赤泥碱性阴离子浸出优化及溶解行为
来源期刊:中国有色金属学报(英文版)2018年第6期
论文作者:李晓飞 叶羽真 薛生国 江钧 吴川 孔祥峰 William Hartley 李义伟
文章页码:1248 - 1255
关键词:赤泥;碱性阴离子;碳酸盐;浸出优化;溶解行为
Key words:bauxite residue; alkaline anions; carbonate; leaching optimization; dissolution behavior
摘 要:赤泥是氧化铝工业生产过程中产生的高碱性固体废弃物。基于单因素-正交实验开展赤泥碱性阴离子浸出特性研究,结合多级浸出实验探讨最佳浸出条件、碱性阴离子分布特征、关键碱性离子类型及其溶解行为。结果表明:在液固比2 mL/g、浸出温度30 °C、浸出时间23 h、2次浸出条件下,可溶性碱性阴离子( , , , OH-)的最佳浸出率达86%;赤泥1次浸出液中,88%的阴离子来源于可溶性碱(NaOH、碳酸盐、碳酸氢盐、NaAl(OH)4),12%的阴离子来源于化学结合碱(方解石、钙霞石、水化石榴石);在最佳浸出条件下,可溶性碱性离子浸出总浓度为69.78 mmol/ L, 约占75%;碳酸盐溶解反应的表观活化能为10.24 kJ/mol,这主要受固膜扩散控制。
Abstract: Bauxite residue is a highly alkaline waste containing soluble alkaline anions, which can cause environmental concerns. The optimal leaching conditions, distribution of alkaline anions, types of pivotal alkaline anions and their dissolution behaviors were investigated based on the combination of single factors-orthogonal experiments and leaching stage experiment. Using a two-stage leaching, 86% of the soluble alkaline anions ( , , , OH-) were leached with a L/S ratio of 2 mL/g, at 30 °C, over 23 h. During the first stage of leaching, approximately 88% of alkaline anions were leached from the dissolution of free alkali (NaOH, carbonate, bicarbonate, NaAl(OH)4) with the rest originating from the dissolution of alkaline minerals (calcite, cancrinite and hydrogarnet). Supernatant alkalinity was 69.78 mmol/L with accounting for 75%. Furthermore, carbonate leaching was controlled by solid film diffusion using the Stumm Model with an apparent activation energy of 10.24 kJ/mol.
Trans. Nonferrous Met. Soc. China 28(2018) 1248-1255
Xiao-fei LI1, Yu-zhen YE1, Sheng-guo XUE1,2, Jun JIANG1,2, Chuan WU1,2, Xiang-feng KONG1, William Hartley3, Yi-wei LI1
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Chinese National Engineering Research Center for Control & Treatment of Heavy Metal Pollution, Central South University, Changsha 410083, China;
3. Crop and Environment Sciences Department, Harper Adams University, Newport, Shropshire, TF10 8NB, United Kingdom
Received 21 February 2017; accepted 21 June 2017
Abstract: Bauxite residue is a highly alkaline waste containing soluble alkaline anions, which can cause environmental concerns. The optimal leaching conditions, distribution of alkaline anions, types of pivotal alkaline anions and their dissolution behaviors were investigated based on the combination of single factors-orthogonal experiments and leaching stage experiment. Using a two-stage leaching, 86% of the soluble alkaline anions (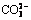 ,
,  ,
,  , OH-) were leached with a L/S ratio of 2 mL/g, at 30 °C, over 23 h. During the first stage of leaching, approximately 88% of alkaline anions were leached from the dissolution of free alkali (NaOH, carbonate, bicarbonate, NaAl(OH)4) with the rest originating from the dissolution of alkaline minerals (calcite, cancrinite and hydrogarnet). Supernatant alkalinity was 69.78 mmol/L with
, OH-) were leached with a L/S ratio of 2 mL/g, at 30 °C, over 23 h. During the first stage of leaching, approximately 88% of alkaline anions were leached from the dissolution of free alkali (NaOH, carbonate, bicarbonate, NaAl(OH)4) with the rest originating from the dissolution of alkaline minerals (calcite, cancrinite and hydrogarnet). Supernatant alkalinity was 69.78 mmol/L with 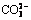 accounting for 75%. Furthermore, carbonate leaching was controlled by solid film diffusion using the Stumm Model with an apparent activation energy of 10.24 kJ/mol.
accounting for 75%. Furthermore, carbonate leaching was controlled by solid film diffusion using the Stumm Model with an apparent activation energy of 10.24 kJ/mol.
Key words: bauxite residue; alkaline anions; carbonate; leaching optimization; dissolution behavior
1 Introduction
Bauxite residue (BR or red mud) is a hazardous alkaline solid waste originating from the Bayer process [1,2], which is used to extract alumina from bauxite ore. Bauxite is digested in a caustic liquor of sodium hydroxide and slaked lime to enhance refining [3,4]. Currently, approximately 120 million tons of BRs are produced per year and the global inventory reached more than 4 billion tons in 2015 [5,6]. Numerous methods for the treatment and reuse of BR have been investigated [7-10], but the volume recycled is still insignificant due to its high alkalinity and salinity [11,12]. Consequently, large volumes of BR are stored in disposal areas (BRDAs), leading to potential contamination of the surrounding environment [13]. The dust formed from the surface of BRDAs, contains elevated concentrations of free alkali (soluble) that may threaten the surrounding ecology [14-16]. Furthermore, storage of residues may cause dam collapse, whilst dissolution of soluble alkaline compounds will also cause environmental risk [17].
