
Effective removal of heavy metals from aqueous solutions by orange peel xanthate
LIANG Sha(梁 莎)1, GUO Xue-yi(郭学益)1, FENG Ning-chuan(冯宁川)2, TIAN Qing-hua(田庆华)1
1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Basic Medical Science, Ningxia Medical College, Yinchuan 750004, China
Received 6 July 2009; accepted 30 December 2009
_____________________________________________________________________________________________________
Abstract: Preparation of orange peel xanthate and its adsorption behaviors of five heavy metals (Cu2+, Cd2+, Pb2+, Zn2+ and Ni2+) were studied. FTIR spectra, Zeta potentials and TG analysis were used to characterize prepared orange peel xanthate. Effects of various parameters including equilibrium pH, initial metal ion concentration and adsorption time on the adsorption processes for the five metal ions were investigated. It was found that for all five metal ions, the adsorption isotherms agreed Langmuir model very well and the maximum adsorption capacities of Cu2+, Cd2+, Pb2+, Zn2+ and Ni2+ were obtained as 77.60, 76.57, 218.34, 49.85 and 15.45 mg/g, respectively. All adsorption processes can attain equilibrium within 20 min and kinetics was well fitted by psesudo-second order equation. It is proposed that the adsorption mechanism was complexation.
Key words: orange peel xanthate; adsorption; heavy metals
_____________________________________________________________________________________________________
1 Introduction
Heavy metal wastewater exists in various industries, such as metal finishing, electroplating, plastics, pigments and mining, which threatens to the environment and human lives severely[1]. Therefore, it is urgent to remove those toxic heavy metals from wastewater. Although heavy metal removal from aqueous solutions can be achieved by conventional methods, including chemical precipitation, oxidation/reduction, electro- chemical treatment, evaporative recovery, filtration, ion exchange and membrane technologies, they may be ineffective or cost-expensive, especially when the metal ion concentrations in solution are in the range of 1-100 mg/L[2-4].
Recently, adsorption technology has become one of the alternative treatments[5-6], especially the widespread industrial use of low-cost adsorbents for wastewater treatment is strongly recommended due to their local availability, technical feasibility, engineering applicability and cost effectiveness. However, using the biological adsorbents directly may suffer from lack of specificity and poor adsorption capacity. It is observed that appropriated modification of the raw adsorbents by crown esters, amines, polyethylamine and sulphur bearing groups like sulfides, thiols, dithiocarbamates, dithiophosphates and xanthates can eliminate the drawbacks and improve their performances significantly [7]. Xanthates are most prominent because they are highly insoluble, easy to prepare with relatively inexpensive reagents and have high stability constant values of the metal complexes formed. Xanthates are formed by reacting an organic hydroxyl-containing substrate with carbon disulfide under caustic condition [8-9].
Orange peel is abundant in soft drink industries and usually treated as wastes. It is mostly composed of cellulose, pectin, hemi-cellulose, lignin, chlorophyll pigments and other low relative-molecular-mass hydro- carbons [10-13]. These components contain many hydroxyl functional groups (―OH), which makes it a potential matrix to synthesize xanthate. The main objective of this work was to investigate the potential adsorption ability of orange peel xanthate for the removal of five heavy metal ions (Cu2+, Cd2+, Pb2+, Zn2+ and Ni2+).
2 Experimental
2.1 Chemicals
All chemicals used in this study were of analytical purity. Stock solutions of single heavy metal were prepared by dissolving CuSO4・5H2O, 3CdSO4・8H2O, Pb(NO3)2, Zn(NO3)2・7H2O and Ni(NO3)2・6H2O in double distilled water, respectively, and diluted to desired concentrations when being used. 0.1 mol/L solution of 2-[4-(2-hydroxyethyl)-1-piperazinyl] ethanesulphonic acid (HEPES) purchased from Sigma Aldrich was used for the purpose of buffering. HCl and NaOH were used to adjust the pH value of experimental solutions.
2.2 Preparation of orange peel xanthate
Orange peel was washed with double distilled water and dried at 70 ℃ inside a convection oven for 24 h. The product was crushed and named as OP for further treatment process.
