Characterization of terbium-doped nano-hydroxyapatite and surface modification
来源期刊:中南大学学报(英文版)2016年第7期
论文作者:赵颜忠 杨敏 张海斌 朱军 周科朝
文章页码:1548 - 1555
Key words:hydroxyapatite; hydrothermal synthesis; arginine modification; rare-earth doping
Abstract: The control synthesis of nanoparticles was the implementation process of material ideal design. Nano-hydroxyapatite (HAP) was prepared by a hydrothermal method with calcium nitrate and diammonium hydrogen phosphate as raw material, to study its characteristics for morphology modification by arginine-functionalization and doping with rare earth such as Tb3+. The crystallization, grain size and dispersibility of the sample HAP were analyzed and discussed. The results show that the surface Zeta potential of arginine-functionalized HAP is changed, and the growth rate of HAP is inhibited to a certain extent during the synthesis. The structure of HAP/Arg is not affected during the synthesis by a small amount of rare earth ions doped such as Tb3+, and has a single phase of HAP with good dispersibility. The synthesized HAP is also of nano-sized level. Nano-hydroxyapatite arginine- functionalized and doped with rare earth such as Tb3+, is suitable for the application of gene delivery as a gene carrier.
J. Cent. South Univ. (2016) 23: 1548-1555
DOI: 10.1007/s11771-016-3207-9

ZHAO Yan-zhong(赵颜忠)1, 2, 3, YANG Min(杨敏)1, ZHANG Hai-bin(张海斌)2,
ZHU Jun(朱军)2, ZHOU Ke-chao(周科朝)2
1. The Third Xiangya Hospital, Central South University, Changsha 410013, China;
2. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
3. Research Center for Medical Material and Instruments, Central South University, Changsha 410013, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: The control synthesis of nanoparticles was the implementation process of material ideal design. Nano-hydroxyapatite (HAP) was prepared by a hydrothermal method with calcium nitrate and diammonium hydrogen phosphate as raw material, to study its characteristics for morphology modification by arginine-functionalization and doping with rare earth such as Tb3+. The crystallization, grain size and dispersibility of the sample HAP were analyzed and discussed. The results show that the surface Zeta potential of arginine-functionalized HAP is changed, and the growth rate of HAP is inhibited to a certain extent during the synthesis. The structure of HAP/Arg is not affected during the synthesis by a small amount of rare earth ions doped such as Tb3+, and has a single phase of HAP with good dispersibility. The synthesized HAP is also of nano-sized level. Nano-hydroxyapatite arginine- functionalized and doped with rare earth such as Tb3+, is suitable for the application of gene delivery as a gene carrier.
Key words: hydroxyapatite; hydrothermal synthesis; arginine modification; rare-earth doping
1 Introduction
Hydroxyapatite (HAP), firstly discovered in 1987 by two American dentists, BROWN and CHOW [1], is the most common bioactive material which has been applied widely in bio-medical field due to its advantages such as good chemical stability, biocompatibility and biological activity. Besides, HAP has neither noxious nor side effect upon human body and shows similarity to inorganic components of human hard tissues [2]. Previous researches have shown that many properties of HAP are closely related to the particle size [3]. Thus, with the development of nanotechnology, many researches about HAP have been gradually transferred to nanometer biological field. Compared to traditional HAP, the nanoparticles of HAP present unique biological properties such as larger specific surface area and surface energy, higher solubility, and better biological activity, which makes it easier to combine with anti-cancer drug, nucleic acid and protein [4-5]. Moreover, the HAP nanoparticles show great antitumor activity, by which it can inhibit the growth of cancer cell [6]. Therefore, the researches about preparation of HAP nanoparticles and its clinical application have caused close attentions of more and more scientists and researchers. Simultaneously, present studies have found that HAP nanoparticles are deemed to be ideal gene vectors lying in its good biological stability, easy preparation and modification [7-10]. And more importantly, the nanoparticles also can load, concentrate and protect genes. However, the genetic transmission mechanism and the influence on gene transfection rate of the particles size, morphology and surface state still remain to be further studied [11]. As a consequence, it is of great importance to improve the transfection efficiency for applying the HAP nanoparticles as gene carriers to clinical gene therapy by studying the transfection mechanism of HAP nanoparticles and improving the preparation process [12].
