Trans. Nonferrous Met. Soc. China 22(2012) s678-s682
Effect of processing temperatures on characteristics of surface layers of low temperature plasma nitrocarburized AISI 204Cu austenitic stainless steel
Insup LEE
Department of Advanced Materials Engineering, Dongeui University, Busan 614-714, Korea
Received 21 May 2012; accepted 5 October 2012
Abstract: The influence of processing temperatures on the surface characteristics of AISI 204Cu austenitic stainless steel was investigated during a low temperature plasma nitrocarburizing. The resultant layer was a dual-layer structure, which comprises a N-enriched layer on the top of C-enriched layer. The surface hardness and the layer thickness increase up to about HV0.05 1000 and 20 μm with increasing temperature. The specimen treated at 400 °C shows a much enhanced corrosion resistance compared to the untreated steel. A loss in corrosion resistance was observed for specimens treated at temperatures above 430 °C due to the formation of Cr2N.
Key words: low temperature plasma nitrocarburizing; corrosion resistance; expanded austenite; AISI 204Cu stainless steel; hardness.
1 Introduction
The nickel-manganese-nitrogen contained stainless steels are austenitic and classified as AISI 200 series. Normally, type 200 series have a low nickel content compared with AISI 300 series, and austenite stabilizers such as nitrogen, copper and cobalt are added in producing an austenitic stainless steel. These steels became popular as a substitute for type 300 series in the early 1950s during The Korean War due to the nickel shortage and has been used in various fields of industry due to their own merits rather than as a substitute material. The primary objective of the incorporation of nitrogen is to stabilize the austenite against ferrite formation at the elevated temperatures and against martensite formation at low temperatures. It has also been recognized that nitrogen has an capability to strengthen the austenite phase by solid solution strengthening and by reducing the grain size in recrystallized material. Additionally, manganese has an effect on increasing the solubility of nitrogen in the austenite phase. It has been known that the incorporation of copper leads to improve cold forging and cold formability of steels by reducing the work hardening rate [1].
Austenitic stainless steels have good corrosion resistance in various environmental situations, but they have low hardness and low wear resistance and poor friction. Many attempts have thus been made during the past decade to develop surface treatment for improving surface hardness and wear resistance of stainless steel. For example, plasma ion nitriding is a thermochemical process to improve the surface properties, such as wear resistance, corrosion resistance, and fatigue strength of various engineering steels. Conventional plasma nitriding of austenitic stainless steels at temperatures higher than 500 °C results in an increase of hardness and a deterioration of corrosion resistance due to the formation of chromium nitrides which consumes the chromium to keep a passive state. However, a low temperature plasma surface treatment can improve the surface hardness without deteriorating the corrosion resistance.
Low-temperature plasma nitriding, which is carried out at temperatures lower than 450 °C for up to several tens of hours, produces a nitrogen-enriched (N-enriched) layer up to about 20 μm in thickness [2-6]. Such a precipitate-free N-enriched layer, where nitrogen atoms occupy in the interstitial sites in face-centered cubic (FCC) lattice, shows high hardness and excellent corrosion resistance, both of which are better than the values for untreated austenitic stainless steel. However, the nitrided layer suffers from some technical problems, including a low-load bearing capacity due to its shallow thickness, an abrupt hardness drop at the layer-core interface, a poor toughness of the layer due to its extremely high hardness, and a nonuniformity of the layer thickness throughout the treated surface.
In order to overcome these problems, the addition of a small amount of CH4 to the nitriding atmosphere has been attempted. This process, the termed low- temperature plasma nitrocarburizing, introduces nitrogen and carbon species to the plasma media by the simultaneous incorporation of nitrogen and carbon atoms into the surface of austenitic stainless steels, resulting in a dual-layer structure composed of a N-enriched layer (γN) with a high nitrogen content on top of a C-enriched layer (γC) with a high carbon content. Such a dual-layer structure possesses not only much increased hardness and corrosion resistance, but also better uniformity of thickness and load-bearing capacity compared with nitrided layers [2-6].