About 50 g of dried OP were pretreated by soaking in 250 mL ethanol and 250 mL 1% NaOH solution at room temperature (25 ℃) for 24 h to remove chlorophyll pigments and other low relative-molecular- mass compounds. After decantation and filtration, the treated biomass was washed with double distilled water until the solution reached a neutral pH value around 7.0 and then dried at 70 ℃ inside a convection oven. This dried product was named as AOP.
Xanthation process was carried out basically according to WING’s procedure[14] with some modifications. About 15 g of AOP was taken in a 300 mL Erlenmeyer flask and 200 mL 4 mol/L NaOH was added into it. The flask was stirred at room temperature (25 ℃) for 3 h and for another 3 h after addition of 10 mL carbon disulphide (CS2). This mixture was then allowed to settle for 1 h. After that, the supernatant was decanted and excess alkali was removed by washing with double distilled water. The mixture was then washed with acetone and dried in vacuum condition. The final product was abbreviated as XOP and stored at low temperature (6-8 ℃) until use.
2.3 Batch adsorption experiment
All of the adsorption tests were carried out batch wise and for single metal ion adsorption. For pH and isotherm experiments, 50 mg of XOP was taken into each conical flask which contained 10 mL of M2+ solution with initial mass concentration of 50 mg/L (for Pb2+, 100 mg/L was used). The pH values of the aqueous solutions were measured with a pHs-3C model acidity meter (made in China). The flasks were shaken at 120 r/min in a thermostatic shaker for 1.5 h at 25 ℃. After adsorption, the mixtures were filtered and the metal ion concentrations in the filtrate were determined by using an AA-6650 atomic absorption spectro-photometer (Shimadzu). The kinetics was examined inside a 150 mL conical flask, containing 0.5 g adsorbent and 100 mL of M2+ solution with the initial mass concentration of 50 mg/L (for Pb2+, 100 mg/L was used) at 25 ℃. The mixture was shaken continuously, then samples were taken at desired time intervals and filtered immediately.
The amount of adsorbed metal (qe) was determined by using the following equation:
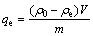 (1)
(1)
where ρ0 and ρe represent the initial and equilibrium metal ion concentrations (mg/L), respectively; V is the volume of the solutions (mL) and m is the mass of adsorbent (mg).
2.4 Characterization of adsorbents
FTIR spectra of the adsorbents (OP and XOP) were taken with a Fourier-transform infrared spectro- photometer (JASCO FT/IR-410, Japan). Pressed pellets were prepared by grinding the powder specimens with spectroscopic grade KBr for FTIR spectra tests. Zeta potentials of XOP were measured over the pH range of 2.0-7.0 on a Malvern Zetasizer (Model ZEN 2010, Malvern Instrument, Co., UK). The thermal analysis was carried out by TG peaks using a SDT Q600 instrument in the range of 10-810 ℃ under N2 atmosphere.
3 Results and discussion
3.1 Characterization of adsorbents
The FTIR spectra of OP and XOP were carried out as a qualitative analysis to determine the main functional groups present in the adsorbent. Usually, the band of 3 640-3 510 cm-1 is O―H stretching vibration. The band 3 000-2 800 cm-1 is stretching vibration of C―H. The peaks at 1 740-1 725 cm-1 is stretching vibration of COO― and C=O. The 1 130-1 000 cm-1 is vibration of C―O―C, C―O―P and O―H of polysaccharides [15-18]. The FTTR spectra details of OP and XOP are given in Table 1. Some distinct changes are noted after modification of OP, and some peaks disappeared and some new peaks are observed. The O―H bond at around 3 420 cm-1 in OP spectra shifts to 3 409 cm-1 in XOP spectra, indicating the hydroxyl groups in OP have combined with CS2. The presence of sulphur groups in XOP has been identified by the appearance of new peaks at 538.3, 1 019.6 and 1 151.3 cm-1 corresponding to stretching vibrations of C―S, C=S and S―C―S, respectively[7].
Fig.1 shows the Zeta potentials of XOP at pH range of 2.0-7.0. Apparently, the Zeta potentials of XOP exhibit all negative values within the pH range tested, which indicates that the surface of XOP is favorable for binding with metal cations. In addition, at pH values lower than 6.0, as the pH increased, the Zeta potential increased, which means the ability to combine M2+ was enhanced.
