DOI:10.19476/j.ysxb.1004.0609.2019.07.22
MFC-MEC生物电化学耦合系统回收钴
吴丹菁,潘璐璐,刘维平
(江苏理工学院 化学与环境工程学院,常州 213001)
摘 要:为回收含Co(Ⅱ)溶液中的金属钴,以微生物燃料电池(Microbial fuel cell,MFC)降解有机物产生的电能驱动微生物电解池(Microbial electrolysis cell,MEC),实现MFC-MEC耦合系统自驱动运行。同时,在MEC的阴极还原Co(Ⅱ),研究不同MEC阴极电极材料、pH值以及电极极距等条件对Co(Ⅱ)处理的影响。结果表明:以碳纸为MEC阴极电极材料时,可得到较小的内阻1359.6 Ω,库伦效率CE和阴极效率ηca分别为33.1%和21.6%,钴比收率为2.99 gCo/gCOD,阴极Co(Ⅱ)去除率为62.5%;MEC阴极液pH为3时,ηca为43.9%,钴比收率为0.73 gCo/gCOD,Co(Ⅱ)去除率为88%;MEC极距为16cm时,可得到较高的阳极库伦效率20.5%和钴比收率0.85 gCo/gCOD,阴极Co(Ⅱ)去除率可以达到97.4%。经XRD分析,阴极产物为金属钴。
关键词:微生物燃料电池;微生物电解池;生物电化学;耦合系统;钴回收
文章编号:1004-0609(2019)-07-1536-07 中图分类号:TQ150.9 文献标志码:A
钴是生产锂离子电池的重要原料,从废锂离子电池中回收钴是解决钴资源短缺的重要途径。废锂离子电池正极材料中含有丰富的钴资源,很多研究人员对从废锂离子电池正极材料中回收钴进行了大量研究。研究者们将废电池破碎分选后分离出含钴材料,通过酸浸,碱浸和生物浸出等方式[1-4],将三价钴降价浸出,从含钴浸出液中回收钴。TORKAMAN等[5]研究了3种酸性和两种碱性萃取剂从氯化物酸性水溶液中萃取钴。DUTTA等[6]使用20%的Cyanex 272在pH4.8条件下提取钴。NGUYEN等[7]从硫酸盐浸出液中萃取出99.8%Co。NAYAKA等[8]以甘氨酸和抗坏血酸为浸出剂,以草酸钴的形式回收钴。徐筱群等[9]采用电解剥离-生物质酸浸回收钴,钴的沉淀率92%以上。NAYL等[10]使用NaOH溶液从浸出液中沉淀Co(OH)2。PEREZ等[11]以磷酸盐有机连接剂作为沉淀剂从模拟废锂电池浸出液中沉淀锰和钴。SUN等[12]利用电解法在电极上获得了具有细粒结构致密的钴沉积物。通过萃取、化学沉淀或电解等方法从废锂离子电池正极材料中回收钴需要使用大量的化学试剂,成本较高,同时还会造成二次污染等问题[13-14]。
近年来,电子废弃物处理的发展趋势从传统处理方法向低能耗和环境友好的方向发展。其中,生物法以其环境友好、能耗低、造成的二次污染小和处理效果好等优点备受关注[15-16]。生物电化学系统是一种基于微生物产电的新型电化学系统,主要包括微生物燃料电池(Microbial fuel cell,MFC)和微生物电解池(Microbial electrolysis cell,MEC),由于其环境友好性,且同时具有产能、废弃物处理和金属回收等多重功效,目前在新能源、废水处理、金属回收等领域受到极大关注。MOHAMED等[17]研究了阳离子交换膜扩散层对微生物燃料电池中阴极电位行为的影响。罗净净 等[18]研究了单室MFC的产电性能及其对废水中有机物、氮和磷的去除效果。LIU等[19]以MFC系统处理富含铵/有机物的废水,系统最大电压输出为816.8 mV,功率密度为62.94 mW/m3,阳极COD和总氮去除率分别为88.53%和71.35%。ABOURACHED等[20]使用空气阴极MFC得到了3.6W/m2的发电功率,Cd2+和Zn2+的去除率分别为90%和97%。LI等[21]发现氧化态金属Fe(III)和Cr(VI)作为单室MFC阴极电子受体时,最大功率密度达到658±6 mW/m2,Fe(Ⅲ)和Cr(Ⅵ)的转化效率大于89%。GANGADHARAN等[22]使用MFC技术处理废水中的Cr(Ⅵ)并发电,MFC最大功率密度达767.01mW/m2,Cr(Ⅵ)还原成无毒的Cr(Ⅲ)。王鲁宁等[23]以人工配成的蜜糖废水为阳极底物时,COD去除率为78%,可在阴极回收Cu、Cd和Ni单质,Cr(Ⅵ还原成Cr2O3。FRADLER等[24]使用MFC处理有机物和锌污染物,在72 h内去除了93%±4%的锌离子。LUO等[25]成功利用MEC从酸性矿山废水中回收CuO和NiO,并产生H2。HUANG等[26]利用自驱动耦合系统从锂钴氧化物中回收钴。
