Separation of sulfide lead-zinc-silver ore under low alkalinity condition
来源期刊:中南大学学报(英文版)2012年第8期
论文作者:孙伟 苏建芳 张刚 胡岳华
文章页码:2307 - 2315
Key words:lead-zinc-silver sulfide; low alkalinity; new flotation reagents; lead-zinc separation; silver recovery
Abstract: A complex lead-zinc-silver sulfide ore containing 2.98% Pb, 6.49% Zn and 116.32×10-4 % Ag (mass fraction) from Yunnan Province, China, was subjected to this work. Research on mineral processing was conducted according to the properties of the lead-zinc-silver ore. Under low alkalinity condition, the lead minerals are successfully separated from the zinc minerals with new reagent YZN as zinc depressant, new reagent BPB as lead collector, CuSO4 as zinc activator and ethyl xanthate as zinc collector. The associated silver is mostly concentrated to the lead concentrate. With the process utilized in this work, a lead concentrate of 51.90% Pb with a recovery of 82.34% and a zinc concentrate of 56.96% Zn with a recovery of 81.98% are produced. The silver recovery in the lead concentrate is 80.61%. Interactions of flotation reagents with minerals were investigated, of which the results indicate that the presence of proper amount of Na2S can precipitate Pb2+ and has a sulfidation on oxidized lead minerals. The results also show that Na2CO3 and YZN used together as combined depressants for sphalerite can signally improve the depressing effect of new reagent YZN on sphalerite.
基金信息:the National Natural Science Foundation of China
J. Cent. South Univ. (2012) 19: 2307-2315
DOI: 10.1007/s11771-012-1276-y![]()
SUN Wei(孙伟), SU Jian-fang(苏建芳), ZHANG Gang(张刚), HU Yue-hua(胡岳华)
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: A complex lead-zinc-silver sulfide ore containing 2.98% Pb, 6.49% Zn and 116.32×10-4 % Ag (mass fraction) from Yunnan Province, China, was subjected to this work. Research on mineral processing was conducted according to the properties of the lead-zinc-silver ore. Under low alkalinity condition, the lead minerals are successfully separated from the zinc minerals with new reagent YZN as zinc depressant, new reagent BPB as lead collector, CuSO4 as zinc activator and ethyl xanthate as zinc collector. The associated silver is mostly concentrated to the lead concentrate. With the process utilized in this work, a lead concentrate of 51.90% Pb with a recovery of 82.34% and a zinc concentrate of 56.96% Zn with a recovery of 81.98% are produced. The silver recovery in the lead concentrate is 80.61%. Interactions of flotation reagents with minerals were investigated, of which the results indicate that the presence of proper amount of Na2S can precipitate Pb2+ and has a sulfidation on oxidized lead minerals. The results also show that Na2CO3 and YZN used together as combined depressants for sphalerite can signally improve the depressing effect of new reagent YZN on sphalerite.
Key words: lead-zinc-silver sulfide; low alkalinity; new flotation reagents; lead-zinc separation; silver recovery
1 Introduction
As an important mineral resource of nonferrous metals, lead-zinc ore plays an important role in Chinese economy. Lead and zinc metals are widely used in the fields such as electrical industry, mechanical industry, military industry, metallurgical industry, chemical industry, light industry and medical industry. China is rich in lead and zinc resources which approximately account for 20% of the world gross. Lead and zinc resources in China are mainly distributed in Yunnan, Gansu, Guangxi, Guangdong, Hunan, Jiangxi, Shanxi, Sichuan and Xinjiang Provinces, China [1-2].
In the past decades, many studies on flotation separation of lead-zinc-silver sulfide ores have been carried out in China, most of which were conducted under high pH condition. Fankou Lead-Zinc Mine, China, has developed a fine grinding-preferential flotation technology under high pH condition after years of technical improvement for separation of refractory lead-zinc ore with high pyrite content [3]. MO et al [4] put regulator CaO, collector diethyl dithiocarbamate and butyl xanthate into mill, utilize CaO to regulate and stabilize the pulp potential to achieve efficient separation of lead, zinc and pyrite minerals. QIN et al [5] improved the flotation indexes of Mengzi Lead-silver-zinc Ore, China, by pulp potential controlled flotation. In practice, the commonly used method for the recovery of sulfide lead and zinc minerals is high pH-preferential flotation technology [6].
The traditional high pH technology has disadvantages such as slow flotation speed, large CaO consumption, which makes the pipeline jammed easily, and low recovery of associated silver and gold. In order to preclude the disadvantages above, research on mineral processing was conducted according to the properties of a lead-zinc-silver ore from Yunnan Province, China. A low alkalinity-preferential flotation process was employed to achieve efficient flotation separation of lead and zinc minerals, during which the associated silver was mostly concentrated to the lead concentrate. Interactions of flotation reagents with minerals were also studied.