In this work, calcium nitrate and ammonium hydrogen phosphate as raw materials were used to prepare HAP nanoparticles by hydrothermal synthesis method. The impact on the morphology of HAP particles by adding arginine was investigated, and test analysis to crystalline, particle size and dispersion of the fabricated samples was provided. The objective of this work is to study the HAP nanoparticles, a new type of modified non-viral vector by hydrothermal synthesis method, to characterize the nanoparticles morphology modified by adding arginine with rare earth metal terbium, and to confirm that it is suitable for application in the field of gene transfection vectors.
2 Materials and experimental procedures
2.1 Materials
In this experiment, the following materials were used: Ca(NO3)2・4H2O, (NH4)3PO4・3H2O, Na2HPO4 (AR, China Medicine Group Shanghai Chemical Reagent Co., Ltd.), arginine (Sigma company), Tb2O3 (99.995%, Hunan Institute of Rare Earth Metals), stronger ammonia water (AR, Shandong Tengzhou Auspicious Embellish Chemical Co., Ltd.), aqua fortis (AR, Nanjing ZhangYang Petrochemical Co., Ltd.), PBS and HBS.
2.2 Preparation of HAP nanoparticles
Take 5.900 g of Ca(NO3)2・4H2O and 30 mL of distilled water into the beaker, stir the mixed liquor fully until it became transparent, and then keep adding distilled water to 50 mL. Meanwhile, 1.447 g of phosphoric acid solution (mass fraction was greater than or equal to 85%) was diluted to 30 mL and turned into a solution with pH value of 1. And then adjust the pH value to 10 by adding ammonia water, and at last, dilute the solution to 50 mL. Subsequently, ammonium phosphate solution was added rapidly into the Ca(NO3)2 solution and stirred fully. The mixed solution was white emulsion and adjusted to a solution with pH value of about 9-10 by adding ammonia water. Pour the mixed solution into 3-mouth flask which was fixed on trestle table and keep the lower section of flask into 80 °C of water. And the external water should be higher than the reaction liquid level in the flask. Adjusting the stirrer made the reaction liquid mix in the appropriate rate. At the beginning of stirring, the reaction liquid remained ivory-white until the end of aging process. After stirring evenly, the mixed solution was transferred into the high-pressure hydrothermal synthesis reactor which was subsequently sealed and put into the constant- temperature dry box where the reaction was performed for 3 h under the condition of 160 °C. Then take out the sediment of the hydrothermal synthesis reactor after cooling, and wash the sediment several times by the deionized water and ethanol. Finally, HAP nanoparticles were obtained after vacuum drying.
2.3 Preparation of HAP nanoparticles modified by arginine
Take 8.710 g of arginine and 5.900 g of Ca(NO3)2・4H2O into the beaker, and then add distilled water into the beaker to 30 mL. After stirring fully, the solution had obvious stratification phenomenon: the bottom was clear solution and the upper was white suspension. Subsequently, ammonium phosphate solution was added rapidly into [Ca2+]/arginine solution and stirred fully, and the pH value of the mixed solution above was adjusted to 9. Pour the mixed solution into 3-mouth flask, put the flask into 80 °C of water, and then stir fully the mixed solution for 16 h. At the beginning of stirring, there was a small number of white foam appearing on the surface of liquid and the liquid was ivory-white. This phenomenon remained until the end of aging process. After aging process, the mixed solution was put into the high-pressure hydrothermal synthesis reactor which was subsequently placed in the constant-temperature dry box where the reaction was performed for 3 h under the condition of 160 °C. Then take out the sediment of the hydrothermal synthesis reactor after cooling, and wash the sediment several times by the deionized water and ethanol. Finally, arginine-modified HAP nanoparticles were obtained after vacuum drying and grinding.
