Trans. Nonferrous Met. Soc. China 27(2017) 1856-1861
Kinetic study on carbothermic reduction of ilmenite with activated carbon
Hai-peng GOU1,2, Guo-hua ZHANG1, Xiao-jun HU1, Kuo-chih CHOU1,2
1. State Key Laboratory of Advanced Metallurgy, University of Science and Technology Beijing, Beijing 100083, China;
2. Collaborative Innovation Center of Steel Technology, University of Science and Technology Beijing, Beijing 100083, China
Received 10 May 2016; accepted 22 November 2016
Abstract: The carbothermic reduction of Panzhihua ilmenite with various additions of activated carbon was investigated by isothermal experiments over the temperature range of 1373 to 1773 K in the argon atmosphere. According to the reaction kinetics recorded by the infrared gas analyzer, it was found that the amount of carbon addition had little influence on the reaction rates at various temperatures except 1473 K. When the reaction temperature was above the eutectic temperature of 1427 K of Fe-C binary system, part of carbon would dissolve into Fe to form a liquid phase, which made the liquid Fe as a diffusion channel of carbon to diffuse to the reaction interface. The carbothermic reduction above 1573 K obeyed the shrinking-core model. The mass fraction of TiC could be determined by the standard addition technique.
Key words: ilmenite; carbothermic reduction; activated carbon; titanium carbide; solid-liquid interface
1 Introduction
As is known to all, ilmenite is one of the major sources for titanium. In China, more than 90% (mass fraction) of ilmenite is located in Panzhihua, Sichuan Province. The content of titanium dioxide in ilmenite is higher than 40%. Although the ilmenite is expressed as FeO・TiO2, it can also accommodate many other components in its structure, for instance, Fe2O3, MgO, Al2O3, MnO and CaO [1]. With the exhaustion of the high grade titanium ores, such as rutile, more attention has been paid on ilmenite [2-7]. Nowadays, ilmenite is widely used as an alternative raw material for rutile in producing titanium metal and titanium containing compounds, for example, titanium sponge, synthetic rutile, titanium tetrachloride, titanium carbide, titanium nitride and so on. Therefore, the carbothermic reduction of ilmenite has a crucial influence on the commercial productions of synthetic rutile, titanium carbide, titanium nitride or titanium carbonitride.
Carbothermic reduction of ilmenite was investigated by Welham and Williams from the ambient temperature to 1773 K in Ar atmosphere [8]. It is demonstrated that iron and rutile initially appeared during the carbothermic reduction. Then, rutile was reduced to a series of oxides (TinO2n-1, n>3), Ti3O5, Ti2O3 and TiC in sequence. REZAN et al [9] studied the carbothermic reduction and nitridation of ilmenite in 50%H2-50%N2 (volume fraction) atmosphere and N2 atmosphere, respectively. It is found that Ti2O3 did not appear as an intermediate reaction product if the nitrogen was present. In addition, the reduction rate in 50%H2-50%N2 atmosphere was significantly faster than that in pure nitrogen. GUPTA et al [10,11] studied the kinetics of ilmenite with graphite from 1273 to 1373 K in Ar atmosphere. The reduction reaction rate of ilmenite was increased significantly by the addition of FeCl3・6H2O. It is thought that the addition of FeCl3・6H2O promoted the nucleation of iron. WELHAM and WILLIS [12] and EL-HUSSINY and SHALABI [13] studied the carbothermic reduction of ilmenite in N2 atmosphere from 973 to 1673 K and 1073 to 1423 K, respectively. The diffusion of the solid-state carbon was regarded as the rate determining step for both carbide formation from oxide and nitridation of carbide. However, part of carbon would dissolve into Fe to form a liquid phase when the experimental temperature was above the eutectic temperature of Fe-C binary system 1427 K. This led to a solid-liquid reaction in the reduction process, which was ignored by many researchers. The reduction kinetic and mechanism of solid-liquid reaction during the carbothermic reduction is still not sufficient.
In the present work, the carbothermic reduction of Panzhihua ilmenite was investigated under isothermal conditions to illustrate the reaction kinetics and reaction mechanism. The solid-liquid reaction interface was proposed for the first time in the carbothermic reduction of ilmenite, which played an important role in the dynamic diffusion.
