AZ31þ�Ͻ�����þת��Ĥ�ĸ�ʴ����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���5��
�������ߣ�Nguyen Van PHUONG Manoj GUPTA Sungmo MOON
����ҳ�룺1087 - 1095
�ؼ��ʣ�þ�Ͻ�AZ31�Ͻ�����þ��ת��Ĥ����ʴ����
Key words��magnesium alloy; AZ31 alloy; magnesium phosphate; conversion coating; corrosion protection
ժ Ҫ��ͨ���ں�Mg2+�� ���ӵ���Һ�н��н��ݣ���AZ31þ�Ͻ����Ʊ�����þת��Ĥ����ǿ�俹��ʴ���ܡ���������20 min��AZ31þ�Ͻ��ϵ�����þת��Ĥ������״�ṹ����Ⱦ���(ԼΪ2.5 ��m)��X������������X���߹������������������þת��Ĥ������þ��������þ������þ��ɡ�����þת��Ĥ��AZ31þ�Ͻ����ǿ�ҵı������á�����þת��Ĥ�ĸ�ʴ����������ͣ�ԼΪδ��Ĥ���港ʴ������3%����0.5 mol/L NaCl��Һ�еĸ�ʴ��ʱ����10 min�ӳ���24 h��
Abstract: Magnesium phosphate conversion coating (MPCC) was fabricated on AZ31 magnesium alloy for corrosion protection by immersion treatment in a simple MPCC solution containing Mg2+ and ions. The MPCC on AZ31 Mg alloy showed micro-cracks structure and a uniform thickness with the thickness of about 2.5 ��m after 20 min of phosphating treatment. The composition analyzed by energy dispersive X-ray spectroscopy and X-ray photoelectron spectroscopy revealed that the coating consisted of magnesium phosphate and magnesium hydroxide/oxide compounds. The MPCC showed a significant protective effect on AZ31 Mg alloy. The corrosion current of MPCC was reduced to about 3% of that of the uncoated surface and the time for the deterioration process during immersion in 0.5 mol/L NaCl solution improved from about 10 min to about 24 h.
Trans. Nonferrous Met. Soc. China 27(2017) 1087-1095
Nguyen Van PHUONG1, Manoj GUPTA2, Sungmo MOON1,3
1. Korea Institute of Materials Science, Gyeongnam 51508, Korea;
2. Department of Mechanical Engineering, National University of Singapore 119260, Singapore;
3. Korea University of Science and Technology, Daejeon 34113, Korea
Received 7 April 2016; accepted 6 September 2016
Abstract: Magnesium phosphate conversion coating (MPCC) was fabricated on AZ31 magnesium alloy for corrosion protection by immersion treatment in a simple MPCC solution containing Mg2+ and  ions. The MPCC on AZ31 Mg alloy showed micro-cracks structure and a uniform thickness with the thickness of about 2.5 ��m after 20 min of phosphating treatment. The composition analyzed by energy dispersive X-ray spectroscopy and X-ray photoelectron spectroscopy revealed that the coating consisted of magnesium phosphate and magnesium hydroxide/oxide compounds. The MPCC showed a significant protective effect on AZ31 Mg alloy. The corrosion current of MPCC was reduced to about 3% of that of the uncoated surface and the time for the deterioration process during immersion in 0.5 mol/L NaCl solution improved from about 10 min to about 24 h.
ions. The MPCC on AZ31 Mg alloy showed micro-cracks structure and a uniform thickness with the thickness of about 2.5 ��m after 20 min of phosphating treatment. The composition analyzed by energy dispersive X-ray spectroscopy and X-ray photoelectron spectroscopy revealed that the coating consisted of magnesium phosphate and magnesium hydroxide/oxide compounds. The MPCC showed a significant protective effect on AZ31 Mg alloy. The corrosion current of MPCC was reduced to about 3% of that of the uncoated surface and the time for the deterioration process during immersion in 0.5 mol/L NaCl solution improved from about 10 min to about 24 h.
Key words: magnesium alloy; AZ31 alloy; magnesium phosphate; conversion coating; corrosion protection
1 IntroductionMagnesium alloys are very attractive materials for a number of applications in the automotive, aeronautic, electronic and recreational industries, owing to their low density, high specific strength, good castability, machinability and weldability [1]. However, one of the main challenges in the use of Mg alloys, particularly for outdoor applications, is its poor corrosion resistance [2-4]. The standard reduction potential of Mg is  =-2.356 V (vs SHE) at 25 ��C [3], which is the lowest among industrial engineering metals. Attempts have been made to improve the corrosion resistance of Mg alloys using chemical treatment, anodizing, plating, metal coatings and organic coating [5-7]. Among them, chemical conversion coatings (CCCs) are regarded as one of the most effective and cheapest ways to enhance the corrosion resistance of Mg alloys. CCCs are formed by precipitation of corrosion inhibiting chemicals onto the metal substrate from chemical conversion coating solution, which can protect the substrate by acting as a barrier between the metal surface and the environment [5,6].
=-2.356 V (vs SHE) at 25 ��C [3], which is the lowest among industrial engineering metals. Attempts have been made to improve the corrosion resistance of Mg alloys using chemical treatment, anodizing, plating, metal coatings and organic coating [5-7]. Among them, chemical conversion coatings (CCCs) are regarded as one of the most effective and cheapest ways to enhance the corrosion resistance of Mg alloys. CCCs are formed by precipitation of corrosion inhibiting chemicals onto the metal substrate from chemical conversion coating solution, which can protect the substrate by acting as a barrier between the metal surface and the environment [5,6].
