J. Cent. South Univ. (2012) 19: 1517-1521
DOI: 10.1007/s11771-012-1170-7
Preparation and biocompatibility evaluation of poly(D, L-lactide-co-glycine)
HU Yao-ping(��ҫƽ)1, CAO Lei-jian(����)1, GU Li(����)2,
LI Qing-hua(���廪)1, SHEN Hong-xia(���ϼ)2, ZHENG Tao(֣��)2
1. School of Materials Science and Engineering, Changzhou University, Changzhou 213164, China;
2. Institute of Polymer Material Engineering, Jiaxing University, Jiaxing 314001, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: D,L-lactide was prepared from D,L-lactic acid by means of polymerization and depolymerization at low vacuum level. Morpholine-2,5-dione (MD) was synthesized from the cyclization of chloroacetyl glycine which was made from chloroacetyl chloride and glycine in the basic condition. A novel copolymer (PLAMD) with D,L-lactide (D,L-LA) and morpholine-2,5-dione (MD) was synthesized using stannous octoate as initiator, and characterized with FT-IR and 1HNMR. The biocompatibility of PLAMD and PLA was investigated by MTT and microscope. The results show that amino acid is introduced into PDLLA main chain. PLAMD has better cell affinity than PLA, so it is a promising biomaterial.
Key words: morpholine-2,5-dione; lactide; amino acid; biocompatibility
1 Introduction
Biodegradable polymeric biomaterials are preferred candidates for developing therapeutic devices such as sutures, surgical insulation, integral artificial skin, temporary prostheses, three-dimensional porous structures as scaffolds for tissue engineering and as controlled/sustained release drug delivery vehicles. Each of these applications requires materials with specific physical, chemical, biological, biomechanical and degradation properties to provide efficient therapy [1-2]. One such polymer is polylactide (PLA), a biodegradable, bioabsorbable, biocompatible, processable and less energy dependence renewably derived thermoplastic polyester which has been extensively investigated over the last several decades [3]. Although PLA is an eco-friendly biomaterial, it has drawbacks as well, especially as functional scaffolds leading to aching pain and even inflammation for related tissue. Certain chemical modifications for PLA with maleic anhydride [4], diaminoethane [5], ethanediol [6], and glycolic acid [7] have been accomplished. As natural biomolecule, ��-amino acids can be used to improve the biocompatibility of PLA by copolymerization with different ratios of amino acid and lactide. Alanine [8], leucine [9], glutaminic acid [10], valine [11] and methionine [12] were copolymerized with lactide respectively, glycine was copolymerized with lactic acid by direct melting polymerization recently [13], and copolymerization of morpholine-2,5-dione (with aspartic acid protected by ��-benzyloxycarbonyl) and L-lactide was studied [14]. Herein, a preparation method of glycine modified PLA was reported by block copolymerization of morpholine-2,5-dione and D,L- lactide using stannous octoate as initiators, and a preliminary biocompatibility evaluation was performed.
2 Experimental
2.1 Materials
D,L-lactic acid (AP), acetic ester (CP), glycine (AP), chlorocetic chloride (CP), dimethyl formamide (AP) and triethylamine (AP) were purchased from Shanghai Chemical Reagent Company, and composite oxide catalyst (being prepared in our lab) and stannous octoate (CP) were purchased from Sigma Company.
2.2 Preparation of D, L-lactide
D,L-lactide using lactic acid as starting material was prepared by reduced pressure distillation technique [15-16]. D,L-lactic acid with a volume of 300 mL was put into a three-neck flask, the free water was removed by keeping the reaction system at the temperature of 80 ��, by reducing pressure and stirring simultaneously, then 3.06 g of La-Ti composite oxide nanocrystal [17] was added into the system as catalyst to promote polycondensation of lactic acid. The temperature was kept at 140 �� for 4-5 h, and the color of the reaction system turned from white into light yellow. Stop vacuum, stir swiftly and replace the receiving flask, then the reaction system was warmed up to 200 ��, and was kept in high vacuum. The light yellow crude lactide was distilled from reaction flask. White and pure lactide was obtained by recrystallization using acetic ester as solvent.