目前,利用MFC-MEC生物电化学耦合系统回收钴的研究还非常少,本研究基于生物电化学原理,通过构建MFC-MEC生物电化学耦合系统,利用MFC产生的电能驱动MEC,实现MFC-MEC生物电化学耦合系统的自驱动运行,在不消耗外部电能的情况下,研究不同电极材料、pH值及电极极距等因素对钴回收的影响。
1 实验
1.1 实验装置
MFC-MEC生物电化学耦合系统通过MFC和MEC串联组成,MFC由有机玻璃制成的双室长方体构成,阳极室和阴极室的有效容积均为500 mL,采用阴离子交换膜隔离阳极室和阴极室。阳极材料分别采用碳纸和石墨板,阴极材料分别采用碳纸、不锈钢和钛板。MFC-MEC生物电化学耦合系统装置示意图如图1所示。
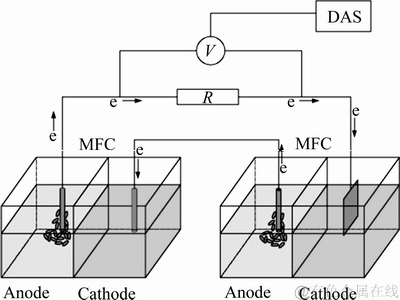
图1 MFC-MEC耦合系统示意图
Fig. 1 Schematic diagram of MFC-MEC coupling system
1.2 实验材料与方法
实验菌种取自污水处理厂的脱水污泥,经驯化后作为阳极接种物。阳极底物及营养液组成成分为1 g/L乙酸钠、0.38 g/L蔗糖、0.21 g/L K2HPO4・3H2O、0.21 g/L KH2PO4、0.12 g/L C6H5O7(NH4)3、0.07 g/L NaCl、0.4 g/L MgCl2・3H2O、0.12 g/L CaCl2、0.4 g/L NH4Cl。
MFC阳极电极材料采用5 cm×5 cm的石墨板,阴极使用5 cm×5 cm的碳纸;阳极室内驯化污泥与底物体积比为1:3;阴极室内为硫酸铜溶液,铜离子浓度500~600 mg/L。MEC阳极采用5 cm×5 cm的碳纸,脱水污泥与底物体积比为1:3,阴极液采用Co(Ⅱ)离子浓度为450~500 mg/L的氯化钴溶液。
实验过程中,当MFC启动完成且输出电压稳定后,将MFC和MEC通过串联方式耦合,整个系统运行过程中保证阳极厌氧环境,实验在常温、常压下进行。
1.3 测试与评价方法
采用数据采集器(MPS-010602,北京,启创莫非电子科技有限公司生产)自动采集MFC输出电压;当输出电压稳定时,采用稳态放电法测定MFC的极化曲线;采用原子吸收光谱仪(200 Series AA,Agilent Technologies Inc,Malaysia.)测定溶液中钴离子浓度;利用X’Pert Powder X射线衍射仪(PW3040/60,PANalytical B.V.,Netherlands)分析阴极产物;通过扫描电镜(S-3400N,Hitachi Limited,Japan)观察阳极表面生物膜的附着情况和MEC阴极产物形貌。
相关参数计算方法如下:
1) 电流密度JA和功率密度PA:
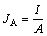 (1)
(1)
 (2)
(2)
式中:I为电流,mA;A为阳极电极表面积,m2;Rex为外电阻,Ω。
2) Co(Ⅱ)去除率ηCo:
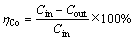 (3)
(3)
式中:Cin为阴极液中Co(Ⅱ)初始浓度,mg/L;Cout为反应后Co(Ⅱ)浓度,mg/L。
3) 库伦效率CE和阴极效率ηca
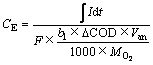 (4)
(4)
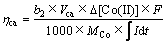 (5)
(5)