The conventional CCCs are based on treatment solutions containing chromium compounds that have been shown to be highly toxic and carcinogens, and are now being restricted in the industry. Many types of CCCs have been reported as the potential replacements for conventional chromate conversion coatings on Mg alloys, including zinc phosphate, stannate, phosphate/ phosphate�Cmanganese, titanate, calcium phosphate, fluoride, rare-earth metal salt (RE), Mg-Al hydrotalcite, ionic liquid, molten salt, vanadium, hexafluorozirconic acid, stearic acid [8-42]. Among potential chromate replacements, phosphate conversion coatings have attracted significantly attention due to their low toxicity, insolubility in neutral pH solution and chemical stability [5-8]. Zinc phosphate conversion coatings have been successfully used as a primer coating on steels and aluminum alloys in automotive industries for many years due to the simplicity in operation, low-cost and low environmental impact. However, for Mg alloys, due to their high electrochemical activity, the zinc phosphate conversion coatings are limited in corrosion protection and still cannot satisfy modern industrial requirements. Other phosphate coating systems on Mg alloys have also been reported in a review by CHEN et al [8]. However, they own limitations in corrosion resistance, operations and cost, and still cannot satisfy for practical use.
Therefore, further investigation for the development of better, simpler, cheaper and environmentally friendly chemical conversion coating solution for Mg alloys is still needed.
Magnesium phosphate compounds such as Mg3(PO4)2 and MgHPO4��3H2O are insoluble in water and chemically stable. These compounds have been reported to be present as an inner layer of zinc, calcium or manganese phosphates conversion coating on Mg alloys [29-34]. The formation of the magnesium phosphate compounds has been explained by the precipitation reactions between Mg2+ and  ions at the near alloy surface during the conversion coating process. Therefore, this suggests that to improve the corrosion resistance of Mg alloys, a coating should contain magnesium phosphate compounds, which can be formed from the chemical conversion coating solution containing Mg2+ and
ions at the near alloy surface during the conversion coating process. Therefore, this suggests that to improve the corrosion resistance of Mg alloys, a coating should contain magnesium phosphate compounds, which can be formed from the chemical conversion coating solution containing Mg2+ and  ions, so-called magnesium phosphate conversion coating (MPCC) solution. At present, MPCC has been considered as a novel chemical conversion coating method on carbon steels [43-46]. The former MPCC mainly consists of newberyite (MgHPO4��3H2O) with a thickness of about 30 ��m which is about three times thicker than that of zinc phosphate conversion coating and provides two times longer stability under salt-spray conditions [45,46]. PHUONG and MOON [47] and ZHAO et al [48] have investigated MPCC on AZ31 Mg alloy and found that the corrosion resistance of MPCC was much better than that of zinc phosphate conversion coating, and about 20 times better than that of the bare surface. However, these studies are limited in the preparation process of MPCC and simply investigating the corrosion behavior using potentiodynamic polarization test. Thus, further work is required to fully understand the composition and corrosion behavior of MPCC on AZ31 Mg alloy.
ions, so-called magnesium phosphate conversion coating (MPCC) solution. At present, MPCC has been considered as a novel chemical conversion coating method on carbon steels [43-46]. The former MPCC mainly consists of newberyite (MgHPO4��3H2O) with a thickness of about 30 ��m which is about three times thicker than that of zinc phosphate conversion coating and provides two times longer stability under salt-spray conditions [45,46]. PHUONG and MOON [47] and ZHAO et al [48] have investigated MPCC on AZ31 Mg alloy and found that the corrosion resistance of MPCC was much better than that of zinc phosphate conversion coating, and about 20 times better than that of the bare surface. However, these studies are limited in the preparation process of MPCC and simply investigating the corrosion behavior using potentiodynamic polarization test. Thus, further work is required to fully understand the composition and corrosion behavior of MPCC on AZ31 Mg alloy.
Accordingly, in this work, MPCC was prepared in a chemical conversion coating solution containing Mg2+ and  ions. The coating was characterized by observation of surface and cross-sectional morphologies, and analyzing compositions using energy dispersive X-ray spectroscopy (EDS) and X-ray photoelectron spectrometer (XPS). The corrosion behavior of MPCC was studied by the open-circuit potential (OCP) measurement, potentiodynamic polarization analysis, electrochemical impedance spectroscopy (EIS) measure- ments and also, immersion test in NaCl solution.
ions. The coating was characterized by observation of surface and cross-sectional morphologies, and analyzing compositions using energy dispersive X-ray spectroscopy (EDS) and X-ray photoelectron spectrometer (XPS). The corrosion behavior of MPCC was studied by the open-circuit potential (OCP) measurement, potentiodynamic polarization analysis, electrochemical impedance spectroscopy (EIS) measure- ments and also, immersion test in NaCl solution.
2 Experimental
AZ31 Mg alloy (Posco, Korea) with composition of Al 2.9%, Zn 0.8%, Mn 0.3%, Si<0.1%, Fe<0.005%, Cu<0.05%, Ni<0.005% (mass fraction). And Mg balance, was used in this work. The samples of 50 mm �� 50 mm �� 2 mm were cut from a rolled AZ31 sheet. The samples were ground in ethanol up to 4000 grit SiC abrasive papers and then rinsed with ethanol.
MPCC was applied on AZ31 by immersion treatment of samples for 20 min in the solution containing 0.1 mol/L of Mg(OH)2 and 0.24 mol/L of H3PO4 at 45 ��C. The formation and growth of MPCC on AZ31 were studied by open circuit potential (OCP) measurement during the phosphating process. Surface morphology, cross-sectional morphology and analysis using energy dispersive X-ray spectroscopy (EDS) were performed using scanning electron microscope (SEM) (Jeol, Japan), operated at an acceleration voltage of 20 kV. X-ray photoelectron spectroscopy (XPS) analysis was carried out on a MultiLab 2000 spectrometer (Thermo Scientific, US) equipped with Al K�� X-ray source, operated at 300 W. The spectra of Mg 2p and O 1s were recorded. The binding energies were referred to the C 1s binding energy at 285.01 eV.
Electrochemical measurements were performed using a computer-controlled potentiostat and a conventional three-electrode cell (Zahner, Germany) with an exposed working electrode area of 1 cm2, using a saturated calomel electrode (SCE) and a platinum sheet as the reference and counter electrodes, respectively. Electrochemical experiments were carried out in 0.1 mol/L NaCl at 25 ��C. Potentiodynamic polarization tests were performed at a scan rate of 1 mV/s on the bare and the MPCC sample after 1 h of exposure to 0.1 mol/L NaCl solution. EIS measurements were measured separately and carried out at the open circuit potential in the frequency range of 100 kHz to 10 mHz with 5 points/decade with an applied sinusoidal signal of 5 mV. A parameter indicative of corrosion resistance, Rcorr, was fitted from the spectra using a complex non-linear least squares fitting program (Thales Z2.12, Germany). The immersion test was conducted in 0.5 mol/L NaCl solution at (25 ��1) ��C.