Recently, strategies to regulate alkalinity have included, amendment with gypsum, neutralization using seawater, carbonation sequestration, and waste acid interaction [18-23]. Although these methods may be effective, they are difficult to conduct on a large scale. The main reason restricting large-scale application may be the limited knowledge of dissolution behavior and distribution of soluble alkaline anions prior to BR disposal. Presently, water leaching may be an effective technique in order to investigate dissolution behavior and composition distribution of soluble alkaline anions in residues. These are the two main factors that effectively regulate alkalinity, and a reduction in soluble alkaline anions in the liquid phase would be a desirable environmental management goal [24]. Nevertheless, the majority of current research has focused on sodium leaching [25-27] that may only be dissolved partially by water, with a dealkalization ratio of less than 50% [6].
This may be caused by existing alkalinity, including free alkali and chemical bonded alkali in the BR [28] with some alkaline compounds being dissolved under an appropriate chemical equilibrium in the solution, as shown in Eqs. (1)-(9).
NaOH→OH-+Na+ (1)
NaAl(OH)4→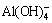 +Na+ (2)
+Na+ (2)
Na2CO3→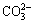 +2Na+ (3)
+2Na+ (3)
NaHCO3→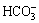 +Na+ (4)
+Na+ (4)
CaCO3→Ca2++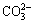 (5)
(5)
3CaO・Al2O3・6H2O→3Ca2++4OH-+Al(OH)4- (6)
[Na6Al6Si6O24]・[2NaX or Na2X]→8Na++6H4SiO4+6Al(OH)3+8X (X: OH- or 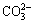 ) (7)
) (7)
[Na6Al6Si6O24]・2[CaCO3]+26H2O→6Na++2Ca2++6H4SiO4+6Al(OH)3+ +8OH- (8)
+8OH- (8)
Ca3Al2(SiO4)x(OH)12-4x→3Ca2++(6-4x)OH-+xH4SiO4+2Al(OH)3 (9)
Equations (6)-(9) were interpreted from Refs. [29-32].
Consequently, in order to elucidate the dissolution of soluble alkalinity, sodium leaching is not accurate, due to dissolution of chemically bonded alkali and other minerals, which makes it difficult to take both dealkalization specification and technical cost into consideration [33].
The objectives of this work are to investigate the optimal leaching conditions of BR (including L/S ratio, temperature, time and leaching stage), to determine alkaline dissolution behavior and reaction equilibrium, to screen alkaline anions and analyze their leaching kinetics, and to comprehend alkaline anion dissolution behavior, composition distribution and alkalinity contribution in BR. Based on the above, we aim to provide a scientific foundation for the effective regulation of alkalinity in BR prior to disposal.
2 Experimental
2.1 Materials
Raw BR (PRM) was collected from the Guangxi Branch, Aluminum Corporation of China Limited. Samples were firstly thoroughly homogenized, dried at 65 °C for 48 h, and subsequently sieved (100 mesh). Soluble cations were determined using inductively coupled plasma atomic emission spectroscopy (ICP-AES). Mineral composition of the PRM was determined by X-ray powder diffraction (XRD) analysis.
2.2 Methods
The following investigations were conducted to assess leaching potential: Firstly, single factor experiments were used to assess L/S ratio, leaching temperature and leaching time, respectively. L/S ratios were 1, 2, 3, 4, 5, 6 and 7 mL/g, temperatures were controlled at 10, 15, 20, 25, 30, 35 and 40 °C, and time was controlled as 1, 2, 3, 5, 8, 13, 18 and 23 h. For the second investigation, three factors and three levels were set based on the results of the single factor experiments in order to obtain the optimized leaching condition. Finally, the leaching stage experiments (denoted as 1, 2, 3, 4, 5 and 6) were operated using the optimized conditions to investigate the source and content of alkaline anions, which were used to obtain the leaching rate of alkaline anions from the dissolution of free alkali.
Bauxite residue (20 g), was mixed with deionized water in a beaker, the mixture was then stirred vigorously for 15 s, and then sealed with a plastic membrane. The experiment was conducted in a temperature controlled water bath. Leachates were filtered through SHZ-D (III) vacuum suction filters and pH, alkaline anions and aluminum concentration were immediately analyzed. pH was carried out using a PHS-3C and the content of hydroxide (OH-) was calculated from pH. Soluble Al was determined by ICP-AES and aluminate  was calculated from the Al concentration [4,26].
was calculated from the Al concentration [4,26].