2.4 Rare earth doping of HAP/Arg nanometer powders
2.0711 g of Tb2O3 was dissolved in 15 mL of deionized water, and the Tb2O3 solution was put into isometric nitric acid solution. The mixed solution above was heated and stirred until the Tb2O3 was dissolved completely. Keep adding a certain amount of deionized water and heat until forming relatively pure (Tb(NO3)3) solution. Cool down the (Tb(NO3)3) solution to room temperature and then transfer the (Tb(NO3)3) solution into 200 mL of volumetric flask which was used to prepare (Tb(NO3)3) solution with concentration of 0.0588 mol/L. Finally, the prepared (Tb(NO3)3) solution was put into the wild-mouth bottle and stored in cool place for subsequent experiments. Subsequently, the (Tb(NO3)3) solution and Ca(NO3)2 solution were used to prepare Tb-doped HAP/Arg nanometer powders. During the process of preparing Tb-doped HAP/Arg nanometer powders, the mole ratio of Tb3+ to Ca2+ was kept a certain value, and the mole ratio of (Ca2++Tb3+) to P was 1.67. The method for preparing Tb-doped HAP/Arg nanometer powders was the same with that of Tb-doped HAP/Arg nanometer powders.
2.5 Preparation of mixed suspension of HAP nanometer powders
Take 0.5 g of prepared HAP nanometer powders above and 20 mL of deionized water into centrifuge tube, and prepare suspension with concentration of 25 mg/mL by ultrasonic dispersion (Ultrasonic Hemogenizer 24710, USA). After standing for 2 h, the suspension had no layering and kept milky. Then, the nanoparticles suspension was placed into 50 mL of glass bottle after autoclaving for subsequent use. Take out 1 mL of nanoparticles suspension which was used to observe the particle size and dispersion of nanoparticles by TEM after ultrasonic treatment for 8 min.
3 Results and discussion
3.1 Morphology of HAP nanoparticles
Figure 1 shows the transmission electron microscopy images of powders prepared by hydrothermal synthesis method. It can be seen that a single particle is about 50-100 nm in length and about 20-30 nm in diameter from Fig. 1(a). But due to the obvious powder agglomeration, there are scarcely any isolated HAP particles in the images and the secondary particles reach a size of hundreds of nanometer and even micron grade. And as shown in Fig. 1(b), the analysis results of SAED accords with the arrangement of HAP crystal structure, which gives evidence for the formation of HAP nanoparticles by hydrothermal synthesis method.

Fig. 1 TEM image of nano-HAP synthesized by hydrothemal method (a) and SAED of sample particle (b)
3.2 Relationship between Zeta potential and pH value
Figure 2 shows the relationship between the Zeta potential and pH value. It can be seen from Fig. 2 that the potential of HAP nanoparticles is almost nothing which is close to the isoelectric point of reaction system when the pH value is equal to 4 or so. With the increase of pH value of reaction system, the absolute value of Zeta potential also increases accordingly. When the pH increases to a value of 7, the absolute value reaches a maximum value of 23.1 mV. And then, the absolute value of Zeta potential reduces gradually but slowly with the pH value keeping rising.
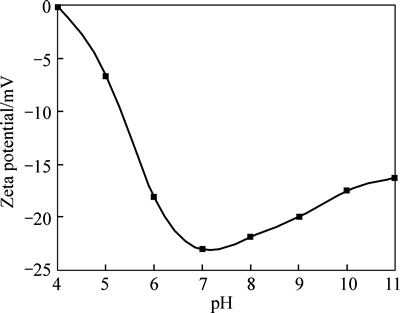
Fig. 2 Effect of pH value on Zeta potential of HAP particle
3.3 Relationship between particle size of nanometer powder and pH value
Figure 3 shows the variation of nanoparticles size versus pH value in the suspension system. It can be seen obviously that the particle size of HAP nanometer powders decreases first and then increases with the pH value of suspension system rising. The HAP particle size is about 470 nm when the pH value is 4, and then decreases to the minimum value of 370 nm as the pH value increases, which demonstrates that powder dispersion gradually increases in the process. Afterwards, when pH value of the suspension system ranges from 7 to 11, the HAP particle size increases gradually but slowly, which indicates that the dispersion effect of HAP in the suspension system is relatively good. On considering conjunctively the relationship between pH value and Zeta potential, the fact that the HAP particle size varies greatly as the Zeta potential keeps changing can be seen obviously. When pH value is 7, the absolute maximum value of Zeta potential is acquired, where HAP particle dispersion in reaction system is the best. And when pH value is about 4, HAP particle surface potential is zero, which leads to a big particle size. In conclusion, when pH value of nanometer HAP sol is in a certain value, a certain amount of charge is adsorbed by HAP particles and then develops into the electric double layer, which leads to electrostatic repulsion between particles of sol. However, the dispersity of HAP nanoparticles obtained by hydrothermal synthesis method cannot meet the requirements of gene carrier. Thus, in this work, the modification of arginine to the HAP particles is studied.