3 Results and discussion
3.1 Formation of MPCC
The formation of MPCC from selected MPCC solution can be explained by using the thermodynamic stability diagram, which shows [Mg2+] and pH levels for precipitation of magnesium phosphate/hydroxide compounds (Fig. 1). The selected MPCC solution at [Mg2+]=0.1 mol/L (or lg[Mg2+]=-1) and pH 3.2 is indicated by asterisk (*), suggesting that it will exist primarily in the form of soluble  ions. The immersion of the AZ31 sample in the acidic MPCC solution induces spontaneous Mg ionization (reaction 1) and hydrogen evolution (reaction 2) on the alloy surface, by which magnesium ions and hydrogen gas bubbles are produced, respectively.
ions. The immersion of the AZ31 sample in the acidic MPCC solution induces spontaneous Mg ionization (reaction 1) and hydrogen evolution (reaction 2) on the alloy surface, by which magnesium ions and hydrogen gas bubbles are produced, respectively.
Mg��Mg2+(aq)+2e (1)
2H++2e��H2(g) (2)
The generation of Mg2+ and the consumption of H+ ions increase both Mg2+ concentration and pH at the near metal surface. As seen in Fig. 1, with increasing Mg2+ concentration and pH, several magnesium compounds such as MgHPO4��3H2O, Mg3(PO4)2 and Mg(OH)2/MgO can be formed (reactions (3-6)).
 +OH�C+2H2O�� MgHPO4��3H2O(s) (3)
+OH�C+2H2O�� MgHPO4��3H2O(s) (3)
 +4OH�C�� Mg3(PO4)2(s)+
+4OH�C�� Mg3(PO4)2(s)+ +4H2O (4)
+4H2O (4)
 +4OH�C��Mg(OH)2(c)+
+4OH�C��Mg(OH)2(c)+ +2H2O (5)
+2H2O (5)
Mg(OH)2��MgO+H2O (6)
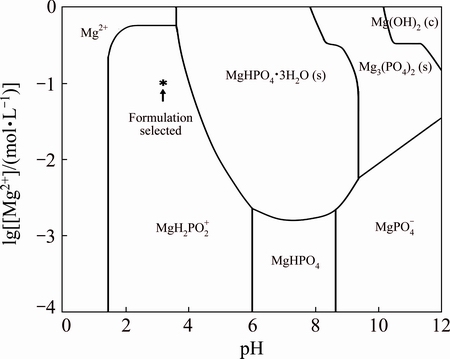
Fig. 1 Thermodynamic stability diagram showing [Mg2+] and pH levels for precipitation of magnesium phosphate compounds, calculated using MEDUSA software package [49] at  =0.24 mol/L, [Mg2+] from 10-4 to 1 mol/L and pH from 0 to 12
=0.24 mol/L, [Mg2+] from 10-4 to 1 mol/L and pH from 0 to 12
Figure 2 shows the OCP transient of the AZ31 Mg alloy immersed in the MPCC solution for 20 min. The shifting of OCP towards a more positive value indicates that the formation process of coating occurs and the coating formed is more thermodynamically stable than the original surface. The OCP transient can be divided into two stages, as indicated in Fig. 2. In the first stage up to 3 min of immersion time, the OCP was rapidly increased from an initial value of approximately -1.92 V to about -1.45 V (vs SCE), which indicated the precipitation and rapid growing process of the coating on the surface. During the second stage, after 3 min of immersion time, the OCP slightly increased, which suggests that the thickness of the MPCC is being further developed. Some potential fluctuations observed in the second stage are associated with the processes of dissolution and repassivation at some parts of the coating during the conversion coating process.
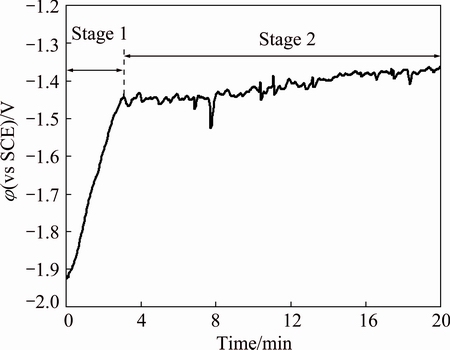
Fig. 2 OCP transient of AZ31 Mg alloy in MPCC solution at 45 ��C
3.2 Surface characterization of MPCC
Figures 3(a) and (b) show SEM images of the surface and cross-sectional morphologies of MPCC coated AZ31 after 20 min of treatment time. The MPCC on AZ31 exhibited micro-cracked structure. The micro-cracked structure is commonly seen on the surface of chemical conversion coated Mg alloys such as with chromium, permanganate and cerium conversion coatings [5,10-13]. Cracks are likely due to the lower molar volume of MPCC resulting from dehydration during the immersion treatment. The coating was otherwise smooth and exhibited a uniform thickness of about 2.5 ��m after 20 min of phosphating treatment.
Figure 3(c) shows the EDS area analyses of the MPCC on AZ31. It was found that the coating consists of Mg (62.11 %), O (31.73 %), P (3.21 %), Al (2.68 %) and Zn (0.27 %). Thus, this result suggests that the coating was mainly composed of MgO/Mg(OH)2 and Mg-PO4 compounds.
To provide better understanding of the chemical composition of MPCC on AZ31, XPS was used to study the surface bonding of the coating (Fig. 4). XPS survey spectrum of coating found that the main constituents of the coating surface included magnesium, aluminum, zinc, oxygen and phosphorous species (Fig. 4(a)). High- resolution XPS scan (Fig. 4(b)) further shows the Mg 2p binding energies appearing at approximately peaks of 50.19, 51.16 and 52.13 eV, which correspond with the bonding of MgO, Mg(OH)2 and Mg-PO4, respectively. For the O 1s binding energy (Fig. 4(c)), three peaks at binding energies of 531.16, 532.39 and 533.67 eV were fitted, which correspond with the bonding of metal oxide (MgO), hydroxide (Mg(OH)2) and phosphate (PO4) compounds, respectively. Thus, both EDS and XPS analyses indicated that the MPCC on AZ31 was composed of MgO/Mg(OH)2 and Mg-PO4 compounds, such as MgHPO4��3H2O and Mg3(PO4)2.