2 Experimental
2.1 Materials
The valuable minerals in the lead-zinc-silver ore sample are galena, sphalerite and silver-containing minerals. The gangue minerals are dolomite, quartz and calcite. The chemical composition and XRD analysis result of the lead-zinc-silver ore sample are shown in Table 1 and Fig. 1, respectively.
Na2S and Na2CO3 as modifiers, new reagent YZN as a depressant for zinc minerals, new reagent BPB as a collector for lead minerals, CuSO4 as an activator for zinc minerals, ethyl xanthate as a collector for zinc minerals and terpineol as frother were used in the flotation tests. The new reagents YZN and BPB were synthesized in laboratory.
Table 1 Chemical composition of lead-zinc-silver ore sample (mass fraction, %)
![]()
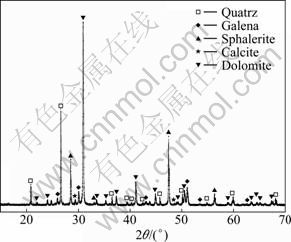
Fig. 1 XRD pattern of lead-zinc-silver ore sample
2.2 Flotation experiments
In the laboratory scale tests, the effects of different parameters such as grinding duration, Na2S dosage, Na2CO3 dosage, YZN dosage, BPB dosage, CuSO4 dosage and ethyl xanthate dosage were investigated in relation to the metals contents and recoveries of concentrates. The flotation experiments were undertaken in a XFD type flotation machine and the impeller speed was 2 000 r/min. The flotation pulp density was about 30%. The flow sheets of lead and zinc flotation tests are shown in Figs. 2(a) and (b), respectively.
3 Results and discussion
3.1 Effects of grinding duration and flotation reagents on lead flotation
3.1.1 Effect of grinding duration on lead flotation
Because most minerals are finely disseminated and intimately associated with the gangue minerals, they must be initially “unlocked” or “liberated” before separation can be undertaken [7]. The effective liberation of lead and zinc minerals is the foundation of separating lead and zinc minerals and improving the concentrates indexes [8-9]. Thus, the effect of grinding duration on lead flotation was investigated, of which the result is shown in Fig. 3. The grinding pulp density was about 66%. The dosages of Na2S, Na2CO3, YZN, BPB and terpineol were 4 000, 4 000, 14 000, 100 and 40 g/t, respectively. The grinding duration increased from 3 to 7 min.
According to the result shown in Fig. 3, increasing grinding duration to 5 min enhances the lead grade and recovery sharply to 37.15% and 78.66%, respectively. However, grinding duration longer than 5 min leads to the decrease in the lead grade and recovery. The zinc recovery in the lead concentrate changes little in the whole grinding duration.
When the grinding duration is less than 5 min, the particles are too coarse to suspend in the pulp and the probabilities of collisions between the particles and bubbles are small, which makes the flotation of lead minerals difficult. When the grinding duration is longer than 5 min, with the extension of grinding duration, the particles are finer and the amount of slime increases. The slime tends to be mixed up with the bubbles because of its small weight and big specific surface area, which decreases the flotation indexes. Thus, 5 min is considered as the optimum grinding duration. When the grinding duration is 5 min, the fraction of particles below 0.074 mm approximately accounts for 75% of the feed.
3.1.2 Effect of Na2S dosage on lead flotation
In order to study the effect of Na2S dosage on lead flotation, 4 000 g/t Na2CO3, 14 000 g/t YZN, 100 g/t BPB and 40 g/t terpineol were used as reagents. Na2S dosage increased from 0 to 6 000 g/t. The lead flotation indexes are illustrated in Fig. 4, as a function of Na2S dosage.
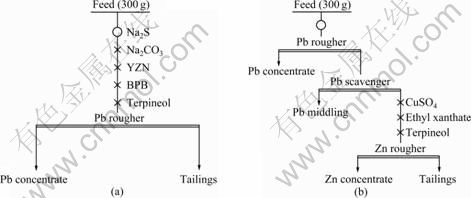
Fig. 2 Flow sheets of lead (a) and zinc (b) flotation tests
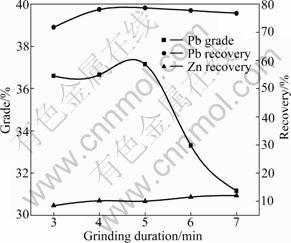
Fig. 3 Effect of grinding duration on lead flotation
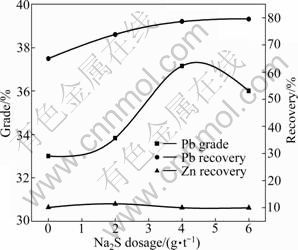
Fig. 4 Effect of Na2S dosage on lead flotation
It is observed from Fig. 4 that increasing Na2S dosage to 4 000 g/t results in the increase in the lead grade and recovery. When Na2S dosage exceeds 4 000 g/t, the lead grade decreases sharply with a slight increase in the lead flotation recovery. In the whole Na2S dosage range, the zinc recovery in the lead concentrate remains low and changes little. The result indicates that proper amount of Na2S addition favors the lead flotation while excessive amount of Na2S has an adverse effect on it. Therefore, the optimum dosage of Na2S is 4 000 g/t and all further experiments will be carried out with this dosage.