式中:I为电流,A;t为反应器运行时间,s;F为法拉第常数;b1为以氧为标准的氧化1 mol有机物转移的电子数;b2为Co(Ⅱ)还原所需要的电子数;ΔCOD为阳极反应初始与终态的COD变化值,mg/L;Δ[Co(Ⅱ)]为MEC阴极室Co(Ⅱ)浓度变化值,mol/L;Van和Vca分别为MEC阳极和阴极有效容积,L;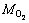 和MCo分别为以氧为标准的有机物摩尔质量和Co的相对分子质量,g/mol。
和MCo分别为以氧为标准的有机物摩尔质量和Co的相对分子质量,g/mol。
4) 钴比收率YCo
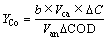 (6)
(6)
式中:b为Co(Ⅱ)与Co的化学计量数之比;Van和Vca分别为MEC阳极室和阴极室有效容积,L;ΔC为阴极反应初始与终态的Co(Ⅱ)浓度变化值,mol/L;ΔCOD为阳极反应初始与终态的COD变化值,mg/L。
2 结果与分析
2.1 MFC-MEC生物电化学耦合系统电性能及钴回收
2.1.1 MFC产电性能
MFC-MEC生物电化学耦合系统中,MFC产电对MEC阴极还原反应具有很大影响,稳定的电压输出能提高金属的析出率[27-28]。MFC启动期间输出电压的变化如图2所示。
从图2看出,在162 h之前电压快速上升达到528 mV,在此阶段,产电微生物在营养物的供养下大量繁殖,代谢增强,产电能力不断提升,随着反应的进行,营养物不断消耗,之后电压上升缓慢并稳定在530 mV左右,在170 h时启动完成。
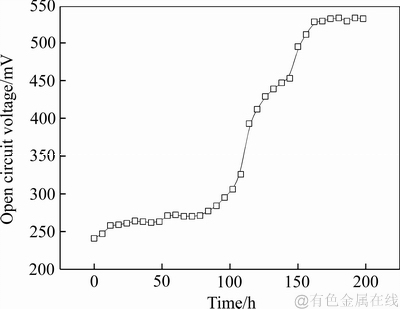
图2 启动期间MFC的输出电压
Fig. 2 Output voltage of MFC during start-up
MFC的极化曲线和功率密度曲线如图3所示,通过对极化曲线的拟合分析,MFC内阻为1887.4 Ω。从图3看出,MFC最大功率密度为1344.8 mW/m2,最大电流密度为28.4 mA/m2,产电性能较好。
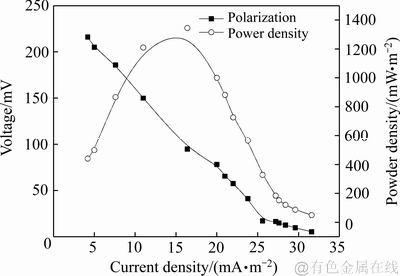
图3 MFC极化曲线和功率密度曲线
Fig. 3 MFC polarization curve and power density curve
2.1.2 Co(Ⅱ)的去除与钴回收
以乙酸钠为阳极底物时,依据能斯特方程,阳极电势为-279 mV,MEC阴极反应为
Co2++2e→Co (7)
阴极电势为-373 mV,平衡电压为-94 mV。从理论上来分析,当阳极底物为乙酸钠时,MEC两端的电压大于94 mV才能发生还原反应,实验中MFC产生的电压达到530 mV,可保证MEC阴极还原反应进行。
耦合系统运行结束后,测定阴极液中Co(Ⅱ)浓度,在MEC极距为16 cm条件下,ηCo达到97.4%。在MEC阴极表面观察到黑褐色沉积物,其形貌如图4所示。阴极产物XRD分析如图5所示,在2θ为51.53°得到一个衍射峰,与标准卡片(01-089-7093)中钴单质的特征峰一致。

图4 MEC阴极表面沉积物的SEM像
Fig. 4 SEM image of deposits on surface of MEC cathode
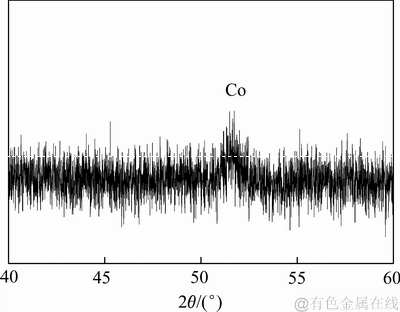
图5 MEC阴极沉积物的XRD谱
Fig. 5 XRD pattern of deposits on MEC cathode
2.2 阴极材料对钴回收的影响
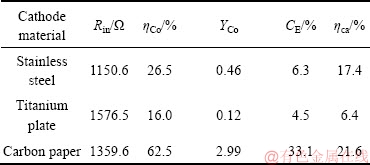
MFC中阳极产生的电子通过外电路到达阴极,在阴极上与金属离子发生还原反应,电极材料的不同会影响金属还原速率[29-30]。实验分别以不锈钢、钛板和碳纸作为MEC阴极电极,研究阴极电极材料对钴回收的影响,结果如表1所列。从表1看出,以碳纸为阴极时,Co(Ⅱ)去除率最高,分别是不锈钢和钛板的2.4倍和3.9倍;从钴回收效率分析,以碳纸为MEC阴极时,钴比收率为2.99 gCo/gCOD,CE和ηca分别达到33.1%和21.6%,均高于不锈钢阴极和钛板阴极的。这是由于碳纸表面粗糙,相对于不锈钢和钛板电极拥有较大的比表面积,会得到较高的回路电流,有利于Co(Ⅱ)的还原。