Fig. 3 SEM images of surface (a) and cross-sectional morphologies (b), and EDS area analysis (c) of MPCC on AZ31 after 20 min treatment time in MPCC solution at 45 ��C
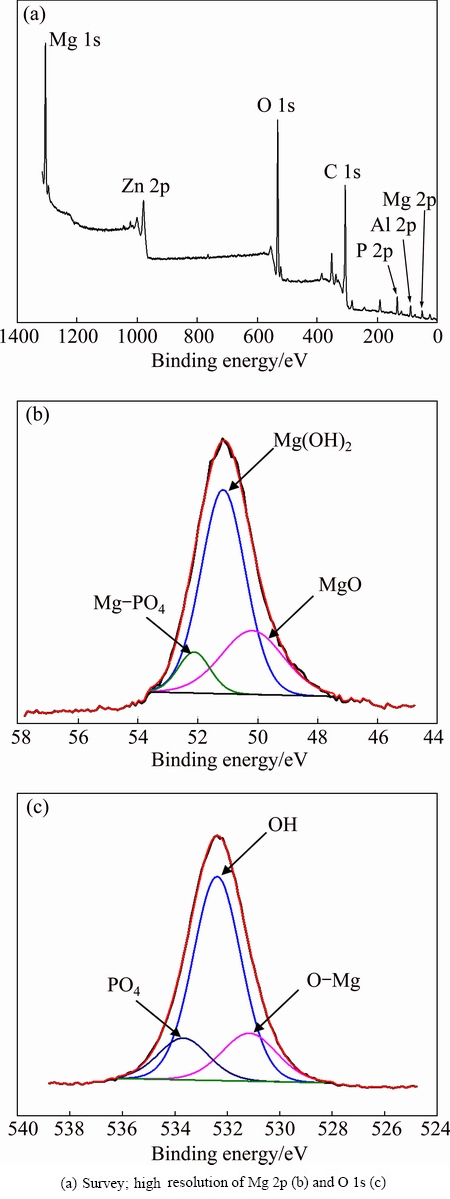
Fig. 4 XPS analyses of MPCC coated AZ31
3.3 Corrosion studies
Figure 5 shows the OCPs of bare and MPCC coated AZ31 as a function of time immersed in 0.1 mol/L NaCl solution at 25 ��C. The measurement of OCP transients is typically used to study some aspects of the chemical stability and corrosion processes of the surface layers on Mg alloys. It is the most rapid and sensitive way of detecting deterioration processes of the film, and the localized corrosion of Mg alloys in a corrosive solution. Localized corrosion of Mg alloys in an electrolyte containing chloride ions is characterized by the appearance of roughly circular blackened regions (pits), which expand radially with time and vigorously evolve hydrogen [3,4]. The corrosion reaction of Mg alloys in aqueous environments generally progresses by electrochemical reaction with water to produce hydrogen gas and magnesium hydroxide (reactions (7-10)) [3-6]:
Mg(s)��Mg2+(aq)+2e (anodic reaction) (7)
2H2O+2e��H2+2OH�C(aq) (cathodic reaction) (8)
Mg2+(aq)+2OH�C(aq)��Mg(OH)2(s) (product formation) (9)
Mg+2H2O��Mg(OH)2(s)+H2 (overall reaction) (10)
The hydroxide film formed on Mg alloys is much less stable than the passive films formed on aluminum and stainless steels [3-6]. Therefore, Mg alloys show poor corrosion resistance.
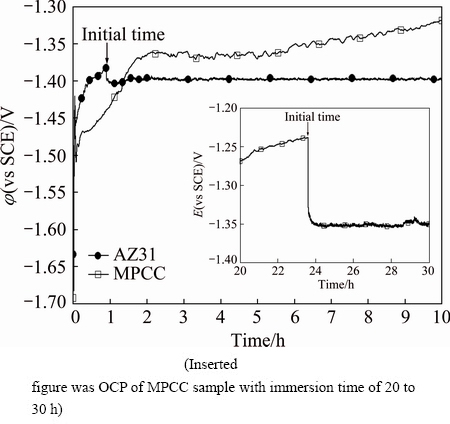
Fig. 5 OCPs of bare and MPCC coated AZ31 with immersion time immersed in 0.1 mol/L NaCl solution at 25 ��C
The OCP transient of bare AZ31 in 0.1 mol/L NaCl solution disclosed two stages. In the first stage, within 1 h of immersion, the OCP increased from an initial negative value (about -1.6 V (vs SCE)) towards a more positive value (about -1.38 V (vs SCE)), indicating the formation and growth process of Mg(OH)2 film on the AZ31 surface [3,4]. In the second stage, after 1 h of immersion, the OCP dropped suddenly from -1.38 V (vs SCE) to about -1.4 V (vs SCE), indicating the deterioration of the hydroxide film on the surface, leading to a localized corrosion reaction.
In contrast, the OCP transient of the MPCC sample was divided into three stages: a rapid increase from -1.5 to -1.36 V (vs SCE) within 2 h; a slight increase between 2 h and 24 h, dropped to a stable value of about -1.35 V (vs SCE) after about 24 h. In the first stage, the increased OCP indicates the decreased chemical activity on the MPCC AZ31 surface. This can be explained by the sealing effect produced by the MPCC during immersion in the NaCl solution. Since the MPCC on the AZ31 included micro-cracks (Fig. 3), the electrolyte could reach the substrate to react with the Mg alloy. The corroded product Mg(OH)2 (reaction (9)) is formed, and it can seal the cracks which resulted in the rapid increase of OCP during the first 2 h of immersion (Fig. 5). In the second stage, the slow increase in OCP suggested that a dynamic equilibrium between the formation and dissolution of the film was reached. In the third stage, the OCP suddenly dropped, indicating that the film had damaged and the continuous corrosion reaction occurred on the surface. Thus, the initial pitting corrosion of MPCC was 24 h, which is much longer than that of the bare surface (about 1 h).