Carbonate (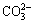 ) and bicarbonate (
) and bicarbonate (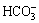 ) were determined by double indicator-neutralization titration using H2SO4 (0.005 mol/L) [23]. Initially, leachate (5 mL) was diluted with deionized water (10 mL), and phenolphthalein (0.05 mL) was added until the solution was titrated to pH 8.3 (T1). Next, bromophenol blue (0.1 mL) was added and the solution was then titrated to pH 4.1 (T2). From T1 and T2, the concentrations of
) were determined by double indicator-neutralization titration using H2SO4 (0.005 mol/L) [23]. Initially, leachate (5 mL) was diluted with deionized water (10 mL), and phenolphthalein (0.05 mL) was added until the solution was titrated to pH 8.3 (T1). Next, bromophenol blue (0.1 mL) was added and the solution was then titrated to pH 4.1 (T2). From T1 and T2, the concentrations of 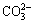 and
and 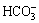 were determined, respectively.
were determined, respectively.
3 Results and discussion
Sodium salt was the main component of soluble alkali, with Al originating from dissolution of aluminate (Table 1). XRD patterns of raw bauxite residue revealed that the main minerals consisted of calcite (CaCO3), cancrinite (Na6CaAlSi(CO3)O24・2H2O), hydrogarnet (Ca3AlFe2(SiO4)(OH)4)), hematite (Fe2O3), goethite (FeOOH), diaspore (AlO(OH)), gibbsite (Al(OH)3) and perovskite (CaTiO3)) (Fig. 1).
Table 1 Cation concentrations under leaching of soluble alkali in raw bauxite residue (mg/L)

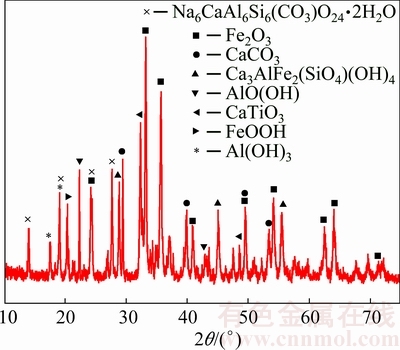
Fig. 1 XRD patterns of raw bauxite residue
3.1 Effect of L/S ratio
Effects of L/S ratio on basic alkaline parameters (pH, alkaline anion concentration and leachate alkalinity) of BR under the leaching process are presented in Tables 2 and 3 (25 °C, 23 h, the 1st stage leaching). Supernatant pH decreased with increasing L/S ratio, probably due to a dilution effect from the water. Alkaline anions reached a maximum concentration when the L/S ratio was 2 mL/g, and the concentrations of 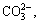
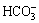 and
and 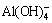 reached 37.20, 9.90 and 1.90 mmol/L, respectively. When the L/S ratio continuously increased, the concentration of alkaline anions slowly decreased. And the alkaline anion concentrations were in the following order:
reached 37.20, 9.90 and 1.90 mmol/L, respectively. When the L/S ratio continuously increased, the concentration of alkaline anions slowly decreased. And the alkaline anion concentrations were in the following order: 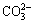 >
>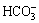 >
>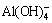 >OH-. Thus, the L/S ratio of 2 mL/g may be used to regulate alkalinity due to the high concentration of alkaline anions, which is beneficial to the reaction rate.
>OH-. Thus, the L/S ratio of 2 mL/g may be used to regulate alkalinity due to the high concentration of alkaline anions, which is beneficial to the reaction rate.
Leaching amounts of alkaline anions are shown in Table 3, which were used to investigate the effect of L/S ratio on the dissolution behavior of alkaline anions. These anions were largely dissolved when the L/S ratio reached 2 mL/g. According to the investigations, L/S ratios of 2, 4 and 5 mL/g should be considered for the orthogonal experimental conditions.
3.2 Effect of leaching temperature
Dissolution behaviors of alkaline anions related to leaching temperature are presented in Fig. 2 (L/S ratio of 4 mL/g, 23 h, the 1st stage leaching). Carbonate revealed a positive correlation with leaching temperature from 15 to 25 °C (Fig. 2), with 18.2 mmol/L obtained at 25 °C. Concentrations of 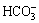 ,
, 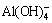 and OH- were lower than that of
and OH- were lower than that of 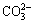 , with no change observed with increasing temperature. This demonstrated that
, with no change observed with increasing temperature. This demonstrated that 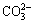 may dissolve continuously with increasing temperature until both dissolution reactions and hydrolysis gradually reached equilibrium after 25 °C. Supernatant pH rapidly increased with increasing temperature (<30 °C) (Fig. 3) eventually stabilizing. In addition, alkaline anion concentrations increased slowly above 25 °C (27.11 mmol/L at 25 °C). This development may be attributed to the dynamic balance of dissolution reactions, ion conversion and adsorption in the mixture above 25 °C. Leaching temperatures of 20, 25 and 30 °C may therefore be considered for the orthogonal experimental conditions.
may dissolve continuously with increasing temperature until both dissolution reactions and hydrolysis gradually reached equilibrium after 25 °C. Supernatant pH rapidly increased with increasing temperature (<30 °C) (Fig. 3) eventually stabilizing. In addition, alkaline anion concentrations increased slowly above 25 °C (27.11 mmol/L at 25 °C). This development may be attributed to the dynamic balance of dissolution reactions, ion conversion and adsorption in the mixture above 25 °C. Leaching temperatures of 20, 25 and 30 °C may therefore be considered for the orthogonal experimental conditions.