3.1.3 Effect of Na2CO3 dosage on lead flotation
The floatability of pyrite is similar to galena under low pH condition, which is harmful to the separation of Pb, Zn and S minerals. The galena will be depressed as well as pyrite if the pH is too high, which has a bad effect on the lead flotation indexes [10]. Thus, the pH value of the flotation pulp should neither be too high nor too low. In practice, Na2CO3 is commonly used as pH regulator. In order to investigate the effect of Na2CO3 dosage on lead flotation, 4 000 g/t Na2S, 14 000 g/t YZN, 100 g/t BPB and 40 g/t terpineol were used as reagents. The Na2CO3 dosage increased from 0 to 6 000 g/t. The result is shown in Fig. 5.
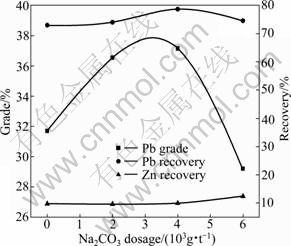
Fig. 5 Effect of Na2CO3 dosage on lead flotation
It can be seen from the result shown in Fig. 5 that increasing Na2CO3 dosage to 4 000 g/t improves the grade and recovery of the lead concentrate. However, Na2CO3 dosage more than 4 000 g/t leads to the decrease in the lead grade and recovery. When the Na2CO3 dosage increases from 4 000 to 6 000 g/t, the lead grade and recovery decrease from 37.15% and 78.66% to 29.20% and 74.61%, respectively. In the whole Na2CO3 dosage range, the zinc recovery in the lead concentrate remains low and changes little. According to Fig. 5, 4 000 g/t is considered as the optimum Na2CO3 dosage.
3.1.4 Effect of YZN dosage on lead flotation
Effect of YZN dosage on lead flotation was investigated extensively, of which the result is shown in Fig. 6. Reagents and their dosages were as follows: Na2S 4 000 g/t, Na2CO3 4 000 g/t, BPB 100 g/t and terpineol 40 g/t. The dosages of YZN were 10 000, 12 000, 14 000 and 16 000 g/t, respectively.
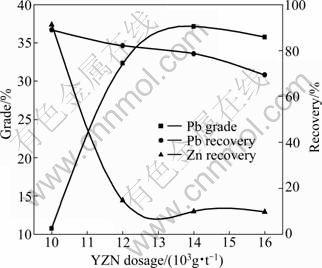
Fig. 6 Effect of YZN dosage on lead flotation
Depressant dosage should be proper in flotation or else the flotation indexes will decline. Excessive depressant will make the valuable minerals depressed while insufficient depressant could not inhibit the gangue minerals completely [11]. It is shown in Fig. 6 that the lead flotation is greatly affected by YZN addition. When the YZN dosage is less than 14 000 g/t, with the increase of YZN dosage, the lead grade increases sharply while the zinc recovery in the lead concentrate decreases significantly. When the YZN dosage exceeds 14 000 g/t, the zinc recovery in the lead concentrate and the lead grade change little. In the whole YZN dosage range, the lead recovery decreases gradually. Figure 6 indicates that the zinc minerals are depressed successfully with the newly synthesized depressant YZN. But the YZN dosage shouldn’t be excessive, or else the lead minerals will be depressed and the lead flotation indexes will decline. According to the results above, 14 000 g/t is considered to be the optimum YZN dosage.
3.1.5 Effect of BPB dosage on lead flotation
Within a certain range, the increase of collector dosage has an improving effect in terms of flotation velocity and indexes while the excessive or insufficient dosage of collector is deleterious for flotation [11]. Therefore, it is necessary to study the effect of BPB dosage on lead flotation.