Figure 6 shows the potentiodynamic polarization curves of bare and MPCC on AZ31 obtained after 1 h exposure to 0.1 mol/L NaCl solution at 25 ��C. The bare AZ31 surface had a more negative corrosion potential (��corr=-1.61 V (vs SCE)) than that of the MPCC sample (��corr=-1.41 V (vs SCE)). The corrosion current density (Jcorr) of MPCC sample was 6.9��10�C3 mA/cm2, which was reduced to about 3% of the bare surface (Jcorr=2.23��10�C1 mA/cm2). Compared to zinc phosphate conversion coatings, MPCC showed more negative corrosion potential and had a little higher corrosion current density, but the MPCC was much more stable than zinc phosphate conversion coatings against corrosion under the salt-spray conditions, due to its compact coating [47].
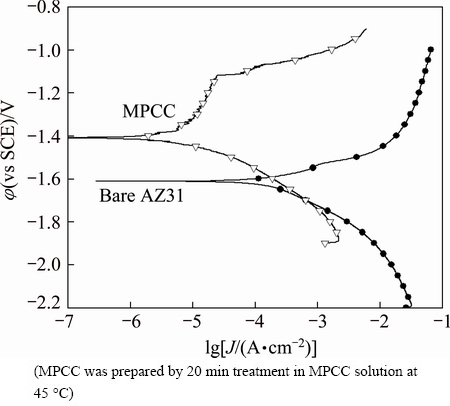
Fig. 6 Potentiodynamic polarization curves of bare and MPCC coated AZ31 samples at scan rate of 1 mV/s in 0.1 mol/L NaCl
To further understand the corrosion behavior and the associated deterioration process of the bare and MPCC on AZ31, EIS was employed. Figure 7(a) shows the Nyquist plots of the bare AZ31 obtained after 0 min, 20 min, 1 h, 3 h, 10 h and 20 h exposure to 0.1 mol/L NaCl solution at 25 ��C. EIS experiments of the bare AZ31 showed two capacitive loops at the high and middle frequencies (HF and MF) in combination with an inductive loop at low frequencies (LF). HF capacitive loops are usually attributed to both charge transfer and surface film effect [50,51]. MF capacitive loops are attributed to the relaxation of mass transport in the growing solid oxide phase. LF inductive loops are attributed to the phenomena of adsorption and desorption of Mg+ species on the surface of the Mg substrate, suggesting the slow corrosion reaction at the interface of AZ31 [11,14,50,51]. The typical EIS experiments of MPCC coated AZ31 consisted of two capacitive loops at HF and MF. According to SEM surface and cross- sectional morphologies, the MPCC on AZ31 consisted of cracks, which reach deeply to the metal surface (Fig. 3). The cracks can be sealed by the corrosion product during immersion in the NaCl solution to form layers: a sealed layer and an unsealed layer. Thus, these two layers correspond to the two capacitive loops of the EIS experiment. The appearance of an inductive loop in the LF region after 24 h immersion is attributed to the localized corrosion of AZ31 due to the deterioration process of the MPCC [52].
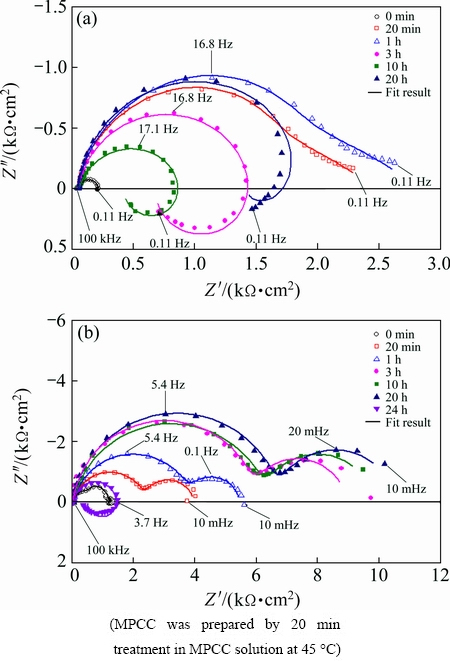
Fig. 7 Nyquist plots of bare (a) and MPCC coated (b) AZ31 samples after different immersion time in 0.1 mol/L NaCl solution at (25��1) ��C
Based on the impedance plots, the microstructure of MPCC, and the EIS studies of bare Mg alloy [50-52], two appropriate equivalent circuits were proposed for fitting these plots, as shown in Fig. 8. The equivalent circuits consist of two R/CPE components in series with Rs, with or without inductive loop (R/L). The equivalent circuit presented in Fig. 8(a) was used to fit the EIS spectrum of the bare AZ31. The element Rs was the solution resistance, the R1/CPE1 and R2/CPE2 pairs were suggested to represent two capacitive loops at HF and MF as explained above. The R3/L pair was suggested to represent inductive loops. The equivalent circuit presented in Fig. 8(b) was used to fit the EIS spectra of the MPCC coated AZ31. The Rf1/CPEf1 and Rf2/CPEf2 pairs were suggested to represent the two layers of MPCC during immersion in NaCl solution. The model presented in Fig. 8(a) with an inductive loop was also used to fit the EIS spectra of MPCC on AZ31 with immersion time 24 h due to the occurrence of the localized corrosion process. The fittings were performed by using the Thales Z2.12 software and the fitted results are drawn as solid lines, together with experimental data, in Fig. 7.