2 Experimental
2.1 Carbothermic reduction of ilmenite with activated carbon
The ilmenite was supplied by Panzhihua Iron and Steel (Group) Co., Ltd, China. It was analyzed by the National Analysis Center for Iron and Steel (NACIS), as listed in Table 1. The activated carbon (analytical reagent, >97%) was supplied by Sinopharm Chemical Reagent Beijing Co., Ltd, China. Both ilmenite and activated carbon were characterized using X-ray diffraction (XRD, Rigaku Ultima IV) with Cu Kα radiation (λ=1.5406  ). The accelerating voltage and the applied current were 40 kV and 40 mA, respectively. XRD patterns of ilmenite and activated carbon are shown in Fig. 1. The ilmenite and the activated carbon were mixed evenly with the addition of polyvinyl alcohol solution (PVA, 2%, mass fraction) in an agate mortar. Then, the mixtures were made into cylindrical briquettes (diameter 18 mm, thickness 4 mm) using a stainless steel mold under a pressure of 230 MPa. All cylindrical briquettes were roasted in an electric resistance furnace (BLMT-1973 K) at 673 K for 2 h under a flowing Ar atmosphere (300 mL/min) to get rid of the PVA. The mass of each cylindrical briquette was 2 g. As shown in Table 2, the mass ratios of activated carbon to ilmenite were set to be 0.276, 0.316 and 0.395, respectively. The reaction temperatures were 1373, 1473, 1573, 1673 and 1773 K, respectively.
). The accelerating voltage and the applied current were 40 kV and 40 mA, respectively. XRD patterns of ilmenite and activated carbon are shown in Fig. 1. The ilmenite and the activated carbon were mixed evenly with the addition of polyvinyl alcohol solution (PVA, 2%, mass fraction) in an agate mortar. Then, the mixtures were made into cylindrical briquettes (diameter 18 mm, thickness 4 mm) using a stainless steel mold under a pressure of 230 MPa. All cylindrical briquettes were roasted in an electric resistance furnace (BLMT-1973 K) at 673 K for 2 h under a flowing Ar atmosphere (300 mL/min) to get rid of the PVA. The mass of each cylindrical briquette was 2 g. As shown in Table 2, the mass ratios of activated carbon to ilmenite were set to be 0.276, 0.316 and 0.395, respectively. The reaction temperatures were 1373, 1473, 1573, 1673 and 1773 K, respectively.
Table 1 Chemical compositions of ilmenite (mass fraction, %)

A schematic diagram of the experimental apparatus is shown in Fig. 2. It consists of a mass flowmeter (Alicat 21-1-10-0-500-Km0410), an electric resistance furnace and an infrared gas analyzer (XLZ-1090). CO2, CO and O2 can be analyzed by the infrared gas analyzer over ranges of 0-30 % (±0.1%), 0-40% (±0.1%) and 0-60% (±0.1%), respectively. Before the experiment, the infrared gas analyzer had been calibrated by a gas mixture of CO2, CO, O2 and Ar under a flow rate of 300 mL/min. In order to diminish the effect of the alumina crucible (diameter 25 mm, height 25 mm) on the reaction temperatures, it was put into the tube at ambient temperature in advance. The furnace was accurately controlled within ±1 K under a flowing Ar atmosphere (300 mL/min). When the furnace temperature reached the desired value, a cylindrical briquette was added into the alumina crucible quickly through a hollow quartz tube (diameter 20 mm). The volume fraction 0of CO was recorded by the infrared gas analyzer every 5 s during the carbothermic reduction process until its value decreased to 0.1%. The reduction products were examined by scanning electron microscope (SEM, Mineral Liberation Analyzer 250, voltage 200 V-30 kV).