In these experiments, BPB dosage increased from 60 to 120 g/t while 4 000 g/t Na2S, 4 000 g/t Na2CO3, 14 000 g/t YZN and 40 g/t terpineol were kept constant in each flotation test. The effect of BPB dosage on lead flotation is shown in Fig. 7. Up to a BPB dosage of 100 g/t, the lead grade and recovery increase from 35.2% and 72.93% to 37.15% and 78.66%, respectively. For higher BPB dosages, the lead grade and recovery decrease sharply. In the whole BPB dosage range, the zinc recovery in the lead concentrate changes little. If the BPB dosage is excessive, the flotation selectivity will decline and the competitive conglutination of the lead and zinc minerals on the bubbles will reduce the lead recovery. Figure 7 indicates that 100 g/t can be considered as the appropriate BPB dosage.
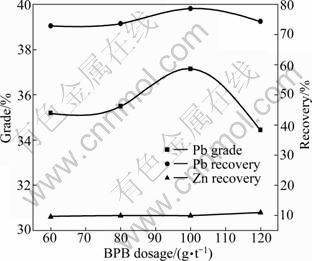
Fig. 7 Effect of BPB dosage on lead flotation
3.2 Effects of flotation reagents on zinc flotation
3.2.1 Effect of CuSO4 dosage on zinc flotation
Zinc sulfide minerals always occur in association with other sulfide minerals. The concentration and separation of these minerals by flotation require the use of modifying procedures: activation to render the zinc mineral susceptible to reaction with sulphydryl collectors, deactivation and depression to prevent their inadvertent flotation together with other components of the ores [12-13]. Preferential flotation technology was employed in this work, in which the lead flotation was followed by zinc flotation. The zinc minerals were depressed during lead flotation and must be activated before zinc flotation.
CuSO4 is commonly used as an activator for zinc minerals. In these experiments, CuSO4 dosage increased from 200 to 800 g/t, while 100 g/t ethyl xanthate and 20 g/t terpineol were kept constant in each flotation test. The effect of CuSO4 dosage on zinc flotation is shown in Fig. 8. It is shown in Fig. 8 that the zinc flotation is greatly affected by the CuSO4 dosage. The zinc grade increases from 25.35% to 49.27% when the CuSO4 dosage increases from 200 to 600 g/t, while the zinc recovery increases from 21.27% to 75.91%. There is no significant increase in the zinc grade and recovery with a further increase in the CuSO4 dosage. Thus, 600 g/t is considered to be the optimum CuSO4 dosage.
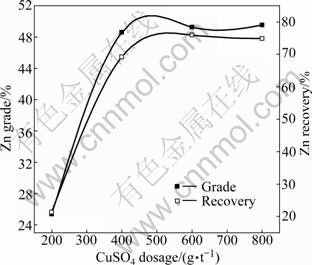
Fig. 8 Effect of CuSO4 dosage on zinc flotation
3.2.2 Effect of ethyl xanthate dosage on zinc flotation
In these experiments, ethyl xanthate dosage increased from 60 to 120 g/t while 600 g/t CuSO4 and 20 g/t terpineol were kept constant in each experiment. The zinc flotation indexes are shown in Fig. 9 as a function of the ethyl xanthate dosage. Increasing ethyl xanthate dosage from 0 to 100 g/t enhances the zinc recovery from 69.48% to 75.91%, while ethyl xanthate dosage more than 100 g/t leads to a decrease in the zinc recovery. In the whole ethyl xanthate dosage range (from 60 to 120 g/t), the increase in zinc grade is not significant. According to Fig. 9, 100 g/t is considered to be the right ethyl xanthate dosage.
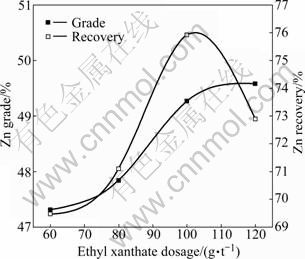
Fig. 9 Effect of ethyl xanthate dosage on zinc flotation
3.3 Flotation circuit
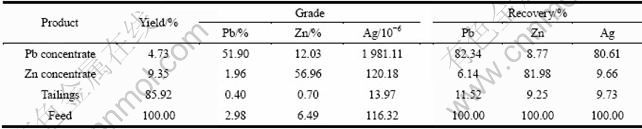
Based on the best flotation conditions achieved through the experiments above, a flow sheet was designed for the treatment of the sulfide lead-zinc-silver ore from Yunnan Province, China. The flow sheet adopted for treatment of the lead-zinc-silver ore included the productions of a lead concentrate and a zinc concentrate. This flow sheet is beneficial for the lead and zinc flotation indexes and the recovery of associated silver. The flotation circuit and result are given in Fig. 10 and Table 2, respectively.