Fig. 3 Effect of pH value on particle size of HAP
3.4 Analysis on HAP/Arg by TEM
The crystal morphology, powder size and disperse state are characterized in Fig. 4 by transmission electron microscope (TEM).

Fig. 4 TEM images of HAP crystal synthesized by hydrothermal method:
It can be seen from Fig. 4(a) that HAP particles present a short column and equiaxed shape without adding arginine. The short rod-shaped particles have uniform cross-section of about 30-70 nm and a length of about 60-180 nm, and the length-diameter ratio is about 2-4. The diameter of equiaxed particles is about 30 nm. HAP presents a close-packed hexagonal structure and tends to grow into rod- and needle-shaped particles. As shown in Fig. 4(b), when adding the arginine into the reaction system, the particles size decreases more obviously and also performs a nanometer-scale. The length of rod-shaped particles reduces to about 50-80 nm. The particles show more homogeneous state in each different direction, and the uniformity of particle size and shape slightly decreases but the dispersion of particles is better.
The Zeta potential of pure HAP powders under acidic conditions is usually positive which can combine weakly with electronegative DNA through electrostatic interactions. And under neutral or weak alkaline conditions, the Zeta potential is negative and it becomes difficult for HAP powders to combine with electronegative DNA. In the present work, it is observed that HAP powders with Zeta potential (-23.1±3.5) mV is hardly combined with DNA when pH value is 7.4. And as shown in Fig. 5, arginine is considered as modified compound in this experiment for two reasons. Firstly, the hydrophilic arginine with guanidine groups ―(CH2)3NHC(NH2)+ is alkaline amino acid. And its isoelectric point of about 10.76 is higher than pH value of reaction system in hydrothermal synthesis experiment, which leads to a positive arginine in the whole process of hydrothermal synthesis. Secondly, in previous study [12], it was found that arginine and guanidine functional groups could penetrate effectively into the cell membrane, which played a critical role in transferring DNA into cell. This implies that arginine-rich short peptide can effectively improve the efficiency of gene transfection, but the transfection mechanism of arginine-rich short peptide still remains unclear which significantly differs from receptor mediated endocytosis mechanism [13].
The experimental data demonstrate that the addition of arginine can change obviously the morphology of HAP particles, especially when increasing the concentration of arginine, which can be attributed to thechange of growth behavior of HAP crystal due to the addition of arginine. In the process of HAP synthesis, it is the preferred adsorption of arginine on the different HAP crystal surfaces that affects the HAP grain growth behavior. As is well known, under thermodynamic equilibrium conditions, HAP crystals prefer to grow on (001). According to the crystal structure of HAP, there is a point (P) of easy adsorption where calcium ion vacancy appears occasionally, which leads to the negatively charged crystal surface (001). In hydrothermal condition, the positively charged guanidine groups ―(CH2)3NHC(NH2)+ of arginine can produce electrostatic interaction with the negatively charged hydroxyl (―OH) at the crystal surface (001) of HAP, where the arginine prefers to adsorb. As a result, the formation of compositions is inhibited on the crystal surface (001) of HAP due to the adsorption of arginine, which makes the growth of HAP crystals more homogeneous in different directions.