Fig. 1 XRD patterns of ilmenite (a) and activated carbon (b)
Table 2 Experimental conditions of carbothermic reduction

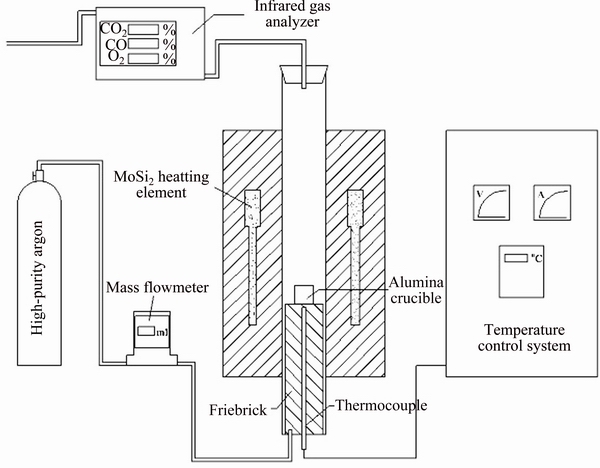
Fig. 2 Schematic diagram of experimental apparatus
2.2 Quantitative determination of titanium carbide in reduction products
The sample with a mass ratio of 0.316 was reduced at 1773 K in the electric furnace under a flowing Ar atmosphere (300 mL/min). After 6 h, the sample was taken out from the furnace and cooled in a flowing Ar atmosphere (300 mL/min). Various mass fractions of pure titanium carbide (≥99.5%, 10 μm) were added in the reduction products. The mass fractions of the added titanium carbide were 5%, 10%, 20%, 30% and 40%, respectively. All the samples were mixed homogenously in an agate mortar and analyzed by XRD. The peak intensities in the XRD patterns reflect the information of the phase’s crystal structure and the distribution of particular atoms in the structure. In the present case, the standard addition technique [14] was employed to determine the mass fraction of TiC in the reduction products.
3 Results
3.1 Influences of mass ratio and reaction temperature on carbothermic reduction of ilmenite
The variation of volume fraction of CO generated by per gram of ilmenite during the carbothermic reduction reaction is shown in Fig. 3. It is important to note that the variation trends of volume fraction of CO with various mass ratios of activated carbon at 1373, 1573, 1673 and 1773 K were overlapping like Fig. 3(b). It is clearly shown that the volume fraction of CO first increased and then decreased as time went by. At the beginning of the reaction, the reactants were rapidly heated up from the ambient temperature to the reaction temperature. At the same time, the generated CO gradually filled the furnace tube evenly and then was carried out by Ar gas to the infrared gas analyzer, which explained why the volume fraction of CO increased rapidly at the very beginning. With the increase of the reduction degree, the unreacted reactants were less and less, which led to the decrease of the volume fraction of CO. It can be seen that the mass ratios had little influence on the reaction rates at various temperatures except 1473 K. The possible reasons for the distinction at 1473 K were given in the section of discussion. The reaction rates were obviously increased by increasing the reaction temperatures.

Fig. 3 Variation of volume fraction of CO generated by per gram of ilmenite
3.2 Mass fraction of TiC in reduction products
The XRD patterns of the reduction products with adding various mass fractions of titanium carbide are shown in Fig. 4(a). The intensity of (200) reflection from TiC (2θ=41.9°) is chosen as comparison because it is the most intense of all reflections from TiC. When plotting the integrated intensities of the (200) reflection of TiC (from 2θ=41.5° to 42.5°) as a function of concentration, a linear relationship is obtained as shown in Fig. 4(b). The x-intercept is the mass fraction of TiC in the reduction products. The obtained relationship is y= 22.44x+879.08, which gives the x-intercept of -39.17. According to Table 1, the theoretical value of the mass fraction of TiC in the reduction products is 41.18%, which provides that all the TiO2 and iron oxide are reduced to TiC and iron, respectively. The result calculated by the standard addition technique is approximate to the theoretical value.

Fig. 4 XRD patterns (a) of reduction products with various mass fractions of titanium carbide and integrated intensities (b) of (200) reflection of TiC as function of concentration
4 Discussion
4.1 Mechanism of carbothermic reduction reaction
The main reactions that occur during the carbothermic reduction are shown as Eq. (1) and Eq. (2). The change of standard Gibbs free energy ΔGΘ for Eqs. (1) and (2) are calculated by a thermodynamic database Factsage 6.4. The reactions of Eq. (1) and Eq. (2) occur when the reaction temperatures are higher than 1127 and 1573 K, respectively.