It is observed from Table 2 that with the flotation circuit shown in Fig. 10, a lead concentrate with 51.90% Pb is produced with 82.34% recovery while a zinc concentrate with 56.96% Zn is produced with 81.98% recovery. The silver grade and recovery in the lead concentrate are 1 981.11 g/t and 80.61%, respectively. The silver grade and recovery in the zinc concentrate are 120.18 g/t and 9.66%, respectively. Thus, the total recovery of associated silver in the lead and zinc concentrate can be above 90%.
4 Interactions of flotation reagents with minerals
4.1 Sulfidation mechanism of Na2S on oxidized lead minerals
The flotation and separation of sulfide minerals can be detrimentally affected by the oxidation of the surfaces of these minerals. The amount of surface oxidation will reduce the hydrophobicity of the mineral and make the adsorption of collectors less selective [14]. To preclude this, Na2S is usually used as the sulfidation agent for lead and zinc oxides [15]. Na2S can be hydrolyzed and then dissociated in solution [16]:
Na2S+2H2O![]() H2S+2NaOH (1)
H2S+2NaOH (1)
S2-![]() HS-,
HS-,![]() (2)
(2)
HS2-+H+![]() H2S,
H2S,![]() (3)
(3)
![]() (4)
(4)
c(S)′=c(S2-)+c(HS-)+c(H2S) (5)
![]()
![]() (6)
(6)
![]() (7)
(7)
![]() (8)
(8)
where K1 and K2 are the first and second proton-addition constants, respectively; ![]() is the second cumulative proton-addition constant; φ0, φ1 and φ2 are the distribution coefficients of various species containing sulfur atom as a function of the pH value.
is the second cumulative proton-addition constant; φ0, φ1 and φ2 are the distribution coefficients of various species containing sulfur atom as a function of the pH value.
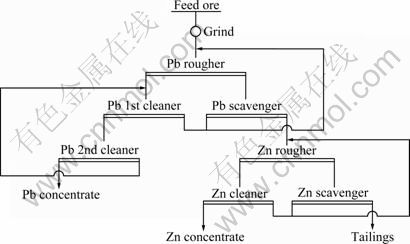
Fig. 10 Flotation circuit of sulfide lead-zinc-silver ore
Table 2 Flotation results
From the equations above, the distribution coefficients of various species containing the sulfur atom can be calculated as a function of the pH value, as shown in Fig. 11.
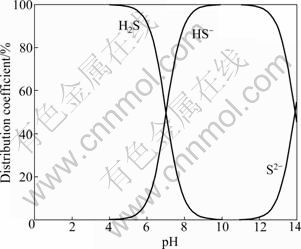
Fig. 11 Distribution coefficients of various species containing sulfur atom in Na2S solution as function of pH
It is shown in Fig. 11 that when pH<7.0, H2S is the dominant species, while S2- and HS- are the dominant species when pH>13.90 and 7.0
- is the main component which has a sulfidation on oxidized lead-zinc ores. The pH value in this work was kept between 8 and 10, which is located in the range where HS- is the predominant species. The lead-zinc- silver ore treated in this work contains some oxidized lead minerals and some sulfide minerals of the ore sample could be oxidized during grinding and flotation. The HS- species interacts with metallic ions on the mineral surface and forms a sulfide film which could activate the flotation of the oxidized minerals. Therefore, the presence of appropriate amount of Na2S is beneficial to improving the flotation indexes of the lead minerals.
4.2 Depressing mechanism of Na2S on sphalerite
During the flotation of complex lead-zinc-silver sulfide minerals, a certain amount of Pb2+ can be dissolved out because of the oxidization of some sulfide minerals. The Pb2+ has a bad effect on lead-zinc flotation indexes because of its activation on sphalerite.
The activation mechanism of sphalerite indicates that reducing the Pb2+ concentration in pulp can prevent sphalerite being activated. The ion precipitation method can be employed to eliminate the influence of Pb2+ in the minerals pulp. Heavy metal ion sulfides are more difficult to be dissolved than their other corresponding compounds, thus the sulfide precipitation method was introduced to eliminate Pb2+ in flotation pulp.
If the concentration of Pb2+ in solution is 1×10-3 mol/L, a Na2S solution with the concentration of 1.1×10-3 mol/L can be used to precipitate Pb2+. The residual Pb2+ concentration in the excessive Na2S solution as a function of pH can be calculated through Eq. (9), of which the result is shown in Fig. 12.
![]() (9)
(9)
where c(Pb2+) is the concentration of residual Pb2+; KPbS is the solubility product of PbS; ![]() is the additional reaction coefficient for the hydrolysis of Pb2+;
is the additional reaction coefficient for the hydrolysis of Pb2+; ![]() is the additional reaction coefficient for the proton-addition reaction of S2-; ci(Na2S) and ci(Pb2+) are the initial concentrations of Na2S and Pb2+, respectively.
is the additional reaction coefficient for the proton-addition reaction of S2-; ci(Na2S) and ci(Pb2+) are the initial concentrations of Na2S and Pb2+, respectively.