Fig. 5 Structural formula of arginine
On the other hand, the ―NH2 groups of arginine can combine with each crystal surface of HAP through hydrogen bonding [14]. Many studies show that HAP crystal surfaces are usually covered by a film of water, and due to the fact that there is stronger interaction force or lower effect energy between HAP crystals and arginine, the added arginine tends to be adsorbed at the HAP crystal surfaces especially on the surface (001) where the adsorbed water molecular is replaced by arginine. When a certain amount of arginine is adsorbed on each crystal surface of HAP, the crystal growth is inhibited and in order to reduce the degree of supersaturation of the solution, more HAP particle nucleation is required. Considering that improving the nucleation is helpful to obtain finer grain, when the concentration of arginine increases to a certain extent, each crystal surface of HAP would adsorb different numbers of arginine, which may lead to differences of size and shape between particles as shown in Fig. 4(b).
3.5 Zeta potential of HAP/Arg powders
Figure 6 shows the Zeta potential of synthetic HAP powders with different concentrations of arginine (0.05 mol/L and 0.10 mol/L, respectively) added when the pH value is 7.4. It can be seen from Fig. 6(a) that under the condition of weak alkaline (pH=7.4), the Zeta potentials of prepared HAP powders with and without 0.05 mol/L arginine added for modification are about (30.9±8.2) mV and (-23.1±6.5) mV, respectively, which indicates that in the process of preparing HAP powders the addition of arginine turns the usual negative potential into positive value. And compared with the Zeta potential of HAP powders with 0.05 mol/L arginine added, it increases slightly when increasing the arginine to 0.1 mol/L, as shown in Fig. 6(b), which can be attributed to theover-saturation of arginine. All the results show that a certain extent of arginine can change fundamentally the charge state of HAP/Arg powders. Thus, the greater absolute value of Zeta potential is obtained, which demonstrates that the dispersity of prepared HAP/Arg powders is better. As a result, it is easier for the electrostatic bonding between HAP nanoparticles and DNA due to the arginine or residues adsorption on the HAP/Arg particle surfaces.
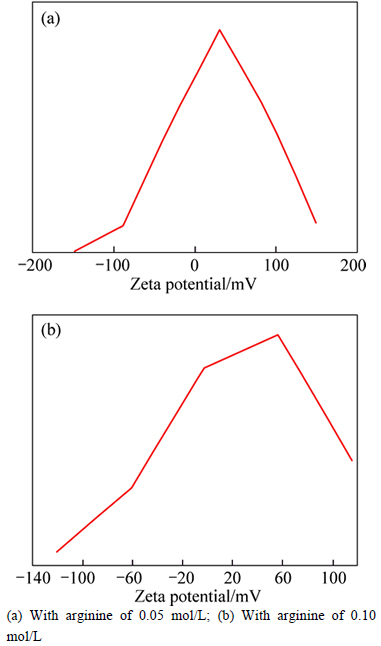
Fig. 6 Zeta potential distributions of HAP/Arg particles:
3.6 Phase composition of HAP/Arg powders
The HAP/Arg powders with/without adding the arginine prepared by hydrothermal synthesis method were analyzed by X-ray diffraction (scanning angle 25°-55o, speed 2.4 (°)/min), and the results are shown in Fig. 7. The XRD pattern of HAP powders with 0.05 mol/L arginine is basically identical to that of pure HAP powders and both characteristic peaks are very sharp, which indicates that the prepared powders have high crystallinity. The calibration result of crystal faces of characteristic peaks presents good consistency to the International Standard Card JCPDs 9-432 of HAP/Arg powders, which indicates that the prepared hydroxyapatite powders are well-crystallized. However, the intensity of characteristic peaks shows subtle differences. As shown in Fig. 7, the diffraction peak intensity on (300) of HAP/Arg powders is higher than that of pure HAP powders, and the diffraction peak intensity on (002) and (211) of HAP/Arg powders is lower than that of pure HAP powders.