2.3 Preparation of morpholine-2,5-dione
Based on Ref. [18], glycine (C2H5NO2, 0.1 mol) and sodium hydroxide solution (NaOH, 4 mL/mol, 26 mL) were added into three-neck flask (250 mL). The reaction system was kept at temperature of 0 �� and stirred slowly. Meanwhile, chloroacetic chloride (C2H2Cl2O, 9.6 mL) and sodium hydroxide solution (NaOH, 4 mL/mol, 32 mL) were mixed and dropped into the reaction system. The reaction was kept for 2 h at the temperature of less than 10 ��. The pH of the reaction system was regulated to be 2 by sulfuric acid, then reaction solution was extracted for three times by acetic ester and rinsed by sodium chloride, desiccated by anhydrous magnesium sulfate, and recrystallized by acetic ester, finally, N-chloroacetylglycine (C4H6ClNO3) was obtained.
N-chloroacetylglycine (2.0 g) and N,N-dimethyl- formamide (HCON(CH3)2, 34 mL) were added into three-neck flask with thermometer and constant pressure funnel. The reaction system was stirred and warmed up to 80 ��, then the mixture of DMF (66 mL) and triethylamine ((CH3CH2)3N, 1.95 mL) was dropped into reaction system slowly. After keeping reaction for 4 h, stop the heating and remove DMF by decompressing. Then, the residue in the flask was dissolved by chloroform (CHCl3, 30 mL), and recrystallized by acetonitrile (CH3CN) [19]. Finally, white morpholine-2, 5-dione prismatic crystal was obtained.
2.4 Preparation of glycine modified PLA
Certain amount of D,L-lactide and morpholine-2,5- dione were mixed evenly and put into a flask, then chloroform solution with stannous octoate was added as initiator. After being kept under ultrasonic vibration for 20 min, the flask was depressed at 35 �� for 2 h and sealed under vacuum, then the flask was kept in vacuum drying oven at 160 �� for 28 h. After being purified by tetrahydrofuran (C4H8O) and deposited by water, the brown yellow sample (PLAMD) was accomplished.
2.5 Analysis and characterization
NMR experiments were conducted in a Varian Inova 400 with tetralmethylsilane as internal standard. Samples were solved in dimethylsulfoxide-d6. IR spectrum of the as-prepared morpholine-2,5-dione and PLAMD was recorded by a Nicolet-IR550 infrared spectrometer.
The tests of biocompatibility of samples were performed by Tetrazolium Salts colorimetry assay and microscopy. Certain amounts of PLAMD and PLA were dissolved in tetrahydrofuran to form solution with 5% (mass fraction) of samples [20]. After being poured onto the surface of culture dish and vacuum dried, a sheet of membrane was obtained. After being sterilized by ultraviolet radiation, the membranes of PLAMD and PLA were put into 96-well plates (four samples per group). Vero cells were inoculated into the 96-well plates, then were incubated in carbon dioxide incubator for 2 d at (37��1) ��. The specimens were taken out and tested by enzyme-labelled meter (with detecting wave length of 490 nm, Bio-RAD680). The microscopy analysis was performed by inverted phase-contrast microscope (OlympusCKX31SF). Vero cells were incubated with PLAMD and PLA membranes for 2 d, and membranes were taken out and observed under microscope.
3 Results and discussion
3.1 Chemical structure identification of morpholine-2, 5-dione
In order to verify the actual chemical compositions of the prepared samples, the NMR and IR spectra were obtained. Figure 1 shows the IR spectrum of the prepared morpholine-2,5-dione. The peak at 3 410.29 cm-1 originates from the stretching vibration of free stated secondary amide (N��H). The peak centered at 3 204.82 cm-1 originates from the stretching vibration of associated secondary amide (N��H). The peak centered at 2 936.25 cm-1 originates from symmetrical and asymmetrical stretching vibration of C��H bonds. The peak centered at 1 754.76 cm-1 originates from the stretching vibration of C=O of ester bonds. The peak centered at 1 694.15 cm-1 originates from the stretching vibration of C=O of amide I, and the peak centered at 1 502.25 cm-1 originates from the stretching vibration of C=O of amide II. In the fingerprint regions, more absorption bands are found, which are consistent with the structure of cycloester. Consequently, IR analysis confirms that the products are morpholine-2, 5-dione.