Table 1 Comparison of infrared bands in 4 000-400 cm-1 spectral region
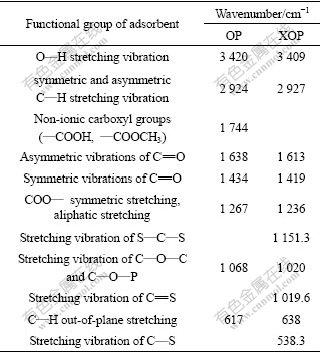
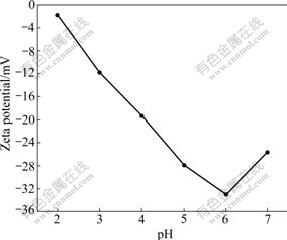
Fig.1 Zeta potentials of XOP
Fig.2 presents the thermogravimetric profile obtained from XOP. This figure shows three stages of mass losses, which are represented as regions Ⅰ, Ⅱ and Ⅲ. The first stage extends from the beginning of the analysis until about 110 ℃, corresponding to water loss. The second stage, ranging from 110 ℃ to 600 ℃, corresponds to organic matter decomposition as well as volatile substance releases proceeding from inorganic compound decomposition. During this stage, the complete degradation of the cellulose occurs (at about 300 ℃). Lignin pyrolysis occurs at about 400 ℃, while hemicellulose decomposes at a considerably lower temperature. Finally, region Ⅲ represents the ash residues[17]. No mass loss was detected at temperature above 800 ℃.
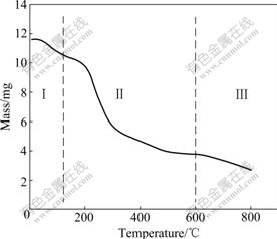
Fig.2 Thermogravimetric curve from XOP obtained under N2 atmosphere
3.2 Effect of equilibrium pH on adsorption
The metal ion adsorption is affected by the pH value of the solution, thereby changing the surface charge of the adsorbent and metal speciation. Fig.3 shows the adsorption efficiency of M2+ as function of equilibrium pH. It can be seen from this figure, the percent of adsorbed metal ions increases with the increase in pH, which is consistent with the phenomenon in Fig.1. Almost all metal ions can be absorbed to the extent of almost 80%-100% at weakly acidic conditions and the order of adsorption affinity among the tested metal ions is as follows: Pb2+>Cu2+>Cd2+>Zn2+>Ni2+. The pH range of 5.0-5.5 was chosen as adsorption condition for the following experiments.
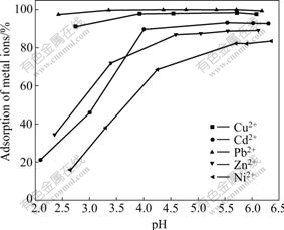
Fig.3 Effect of pH on adsorption of heavy metals on XOP
3.3 Adsorption isotherms
Initial mass concentrations of metal ions were varied (50-1 000 mg/L) for sorption by the constant 50 mg of XOP for the adsorption time of 1.5 h. The influence of metal ion concentrations on adsorption were examined and the results are shown in Fig.4. It appears that at first the adsorption capacities increase with the increase of ion concentration in equilibrium metal and then tend to approach constant values, suggesting these metal ions are absorbed onto the orange peel xanthate according to the Langmuir adsorption[4].
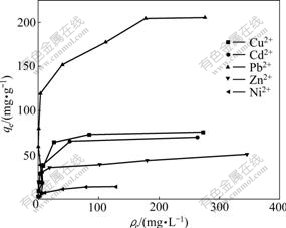
Fig.4 Adsorption isotherms of metal ions on XOP
Langmuir adsorption model can be expressed as [19]:
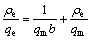 (2)
(2)
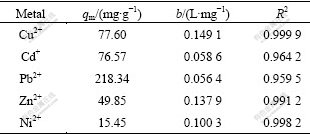
where qm is the maximum amount of adsorption (mg/g), b is the adsorption constant (L/mg). Fig.5 shows the plots of the experimental data in Fig.4 which is rearranged according to the Eq.(2). The values of the adsorption capacity qm, and the Langmuir constant b were obtained from linear regression and the results are presented in Table 2. It can be seen that the R2 values of all metal ions were close to 1, revealing the extremely good applicability of the Langmuir model to these adsorption processes. The maximum adsorption capacities of Cu2+, Cd2+, Pb2+, Zn2+ and Ni2+ were calculated to be 77.60, 76.57, 218.34, 49.85 and 15.45 mg/g, respectively. Therefore, we obtained that the order of maximum adsorption capacity of these metal ions on XOP was Pb2+>Cu2+>Cd2+>Zn2+>Ni2+.