Table 2 Effect of L/S ratio on pH, alkaline anions and alkalinity of leachates
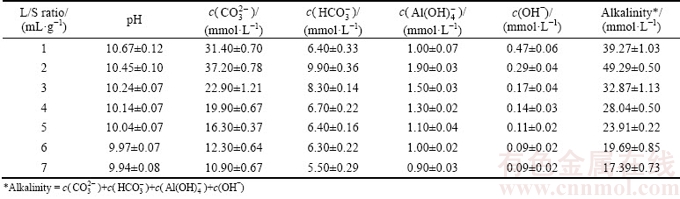
Table 3 Effect of L/S ratio on leaching amount of alkaline anions
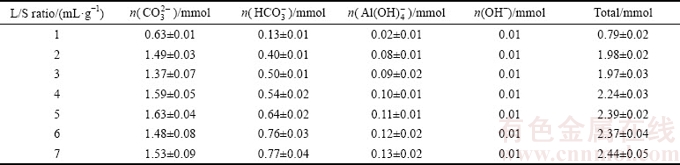

Fig. 2 Effect of leaching temperature on change in alkaline anion concentration
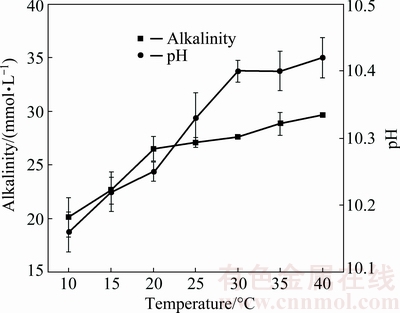
Fig. 3 Effect of leaching temperature on change in pH and alkalinity
3.3 Effect of leaching time
Effects of leaching time on the dissolution behavior of alkaline anions are presented in Fig. 4 (L/S ratio of 4 mL/g, 25 °C, the 1st stage leaching). Leaching time appeared to affect 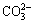 concentration, reaching 17.30 mmol/L at 13 h, but taper off after this point (Fig. 4). Concentrations of
concentration, reaching 17.30 mmol/L at 13 h, but taper off after this point (Fig. 4). Concentrations of 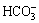 ,
, 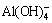 and OH- altered only slightly with increasing leaching time, being almost completely leached within 1 h.
and OH- altered only slightly with increasing leaching time, being almost completely leached within 1 h. 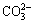 is a critical alkaline anion in BR, but alkaline anions originating from the dissolution of chemically bonded alkali or free alkali may also be involved. pH of leachates persisted at 10.30 with alkalinity of 17.30 mmol/L at 13 h (Fig. 5). Leaching time of 13, 18 and 23 h may be considered for the orthogonal experimental conditions.
is a critical alkaline anion in BR, but alkaline anions originating from the dissolution of chemically bonded alkali or free alkali may also be involved. pH of leachates persisted at 10.30 with alkalinity of 17.30 mmol/L at 13 h (Fig. 5). Leaching time of 13, 18 and 23 h may be considered for the orthogonal experimental conditions.
3.4 Orthogonal experimental analysis for optimized leaching conditions
Orthogonal experiments were used to optimize alkaline anion leaching conditions (Table 4), and the results are presented in Table 5. The effect of leaching factors was as follows: leaching temperature > L/S ratio > leaching time (Table 5). This implied that leaching temperature was the main factor to influence leaching rate as demonstrated by dissolution and diffusion of alkaline anions, which is closely related to temperature.