表1 电极材料对钴回收的影响
Table 1 Effect of electrode materials on cobalt recovery
2.3 阴极液pH对钴回收的影响
调节MEC阴极液pH,分别为1、3和5,实验结果如表2所列。
表2 阴极pH值对钴回收影响
Table 2 Effect of cathode pH on cobalt recovery
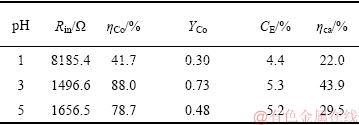
分析表2数据发现,当pH从5降到3时,CE仅提高了0.1%,但ηca和钴比收率都有较大提高,达到43.9%和0.73 gCo/gCOD,Co(II)在阴极液中的去除率也提高了10%。pH值的降低,阳极库伦效率和阴极效率均有明显提高,说明较低的pH值能够提高电子利用率,并促进Co(Ⅱ)还原。但是pH值过低时,氢离子易吸附在生成的钴单质上发生产氢反应:
H++Co+e→CoH (8)
H++CoH+e→Co+H2 (9)
在产氢过程中一部分电子用于生成氢气,会降低Co(Ⅱ)的还原效率。当阴极液pH值降低到1时,阴极效率降为22.0%,钴的比收率和阴极液中Co(Ⅱ)的去除率仅为pH=3时的一半。较低的pH也会影响离子交换膜的通透性和阳极微生物活性,图6所示为阳极微生物附着情况。
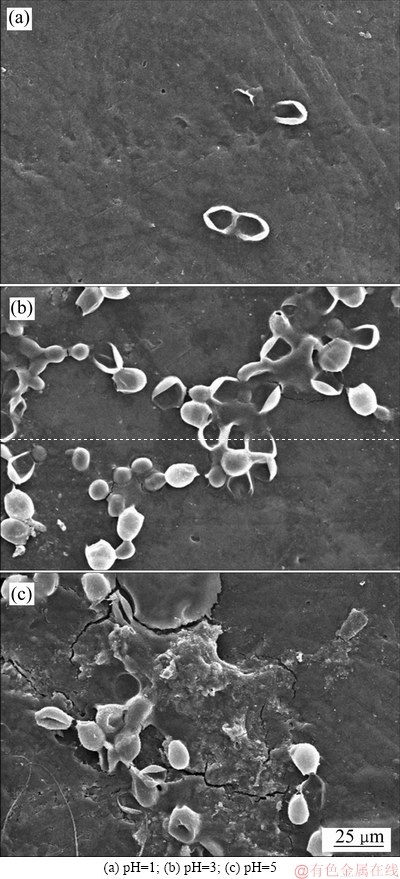
图6 阳极微生物表面附着物的形貌
Fig. 6 Morphologies of microbial attachment on anode surface
从图6看出,pH=1时微生物附着最差,只有少量的微生物附着在电极上;pH为3和5时,电极上能明显看到微生物附着,说明过酸的条件不利于微生物生长,微生物的活性降低导致生物氧化有机物的过程受到影响,同时,电子在极板上的转移速率减缓,当阳极微生物呼吸作用释放电子的速度小于电子流入外电路的速度时,正电荷在阳极上积累,阳极电位向正的方向移动会造成阳极极化,而极化现象会导致内阻增加。从表2数据看出,pH为1的MEC内阻高达8185.4 Ω,是pH为3和5的5.5和4.9倍,此时的库伦效率最低,对钴的回收效果也较差,对比Co(Ⅱ)去除率和钴比收率,可以看出,pH为3时最优,过高和过低的pH值均不利于钴的回收。
2.4 电极极距对钴回收的影响
MEC阳极和阴极间距分别设定为4 cm、8 cm和16 cm,研究不同电极极距对钴回收的影响,其结果如表3所列。
表3 电极极距对钴回收的影响
Table 3 Effect of electrode spacing on cobalt recovery
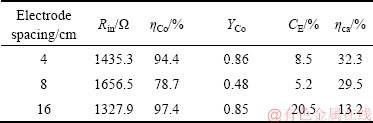
电极极距会影响系统的内阻,电极极距小于最佳值时,可能使阳极微生物活性降低,影响阳极生物膜厌氧微生物的活性及系统的库仑效率。从表3看出,当极距为16 cm时,内阻最小,此时,CE达到20.5%,Co(Ⅱ)去除率达到97.4%。
3 结论
1) 在MFC-MEC生物电化学耦合系统中,MEC可原位利用MFC降解有机物产生的电能,实现MFC-MEC耦合系统自驱动运行,在不消耗外部电能的情况下,Co(Ⅱ)在MEC阴极还原析出。
2) 电极材料、电极极距以及阴极液pH值均会对钴的回收和MFC-MEC耦合系统性能产生影响。MEC阴极采用碳纸时,对钴的回收效果较好;MEC阴极液pH=3,电极极距为16 cm时,系统内阻较小,有利于钴的回收。
REFERENCES
[1] PENG C, HAMUYUNI J, WILSON B P, LUNDSTROM M. Selective reductive leaching of cobalt and lithium from industrially crushed waste Li-ion batteries in sulfuric acid system[J]. Waste Management, 2018, 76: 582-590.
