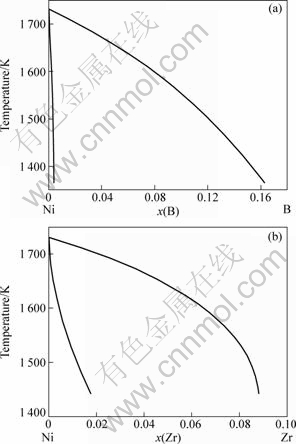
Fig.1 Equilibrium phase diagram of Ni-B (a) and Ni-Zr (b) in nickel rich part obtained from Thermo-Calc
As shown in Fig.2, the present model is adopted to predict V as a function of DT. The used parameters for the Ni-0.7%B alloy is given in Ref.[20]: for Ni-1%Zr, ΔHf=1.623×104 J/mol[11], cp=41 J/(mol?K)[11], αL= 4.2×10-5 m2/s[11], D=5×10-9 m2/s, Γ=8.5×10-8 K?m, VD=21.5 m/s, VDI=19 m/s and V0=550 m/s. The experimental data are from Refs.[7-8, 11]. The present model gives a satisfactory agreement with the experimental data in the transition from the power law to linear law growth process for both undercooled Ni-0.7%B and Ni-1%Zr alloy melts. Moreover, less adjust parameters is needed for the present model, which makes the prediction more physically realistic.
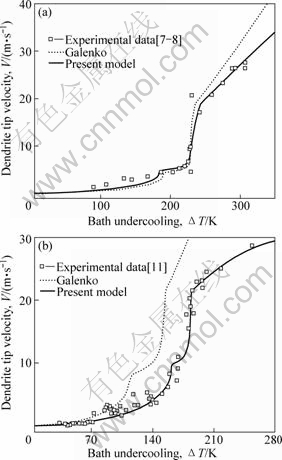
Fig.2 Dendrite tip velocity as function of bath undercooling for Ni-0.7%B (a) and Ni-1%Zr (b) alloys
3.2 Characteristic velocities and mechanism controlling solidification
Further, a plateau is found at the intermediate undercooling range for both undercooled Ni-0.7%B and Ni-1%Zr alloy melts in the model predictions. To explain the plateau, a definition of absolute solute stability velocity, VC, is introduced for dendritic growth. The absolute solute stability velocity indicates that the growth is purely solute-controlled without thermal effect, and the interface will always keep stable if V>VC. Following the T-K method for high Pe value[6], e.g. Pe>> 1, VC can be expressed as[20]
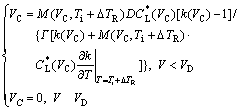 (10)
(10)
Clearly, VC is also dependent on the interface temperature Ti. This is different from the models assuming linear liquidus and solidus, such as M-S[21], T-K[6] and GALENKO and DANILOV’s model[22], where a constant value, or a value dependent solely on the dendrite tip velocity, V, is obtained. Fig.2 shows the prediction of the evolution of VC with DT. One can see that the initiating point of the plateau coincides with the dendrite tip velocity at V=VC*, which is defined as the critical velocity of the absolute solute stability.
Fig.3 exhibits four cases of evolutions of tip radius with DT, i.e. solute- and thermal-controlled case, solute-controlled case with solute trapping, solute- controlled case without solute trapping, and pure thermal case. From Figs.3(a) and (b), the solid line and the dotted line coincide if V≤VC*, indicating that VC* stands up for the ending point of the mainly solute-controlled case.
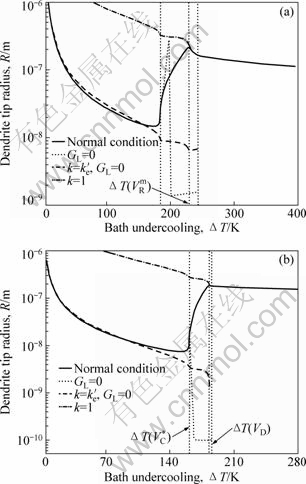
Fig.3 Evolution of dendrite tip radius as function of bath undercooling for Ni-0.7%B (a) and Ni-1%Zr (b) alloys
Another characteristic velocity, VRm, is defined as the velocity where the maximal dendrite tip radius is achieved. From Fig.3, the dash-dotted line and the solid line almost (i.e. not exactly) coincide if V≥VRm. This implies that the mainly thermal-controlled (i.e. instead of pure thermal-controlled) growth initiates once the tip radius R reaches the maximal value, i.e. V=VRm. But if V≥VD, the solid line and the dash-dotted line exactly coincide from Figs.3(a) and (b). This indicates an occurrence of complete solute trapping, i.e. the initiation of a purely thermal-controlled case if V=VD. Note that the phase diagram for the Ni-Zr alloy changes greatly, at high undercooling, which makes the prediction for the purely thermal-controlled growth, i.e. when V≥VD, deviate a little from linear growth, as shown in Fig.3(b). In other words, the linear growth process is not completely linear assuming non-linear liquidus and solidus in the present dendrite growth model.
Regarding the three characteristic velocities, the overall solidification process can be categorized as follows: 1) mainly solute-controlled growth, i.e. ?T<?T(VC*); 2) transition from mainly solute-controlled to mainly thermal-controlled growth, i.e. ?T(VC*)≤?T<?T(VRm); 3) mainly thermal-controlled growth, i.e. ?T(VRm)≤?T<?T(VD); and 4) purely thermal-controlled growth, i.e. ?T≥?T(VD).