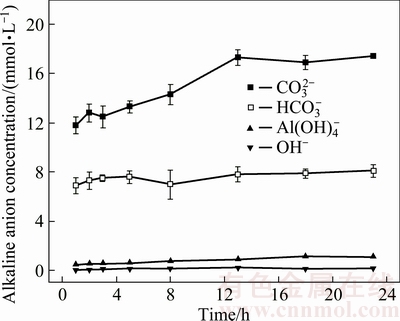
Fig. 4 Effect of leaching time on changes in alkaline anion concentration
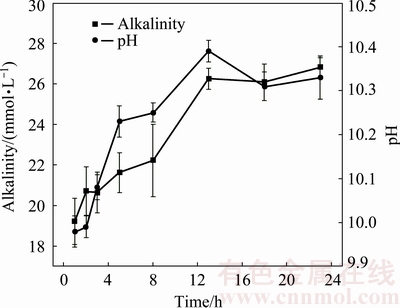
Fig. 5 Effect of leaching time on change in pH and alkalinity
Table 4 Factors and levels of orthogonal experiment

3.5 Dissolution equilibrium of multiple leaching stages
Table 5 Orthogonal experiment results


Fig. 6 Effect of leaching stage on amount of total alkaline anions (a) and free alkaline anions (b)
With increasing leaching stage, the amount of alkaline anions rapidly reduced (Fig. 6(a)). Amounts of 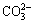 and
and 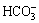 reached 1.52 and 0.49 mmol in leachates at the first stage, respectively. However, after the third stage, these alkaline anions showed negligible changes. According to alkali dissolution equilibrium in the solid-liquid phase, alkaline anions from dissolution of free alkali were completely leached after the sixth stage. The concentrations of alkaline anions generated from the dissolution equilibrium of the chemically bonded alkali are shown in Table 6. Dissolutions of chemically bonded alkalis (CaCO3, Na6CaAlSi(CO3)O24・ 2H2O, Ca3AlFe2(SiO4)(OH)4) were unaffected by the higher concentration of alkaline anions when soluble alkalinity was completely removed. Only a few alkaline anions were leached at the 6th stage. Concentrations of
reached 1.52 and 0.49 mmol in leachates at the first stage, respectively. However, after the third stage, these alkaline anions showed negligible changes. According to alkali dissolution equilibrium in the solid-liquid phase, alkaline anions from dissolution of free alkali were completely leached after the sixth stage. The concentrations of alkaline anions generated from the dissolution equilibrium of the chemically bonded alkali are shown in Table 6. Dissolutions of chemically bonded alkalis (CaCO3, Na6CaAlSi(CO3)O24・ 2H2O, Ca3AlFe2(SiO4)(OH)4) were unaffected by the higher concentration of alkaline anions when soluble alkalinity was completely removed. Only a few alkaline anions were leached at the 6th stage. Concentrations of 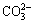 ,
, 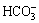 ,
, 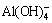 and OH- were 3.10, 3.80, 0.21, 0.04 mmol/L with pH reaching 9.57, respectively. Assuming the dissolution balance of chemically bonded alkali would not be affected by leaching stages, the contents of alkaline anions originating from the dissolution of free alkali were calculated using the following equations:
and OH- were 3.10, 3.80, 0.21, 0.04 mmol/L with pH reaching 9.57, respectively. Assuming the dissolution balance of chemically bonded alkali would not be affected by leaching stages, the contents of alkaline anions originating from the dissolution of free alkali were calculated using the following equations:
 (10)
(10)
 (11)
(11)
 (12)
(12)
 (13)
(13)
where n is the leaching stage (denoted as 1, 2, 3, 4, 5, and 6); Fa is the total concentration of alkaline anions from the dissolution of free alkali; cb is the concentration of alkaline anions from the dissolution of chemically bonded alkali; Ls is the concentration of alkaline anions from the different leaching stages; and Rn(%) is the leaching ratio.
Calculated results are shown in Table 6. Alkalinity of the liquid phase in the BR was improved by alkaline anions from the dissolution of free alkali. The effect of leaching stage on the dissolution of free alkali is shown in Fig. 6(b). During the first stage, the concentration of 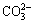 and
and 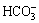 reached 1.40 and 0.34 mmol in leachates, respectively. Alkaline anions were almost unchanged after the third stage. Alkalinity reached 69.78 mmol/L (Table 6), and the leaching ratio was calculated using Eq. (13) (Fig. 7). Leaching ratio reached 67% during the first stage, reducing to 19%, 9%, 4%, 2% and 0%, at the second to sixth stages, respectively. The total leaching ratio was 86% within two leaching stages. Table 6 reveals that the critical alkaline anion from dissolution of free alkali was
reached 1.40 and 0.34 mmol in leachates, respectively. Alkaline anions were almost unchanged after the third stage. Alkalinity reached 69.78 mmol/L (Table 6), and the leaching ratio was calculated using Eq. (13) (Fig. 7). Leaching ratio reached 67% during the first stage, reducing to 19%, 9%, 4%, 2% and 0%, at the second to sixth stages, respectively. The total leaching ratio was 86% within two leaching stages. Table 6 reveals that the critical alkaline anion from dissolution of free alkali was 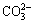 and its concentration reached 52.10 mmol/L, followed by
and its concentration reached 52.10 mmol/L, followed by 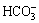 (13.60 mmol/L).
(13.60 mmol/L).
3.6 Leaching kinetics of critical anions
Due to the highly soluble alkalinity, the Stumm Model may be applied to evaluating leaching kinetics of critical anions. The Shrinking Core Model (SCM) was used to assess the dissolution process of the solid phase whilst analyzing the leaching process of sodium [6,34]. The critical anion 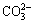 was the main source of high alkalinity in the liquid phase. It is therefore necessary to analyze its leaching process. The following two expressions of leaching kinetics can be used.
was the main source of high alkalinity in the liquid phase. It is therefore necessary to analyze its leaching process. The following two expressions of leaching kinetics can be used.