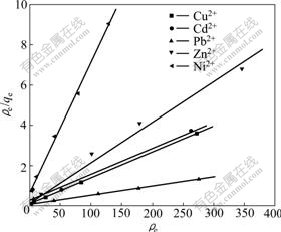
Fig.5 Langmuir plot of adsorption of metal ions on XOP
Table 2 Langmuir adsorption isotherm model parameters
The reason why the binding capacities of XOP were not identical for five heavy metals could be explained with two possible adsorption mechanisms: ordinary ion exchange and complex formation. By ion exchange, two negatively charged sulfur atoms of XOP participate in capturing one divalent metal ion. However, complex formation occurs between four sulfur atoms and one divalent metal ion. It is postulated that each heavy metal used in this study could undergo both ion exchange and complex formation with XOP and the fractions of those two mechanisms were different for the five metals[8, 20]. However, because Cu, Cd, Pb, Zn and Ni are all transition metals which have empty orbital that can be occupied by extra electrons of S or N in XOP, the major mechanism would be a complex formation:
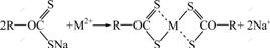 (3)
(3)
3.4 Adsorption Kinetics
The adsorption kinetics was investigated for better understanding of the dynamics of adsorption of heavy metal ions on XOP. Fig.6 shows the effect of time on the amount of absorbed capacity of the five metal ions on XOP. It can be seen that the adsorption process proceeds rapidly and the adsorption equilibrium can be attained within 20 min.
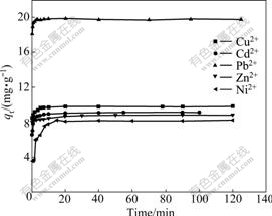
Fig.6 Adsorption kinetics of metal ions on XOP
Kinetics data were modeled using pseudo-second-order model, which assumes that the rate is proportional to the square of the number of remaining free surface sites. This model can be written as[21]:
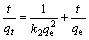 (4)
(4)
where k2 (g/(mg・min)) is the constant of pseudo-second- order rate. The relationship between t/qt and t is linear with a slope of 1/qe and an intercept of 1/(k2qe2). Fig.7 shows the application of pseudo-second-order model for the experimental data in Fig.6. Obviously, the adsorption process could be well described by the pseudo-second- order equation, indicating the process mechanism to be chemical adsorption[12].
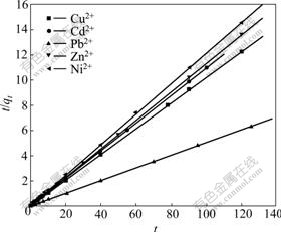
Fig.7 Pseudo second-order kinetics plots of adsorption kinetics of metal ions on XOP
4 Conclusions
1) Orange peel, through simple chemical modification of being crosslinked with CS2 in alkali condition to prepare orange peel xanthate, can exhibit excellent adsorption characteristics for the five metal ions tested.
2) FTIR spectra confirm the presence of sulphur groups on the orange peel xanthate and negative Zeta potentials within 2.0-7.0 mV make sure the adsorption process available.
3) For all five metal ions, pH has a positive effect on adsorption efficiencies; adsorption isotherms fit Langmuir model; and kinetics conforms to pesudo- second equation. The selectivity order for metal ion removal by XOP is Pb2+>Cu2+>Cd2+>Zn2+>Ni2+. The adsorption mechanism is proposed as complexation.
References
[1] KURNIAWAN T A, CHAN G Y S, LO W, BABEL S. Comparisons of low-cost adsorbents for treating wastewaters laden with heavy metals [J]. Science of the Total Environment, 2006, 366: 409-426.
[2] LIANG S, GUO X Y, FENG N C, TIAN Q H. Application of orange peel xanthate for the adsorption of Pb2+ from aqueous solutions [J]. Journal of Hazardous Materials, 2009, 170(1): 425-429.