FeTiO3+C→Fe+TiO2+CO, ΔGΘ=174325.3-154.7T (1)
TiO2+3C→TiC+2CO, ΔGΘ=533760-339.4T (2)
When the reaction temperatures were 1373 and 1473 K, the reduction products were Fe and titanium suboxides (TinO2n-1, n=2, 3) [15]. The reduction reaction that occurred at 1373 K was solid-solid reaction or gas-solid reaction by way of CO2. The reaction rate at 1373 K was too slow to consume the solid-state carbon around the ilmenite. The reactant carbon was sufficient even for group A1. As a result, the increase of amount of carbon addition had little influence on the reaction rate at 1373 K. When the experimental temperatures were above the eutectic temperature of Fe-C binary system 1427 K, a liquid phase composed of Fe and C was formed. The products reduced at 1473 K for 1 h are shown in Fig. 5(a). It can be seen that there were a large number of Fe-C droplets on the surface of samples. The carbon participated into the reaction in both forms of solid state and liquid state in liquid iron. The liquid Fe-C droplets had a better fluidity than the solid carbon particles, which made liquid Fe form a diffusion channel for carbon to contact with titanium suboxides. The reaction rate at 1473 K was much higher than that at 1373 K. It is speculated that the carbon around the ilmenite was rapidly consumed by the carbothermic reduction of ilmenite and the formation of Fe-C droplets. As a result, the increase of amount of carbon addition could increase the contact area between the reactants, which increased the reaction rates at 1473 K.
When the reaction temperatures were above 1573 K, TiC appeared in the reduction products [15]. Figures 5(b) and (c) show the reduction products reduced for 1 h at 1773 K. The reduction products were porous due to the generation of CO. Equation (1) reacted so fast that the generated Fe-C droplets connected together to form a continuous liquid phase. Particles of TiC and Mg2SiO4 were wrapped in the liquid iron. The carbon dissolved in the Fe so fast that most of the carbon participated into the reaction through the liquid phase. In this case, the real concentration of the carbon was not controlled by the amount of carbon addition but the solubility of carbon in the Fe-C liquid phase. Furthermore, the solubility of carbon in the Fe-C liquid phase was only decided by the reaction temperature. This explained the overlaps of the volume fraction of CO curves with various mass ratios above 1573 K like Fig. 3(b).

Fig. 5 SEM images of reduction products reduced for 1 h at different temperatures

Fig. 6 Mechanism of carbothermic reduction of ilmenite above 1573 K
4.2 Shrinking-core model
The carbothermic reduction of ilmenite above 1573 K was the typical solid-liquid interface. As shown in Fig. 5(b), the formation and growth of pore and crack on the unreacted particles were clearly visible. The reaction mechanism was analyzed by using the shrinking-core model [16,17]. To simplify the analyses, the particle shapes of all phases are assumed to be spherical. The shrinking-core model scheme for the carbothermic reduction of ilmenite above 1573 K is shown in Fig. 6. At the beginning of the carbothermic reduction of ilmenite, the carbon participated into the reduction reaction in the form of solid particles. When the reduction product Fe appeared on the surface of the mineral particles, part of carbon dissolved in Fe to form a liquid phase. Due to the good fluidity, the Fe-C droplets gathered and formed a liquid layer out of the unreacted core, which cut off the direct contact between the solid carbon and the unreacted core. Although the content of dissolved carbon in the liquid layer was small, there was a large amount of solid carbon on the outside of product layer. Once the dissolved carbon in the liquid iron was consumed by the titanium suboxides (TinO2n-1, n=2, 3) at the reaction interface, there would be a diffusion of carbon from solid carbon to liquid phase continuously.
5 Conclusions
1) The reaction rate and extent of the carbothermic reduction increase with increasing the reaction temperature.
2) The amount of carbon addition has little influence on the reaction rates at various temperatures except 1473 K.
3) When the reaction temperature is above 1573 K, the reaction rate is dominated by the solid-liquid reaction and the carbothermic reduction obeyed the shrinking-core model.
4) The Fe-C droplets gather and form a liquid layer out of the unreacted core, which make the liquid phase form a diffusion channel for carbon to transport to the reaction interface.
References
[1] BERMAN R G, ARANOVICH L Y. Optimized standard state and solution properties of minerals: Contributions to mineralogy and petrology [J]. Contributions to Mineralogy and Petrology, 1996, 126: 1-24.
[2] XIAO Wei, LU Xiong-gang, ZOU Xing-li, WEI Xue-mei, DING Wei-zhong. Phase transitions, micro-morphology and its oxidation mechanism in oxidation of ilmenite (FeTiO3) powder [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 2439-2445.
[3] GOU Hai-peng, ZHANG Guo-hua, CHOU Kuo-chih. Influence of pre-oxidation on carbothermic reduction process of ilmenite concentrate [J]. ISIJ International, 2015, 55: 928-933.