Fig. 7 XRD patterns of HAP powder synthesized by hydrothermal method at 160 °C:
3.7 Infrared spectra characteristics of HAP/Arg powders
The HAP/Arg powders with/without adding the arginine was analyzed by FTIR spectrometer, and the results are shown in Fig. 8. Both of two different HAP powders with/without adding the arginine respectively have similar infrared spectra characteristics such as the main peaks having same location. The relatively stronger peaks appear at locations of 565.25, 604.21, 1035.78 and 3441.75 cm-1 and the relatively weaker or wider peaks appear at locations of 1106.57, 1420.30, 1631.24 and 3570.12 cm-1. Theoretically, the positions of peaks corresponding to four kinds of vibration mode of phosphate radical (PO43-) are as follows: ν1 peak close to 960 cm-1, ν2 peak located in the range from 470 to 440 cm-1, ν3 peak located in the range from 1190 to 976 cm-1, and ν4 peak located in the range from 600 to 560 cm-1. Therefore, the strong peaks (at 565.25, 604.21 and 1035.78 cm-1) and the weak peak (at 1106.57 cm-1) are produced due to the existence of phosphate radical (PO43-) in HAP. The water molecules and hydroxyl apatite in the crystal lattice lead to the peaks at 3441.75 and 3570.12 cm-1. The peak at 1631.24 cm-1 can be attributed to the vibration of H2O, which demonstrates that the powders surfaces adsorb amounts of water. The characteristic peak of amidogen (―NH2) appears in the range of 1400-1420 cm-1. And the peak at 1420.30 cm-1 may be due to the residues adsorbed on HAP which comes from ammonium (NH4+) and amino acid of diammonium phosphate. Meanwhile, as shown in Fig. 8, the peak at 1420.30 cm-1 is stronger when adding the arginine, which indicates the presence of arginine residues. By the analysis of FTIR spectrometer, the absorption peak of HAP nanoparticles with adding the arginine accords with the characteristics of HAP, which proves their structural consistency whether adding the arginine or not.

Fig. 8 FTIR spectra of HAP prepared by hydrothermal synthesis:
3.8 Phase composition of rare earth doped HAP/Arg powders
The HAP/Arg powders with 2%Tb (molar fraction) doped were analyzed by X-ray diffraction, and the results are shown in Fig. 9. It can be seen that the main diffraction peaks are very sharp which accord completely with the peak positions of HAP on International Standard Powder Card (JCPDs 9-432), and any other compositions are not detected such as TbPO4 and Tb(OH)3, which indicates that the synthesized products are also single phase HAP.
By calculating and analyzing the diffraction data of HAP/Arg powders with/without Tb-doping, the lattice constant of HAP/Arg powders with 2% Tb-doped is obtained (a=0.9437 nm and c=0.6899 nm) and that without Tb-doped is also obtained (a=0.9454 nm and c=0.6903 nm). It is obvious that the doping of Tb3+ into HAP/Arg powders also makes the lattice constantdecrease, which can also be attributed to the smaller radius of Tb3+ (0.092 nm) than that of Ca2+ [15]. The fact that the Tb3+ replaces parts of Ca2+ in the lattice makes the lattice constant and unit-cell volume decrease. The analysis results by ICP-AES show that the segregation coefficient of Tb3+ in the HAP lattice is 0.96, which presents that Tb3+ is easier to ingress into the HAP lattice due to its smaller radius.