Systematic studies have been conducted on low temperature plasma nitrocarburizing of AISI 300 series of austenitic stainless steels. However, very little work has been done concerning 200 series of austenitic stainless steels. Thus, a more rigorous research is required to establish the optimum conditions for low temperature plasma nitrocarburizing of 200 series of austenitic stainless steels for industrial applications. In the present work, the effect of processing temperatures on the characteristics of surface layers formed on AISI 204Cu austenitic stainless steel by plasma nitrocarburizing process was investigated.
2 Experimental
The material selected for this research was AISI 204Cu austenitic stainless steel of which composition is listed in Table 1. The surfaces of the disc samples (diameter × height is 20 mm × 5 mm) to be exposed to the plasma were polished and then cleaned. The samples were placed on the cathode table in the pulsed plasma ion nitriding system. The vacuum chamber was closed and pumped down to at least 6.65 Pa and then Ar and H2 ions sputtering were performed at 300 °C for 40 min for further surface cleaning (voltage: 350 V, gas composition: V(Ar)/V(H2)=25/75, pulse: on/off time= 135 s and 15 s, time: 40 min). After cleaning, the low temperature plasma nitrocarburizing process was immediately carried out with a pulsed D.C. potential at temperatures between 300 °C and 460 °C for 15 h in the glow discharge of a gas mixture of N2, H2, and CH4. After plasma treatment, the vacuum chamber was evacuated again and the samples were cooled in the vacuum chamber to room temperature.
The plasma treated samples were sectioned for metallographic examination and hardness profile determination. The polished cross-sectional surfaces were first etched with the special reagent composed of (volume fraction) 50% HCl, 30% HNO3 and 20% H2O. The microstructures of low temperature nitrocarburizd layers on the surface were observed by optical microscope. A Rigaku D/Max-200 X-ray diffractometer was applied to analyze the phases formed on the plasma treated surfaces of the specimens. Microhardness measurements were carried out with Mitutoyo Micro Vickers Hardness Tester using an indentation load of 0.49 N (50 g) and loading time 15 s. A potentiostat polarization technique was employed to estimate the corrosion characteristics of the low temperature nitrocarburizd layer in a 3.5% NaCl solution. The 3.5% KCl Ag/AgCl was selected for reference electrode and Pt was used for counter electrode. The anodic polarization curves were recorded with a sweep speed of 0.166 mV/s by using a Princeton Applied Research model 273 A. Glow discharge optical spectrometry was used to analyze the nitrogen and carbon concentration depth profiles across the hardened layer.
Table 1 Chemical composition of AISI 204Cu austenitic stainless steel (mass fraction, %)
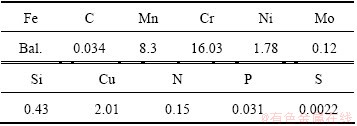
3 Results and discussion
Figure 1 represents the X-ray diffraction patterns generated from the hardened layers over a range of processing temperatures (300-460 °C) under a fixed pressure (5.32 × 102 Pa), gas composition (φ(N2): φ(H2): φ(CH4)=25:70:5) and time (15 h). It can be seen that the diffraction peaks from the treated surfaces are shifted towards lower Bragg angles compared with the corresponding austenite (face-centered cubic) peaks of the untreated surface, and move to much lower angles with increasing treatment temperatures up to 400 °C. This indicates that the incorporation of nitrogen and carbon atoms into FCC austenite structure causes lattice expansion and distortion, leading to the formation of new phases, termed as both a nitrogen-enriched expanded austenite phase (γN) in the near surface region, and a carbon-enriched expanded austenite phase (γC), beneath the nitrogen-enriched layer and not detected by X-ray diffractometer, which can reveal the region of up to 2-3 μm depth of any surface. In addition, it is clear that the characteristics of a diffusion-controlled process leads to the supersaturation of larger amounts of nitrogen and carbon atoms with increasing treatment temperature. On the other hand, at 430 °C, the diffraction peaks move to slightly higher angles compared with corresponding diffraction peaks from the specimen treated at 400 °C due to the formation of chrominum nitride Cr2N in the N-enriched layer, which reduces lattice expansion and distortion of the fcc lattices by the release of nitrogen atoms from the lattice. The crystal quality of austenite phase is further improved by raising the nitrocarburizing temperature up to 460 °C.