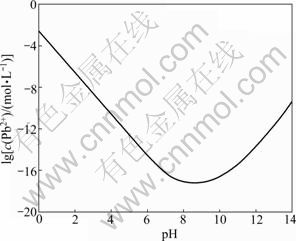
Fig. 12 Residual Pb2+ concentration in excessive Na2S solution as function of pH (ci(Pb2+)=1×10-3 mol/L, ci(S2-)=1.1×10-3 mol/L)
It can be seen from Fig. 12 that when the pH value is between 7 and 11, the Pb2+ is precipitated thoroughly in excessive Na2S solution. The pH value in this work was maintained between 8 and 10 with which the Pb2+ can be precipitated to prevent the sphalerite being activated. Therefore, the lead-zinc flotation indexes can be improved because of the addition of Na2S.
Precipitation effect of Na2S on Pb2+ during flotation was confirmed through sulfide precipitation tests. In each test, 500 g of lead-zinc ore sample which had been crushed to <1 mm was put into a ball mill to be ground for 8 min. The mineral pulp and some certain amount of Na2S were put into a 1.5 L flotation cell where water was added to the same calibration tails. The sample was agitated for 3 min and then deposited for 10 min. The upper layer liquid was decanted to measure the Pb2+ concentration. The result of the test is shown in Fig. 13.
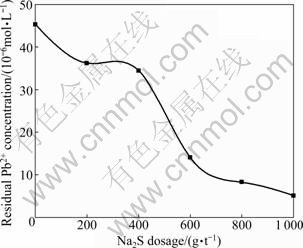
Fig. 13 Residual Pb2+ concentration in upper layer liquid as function of Na2S dosage
From Fig. 13, it is observed that the Pb2+ concentration in the upper layer clarified liquid gradually decreases with the increase of Na2S dosage. The increase of Na2S dosage from 0 to 1 000 g/t leads a decrease of the residual Pb2+ concentration from 45.36×10-6 to 5.16×10-6 mol/L. The result shows that Na2S has a positive effect on precipitation of Pb2+. The utilization of Na2S reduces the Pb2+ concentration and weakens the activation of Pb2+ on sphalerite.
4.3 Depressing mechanism of Na2CO3 and ZnSO4 on sphalerite
Zinc depressant YZN used in this work was recently synthesized in laboratory. ZnSO4 is one of the main components of YZN. In order to improve the depressing effect of ZnSO4 on sphalerite, Na2CO3 and ZnSO4 are commonly used as the joint zinc depressants, the depressing mechanism of which can be discussed through dominant species diagram [15].
In Na2CO3-ZnSO4 solution system, the following reactions take place:
ZnCO3(s)=Zn2++![]() , Ksp1=10-10.0 (10)
, Ksp1=10-10.0 (10)
Zn2++![]() =ZnCO3(aq), K1=105.3 (11)
=ZnCO3(aq), K1=105.3 (11)
H++![]() =
=![]()
![]() 1010.33 (12)
1010.33 (12)
H++![]() =
=![]()
![]() 106.36 (13)
106.36 (13)
Zn2++OH-=ZnOH+, β1=105.0 (14)
Zn2++2OH-=Zn(OH)2(l), β2=1011.1 (15)
Zn2++3OH-=![]() β3=1013.6 (16)
β3=1013.6 (16)
Zn2++4OH-=![]() β4=1014.3 (17)
β4=1014.3 (17)
Zn(OH)2(s)=Zn2++2OH-, Ksp2=10-16.2 (18)
where Ksp1 and Ksp2 are the solubility products of ZnCO3(s) and Zn(OH)2(s), respectively; K1 is the equilibrium constant for formation of ZnCO3(aq); ![]() and
and ![]() are the first and second proton-addition constants; β1, β2, β3 and β4 are the hydroxylation constants of the first to the fourth hydroxyl zinc complexes.
are the first and second proton-addition constants; β1, β2, β3 and β4 are the hydroxylation constants of the first to the fourth hydroxyl zinc complexes.
According to the equations above, the demarcation lines for dominant species can be obtained as follows:
1) Dividing line between ZnCO3(s) and Zn(OH)2(s):
Zn(OH)2(s)+![]() =ZnCO3+2OH- (19)
=ZnCO3+2OH- (19)
logc(Na2CO3)=logKsp1-logKsp2-2pKW+2logαCO3+2pH (20)
where KW is the ion product of OH- and H+ of water; αCO3 is the additional reaction coefficient for the proton- addition reaction of ![]()
2) Dividing line between Zn2+ and ZnCO3:
logc(Na2CO3)=(1/2)(logKsp1+logαCO3+logαZn) (21)
where αZn is the additional reaction coefficient for the hydrolysis of Zn2+.