[4] YUAN Z F, WANG X Q, XU C, LI W B, KWAUK M. A new process for comprehensive utilization of complex titania ore [J]. Minerals Engineering, 2006, 19: 975-978.
[5] KUCUKKARAGOZ C S, ERIC R H. Solid state reduction of a natural ilmenite [J]. Minerals Engineering, 2006, 19: 334-337.
[6] CHEN Min, TANG Ai-tao, XIAO Xuan. Effect of milling time on carbothermic reduction of ilmenite [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 4201-4206.
[7] BABA A A, SWAROOPA S, GHOSH M K, ADEKOLA F A. Mineralogical characterization and leaching behavior of Nigerian ilmenite ore [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 2743-2750.
[8] WELHAM N J, WILLIAMS J S. Carbothermic reduction of ilmenite (FeTiO3) and rutile (TiO2) [J]. Metallurgical and Materials Transactions B, 1999, 30: 1075-1081.
[9] REZAN S A, ZHANG G Q, OSTROVSKI O. Carbothermal reduction and nitridation of ilmenite concentrates [J]. ISIJ International, 2012, 52: 363-368.
[10] GUPTA S K, RAJAKUMAR V, GRIEVESON P. Kinetics of reduction of ilmenite with graphite at 1000 to 1100 oC [J]. Metallurgical Transactions B, 1987, 18: 713-718.
[11] GUPTA S K, RAJAKUMAR V, GRIEVESON P. The influence of weathering on the reduction of ilmenite with carbon [J]. Metallurgical Transactions B, 1989, 20: 735-745.
[12] WELHAM N J, WILLIS P E. Formation of TiN/TiC-Fe composites from ilmenite (FeTiO3) concentrate [J]. Metallurgical and Materials Transactions B, 1998, 29: 1077-1083.
[13] EL-HUSSINY N, SHALABI M. Studying the pelletization of rosseta ilmenite concentrate with coke breeze using molasses and reduction kinetics of produced pellets at 800-1150 °C [J]. Science of Sintering, 2012, 44: 113-126.
[14] ALEXANDER L, KLUG H P. Basic aspects of X-ray absorption in quantitative diffraction analysis of powder mixtures [J]. Analytical Chemistry, 1948, 20: 886-889.
[15] GOU Hai-peng, ZHANG Guo-hua, CHOU Kuo-chih. Phase evolution during the carbothermic reduction process of ilmenite concentrate [J]. Metallurgical and Materials Transactions B, 2014, 46: 48-56.
[16] YOSHIOKA T, MOTOKI T, OKUWAKI A. Kinetics of hydrolysis of poly (ethylene terephthalate) powder in sulfuric acid by a modified shrinking-core model [J]. Industrial & Engineering Chemistry Research, 2001, 40: 75-79.
[17] YOSHIOKA T, OKAYAMA N, OKUWAKI A. Kinetics of hydrolysis of PET powder in nitric acid by a modified shrinking-core model [J]. Industrial & Engineering Chemistry Research, 1998, 37: 336-340.
活性炭碳热还原钛铁矿的动力学研究
苟海鹏1,2,张国华1,胡晓军1,周国治1,2
1. 北京科技大学 钢铁冶金新技术国家重点实验室,北京 100083;
2. 北京科技大学 钢铁共性技术协同创新中心,北京 100083
摘 要:研究氩气气氛下,钛铁矿与不同质量分数的活性炭在1373~1773 K温度区间内的等温碳热还原反应。通过红外气体分析仪得到了碳热还原过程中的动力学曲线。研究结果表明,除1473 K外,在其他反应温度下改变活性碳的添加量几乎不影响反应速率。当反应温度高于Fe-C二元系共晶温度1427 K时,部分碳会溶解在铁中形成液相。此时,碳能通过液态铁传输扩散到反应界面。当反应温度高于1573 K时,碳热还原钛铁矿符合收缩核模型。还原产物中碳化钛的质量分数可以通过标准添加法测量。
关键词:钛铁矿;碳热还原;活性炭;碳化钛;固液反应界面
(Edited by Xiang-qun LI)
Foundation item: Project (FRF-TP-15-009A3) supported by the Fundamental Research Funds for the Central Universities, China; Project (U1460201) supported by the National Natural Science Foundation of China
Corresponding author: Guo-hua ZHANG; Tel: +86-10-62333703; E-mail: ghzhang_ustb@163.com
DOI: 10.1016/S1003-6326(17)60209-7