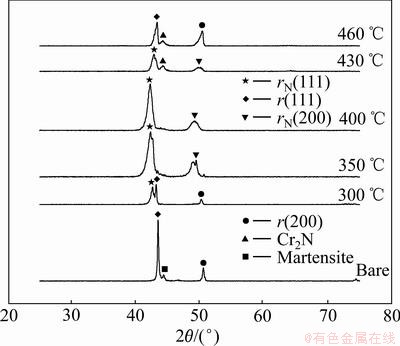
Fig. 1 XRD patterns of nitrocarburized layers formed on AISI 204Cu austenitic stainless steel at various processing temperatures
The nitrogen and the carbon concentration profiles of the nitrocarburized layer produced on AISI 204Cu stainless steel for various temperatures are illustrated in Fig. 2. It is evident that a dual-layer structure, which comprises a N-enriched layer (γN) with a high nitrogen content on top of a C-enriched layer (γC) with a high carbon content, has been produced. With increasing temperature up to 400 °C, the nitrogen and carbon concentrations increase at the surface and their depth of penetration. This indicates that the formation of the dual-layer structure by a low temperature plasma nitrocarburizing is a thermally activated process. At 430 °C, the nitrogen and the carbon concentrations at the surface decrease, while the depth of nitrogen and carbon penetration slightly increases compared with those treated at 400 °C. This implies that at this temperature, the diffusion rate of the nitrogen and carbon atoms towards the interior of the steel is much higher than the rate of supply of those atoms from the plasma to the surface. It can be seen that further penetration of nitrogen and carbon atoms into the substrate materials occurs at treatment temperature of 460 °C due to the fast diffusion of nitrogen and carbon atoms at higher temperature. Since carbon diffuses faster than nitrogen in austenite, the C-enriched layer (γC) is formed in the subsurface beneath the N-enriched layer (γN). The high solubility of nitrogen in austenite leads to the high nitrogen level in the near surface and greatly contributes to the penetration of carbon atoms towards the interior of the steel as if carbon atoms were pushed into the lower part of the hardened layer by nitrogen atoms, resulting in a low carbon level in the N-enriched layer. Thus, the low-temperature plasma nitrocarburizing process is a successful integration of low-temperature nitriding and low-temperature carburizing processes.
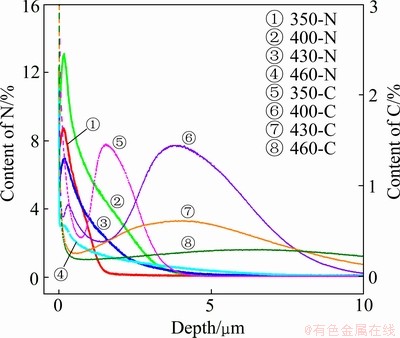
Fig. 2 Contents of nitrogen and carbon profiles of nitrocarburized layers formed on AISI 204Cu austenitic stainless steel at various processing temperatures
The cross-sectional morphology of the plasma nitrocarburized AISI 204Cu austenitic stainless steel is revealed in Fig. 3. As shown in nitrogen and carbon concentration profiles measured by GDS (Fig. 2), the corresponding two sub-layers, i.e. the N-enrichesd outer layer and the C-enriched inner layer, can be clearly seen in the hardened layer. It is also clear that the hardened layer is resistant to the etchant (30% HNO3 + 50% HCl + 20% H2O, volume fraction) due to their bright appearance under an optical microscope, implying their good corrosion resistance compared with the substrate material [7-13]. For the treatment temperatures above 430 °C, some dark regions are observed in the N-enriched layer, which are associated with the precipitation of Cr2N, confirmed by XRD analysis (Fig .1). The formation of chromium nitride precipitates leads to the decrease in corrosion resistance, resulting in the heavily etching of the associated region as illustrated in the N-enriched layer of microstructure of the specimen treated at 460 °C. The thickness of the hardened layer increases with increasing treatment temperature, which is in good agreements with N and C concentration profiles of Fig. 2.