4 Conclusions
1) On the basis of nonlinear liquidus and solidus, an extended model for dendrite growth in bulk undercooled melts is developed under local non-equilibrium conditions both at the interface and in the bulk liquid.
2) In the extended steady-state dendrite growth model, the kinetic effect plays an important role in the current marginal stability criterion. Accordingly, the liquidus slope in the kinetic phase diagram assuming non-linear solidus and liquidus, i.e. M(V, Ti), is dependent on both the interface temperature Ti and the interface velocity V.
3) Adopting three characteristic velocities, i.e. the critical velocity of absolute solute stability, VC*, the velocity of maximal tip radius, VRm, and the velocity of bulk liquid diffusion, VD, application of the present model to rapid solidification of the undercooled Ni-0.7%B and Ni-1%Zr (molar fraction) alloys shows a quantitative agreement between the model predictions and the experimental results.
References
[1] BOETTINGER W J, CORIELL S R, TRIVEDI R. Rapid solidification processing: Principles and technologies Ⅳ [M]. Baton Rouge (LA): Claitor’s, 1988: 13-24.
[2] BACKER J C, CAHN J W. Solidification [M]. Ohio: American Society Metals, Metals Park, 1970: 23-58.
[3] AZIZ M J. Model for solute redistribution during rapid solidification [J]. Journal of Applied Physics, 1982, 53(2): 1158-1168.
[4] AZIZ M J, KAPLAN T. Continuous growth model for interface motion during alloy solidification [J]. Acta Metallurgica, 1988, 36(8): 2335-2347.
[5] TURNBULL D. On the relation between crystallization rate and liquid structure [J]. The Journal of Physical Chemistry, 1962, 66: 609-613.
[6] TRIVEDI R, KURZ W. Morphological stability of a planar interface under rapid solidification conditions [J]. Acta Metallurgica, 1986, 34(8): 1663-1670.
[7] ECKLER K, HERLACH D M, AZIZ M J. Search for a solute-drag effect in dendrite solidification [J]. Acta Metallurgica et Materialia, 1994, 42(3): 975-979.
[8] ECKLER K, COCHRANE R F, HERLACH D M, FEUERBACHER B, JURISCH M. Evidence for a transition from diffusion-controlled to thermally controlled solidification in metallic alloys [J]. Physical Review B, 1992, 45(9): 5019-5022.
[9] WILLNECKER R, HERLACH D M, FEUERBACHER B. Evidence of nonequilibrium processes in rapid solidification of undercooled melts [J]. Physical Review Letters, 1989, 62(23): 2707-2710.
[10] ARNOLDC B, AZIZ M J, SCHWARZ M, HERLACH D M. Parameter-free test of alloy dendrite-growth theory [J]. Physical Review B, 1999, 59(1): 334-343.
[11] HERLACH D M, GALENKO P K. Rapid solidification: In situ diagnostics and theoretical modeling [J]. Materials Science and Engineering: A, 2007, 449/451: 34-41.
[12] GALENKO P K, DANILOV D A. Local nonequilibrium effect on rapid dendritic growth in a binary alloy melt [J]. Physical Letters A, 1997, 235: 271-280.
[13] GALENKO P K, DANILOV D A. Model for free dendritic alloy growth under interfacial and bulk phase nonequilibrium conditions [J]. Journal of Crystal Growth, 1999, 197: 992-1002.
[14] SOBOLEV S L. Local-nonequilibrium model for rapid solidification of undercooled melts [J]. Physical Letters A, 1995, 199: 383-386.
[15] SOBOLEV S L. Influence of local nonequilibrium on the rapid solidification of binary alloys [J]. Technical Physics, 1998, 43(3): 307-313.
[16] SOBOLEV S L. Rapid solidification under local nonequilibrium conditions [J]. Physical Review E, 1997, 55(6): 6845-6854.
[17] DIVENUTI A G, ANDO T. A free dendritic growth model accommodating curved phase boundaries and high Peclet number conditions [J]. Metallurgical and Materials Transactions A, 1998, 29: 3047-3056.
[18] GALENKO P. Extended thermodynamical analysis of a motion of the solid-liquid interface in a rapidly solidifying alloy [J]. Physical Review B, 2002, 65: 144103.
[19] CORIELL S R, TURNBULL D. Relative roles of heat transport and interface rearrangement rates in the rapid growth of crystals in undercooled melts [J]. Acta Metallurgica, 1982, 30: 2135-2139.
[20] WANG Hai-fang, LIU Feng, CHEN Zheng, YANG Gen-cang, ZHOU Yao-he. Analysis of non-equilibrium dendrite growth in a bulk undercooled alloy melt: Model and application [J]. Acta Materialia, 2007, 55: 497-506.
[21] MULLINS W W, SEKERKA R F. Stability of a planar interface during solidification of a dilute binary alloy [J]. Journal of Applied Physics, 1964, 35(2): 444-451.
[22] GALENKO P K, DANILOV D A. Linear morphological stability analysis of the solid-liquid interface in rapid solidification of a binary system [J]. Physical Review E, 2004, 69: 051608.
Foundation item: Projects(50501020, 50395103, 50431030) supported by the National Natural Science Foundation of China; Project(NCET-05-870) supported by Program for New Century Excellent Talents in Chinese University; Project(CX200706) supported by the Doctorate Foundation of Northwestern Polytechnical University, China
Corresponding author: LIU Feng; Tel: +86-29-88460374; E-mail: liufeng@nwpu.edu.cn
DOI: 10.1016/S1003-6326(09)60167-9
(Edited by YANG Bing)