[3] DHAKAL R P, GHIMIRE K N, INOUE K. Adsorptive separation of heavy metals from an aquatic environment using orange waste [J]. Hydrometallurgy, 2005, 79: 182-190.
[4] PARAJULI D, INOUE K, OHTO K, OSHIMA T, MUROTA A, FUNAOKA M, MAKINO K. Adsorption of heavy metals on crosslinked lignocatechol: A modified lignin gel [J]. Ractive and Functional Polymers, 2005, 62: 129-130.
[5] KUMAR U, BANDYOPADHYAY M. Sorption of cadmium from aqueous solution using pretreated rice husk [J]. Bioresour Technol, 2006, 97: 104-109.
[6] SINGH K K, TALAT M, HASAN S H. Removal of lead from aqueous solutions by agriculture waste maize bran [J]. Bioresour Technol, 2006, 97: 2124-2130.
[7] PANDA G C, DAS S K, GUHA A K. Biosorption of cadmium and nickel by functionalized husk of Lathyrus sativus [J]. Collids and Surfaces B: Biointerfaces, 2008, 62: 173-179.
[8] KIM H, LEE K. Application of insoluble cellulose xanthate for the removal of heavy metals from aqueous solution [J]. Korean J Chem Eng, 1999, 16(3): 298-302.
[9] BAILEY S E, OLIN T J, BRICKA R M, ADRIAN D D. A review of potentially low-cost sorbents for heavy metals [J]. Water Res, 1999, 33: 2469-2479.
[10] BISWAS B K, INOUE K, GHIMIRE K N, SHINGO O, HARADA H, OHTO K, KAWAKITA H. The adsorption of phosphate from an aquatic environment using metal-loaded orange waste [J] J Colloid Interface Sci, 2007, 312: 214-223.
[11] P?REZ-MAR?N A B, ZAPATA V M, ORTU?O J F, AGUILAR M, S?EZ J, LLOR?NS M. Removal of cadmium from aqueous solutions by adsorption onto orange waste [J]. Journal of Hazardous Materials, 2007, 139(1): 122-131.
[12] FENG N C, GUO X Y, LIANG S. Adsorption study of copper(Ⅱ) by chemically modified orange peel [J]. Journal of Hazardous Materials, 2009, 164(2/3): 1286-1292.
[13] LI X M, TANG Y R, CAO X, LU D, LUO F, SHAO W. Preparation and evaluation of orange peel cellulose adsorbents for effective removal of cadmium, zinc, cobalt and nickel [J]. Collids and Surfaces A: Physicochem Eng Aspects, 2008, 317: 512-521.
[14] WING R E, DOANE W M, RUSSEL C R. Insoluble strach xanthates: Use in heavy metal removal [J]. J Appl Polym Sci, 1975, 19: 847-854.
[15] GNANASAMBANDAM R, PROTOR A. Determination of pectin degree of esterification by diffuse reflectance fourier transform infrared spectroscopy [J]. Food Chem, 2000, 68: 327-332.
[16] LI F T, YANG H, ZHAO Y, XU R. Novel modification pectin for heavy metal adsorption [J]. Chinese Chem Lett, 2007, 18: 325-328.
[17] FARINELLA N V, MATOS G D, ARRUDA M A Z. Grape bagasse as a potential iosorbent of metals in effluent treatments [J]. Bioresource Technol, 2007, 98: 1940-1946.
[18] GUIBAVD G, TIXIER N, BOUJU A, BAUDU M. Relation between extracellular polymer’s composition and its ability to complex Cd, Cu and Pb [J]. Chemosphere, 2003, 52: 1701-1710.
[19] LANGMUIR I. The adsorption of gases on plane surfaces of glass, mica and platinum [J]. Journal of American Chemistry Society, 1918, 40: 1361-1403.
[20] KIM H, LEE K. Application of cellulose xanthate for the removal of nickel ion from aqueous solution [J]. J Kor Soc Eng, 1998, 20: 247-254.
[21] LAGERGERN S. About the theory of so-called adsorption of soluble substances [J]. K Sven Vetenskapsakad Handl, 1898, 24: 1-39.
_______________________
Foundation item: Project(50774100) supported by the National Natural Science Foundation of China
Corresponding author: GUO Xue-yi; Tel: +86-731-88877863; E-mail: xyguo@mail.csu.edu.cn
(Edited by FANG Jing-hua)