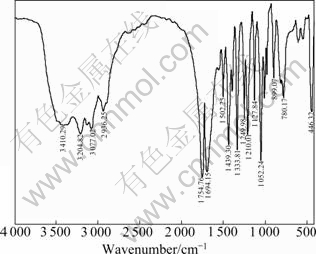
Fig. 1 IR spectrum of morpholine-2,5-dione
As shown in Fig. 2, the 1H NMR peaks at about 8.42��10-6, 4.69��10-6, and 4.00��10-6 are assigned to secondary amide proton, OCH2 protons and NHCH2 protons, respectively. There are not any impurity peaks. The ratios of peak areas are 1:2:2, which are identical with the structure of morpholine-2,5-dione.

Fig. 2 1H NMR spectrum of morpholine-2,5-dione
3.2 Chemical structure identification of poly-lactide- morpholine-2,5-dione (PLAMD)
Figure 3 shows the IR spectrum of the prepared poly-lactide-morpholine-2,5-dione, with the ratio of lactide to morpholine-2,5-dione as 4:1. The peak at 3 399.82 cm-1 originates from the stretching vibration of amide (N��H). The peaks centered at 2 996.29 cm-1 and 2 948.48 cm-1 originate from the stretching vibration of methyl (CH3). The peak centered at 1 757.63 cm-1 originates from the stretching vibration of C=O of ester bond. The peak centered at 1 686.35 cm-1 originates from the stretching vibration of amide I, and the peak centered at 1 544.17 cm-1 originates from the stretching vibration of amide II. The peaks centered at 1 454 and 1 383 cm-1 originate from the bending vibration of methyl. The vibration intensity of C=O is much more than that of amide I. All the characteristic peaks of the poly morpholine-2,5-dione and polylactide can be marked obviously.

Fig. 3 IR spectrum of poly-lactide-morpholine-2,5-dione
With ratio of lactide to morpholine-2,5-dione as 4:1, the copolymerization of lactide and morpholine-2,5- dione was performed and tested by NMR, as shown in Fig. 4, and the 1H NMR peak at about 1.44��10-6 was assigned to methyl protons of dimethylsulfoxide-d6. The peak at 5.18��10-6 was assigned to methylene protons of D, L-lactide. The peak at 8.50��10-6 was assigned to protons of nitrogen in amide group, and the peaks at about 4.60��10-6 and 4.00��10-6 were assigned to OCH2 protons and NHCH2 protons of morpholine-2,5-dione, respectively. All the 1H NMR peaks are characteristic peaks of D, L-lactide and morpholine-2,5-dione, which suggests that the copolymerization of lactide and morpholine-2,5-dione is successful. The mole fraction of all the monomers for the copolymer can be calculated with the principle, i.e. the ratio of integral area of protons peaks is equal to that of relative molar mass of related functional group. With the following equation: cMD= (��(CH2)/4)/((��(CH)+��(CH3))/4) = (4.11/4)/((3.51+11.31)/ 4), the molar ratio (cMD) of MD to LA in prepared coplolymer is 0.227, which is a little more than the initial ratio of MD to LA, suggesting that the activity of morpholine-2,5-dione is better than that of D, L-lactide.
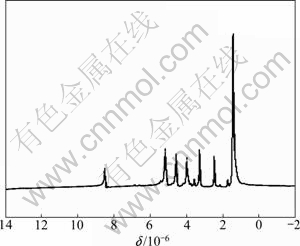
Fig. 4 1H NMR spectrum of poly-lactide-morpholine-2,5-dione
3.3 Biocompatibility evaluation of PLAMD
Cytotoxicity of materials is the most key factor for evaluation of interaction of biomaterials and cells, and cell relative proliferation rate performs as an important parameter for evaluating the material cytotoxicity [21]. In this work, MTT method was employed to calculate the relative proliferation rate of cells, and the cytotoxicity grade was obtained by conversion from cell relative proliferation rate, which was then used to evaluate the cytotoxicity of prepared copolymers [22]. Being co-incubated in carbon dioxide incubator for 2 d, the absorbance of the adherent cells on membranes of PLA and PLAMD was tested by MTT colorimetry, as shown in Fig. 5. The absorbance of Vero cells on PLA membrane amounted to be 0.281, while that of Vero cells on PLAMD membrane was 0.400, and their relative reproduction degrees were 99.42% and 99.46% respectively, indicating that the cytotoxicity of membrane from PLA and PLAMD into grade one and suggesting the safety and innocuity of the prepared materials.

Fig. 5 Absorbance of Vero cells on surface of as-prepared membrane
In addition, based on the direct proportion between cytoactivity and absorbance of cell [23], the absorbance of Vero cells on PLAMD membrane is much more than that on PLA membrane, suggesting the better cytoactivity for Vero cells on the glycine modified PLA membrane than that on pure PLA membrane.