Table 6 Leaching parameters of chemically bonded alkali and free alkali in bauxite residue
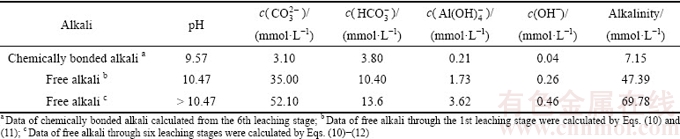
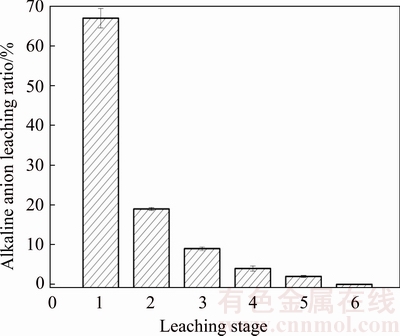
Fig. 7 Effect of leaching stage on leaching ratio from dissolution of free alkali
The Stumm Model [35]:
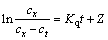 (14)
(14)
where t is the reaction time; cx and ct are the concentrations of 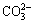 in dissolution equilibrium and at the time of t, respectively; Z is a constant; and Kq is the kinetics rate constant.
in dissolution equilibrium and at the time of t, respectively; Z is a constant; and Kq is the kinetics rate constant.
SCM diffusion model:
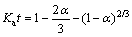 (15)
(15)
where α is the leaching ratio (%); Ka is the rate constant of internal diffusion.
Leaching data at different temperatures exhibited a good fit to the Stumm Model (Fig. 8 and Fig. 9), demonstrating that 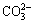 leaching from dissolution of free alkali was controlled by solid film diffusion. Its rate increased with increasing L/S ratio and temperature, whilst another factor was the diffusion rate of water into interior particles. Based on the diffusion constants at different temperatures, the plots of ln Kq versus temperature were established by the Arrhenius equation. Correlation coefficients (R2) were above 0.99 at different observed temperatures (Fig. 10). The apparent activation energy (Ea) was 10.24 kJ/mol, further illustrating that the diffusion rate of
leaching from dissolution of free alkali was controlled by solid film diffusion. Its rate increased with increasing L/S ratio and temperature, whilst another factor was the diffusion rate of water into interior particles. Based on the diffusion constants at different temperatures, the plots of ln Kq versus temperature were established by the Arrhenius equation. Correlation coefficients (R2) were above 0.99 at different observed temperatures (Fig. 10). The apparent activation energy (Ea) was 10.24 kJ/mol, further illustrating that the diffusion rate of 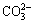 was controlled by solid film diffusion.
was controlled by solid film diffusion.

Fig. 8 Plot of [1-2α/3-(1-α)2/3] vs time at different temperatures

Fig. 9 Plot of ln[cx/(cx-ct)] vs time at different temperatures

Fig. 10 Plot of ln Kq vs temperature of water leaching
4 Conclusions
1) Under the optimized conditions, with a L/S ratio of 2 mL/g, at 30 °C, for 23 h, leaching rates for soluble alkaline anions (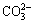 ,
,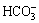 ,
,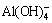 , OH-) of 86% and pH of 9.78 were obtained during a two-stage leaching.
, OH-) of 86% and pH of 9.78 were obtained during a two-stage leaching.
2) Approximately 88% of alkaline anions were leached from the dissolution of free alkali during the first stage, with the remainder originating from alkaline minerals (CaCO3, Na6CaAlSi(CO3)O24・2H2O, Ca3AlFe2(SiO4)(OH)4).
3) The alkaline anion 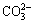 accounted for 75% of the total soluble alkalinity (69.78 mmol/L), with
accounted for 75% of the total soluble alkalinity (69.78 mmol/L), with 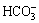 accounting for 19%.
accounting for 19%.
4) Carbonate leaching was controlled by solid film diffusion using the Stumm Model, with an apparent activation energy of 10.24 kJ/mol.
References
[1] KONG Xiang-feng, JIANG Xing-xing, XUE Sheng-guo, HUANG Ling, HARTLEY W, WU Chuan, LI Xiao-fei. Migration and distribution of salinity in bauxite residue during water leaching [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(3): 534-541.
[2] ZHU Feng, HOU Jing-tao, XUE Sheng-guo, WU Chuan, WANG Qiong-li, HARTLEY W. Vermicompost and gypsum amendments improve aggregate formation in bauxite residue [J]. Land Degradation & Development, 2017, 28: 2109-2120.
[3] XUE Sheng-guo, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei, LI Yi-wei. Proposal for management and alkalinity transformation of bauxite residue in China [J]. Environmental Science and Pollution Research, 2016, 13: 12822-12834.
[4] KIRWAN L J, HARTSHORN A, MCMONAGLE J B, FLEMING L, FUNNELL D. Chemistry of bauxite residue neutralization and aspects to implementation [J]. International Journal of Mineral Processing, 2013, 119: 40-50.
[5] XUE Sheng-guo, ZHU Feng, KONG Xiang-feng, WU Chuan, HUANG Ling, HUANG Nan, HARTLEY W. A review of the characterization and revegetation of bauxite residues (red mud) [J]. Environmental Science and Pollution Research, 2016, 23: 1120-1132.
[6] ZHU Xiao-bo, LI Wang, GUAN Xue-mao. An active dealkalization of red mud with roasting and water leaching [J]. Journal of Hazardous Materials, 2015, 286: 85-91.
[7] ZHU Feng, HUANG Nan, XUE Sheng-guo, HARTLEY W, HAN Fu-song, LI Yi-wei. Effects of binding materials on micro-aggregate size distribution in bauxite residues [J]. Environmental Science and Pollution Research, 2016, 23: 23867-23875.