Fig. 9 XRD pattern of HAP/Arg powder doped with Tb
3.9 Infrared spectra characteristics of rare-earth- doped HAP/Arg powders
The FTIR analysis results of Tb-doped HAP/Arg powders are shown in Fig. 10. It can be seen that the infrared spectra characteristics and main peak positions of Tb-doped HAP/Arg powders are basically consistent with those of HAP/Arg powders. Obviously, in Fig. 10, the peak located in the range from 564 to 602 cm-1 corresponds to the ν4 absorption peak of  and the peak at 573 cm-1 corresponds to the ν2 absorption peak of
and the peak at 573 cm-1 corresponds to the ν2 absorption peak of  the peak at 961 cm-1 corresponds to the ν1 absorption peak of
the peak at 961 cm-1 corresponds to the ν1 absorption peak of  the peak located in the range from 1032 to 1092 cm-1 corresponded to the ν3 absorption peak of PO43-, the peak at 1432 cm-1 corresponds to the characteristic peak of H2O, and the peak at 3571 cm-1 corresponds to the peak of lattice water and hydroxyapatite (―OH). Besides, the peak at 1645 cm-1 might be the peak of
the peak located in the range from 1032 to 1092 cm-1 corresponded to the ν3 absorption peak of PO43-, the peak at 1432 cm-1 corresponds to the characteristic peak of H2O, and the peak at 3571 cm-1 corresponds to the peak of lattice water and hydroxyapatite (―OH). Besides, the peak at 1645 cm-1 might be the peak of  It can be concluded that the doping of the Tb into the HAP/Arg nanoparticles does not change the characteristic peak and other absorption peak obviously, which shows that Tb does not change the structure of HAP/Arg nanoparticles [16].
It can be concluded that the doping of the Tb into the HAP/Arg nanoparticles does not change the characteristic peak and other absorption peak obviously, which shows that Tb does not change the structure of HAP/Arg nanoparticles [16].

Fig. 10 FTIR spectra of HAP/Arg powder:
3.10 Observation of rare-earth-doped HAP/Arg powders by TEM
Figure 11 shows the transmission electron microscope (TEM) image of rare-earth-doped HAP/Arg powders fabricated through hydrothermal reaction for 3 h. It can be seen from Fig. 11 that the Tb-doped HAP/Arg particles are composed of a small amount of equiaxial particles and mainly short columnar particles with uniform size. The length of short columnar particles is about 100 nm and the length-to-diameter ratio is about 3-4, which indicates that HAP/Arg particles are at the nano-scale size, and these results are very close to the X-ray diffraction analysis.

Fig. 11 TEM image of HAP/Arg powder doped with Tb
3.11 Fluorescence characteristics of rare-earth-doped HAP/Arg powders
Figure 12 shows the photoluminescence of Tb-doped HAP/Arg nanoparticles and the excitation wavelength is 272 nm. It can be seen from Fig. 12 that the HAP/Arg nanoparticles without doping have no characteristic photoluminescence peaks, and the HAP/Arg nanoparticles with Tb (2%, molar fraction) doped appear photoluminescence peaks near 488.6, 542.4, 583.4 and 619.0 nm, which corresponds to the 5D4-7F6, 5D4-7F5, 5D4-7F4 and 5D4-7F3 transition [17], respectively. Meanwhile, the photoluminescence peak at 542.4 nm has the greatest intensity, which can be used for observing the impact of HAP/Arg nanoparticles on cell and the distribution in cell within the scope of thegreen light. As a result, the Tb3+ doping into HAP/Arg nanoparticles makes the photoluminescence of HAP/Arg nanoparticles happen.

Fig. 12 Emission spectra of Tb: HAP/Arg nanopowder sample
4 Conclusions
1) The HAP nanoparticles with high crystallinity and complete crystal form are prepared, and the Zeta potential of HAP nanoparticles is changed and reaches a value of (30.9±8.2) mV through the modification of the arginine, which can provide theoretical basis for improving the efficiency of HAP nanoparticles as gene transfection vectors.
2) To some extent, the addition of arginine inhibits the growth of HAP. Under the same condition, the length of short rod-shaped HAP particles modified by adding the arginine is about 50-80 nm less than the length (about 60-180 nm) of nanoparticles without adding the arginine. Besides, the former had smaller length-to- diameter and more irregular particle shape.
3) A small amount of rare earth elements doped into HAP/Arg nanoparticles does not change the structure of synthetic products, which are of single phase and nano-sized. The main fluorescence emission peaks of Tb-doped HAP/Arg nanoparticles appear at 488.6 nm and 542.4 nm.
References
[1] BROWN W E, CHOW L C. A new calcium phosphate, watersetting cemen [J]. American Ceramic Society, 1987, 4: 352-379.