[2] GOLMOHAMMADZADEH R, FARAJI F, RASHCHI F. Recovery of lithium and cobalt from spent lithium ion batteries (LIBs) using organic acids as leaching reagents: A review[J]. Resources, Conservation and Recycling, 2018, 136: 418-435.
[3] XIAO S, REN G, XIE M, PAN B, FAN Y, WANG F, XIA X. Recovery of valuable metals from spent lithium-ion batteries by smelting reduction process based on MnO-SiO2-Al2O3 slag system[J]. Journal of Sustainable Metallurgy, 2017, 27(2): 1-8.
[4] GAO W, LIU C, CAO H, ZHENG X, LIN X,WANG H J, ZHANG Y, SUN Z. Comprehensive evaluation on effective leaching of critical metals from spent lithium-ion batteries[J]. Waste Management, 2018, 75: 477-485.
[5] TORKAMAN R, ASADOLLAHZADEH M, TORAB- MOSTAEDI M, MARAGHEH M G. Recovery of cobalt from spent lithium ion batteries by using acidic and basic extractants in solvent extraction process[J]. Separation & Purification Technology, 2017, 186: 318-325.
[6] DUTTA D, KUMARI A, PANDA R,JHA S,GUPYA D,GOEL S,JHA M K. Close loop separation process for the recovery of Co, Cu, Mn, Fe and Li from spent lithium-ion batteries[J]. Separation & Purification Technology, 2018, 200: 327-334.
[7] NGUYEN V T, LEE J C, JEONG J, KIM B S, PANDEY B D. Selective recovery of cobalt, nickel and lithium from sulfate leachate of cathode scrap of Li-ion batteries using liquid-liquid extraction[J]. Metals & Materials International, 2014, 20(2): 357-365.
[8] NAYAKA G P, PAI K V, SANTHOSH G, MANJANNA J. Recovery of cobalt as cobalt oxalate from spent lithium ion batteries by using glycine as leaching agent[J]. Journal of Environmental Chemical Engineering, 2016, 4(2): 2378-2383.