Fig. 3 Optical micrographs of cross-sections of nitrocarburized layers formed on AISI 204 Cu austenitic stainless steel at various processing temperatures
Thickness and surface hardness of nitrocarburized layer are illustrated as a function of treatment temperature under fixed treatment time of 15 h in Fig. 4. The selection of a small indentation load of 50 g is to minimize the influence of the underlying substrate material due to the small thickness of the nitrocatburized layer. It is clear that the thickness of the hardened layer increases with increasing treatment temperature, which is also demonstrated in Figs. 2 and 3. The surface hardness of the layer increases in proportion to the increase of the layer thickness, since the larger thickness of the layer is associated with more supersaturation with nitrogen and carbon and has a less significant influence from the substrate material due to higher load-bearing capacity during hardness measurement. The incorporation of interstitial atoms such as nitrogen and carbon atoms into face centered cubic austenitic stainless steel causes lattice expansion and distortion, leading to the build-up of internal stresses which restrict the motion of dislocation, and resulting in the increase of surface hardness. The surface hardness reaches up to HV0.05 1000 which is about 4 times higher than that of the untreated sample (HV0.05 230).
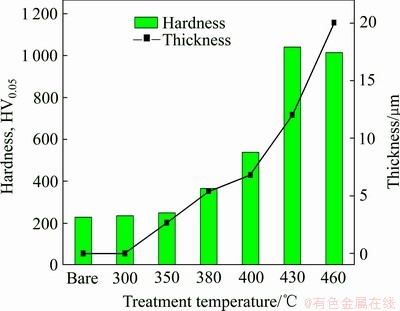
Fig. 4 Surface hardness and thickness of nitrocarburized layers formed on AISI 204Cu austenitic stainless steel at various processing temperatures
Figure 5 shows the anodically potentiodynamic polarization curves of the nitrocarburized layers in a 3.5% NaCl solution. The specimen treated at 400 °C reduces the corrosion current density by about two orders of magnitude and has much higher pitting potential by about 300 mV in the anodic region as compared to the untreated steel, implying an improvement in corrosion resistance. In addition, the specimen treated at 400 °C shows a much lower current density after pitting corrosion by two orders of magnitude as compared to that of the untreated steel. After testing up to a potential of 1500 mV, no pitting was observed in the surface of the specimen treated at 400 °C, while pitting corrosion was confirmed in the untreated steel by microscopic examination. A possible mechanism for the enhancement in corrosion resistance due to nitrogen supersaturation in austenite is that the presence of high concentration of nitrogen on the surface enhances the corrosion resistance by the increase of pH associated with the formation of NH4+ (N + 4H+ + 3e-→ NH4+) in the acid pits, resulting in increasing the passivation ability and retarding the corrosion rate [14-15]. Although no pitting corrosion was observed in the treated surfaces at temperatures of both 350 °C and 380 °C, the type of general corrosion occurred with relatively high corrosion rate. However, the higher corrosion rate was observed for the specimen treated at temperatures above 430°C due to the formation of a small amount of chromium nitride in the nitrogen-enriched layer which will cause chromium depletion in their vicinity and eventually prevent the generation of a very thin Cr2O3 surface film, termed as a passive film.
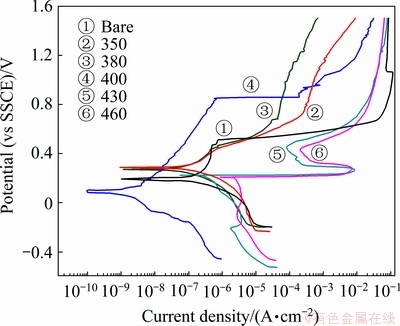
Fig. 5 Potentiodynamic polarization curves of nitrocarburized layers formed on AISI 204Cu austenitic stainless steel at various processing temperatures
4 Conclusions
1) The low-temperature plasma nitrocarburizing of AISI 204 Cu austenitic stainless steel lends to the formation of a dual-layer structure, which comprises a N-enriched layer (γN) with a high nitrogen content on top of a C-enriched layer (γC) with a high carbon content, leading to significant increase in the surface hardness and the corrosion resistance. The employed low temperature around 400 °C leads to the generation of precipitation-free sub-layers with an expanded austenite structure. However, chromium nitride is formed in the N-enriched layer for specimen treated at temperatures above 430 °C.