[2] YANG Chun, GUO Ying-kui, ZHANG Mi-lin. Thermal decomposition and mechanical properties of hydroxyapatite ceramic [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(2): 254-258.
[3] SUEN R B, LIN S C, HSU W H. Hydroxyapatite-based immobilized metal affinity adsorbents for protein purification [J]. Journal of Chromatography A, 2004, 1048(1): 31-39.
[4] LEGEROS R Z. Properties of osteoconductive biomaterials: Calciumphosphates [J]. Clinical Orthopaedics and Related Research, 2002, 395: 81-98.
[5] AOKI H, KUTSUNO T. An in vivo study on the reaction of hydroxyapatite-sol injected into blood [J]. Journal of Materials Science: Materials in Medicine, 2000, 11: 67-72.
[6] LIU Zhi-su, TANG Sheng-li, AI Zhong-li. Effects of hydroxyapatite nanoparticles on proliferation and apoptosis of human hepatoma BEL-7402 cells [J]. World Journal of Gastroenterology, 2003, 9(9): 1968-1971.
[7] ZANG Li-ge. Comparison of the vector capacity of different modified hydroxyapatite nanoparticles as gene vectors [D]. Changsha: Central South University, 2008. (in Chinese)
[8] DO T N, LEE W H, LOO C Y, ZAVGORODNIY A V, ROHANIZADEH R. Hydroxyapatite nanoparticles as vectors for gene delivery [J]. Therapeutic Delivery, 2012, 3(5): 623-632.
[9] MAO Yue-feng. Study on the transfection efficiency of chitosan modified nano hydroxyapatite as gene transfer carrier [D]. Changsha: Central South University, 2009. (in Chinese)
[10] ZUO Gui-fu, WAN Yi-zao, ZHANG Yu. Preparation and characterization of a novel laminated magnetic hydroxyapatite for application on gene delivery [J]. Materials Letters, 2012, 68: 225-227.
[11] LI Yan, YANG Jun, LIU Gui-ying, ZHANG Xin. Research situation and development trend of gene therapy for drug delivery system [J]. Progress in Biochemistry and Biophysics, 2013, 44(10): 998-1007. (in Chinese)
[12] KIKUCHI M. Hydroxyapatite/collagen bone-like nanocomposite [J]. Biological and Pharmaceutical Bulletin, 2013, 36(11): 1666-1669.
[13] LI Shu-li. Preparation and study of hydroxyapatite drug delivery microspheres [D]. Tianjin: Tianjin University, 2008. (in Chinese)
[14] ZHOU Hai-long. Study on the adsorption mechanism of BMP-7 onto HAP surfaces [D]. Zhejiang: Zhejiang University, 2007. (in Chinese)
[15] PAGANO G, GUIDA M, TOMMASI F, ORAL R. Health effects and toxicity mechanisms of rare earth elements-Knowledge gaps and research prospects [J]. Ecotoxicology and Environmental Safety, 2015, 115: 40-48.
[16] FERNANDEZ-OSORIO A, RIVERA C E,VAZQUEZ-OLMOS A, CHAVEZ J. Luminescent ceramic nano-pigments based on terbium-doped zinc aluminate: Synthesis, properties and performance [J]. Dyes and Pigments, 2015, 119: 22-29.
[17] RAMBABU U, AMALNERKAR D P, KALE B B, BUDDHUDU S. Optical properties of LnPO4:Tb3+(Ln=Y, La and Gd) powder phosphors [J]. Materials Chemistry and Physics, 2001, 70(1): 1-6.
(Edited by YANG Bing)
Foundation item: Project(2013SK2024) supported by the Key Projects in Social Development Pillar Program of Hunan Province, China; Project(20130162120094) supported by Specialized Research Fund for the Doctoral Program of Higher Education (SRFDP), Ministry of Education, China; Project supported by State Key Laboratory of Powder Metallurgy, Central South University, China
Received date: 2015-12-16; Accepted date: 2016-04-06
Corresponding author: ZHAO Yan-zhong, Associate Professor, PhD; Tel: +86-731-88618669; E-mail: yanzhongzhao@163.com