[8] XUE Sheng-guo, SHI Li-zheng, WU Chuan, WU Hui, QIN Yan-yan, PAN Wei-song, HARTLEY W, CUI Meng-qian. Cadmium, lead, and arsenic contamination in paddy soils of a mining area and their exposure effects on human HEPG2 and keratinocyte cell-lines [J]. Environmental Research, 2017, 156: 23-30.
[9] LU Qing-Hua, HU Yue-Hua. Synthesis of aluminum tri-polyphosphate anticorrosion pigment from bauxite tailings [J]. Transactions of Nonferrous Metals Society of China, 2012, 22: 483-488.
[10] WU Chuan, HUANG Liu, XUE Sheng-guo, PAN Wei-song, ZOU Qi, HARTLEY W, WONG Ming-hung. Oxic and anoxic conditions affect arsenic (As) accumulation and arsenite transporter expression in rice [J]. Chemosphere, 2017, 168: 969-975.
[11] ZHU Feng, LI Yu-bing, XUE Sheng-guo, HARTLEY W, WU Hao. Effects of iron-aluminium oxides and organic carbon on aggregate stability of bauxite residues [J]. Environmental Science and Pollution Research, 2016, 23: 9073-9081.
[12] MAYES W M, JARVIS A P, BURKE I T, WALTON M, FEIGL V, KLEBERCZ O, GRUIZ K. Dispersal and attenuation of trace contaminants downstream of the Ajka bauxite residue (red mud) depository failure Hungary [J]. Environmental Science & Technology, 2011, 45: 5147-5155.
[13] ZHU Feng, LI Xiao-fei, XUE Sheng-guo, HARTLEY W, WU Chuan, HAN Fu-song. Natural plant colonization improves the physical condition of bauxite residue over time [J]. Environmental Science and Pollution Research, 2016 23: 22897-22905.
[14] LIU Wan-chao, CHEN Xiang-qing, LI Wang-xing, YU Yan-fen, YAN Kun. Environmental assessment, management and utilization of red mud in China [J]. Journal of Cleaner Production, 2014, 84: 606-610.
[15] ZHU Feng, ZHOU Jia-yi, XUE Sheng-guo, HARTLEY W, WU Chuan, GUO Ying. Aging of bauxite residue in association of regeneration: A comparison of methods to determine aggregate stability & erosion resistance [J]. Ecological Engineering, 2016, 92: 47-54.
[16] GOLOTAN J B, CHEN Cheng-rong, PHILLIPS I R, ELSER J J. Shifts in leaf N:P stoichiometry during rehabilitation in highly alkaline bauxite processing residue sand [J]. Scientific Reports, 2015, 5: 1-12.
[17] ZHU Feng, XUE Sheng-guo, HARTLEY W, HUANG Ling, WU Chuan, LI Xiao-fei. Novel predictors of soil genesis following natural weathering process of bauxite residue [J]. Environmental Science and Pollution Research, 2016, 23: 2856-2863.
[18] COURTNEY R, KIRWAN L. Gypsum amendment of alkaline bauxite residue-Plant available aluminium and implications for grassland restoration [J]. Ecological Engineering, 2012, 42: 279-282.
[19] ZHU Feng, CHENG Qing-yu, XUE Sheng-guo, LI Chu-xuan, WU Chuan, TIAN Tao. Influence of natural regeneration on fractal features of residue microaggregates in bauxite residue disposal areas [J]. Land Degradation and Development, 2018, 29: 138-149.
[20] MENZIES N W, FULTON I M, MORRELL W J. Seawater neutralization of alkaline bauxite [J]. Journal of Environmental Quality,2004, 33: 1877-84.
[21] HAN Y S, JI S, LEE P K, OH C. Bauxite residue neutralization with simultaneous mineral carbonation using atmospheric CO2 [J]. Journal of Hazardous Materials, 2017, 326: 87-93.
[22] ZHU Feng, LIAO Jia-xing, XUE Sheng-guo, HARTLEY W, ZOU Qi, WU Hao. Evaluation of aggregate microstructures following natural regeneration in bauxite residue as characterized by synchrotron-based X-ray micro-computed tomography [J]. Science of Total Environmental, 2016, 573: 155-163.
[23] KONG Xiang-feng, LI Meng, XUE Sheng-guo, HARTLEY W, CHEN Cheng-rong, WU Chuan, LI Xiao-fei, LI Yi-wei. Acid transformation of bauxite residue: Conversion of its alkaline characteristics [J]. Journal of Hazardous Materials, 2017, 324: 382-390.
[24] JONES B E H, HAYNES R J. Bauxite processing residue: A critical review of its formation, properties, storage, and revegetation [J]. Environmental Science and Technology,2011, 41: 271-315.
[25] BORGES A J P, HAUSER-DAVIS R A, OLIVEIRA T F D. Cleaner red mud residue production at an alumina plant by applying experimental design techniques in the filtration stage [J]. Journal of Cleaner Production, 2011, 19: 1763-1769.