2) The surface hardness and the layer thickness increase up to about HV0.05 1000 and 20 μm with increasing treatment temperature. The specimen treated at 400 °C shows a much enhanced corrosion resistance in terms of lower corrosion current density and a higher pitting potential as compared to the untreated steel. A drop in corrosion resistance is observed for the specimen treated at temperatures above 430 °C due to the formation of chromium nitrides in the nitrogen-enriched layer.
References
[1] SEDRIKS A J. Corrosion of stainless steels [M]. New York: John Wiley & Sons, InC., 1996: 25-26.
[2] SUN Y. Kinetics of low temperature plasma carburizing of austenitic stainless steels [J]. Mater Proc Tech, 2005, 168: 189-194.
[3] LARISCH B, BRUSKY U, SPIES H J. Plasma nitriding of stainless steels at low temperatures [J]. Surf Coat Technol., 1999, 116-119: 205-211.
[4] SUN Y, BELL T. Dry sliding wear resistance of low temperature plasma carburised austenitic stainless steel [J]. Wear, 2002, 253: 689-693.
[5] SUN Y, HARUMAN E. Effect of carbon addition on low- temperature plasma nitriding characteristics of austenitic stainless steels [J]. Vac, 2006, 81: 114-119.
[6] SUN Y. Enhancement in corrosion resistance of austenitic stainless steels by surface alloying with nitrogen and carbon [J]. Mater Letter, 2005, 59: 3410-3413.
[7] TSUJIKAWA M, NOGUCHI S, YAMAUCHI N, UEDA N, SONE T. Effect of molybdenum on hardness of low-temperature plasma carburized austenitic stainless steel [J]. Surf Coat Technol, 2007, 201: 5102-5107.
[8] CHENG Z, LI C X, DONG H, BELL T. Low temperature plasma nitrocarburizing of AISI 316 austenitic stainless steel [J]. Surf Coat Technol, 2005, 191: 195-200.
[9] SUN Y, LI X, BELL T. Low temperature plasma carburizing of austenitic stainless steels for improved wear and corrosion resistance [J]. Surf Eng, 1999, 15(1): 49-54.
[10] EGAWA M, UEDA N, NAKATA K, TSUJIKAWA M, TANAKA M. Effect of additive alloying element on plasma nitriding and carburizing behavior for austenitic stainless steels [J]. Surf Coat Technol, 2010, 205: S246-S251.
[11] MOLLEJA G, NOSEI J, FERRON L, BEMPORAD J, LESAGE E, CHICOT J, FEUGEAS D. Characterization of expanded austenite developed on AISI 316L stainless steel by plasma carburization [J]. Surf Coat Technol, 2010, 204: 3750-3759.
[12] SUN Y, HARUMAN E. Influence of processing conditions on structural characteristics of hybrid plasma surface alloyed austenitic stainless steel [J]. Surf Coat Technol, 2008, 202: 4069-4075.
[13] SUN Y. Hybrid plasma surface alloying of austenitic stainless steels with nitrogen and carbon [J]. Mater Sci Eng A, 2005, 404: 124-129.
[14] BABA H, KODAMA T, KATADA Y. Role of nitrogen on the corrosion behavior of austenitic stainless steels [J]. Corrosion Science, 2002, 44(10): 2393-2407.
[15] SUN Y. Depth-profiling electrochemical measurements of low temperature plasma carburised 316L stainless steel in 1 M H2SO4 solution [J]. Surf Coat Technol, 2010, 204: 2789-2796.
(Edited by DENG Lü-xiang)
Foundation item: Project (2011AA192) supported by Dongeui University, Korea
Corresponding author: Insup LEE; Tel/Fax: +82-51-890-228512635; E-mail: islee@deu.ac.kr
DOI: 10.1016/S1003-6326(12)61785-3