Observing the interaction between the material and cell is the key factor in studying the biocompatibility of tissue engineering material. With direct contact culturing, cell surface adhesion on biomaterials can be observed directly, where once the toxic matter is released from biomaterials, the shape, number and reproduction state must be affected obviously, which can be used as visual evidence for obtaining objective and effective evaluation [24]. With inverted phase contrast microscope, the Vero cells cultured on the surface of PLA and PLAMD membrane were observed, as shown in Fig. 6. In contrast to PLA membrane, the Vero cells flourished as fusiform shape spreading well on the surface of PLAMD membrane and kept better shape. Therefore, with MTT method and microscope, the results indicated that the glycine modified PLA enjoyed lower cytotoxicity and better cellular affinity than PLA and can be used for biomaterials.

Fig. 6 Photos of Vero cells on surface of as-prepared materials observed under inverted phase contrast microscope: (a) PLA group; (b) PLAMD group
4 Conclusions
1) With chloroacetic chloride and glycine as starting materials, copolymer of lactic acid and glycine is obtained by three reaction steps. With the molar ratio of monomers as 4:1 for lactide and morpholine-2,5-dione, the copolymer appears as a ratio of 3.6:1 for lactide and morpholine-2,5-dione, suggesting better reaction activity of morpholine-2,5-dione than lactide.
2) With solvent evaporation method, membranes of PLA and PLAMD are achieved. With MTT method and microscope, the results indicate that the glycine modified PLA enjoys lower cytotoxicity and better cellular affinity than PLA and can be used for biomaterials.
3) The present work is only a new try on the preparation of glycine modified poly lactide. Some other works are in need of perfection and fulfillment, including thermal stability, molecular mass, and degradation features.
References
[1] RASAL R M, JANORKARC A M V, HIRT D E. Poly(lactic acid) modifications [J]. Progress in Polymer Science, 2010, 35(1): 338- 356.
[2] NAIRA L S, LAURENCIN C T. Biodegradable polymers as biomaterials [J]. Progress in Polymer Science, 2007, 32(8/9): 762- 798.
[3] REZWAN K, CHEN Q Z, BLAKER J J, BOCCACCINI A R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering [J]. Biomaterials, 2006, 27(18): 3413-3431.
[4] [4] CAO Xue-bo, WANG Yuan-liang, PAN Jun, LU Xiao, CAI Shao-xi. The change in mechanical properties of mod if ied polylactide with maleic anhydride [J]. Polymer Materials Science and Engineering, 2002, 18(1): 115-118. (in Chinese)
[5] LUO Yan-feng, WANG Yuan-liang, NIU Xu-feng, WU Yang-lan, PAN Jun, SHI Liang-ping. Synthesis and characterization of ethylene diamine modified poly (D, L-lactic acid) [J]. Polymer Materials Science and Engineering, 2005, 21(2): 139-143. (in Chinese)
[6] RUAN Gang, FENG Si-Shen. Preparation and characterization of poly(lactic acid)�Cpoly(ethylene glycol)�Cpoly (lactic acid) (PLA�CPEG�CPLA) microspheres for controlled release of paclitaxel [J]. Biomaterials, 2003, 24(27): 5037-5044.
[7] RASALA R M, JANORKARC A, HIRT D E. Poly(lactic acid) modifications [J]. Progress in Polymer Science, 2010, 35: 338-356.
[8] HOU Xiao-na, WANG Zhao-yang, YE Rui-rong, MAO Zheng-zhou. Influences of feed ratio on the biodegradable material polylactic acid modified by l-alanine via direct melt copolymerization [J]. Material Review, 2009, 23(6): 36-40. (in Chinese)
[9] PENG Ya-liu, HUANG Yong, CHUANG Hui-ju, KUO Chen-yuan, LIN Chu-chieh. Synthesis and characterization of biodegradable polylactides and polylactide-block-poly(Z-lysine) copolymers [J]. Polymer, 2010, 51(19): 4329-4335.