3) Dividing lines for various hydroxides:
c(Zn2+)=c(Zn(OH)2(l)), pH=8.45 (22)
c(Zn(OH)2(l))=![]() pH=11.5 (23)
pH=11.5 (23)
![]() pH=12.8 (24)
pH=12.8 (24)
The dominant species diagram for Na2CO3-ZnSO4-H2O solution system is given in Fig. 14(a).
Similarly, conditions for PbCO3 formation and the demarcation lines for dominant species are given as follows:
1) Dividing line between PbCO3 and Pb(OH)2:
![]()
![]() (25)
(25)
2) Dividing line between Pb2+ and PbCO3:
![]() (26)
(26)
3) Dividing lines for various hydroxides:
c(Pb2+)=c(Pb(OH)+), pH=7.7 (27)
c(Pb(OH)+)=c(Pb(OH)2(l)), pH=9.3 (28)
c(Pb(OH)2(l))=![]() pH=11.0 (29)
pH=11.0 (29)
The dominant species diagram for Na2CO3-Pb(II) solution system is shown in Fig. 14(b).
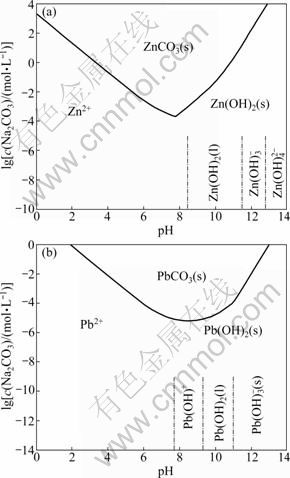
Fig. 14 Dominant species diagram: (a) Dominant species diagram of Na2CO3-ZnSO4-H2O solution system; (b) Dominant species diagram of Na2CO3-Pb(II)-H2O solution system
The Na2CO3 dosage in this work was 4 000 g/t which was approximately equivalent to 1×10-2 mol/L in term of concentration in flotation pulp. It can be seen from Fig. 14 that when pH value is between 8 and 10 and the concentration of Na2CO3 in flotation pulp is 1×10-2 mol/L, the dominant species in Na2CO3-ZnSO4 solution system are ZnCO3(s) and Zn(OH)2(s), while the dominant species in Na2CO3-Pb(II) solution system is PbCO3. Therefore, the depressing mechanism of Na2CO3 and ZnSO4 on sphalerite can be concluded as follows: 1) The formations of ZnCO3(s) and Zn(OH)2(s) as a result of the reaction between Zn2+ and ![]() make sphalerite hydrophilic; 2) The formation of PbCO3(s) as a result of the reaction between Pb2+ and
make sphalerite hydrophilic; 2) The formation of PbCO3(s) as a result of the reaction between Pb2+ and ![]() reduces the concentration of Pb2+ in pulp and prevents sphalerite being activated by Pb2+.
reduces the concentration of Pb2+ in pulp and prevents sphalerite being activated by Pb2+.
4.4 Depressing mechanism of Na2SO3 on sphalerite
Na2SO3 is one of the main components of YZN. The depressing mechanisms of Na2SO3 on sphalerite can be concluded as follows [17-19]:
1) Reduction
The ![]() of Na2SO3 is easy to be oxidized to
of Na2SO3 is easy to be oxidized to ![]() Thus Na2SO3 can reduce the Cu2+ to be Cu+ or Cu and eliminate the activation of Cu2+ on sphalerite. The equation of Cu2+ being reduced to Cu+ is as follows:
Thus Na2SO3 can reduce the Cu2+ to be Cu+ or Cu and eliminate the activation of Cu2+ on sphalerite. The equation of Cu2+ being reduced to Cu+ is as follows:
2Cu2++![]() +H2O=2Cu++2H++
+H2O=2Cu++2H++![]() (30)
(30)
2) Complexation
![]() can make some heavy metal ions form stable complexes. Complex ion
can make some heavy metal ions form stable complexes. Complex ion ![]() can be formed as the reaction of
can be formed as the reaction of ![]() and Cu+, which reduces the concentration of Cu+ and eliminates the activation effect of Cu+ on sphalerite. The reaction equation is as follows:
and Cu+, which reduces the concentration of Cu+ and eliminates the activation effect of Cu+ on sphalerite. The reaction equation is as follows:
Cu++2![]() =
=![]() (31)
(31)
5 Conclusions
1) The sulfide lead-zinc-silver ore subjected to this work contains 2.98% Pb, 6.49% Zn and 116.32×10-4 % Ag (mass fraction). It includes galena as main lead minerals, sphalerite as main zinc minerals and noble silver, quartz, calcite, dolomite as other minerals. When the fraction of <0.074 mm particles accounted for 75% or so of the feed, the galena is mostly liberated from sphalerite and the flotation indexes are comparatively good.