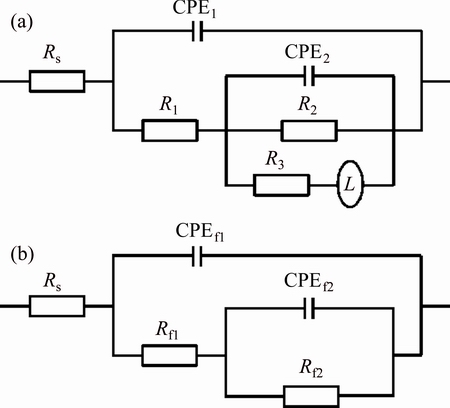
Fig. 8 Equivalent circuit models for simulation of Nyquist plots
Considering the protective film formed on the bare and MPCC coated AZ31, the magnitudes of R1, R2, Rf1 and Rf2 were plotted with immersion time and shown in Fig. 9. For AZ31, the film resistance (R1) is much larger than the mass transport resistance (R2). The increase of R1 during the first 1 h immersion indicates the growth of hydroxide film on AZ31. However, after an immersion time longer than 1 h, the rapid decrease of R1 indicates the process of deterioration of the hydroxide film, where the localized corrosion occurs on the surface. The slight increase of R1 with immersion time up to 20 h indicates that the surface of the AZ31 was completely covered by the corrosion product.
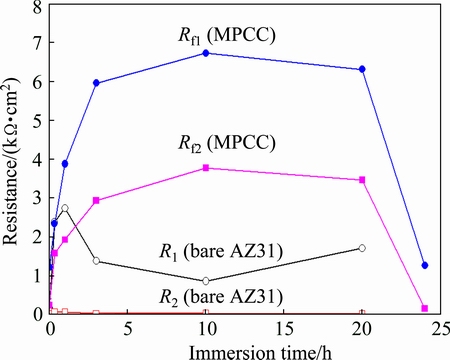
Fig. 9 Resistances of bare and MPCC coated AZ31 resulted from fitting on EIS spectrum by equivalent circuit models in Fig. 8

Fig. 10 Photographs of (a) bare and (b) MPCC coated AZ31 for 20 min with immersion time in 0.5 mol/L NaCl solution at (25��1) ��C
In contrast to bare AZ31, both Rf1 and Rf2 of the MPCC coated AZ31 rapidly increased to much higher value with increasing the immersion time from 0 to 3 h in 0.1 mol/L NaCl solution. As explained before, the corrosion product can be formed and seal the cracks of MPCC to increase the coating resistance. The sealing effect occurred strongly during the first 3 h of immersion in the NaCl solution, and became stabilized with immersion time longer than 3 h. After 24 h immersion, the sudden decreases of both Rf1 and Rf2 indicate the deterioration of MPCC and the initiation of localized corrosion on the surface. This result is in good agreement with OCP measurement (Fig. 5), where, OCP is rapidly increased within the first 3 h, and then, dropped after 24 h of immersion.
Figure 10 shows photographs of bare and MPCC coated AZ31 with immersion time in 0.5 mol/L NaCl solution at 25 ��C. The bare AZ31 showed pitting corrosion with a pit initiation time of about 10 min. The corroded sites rapidly expanded to the entire surface within 3 h immersion. In contrast, the MPCC coated AZ31 showed a filiform corrosion with a much longer pit initiation time of about 24 h. Thus, the immersion test again revealed that the MPCC can significantly increase the deterioration process of AZ31 magnesium alloy in NaCl solution.
4 Conclusions
1) Magnesium phosphate conversion coating can be successfully applied on AZ31 using a solution containing Mg2+ and  ions.
ions.
2) The results of characterization studies show that the coating consists of magnesium phosphate and magnesium hydroxide/oxide compounds with a thickness of about 2.5 ��m after 20 min of phosphating treatment. Cracks observed can be sealed by corrosion products during immersion in NaCl solution.
3) The coating shows a significant protective effect. The corrosion current measured by potentiodynamic polarization curve is reduced to about 3% of that of the bare surface. During the immersion test in 0.5 mol/L NaCl solution, the pitting corrosion was observed after about 24 h for MPCC, which is much longer than that of the bare surface (about 10 min).
Acknowledgements
This research was financially supported by a research grant from Korea Institute of Materials Science (PNK4652).
References
[1] FRIEDRICH H E, MORDIKE B L. Magnesium technology: Metallurgy, design data, applications [M]. Springer, Berlin, 2006.
[2] MAKAR G L, KRUGER J, JOSHI A. Advances in magnesium alloys and composites [C]//International Magnesium Association and the Non-Ferrous Metals Committee, Phoenix, Arizona, US, TMS, 1998.
[3] CRC handbook of chemistry and physics [M]. 60 ed. CRC Press Inc, 1980: 81.
[4] SONG G L, ATRENS A. Understanding magnesium corrosion [J]. Advanced Engineering Materials, 2003, 5: 837-858.
[5] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys��A critical review [J]. Journal of Alloys and Compounds, 2002, 33: 88-113.
[6] CZERWINSKI F. Magnesium alloys��Corrosion and surface treatments [M]. Croatia, InTech, 2011.
[7] CHEN X B, YANG H Y, ABBOTT T B, EASTON M A, BIRBILIS N. Corrosion-resistance electrochemical platings on magnesium alloys: A state-of-the-art review [J]. Corrosion, 2011, 68: 518-535.
[8] CHEN X B, BIRBILIS N, ABBOTT T B. Review of corrosion- resistance conversion coating for magnesium and its alloys [J]. Corrosion, 2011, 67: 1-16.
[9] ELSENTRIECY H H, AZUMI K, KONNO H. Effects of pH and temperature on the deposition properties of stannate chemical conversion coatings formed by the potentiostatic technique on AZ91 D magnesium alloy [J]. Electrochimica Acta, 2008, 53: 4267-4275.
[10] LIU W, XU D D, DUAN X Y, ZHAO G S, CHANG L M, LI X. Structure and effects of electroless Ni-Sn-P transition layer during acid electroless plating on magnesium alloys [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1506-1516.
[11] LEE Y L, CHU Y R, LI W C, LIN C S. Effect of permanganate concentration on the formation and properties of phosphate/ permanganate conversion coating on AZ31 magnesium alloy [J]. Corrosion Science, 2013, 70: 74-81.
[12] ARDELEAN H, FRATEUR I, MARCUS P. Corrosion protection of magnesium alloys by cerium, zirconium and niobium-based conversion coatings [J]. Corrosion Science, 2008, 50: 1907-1918.