[9] 徐筱群, 满瑞林, 张 建, 刘 琦, 徐 娟, 孙祖眉. 电解剥离-生物质酸浸回收废旧锂电池[J]. 中国有色金属学报, 2014, 24(10): 2576-2581.
XU Xiao-qun, MAN Rui-lin, ZHANG Jian, LIU Qi, XU Juan, SUN Zu-mei. Electrolytic stripping-biomass acid leaching-recycling spent Li-ion battery[J]. The Chinese Journal of Nonferrous Metals, 2014, 24(10): 2576-2581.
[10] NAYL A A, ELKHASHAB R A, BADAWY S M, EIKHATEEB M A. Acid leaching of mixed spent Li-ion batteries[J]. Arabian Journal of Chemistry, 2017, 2(10): 3632-3639.
[11] PEREZ E, ANDRE M L, NAVARRO A R, HYVRARD F, CARBONI M, MEYER D. Recovery of metals from simulant spent lithium-ion battery as organophosphonate coordination polymers in aqueous media[J]. Journal of Hazardous Materials, 2016, 317: 617-621.
[12] SUN L, QIU K. Organic oxalate as leachant and precipitant for the recovery of valuable metals from spent lithium-ion batteries[J]. Waste Management, 2012, 32(8): 1575-1582.
[13] ORDONEZ J, GAGO E J, GIRARD A. Processes and technologies for the recycling and recovery of spent lithium-ion batteries[J]. Renewable & Sustainable Energy Reviews, 2016, 60: 195-205.
[14] SWAIN B. Recovery and recycling of lithium: A review[J]. Separation & Purification Technology, 2017, 172: 388-403.
[15] NAYCHARAIAH Y V, VENKATA M, LENS P N L. Metals removal and recovery in bioelectrochemical systems: A review[J]. Bioresource Technology, 2015, 195: 102-114.
[16] ELMEKAWY A, SRIKANTH S, BAJRACHARYA S, HEGAB H M, NIGAM P S, SINGH A, VENKATA MOHAM S, PANT D. Food and agricultural wastes as substrates for bioelectrochemical system (BES): The synchronized recovery of sustainable energy and waste treatment[J]. Food Research International, 2015, 73: 213-225.
[17] MOHAMED H O, ABDELKAREEM M A, PARK M, LEE J, KIM T, OJHA G P, PANT B, PARK S J, KIM H Y, BARAKAT N A M. Investigating the effect of membrane layers on the cathode potential of air-cathode microbial fuel cells[J]. International Journal of Hydrogen Energy, 2017, 42(38): 24308-24318.
[18] 罗净净, 周少奇, 许明熠, 陶琴琴. 单室微生物燃料电池产电与脱氮除磷的研究[J]. 环境科学学报, 2016, 36(6): 1955-1961.
LUO Jing-jing, ZHOU Shao-qi, XU Ming-yu, TAO Qin-qin. Electricity generation and recovery of nitrogen and phosphorus in the single-chamber microbial fuel cell[J]. Acta Scientiae Circumstantiae, 2016, 36(6): 1955-1961.
[19] LIU S, LI L, LI H, WANG H, YANG P. Study on ammonium and organics removal combined with electricity generation in a continuous flow microbial fuel cell[J]. Bioresource Technology, 2017, 243: 1087-1096.
[20] ABOURACHED C, CATAL T, LIU H. Efficacy of single-chamber microbial fuel cells for removal of cadmium and zinc with simultaneous electricity production.[J]. Water Research, 2014, 51(6): 228-233.
[21] LI Y, WU Y, PURANIK S, LEI Y, VADAS T, LI B. Metals as electron acceptors in single-chamber microbial fuel cells[J]. Journal of Power Sources, 2014, 269(4): 430-439.
[22] GANGADHARAN P, NAMBI I M. Hexavalent chromium reduction and energy recovery by using dual-chambered microbial fuel cell[J]. Water Science & Technology, 2015, 71(3): 353-358.
[23] 王鲁宁, 孙彩玉, 王佳瑜, 李永峰. 连续流微生物燃料电池处理含重金属废水研究[J]. 环境科学与技术, 2017(5): 107-113.
WANG Lu-ning, SUN Cai-yu, WANG Jia-yu, LI Yong-feng. Continuous flow of microbial fuel cells for metal wastewater treatment and energy recovery[J]. Environmental Science & Technology, 2017(5): 107-113.