[26] HUANG Yan-fang, CHAI Wen-cui, HAN Gui-hong, WANG Wen-juan, YANG Shu-zhen, LIU Jiong-tian. A perspective of stepwise utilisation of Bayer red mud: Step two-Extracting and recovering Ti from Ti-enriched tailing with acid leaching and precipitate flotation [J]. Journal of Hazardous Materials, 2016, 307: 318-327.
[27] YANG Yang, WANG Xue-wen, WANG Ming-yu, WANG Hua-guang, XIAN Peng-fei. Recovery of iron from red mud by selective leach with oxalic acid [J]. Hydrometallurgy, 2015, 157: 239-245.
[28] KONG Xiang-feng, GUO Ying, XUE Sheng-guo, HARTLEY W, WU Chuan, YE Yu-zhen, CHENG Qing-yu. Natural evolution of alkaline characteristics in bauxite residue [J]. Journal of Cleaner Production, 2017, 143: 224-230.
[29] SANTINI T C, HINZ C, RATE A W, CARTER C M, GILKES R J. In situ neutralisation of uncarbonated bauxite residue mud by cross layer leaching with carbonated bauxite residue mud [J]. Journal of Hazardous Materials, 2011, 194: 119-127.
[30] RADOMIROVIC T, SMITH P, SOUTHAM D, TASHI S, JONES F. Crystallization of sodalite particles under Bayer-type conditions [J]. Hydrometallurgy, 2013, 137: 84-91.
[31] PARADIS M, DUCHESNE J, LAMONTAGNE A, ISABEL D. Long-term neutralisation potential of red mud bauxite with brine amendment for the neutralisation of acidic mine tailings [J]. Applied Geochemistry, 2007, 22: 2326-2333.
[32] KONG Xiang-feng, TIAN Tao, XUE Sheng-guo, HARTLEY W, HUANG Long-bin, WU Chuan, LI Chu-xuan. Development of alkaline electrochemical characteristics demonstrates soil formation in bauxite residue undergoing natural rehabilitation [J]. Land Degradation & Development, 2018, 29: 58-67.
[33] KINNARINEN T, HOLLIDAY L, HAKKINEN A. Dissolution of sodium, aluminum and caustic compounds from bauxite residues [J]. Minerals Engineering,2015, 79:143-151.
[34] FERRIER R J, CAI L P, LIN Q Y, GORMAN G J, NEETHLING S J. Models for apparent reaction kinetics in heap leaching: A new semi-empirical approach and its comparison to shrinking core and other particle-scale models [J]. Hydrometallurgy, 2016, 166: 22-33.
[35] STUMM W, MORGAN J J. Introduction of natural water body chemical balance [M]. TANG Hong-xiao, trasl. Beijing: Science Press, 1987: 129-134. (in Chinese).
李晓飞1,叶羽真1,薛生国1,2,江 钧1,2,吴 川1,2,孔祥峰1,William Hartley3,李义伟1
1. 中南大学 冶金与环境学院,长沙 410083;
2. 中南大学 国家重金属污染防治工程技术研究中心,长沙 410083;
3. Crop and Environment Sciences Department, Harper Adams University, Newport, Shropshire, United Kingdom TF10 8NB
摘 要:赤泥是氧化铝工业生产过程中产生的高碱性固体废弃物。基于单因素-正交实验开展赤泥碱性阴离子浸出特性研究,结合多级浸出实验探讨最佳浸出条件、碱性阴离子分布特征、关键碱性离子类型及其溶解行为。结果表明:在液固比2 mL/g、浸出温度30 °C、浸出时间23 h、2次浸出条件下,可溶性碱性阴离子(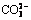 ,
, ,
, , OH-)的最佳浸出率达86%;赤泥1次浸出液中,88%的阴离子来源于可溶性碱(NaOH、碳酸盐、碳酸氢盐、NaAl(OH)4),12%的阴离子来源于化学结合碱(方解石、钙霞石、水化石榴石);在最佳浸出条件下,可溶性碱性离子浸出总浓度为69.78 mmol/ L,
, OH-)的最佳浸出率达86%;赤泥1次浸出液中,88%的阴离子来源于可溶性碱(NaOH、碳酸盐、碳酸氢盐、NaAl(OH)4),12%的阴离子来源于化学结合碱(方解石、钙霞石、水化石榴石);在最佳浸出条件下,可溶性碱性离子浸出总浓度为69.78 mmol/ L,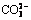 约占75%;碳酸盐溶解反应的表观活化能为10.24 kJ/mol,这主要受固膜扩散控制。
约占75%;碳酸盐溶解反应的表观活化能为10.24 kJ/mol,这主要受固膜扩散控制。
关键词:赤泥;碱性阴离子;碳酸盐;浸出优化;溶解行为
(Edited by Xiang-qun LI)
Foundation item: Project (41371475) supported by the National Natural Science Foundation of China; Project (201509048) supported by the Environmental Protection’s Special Scientific Research for Chinese Public Welfare Industry
Corresponding author: Sheng-guo XUE; Tel: +86-13787148441; E-mail: sgxue70@hotmail.com; sgxue@csu.edu.cn
DOI: 10.1016/S1003-6326(18)64763-6