[13] WANG C, ZHU S L, JIANG F, WANG F H. Cerium conversion coatings for AZ91D magnesium alloy in ethanol solution and its corrosion resistance [J]. Corrosion Science, 2009, 51: 2916-2923.
[14] WAN T T, LIU Z X, BU M Z, WANG P C. Effect of surface pretreatment on corrosion resistance and bond strength of magnesium AZ31 alloy [J]. Corrosion Science, 2013, 66: 33-42.
[15] CHIU K Y, WONG M H, CHENG F T, MAN H C. Characterization and corrosion studies of fluoride conversion coating on degradable Mg implants [J]. Surface & Coatings Technology, 2007, 202: 590-598.
[16] SONG Y, SHAN D, CHEN R, ZHANG F, HAN E H. Formation mechanism of phosphate conversion film on Mg-8.8Li alloy [J]. Corrosion Science, 2009, 51: 62-69.
[17] ZHOU W Q, SHAN D Y, HAN E -H, KE W. Structure and formation mechanism of phosphate conversion coating on die-cast AZ91D magnesium alloy [J]. Corrosion Science, 2008, 50: 329-337.
[18] ZENG R C, LIU Z G, ZHANG F, LI S Q, HE Q K, CUI H Z, HAN E H. Corrosion resistance of in-situ Mg-Al hydrotalcite conversion film on AZ31 magnesium alloy by one-step formation [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1917-1925.
[19] LI J K, UAN J Y. Formation of Mg, Al-hydrotalcite conversion coating on Mg alloy in aqueous  and corresponding protection against corrosion by the coating [J]. Corrosion Science, 2009, 51: 1181-1188.
and corresponding protection against corrosion by the coating [J]. Corrosion Science, 2009, 51: 1181-1188.
[20] ZENG R C , HU Y, ZHANG F, HUANG Y D, WANG Z L, LI S Q, HAN E H. Corrosion resistance of cerium-doped zinc calcium phosphate chemical conversion coatings on AZ31 magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 865-873.
[21] CUI X F, LI Q F, LI Y, WANG F H, JIN G, DING M H. Microstructure and corrosion resistance of phytic acid conversion coatings for magnesium alloy [J]. Applied Surface Science, 2008, 255: 2098-2103.
[22] NG W F, WONG M H, CHENG F T. Stearic acid coating on magnesium for enhancing corrosion resistance in Hanks�� solution [J]. Surface & Coatings Technology, 2010, 204: 1823-1830.
[23] CHEN J, SONG Y, SHAN D Y, HAN E H. In situ growth of Mg-Al hydrotalcite conversion film on AZ31 magnesium alloy [J]. Corrosion Science, 2011, 53: 3281-3288.
[24] CHEN J, SONG Y, SHAN D Y, HAN E H. Study of the corrosion mechanism of the in situ grown Mg-Al- hydrotalcite film on AZ31 alloy [J]. Corrosion Science, 2012, 65: 268-277.
hydrotalcite film on AZ31 alloy [J]. Corrosion Science, 2012, 65: 268-277.
[25] CUI X J, YANG R S, LIU C H, YU Z X, LIN X Z. Structure and corrosion resistance of modified micro-arc oxidation coating on AZ31B magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 814-821.
[26] ADHIKARI S, UNOCIC K A, ZHAI Y, FRANKEL G S, ZIMMERMAN J, FRISTAD W. Hexafluorozirconic acid based surface pretreatments: Characterization and performance assessment [J]. Electrochimica Acta, 2011, 56: 1912-1924.
[27] HE M F, LIU L, WU Y T, TANG Z X, HU W B. Corrosion properties of surface-modified AZ91D magnesium alloy [J]. Corrosion Science, 2008, 50: 3267-3273.
[28] PHUONG N V, LEE K H, CHANG D, KIM M, LEE S, MOON S. Zinc phosphate conversion coatings on magnesium alloys��A review [J]. Metals and Materials International, 2013, 19: 273-281.
[29] PHUONG N V, MOON S, CHANG D, LEE K H. Effect of microstructure on the zinc phosphate conversion coatings on magnesium alloy AZ91 [J]. Applied Surface Science, 2012, 264: 70-78.
[30] PHUONG N V, LEE K H, CHANG D, MOON S. Effects of Zn2+ concentration and pH on the zinc phosphate conversion coatings on AZ31 magnesium alloy [J]. Corrosion Science, 2013, 74: 314-322.
[31] KOUISNI L, AZZI M, ZERTOUBI M, DALARD F. Phosphate coatings on magnesium alloy AM60 part 1: Study of the formation and the growth of zinc phosphate films [J]. Surface & Coatings Technology, 2004, 185: 58-67.
[32] KOUISNI L, AZZI M, DALARD F, MAXIMOVITCH S. Phosphate coatings on magnesium alloy AM60: Part 2: Electrochemical behaviour in borate buffer solution [J]. Surface & Coatings Technology, 2005, 192: 239-246.
[33] LI Q, XU S, HU J, ZHANG S, ZHONG X, YANG X. The effects to the structure and electrochemical behavior of zinc phosphate conversion coatings with ethanolamine on magnesium alloy AZ91D [J]. Electrochimica Acta, 2010, 55: 887-894.
[34] CHEN X B, BIRBILIS N, ABBOTT T B. Effect of [Ca2+] and  levels on the formation of calcium phosphate conversion coatings on die-cast magnesium alloy AZ91D [J]. Corrosion Science, 2012, 55: 226-232.
levels on the formation of calcium phosphate conversion coatings on die-cast magnesium alloy AZ91D [J]. Corrosion Science, 2012, 55: 226-232.
[35] LI G Y, LIAN J S, NIU L Y, JIANG Z H, JIANG Q. Growth of zinc phosphate coatings on AZ91D magnesium alloy [J]. Surface & Coatings Technology, 2006, 201: 1814-1820.
[36] ZOU B,  G H, ZHANG G L, TIAN Y Y. Effect of current frequency on properties of coating formed by microarc oxidation on AZ91D magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1500-1505.