[24] FRADLER K R, MICHIE I, DINSDALE R M, GUWY A J, PREMIER G C. Augmenting microbial fuel cell power by coupling with supported liquid membrane permeation for zinc recovery[J]. Water Research, 2014, 55(2): 115-125.
[25] LUO H, LIU G, ZHANG R, BAI Y, FU S, HOU Y. Heavy metal recovery combined with H2 production from artificial acid mine drainage using the microbial electrolysis cell[J]. Journal of Hazardous Materials, 2014, 270(7): 153-159.
[26] HUANG L, YAO B, WU D, QUAN X. Complete cobalt recovery from lithium cobalt oxide in self-driven microbial fuel cell-microbial electrolysis cell systems[J]. Journal of Power Sources, 2014, 259(7): 54-64.
[27] TENG W, LIU G, LUO H, ZHANG R, XIANG Y. Simultaneous sulfate and zinc removal from acid wastewater using an acidophilic and autotrophic biocathode[J]. Journal of Hazardous Materials, 2016, 304: 159-165.
[28] KARTHIKEYAN R, CHENG K Y, SELVAM A, BOSE A, JWC W. Bioelectrohydrogenesis and inhibition of methanogenic activity in microbial electrolysis cells: A review[J]. Biotechnology Advances, 2017, 35(6): 758-771.
[29] LUO S, HE Z. Ni-coated carbon fiber as an alternative cathode electrode material to improve cost efficiency of microbial fuel cells[J]. Electrochimica Acta, 2016, 222: 338-346.
[30] ASENSIO Y, MONTES I B, FERNANDEZ- MARCHANTE C M, LOBATO L, CANIZARES P, RODRIGO M A. Selection of cheap electrodes for two-compartment microbial fuel cells[J]. Journal of Electroanalytical Chemistry, 2017, 785: 235-240.
Recovery of cobalt by MFC-MEC bioelectrochemical coupling system
WU Dan-jing, PAN Lu-lu, LIU Wei-ping
(School of Chemical and Environmental Engineering, Jiangsu University of Technology, Changzhou 213001, China)
Abstract: In order to recover the metal cobalt in the Co(Ⅱ) solution, a microbial electrolysis cell (MEC) was driven by a microbial fuel cell (MFC), in which the degradation of organic matter produced electricity, the MFC-MEC coupling system can be self-driven, and at the same time, the Co(Ⅱ) was reduced in the MEC cathode chamber. The effects of different MEC cathode electrode materials, pH and electrode pole distance on the treatment of cobalt ions were investigated. The results show that carbon paper can be used as the MEC cathode electrode material to obtain a small internal resistance of 1359.6 Ω,CE and ηca are 33.1% and 21.6%, respectively, the specific yield of cobalt is 2.99 gCo/g COD, and the removal rate of Co(Ⅱ) is 62.5%. When the pH of the MEC catholyte is 3, hca reaches 43.9%, the specific yield of cobalt is 0.73 gCo/g COD, and the removal efficiency of Co(Ⅱ) reaches 88%. When the MEC electrode spacing is 16 cm, the highest anode coulombic efficiency is 20.5% and the cobalt specific yield is 0.85 gCo/g COD, the Co(Ⅱ) removal rate in the cathode reaches 97.4%, and the XRD analysis shows that the cathode product is cobalt.
Key words: microbial fuel cell; microbial electrolysis cell; bioelectrochemistry; coupling system; cobalt recovery
Foundation item: Project(BK20131133) supported by Natural Science Foundation of Jiangsu Province, China; Project (CZ20170020) supported by Changzhou Science and Technology Plan, China; Project(SJCX17-0779) supported by Jiangsu Postgraduate Practice Innovation Program, China
Received date: 2018-07-12; Accepted date: 2018-10-16
Corresponding author: LIU Wei-ping; Tel: +86-519-86953092; E-mail: weiping@jsut.edu.cn
(编辑 王 超)
基金项目:江苏省自然科学基金资助项目(BK20131133);常州市科技计划(国际科技合作)项目(CZ20170020);江苏省研究生实践创新计划项目(SJCX17-0779)
收稿日期:2018-07-12;修订日期:2018-10-16
通信作者:刘维平,教授,博士;电话:0519-86953092;E-mail:weiping@just.edu.cn