[10] YE Rui-rong, WANG Zhao-yang, MO Yang-qing, LUO Shi-he. Study on synthesis of biomaterial poly( lactic acid-co-glutamic acid) by direct melt copolymerization [J]. Materials Review, 2010, 24(4): 94-97. (in Chinese)
[11] HOU Xiao-na, WANG Zhao-yang, LUO Yu-fen, YE Rui-rong. Synthesis of poly(lactic acid-co-va line) via direct melt copolycondensation [J]. Chinese Journal of Applied Chemistry, 2009, 26(3): 273-276. (in Chinese)
[12] YANG Li-chuan, YE Rui-rong, YU Shi-qin, LI Jing-ning, WANG Zhao-yang. Synthesis and characterization of biomaterial poly( lactic acid-co-methionine) [J]. Materials Science and Engineering, 2010, 26(12): 129-132. (in Chinese)
[13] YE Rui-rong, WANG Qun-fang, WANG Zhao-yang, LUO Shi-he, MAO Chao-xu. Studies on the properties of poly lactic acid directly modified by different amino acids [J]. Chemical Research and Application, 2010, 22(9): 1126-1131. (in Chinese)
[14] TANG Zhi-rong, HUANG Hong, RAO Ju, CHENG Shu-jun. Copolymerization of morphel ine-2, 5-dione and lactide [J]. Journal of East China University of Science and Technology, 2002, 28(6): 618-620. (in Chinese)
[15] CUI Guo-zhen, HE Ji-dong, DONG Jing-jing, LI Yong-zhen. Study on optimization of synthesis and purification of L-Lactide [J]. Chemistry & Bioengineering, 2009, 26(12): 41-44. (in Chinese)
[16] GU Li. Study on optimization of technology in preparing lactide by reduced pressure distillation [J]. Journal of Jiaxing University, 2003, 15(6): 18-20. (in Chinese)
[17] GU Li, LI Fei, MA Hong-xia, ZHANG Yang. Synthesis of optically active lactide catalyzed by nanocrystals of La-Ti composite oxides [J]. Journal of Central South University of Technology, 2008, 15(4): 469-473.
[18] SHEN Zheng-rong, CHEN Hui-chuang. Preparation of morpholine-2,5-dione [J]. Chemical Reagent, 1999, 1(5): 311-317. (in Chinese)
[19] NELSON T D, ROSEN J D, BRANDS K M J, CRAIG B, HUFFMAN M A, MCNAMARA J M. Syntheses of morpholine-2,3- diones and 2-hydroxymorpholin-3-ones: Intermediates in the synthesis of aprepitant [J]. Tetrahedron Letters, 2004, 45(22): 8917- 8920.
[20] YAN Qiong-jiao, LI Shi-pu, YIN Yi-xia, LI Juan. Preparation and characterization of RGD peptide modification of poly{(lactic acid)-co-[(glycolic acid)-alt-(L-lysine)]} [J]. Journal of Central South University: Science and Technology, 2008, 39(6): 1190-1195. (in Chinese)
[21] WILLIAMS D F. Systematic aspects of biocompatibility (Vol. 2) [M]. Boca Raton: CRC Press, 1981: 45-80.
[22] CIAPETTI G, CENNI E, PRATELLI L, PIZZOFERRATO A. In vitro evaluation of cell/biomaterial interaction by MTT assay [J]. Biomaterials, 1993, 14(5): 359-364.
[23] SU Ai-hua, WANG Yuan-liang, LUO Yan-feng, TANG Li-ling. Effect of ethylene diamine modified to polylactic acid on osoteoblasts [J]. Chinese Journal of Clinical Rehabilitation, 2004, 8(5): 866-868. (in Chinese)
[24] KATSIKARI A, PATRONIDOU C H R, KIPARISSIDES C, ARSENAKIS M. Uptake and cytotoxicity of poly(D,L-lactide-co- glycolide) nanoparticles in human colon adenocarcinoma cells [J]. Materials Science and Engineering B, 2009, 165(3): 160-164.
(Edited by HE Yun-bin)
Foundation item: Project(21107032) supported by the National Natural Science Foundation of China; Projects(Y406469, Y4110606) supported by the Natural Science Foundation of Zhejiang Province, China; Projects(2008AY2018, 2011AY1048-5, 2011AY1030) supported by the Science Foundation of Jiaxing Science and Technology Bureau, China; Project(2009C21003) supported by the Science and Technology Department of Zhejiang Province, China
Received date: 2011-03-23; Accepted date: 2011-06-16
Corresponding author: GU Li, Professor, PhD; Tel: +86-573-83646195; E-mail: guli@mail.zjxu.edu.cn