G H, ZHANG G L, TIAN Y Y. Effect of current frequency on properties of coating formed by microarc oxidation on AZ91D magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1500-1505.
[37] NIU L Y, JIANG Z H, LI G Y, GU C D, LIAN J S. A study and application of zinc phosphate coating on AZ91D magnesium alloy [J]. Surface & Coatings Technology, 2006, 200: 3021-3026.
[38] AMINI R, SARABI A A. The corrosion properties of phosphate coating on AZ31 magnesium alloy: The effect of sodium dodecyl sulfate (SDS) as an eco-friendly accelerating agent [J]. Applied Surface Science, 2011, 257: 7134-7139.
[39] LI G Y, LIAN J S, NIU L Y, JIANG Z H. Influence of pH of phosphating bath on the zinc phosphate coating on AZ91D magnesium alloy [J]. Advanced Engineering Materials, 2006, 8: 123-127.
[40] ZENG R C, LAN Z D, KONG L H, HUANG Y D, CUI H Z. Characterization of calcium-modified zinc phosphate conversion coatings and their influences on corrosion resistance of AZ31 alloy [J]. Surface & Coatings Technology, 2011, 205: 3347-3355.
[41] ZENG R C, ZHANG F, LAN Z D, CUI H Z, HAN E H. Corrosion resistance of calcium-modified zinc phosphate conversion coatings on magnesium�Caluminium alloys [J]. Corrosion Science, 2014, 88: 452-459.
[42] ZENG R C, SUN X X, SONG Y W, ZHANG F, LI S Q, CUI H Z, HAN E H. Influence of solution temperature on corrosion resistance of Zn-Ca phosphate conversion coating on biomedical Mg-Li-Ca alloys [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 3293-3299.
[43] MORKS M F. Magnesium phosphate treatment for steel [J]. Materials Letters, 2004, 58: 3316-3319.
[44] ISHIZAKI T, SHIGEMATSU I, SAITO N. Anticorrosive magnesium phosphate coating on AZ31 magnesium alloy [J]. Surface & Coatings Technology, 2009, 203: 2288-2291.
[45] FOULADI M, AMADEH A. Comparative study between novel magnesium phosphate and traditional zinc phosphate coatings [J]. Materials Letters, 2013, 98: 1-4.
[46] FOULADI M, AMADEH A. Effect of phosphating time and temperature on microstructure and corrosion behavior of magnesium phosphate coating [J]. Electrochimica Acta, 2013, 103: 1-12.
[47] PHUONG N V, MOON S. Comparative corrosion study of zinc phosphate and magnesium phosphate conversion coatings on AZ31 Mg alloy [J]. Materials Letters, 2014, 122: 341-344.
[48] ZHAO H, CAI S, DING Z, ZHANG M, LI Y, XU G. A simple method for the preparation of magnesium phosphate conversion coatings on a AZ31 magnesium alloy with improved corrosion resistance [J]. RSC Advances, 2015, 5: 24586-24590.
[49] PUIGDOMENECH I. Hydra/medusa chemical equilibrium database and plotting software [J]. KTH Royal Institute of Technology, 2004.
[50] TURHAN M C, LYNCH R, KILLIAN M S, VIRTANEN S. Effect of acidic etching and fluoride treatment on corrosion performance in Mg alloy AZ91D (MgAlZn) [J]. Electrochimica Acta, 2009, 55: 250-257.
[51] KING A D, BIRBILIS N, SCULLY J R. Accurate electrochemical measurement of magnesium corrosion rates: A combined impedance, mass-loss and hydrogen collection study [J]. Electrochimica Acta, 2014, 121: 394-406.
[52] LIANG J, SRINIVASAN P B, BLAWERT C, DIETZEL W. Comparison of electrochemical corrosion behaviour of MgO and ZrO2 coatings on AM50 magnesium alloy formed by plasma electrolytic oxidation [J]. Corrosion Science, 2009, 51: 2483-2492.
Nguyen Van PHUONG1, Manoj GUPTA2, Sungmo MOON1,3
1. Korea Institute of Materials Science, Gyeongnam 51508, Korea;
2. Department of Mechanical Engineering, National University of Singapore 119260, Singapore;
3. Korea University of Science and Technology, Daejeon 34113, Korea
ժ Ҫ��ͨ���ں�Mg2+�� ���ӵ���Һ�н��н��ݣ���AZ31þ�Ͻ����Ʊ�����þת��Ĥ����ǿ�俹��ʴ���ܡ���������20 min��AZ31þ�Ͻ��ϵ�����þת��Ĥ������״�ṹ����Ⱦ���(ԼΪ2.5 ��m)��X������������X���߹������������������þת��Ĥ������þ��������þ������þ��ɡ�����þת��Ĥ��AZ31þ�Ͻ����ǿ�ҵı������á�����þת��Ĥ�ĸ�ʴ����������ͣ�ԼΪδ��Ĥ���港ʴ������3%����0.5 mol/L NaCl��Һ�еĸ�ʴ��ʱ����10 min�ӳ���24 h
���ӵ���Һ�н��н��ݣ���AZ31þ�Ͻ����Ʊ�����þת��Ĥ����ǿ�俹��ʴ���ܡ���������20 min��AZ31þ�Ͻ��ϵ�����þת��Ĥ������״�ṹ����Ⱦ���(ԼΪ2.5 ��m)��X������������X���߹������������������þת��Ĥ������þ��������þ������þ��ɡ�����þת��Ĥ��AZ31þ�Ͻ����ǿ�ҵı������á�����þת��Ĥ�ĸ�ʴ����������ͣ�ԼΪδ��Ĥ���港ʴ������3%����0.5 mol/L NaCl��Һ�еĸ�ʴ��ʱ����10 min�ӳ���24 h
�ؼ��ʣ�þ�Ͻ�AZ31�Ͻ�����þ��ת��Ĥ����ʴ����
(Edited by Yun-bin HE)
Corresponding author: Sungmo MOON; Tel: +82-55-280 3549; E-mail: Sungmo@kims.re.kr
DOI: 10.1016/S1003-6326(17)60127-4