2) With the flotation circuit proposed, a lead concentrate of 51.90% Pb with 82.34% recovery and a zinc concentrate of 56.96% Zn with 81.98% recovery are produced. The silver grade and recovery in the lead concentrate are 1 981 g/t and 80.61%, respectively.
3) A low alkalinity-preferential flotation technology is utilized to achieve efficient flotation separation of the sulfide lead-zinc-silver ore. The low alkalinity technique has the advantages, such as short flotation process, good flotation indexes, and high silver recovery.
4) The disadvantage of the low alkalinity technique employed is the large consumption of flotation reagents. Therefore, the subject of reducing reagents consumption deserves further investigation.
5) Interaction of flotation reagents with minerals indicates that the presence of proper amount of Na2S could precipitate Pb2+ in flotation pulp to prevent sphalerite being activated. The presence of Na2S also has a sulfidation on oxidized lead minerals, which could improve lead flotation. Na2CO3 and YZN are used as combined depressants which can improve the depressing effect of YZN.
References
[1] LIU Shou-xin. Experimental research on mineral processing of lead-zinc polymetallic sulfide ore from Yunnan [D]. Kunming: Faculty of Land Resource Engineering, Kunming University of Science and Technology, 2007: 24-28. (in Chinese)
[2] U.S.Geological Survey, Mineral Commodity Summaries [Z]. 2007.
[3] ZHENG Lun, ZHANG Du, LIU Yun-cai. Study on flotation properties of sphalerite in high alkalinity medium in Fankou lead-zinc mine [J]. Mining and Metallurgical Engineering, 2005, 25(3): 37-40. (in Chinese)
[4] MO Xue-shi, HUANG Wei-zhong, ZHANG Yan-sheng, QIN Wen-qing. Study and practice on flotation of complex lead-zinc sulfide minerals for increasing flotation index [J]. Nonferrous Metals, 2007, 19(1): 9-12. (in Chinese)
[5] QIN Wen-qing, HE Ming-fei, CHEN Yu-ping. Improvement of flotation behavior of Mengzi lead-silver-zinc ore by pulp potential control flotation [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(4): 949-954.
[6] HU Wei-bo. Flotation [M]. Beijing: Metallurgical Industry Press, 1983. (in Chinese)
[7] Barry A. Wills, Tim Napier-Mum. Mineral processing technology [M]. United Kingdom: Butter Worth-Heinemann Ltd., 2006: 267- 344.
[8] PENG Y, GRANO S, FORNASIERO D, RALSTON J. Control of grinding conditions in the flotation of galena and its separation from pyrite [J]. International Journal of Mineral Processing, 2003, 70(1): 67-82.
[9] PENG Y, GRANO S, FORNASIERO D, RALSTON J. Control of grinding conditions in the flotation of chalcopyrite and its separation from pyrite [J]. International Journal of Mineral Processing, 2003, 69(1): 87-100.
[10] CHENG De-ming. Mineral processing techniques present status and prospect of lead-zinc ore sulfide in China [J]. Journal of Guangdong Non-ferrous Metals, 1994(1): 6-12. (in Chinese)
[11] HU Yue-hua, FENG Qi-ming. Mineral resources processing technology and equipment [M]. Beijing: Science Press, 2006: 209-210. (in Chinese)
[12] FINKELSTEIN N P. The activation of sulphide minerals for flotation: A review [J]. Mineral Processing, 1997, 52: 81-120.
[13] EL-SHALL H E, ELGILLANI D A, ABDEL-KHALEK N A. Role of zinc sulfate in depression of lead-activated sphalerite [J]. International Journal of Mineral Processing, 2000, 58(1): 67-75.
[14] CLARKE P, FORNASIERO D, RALSTON J, SMART R S T. A study of the removal of oxidation products from sulfide mineral surfaces [J]. Minerals Engineering, 1995, 8(11): 1347-1357.
[15] SHI Dao-min, Yang-ao. Flotation of oxidized lead and zinc ores [M]. Yunnan: Yunnan Science and Technology Press, 1996: 215-320. (in Chinese)
[16] WANG Dian-zuo, HU Yue-hua. Solution chemistry of flotation [M]. Changsha: Hunan Science and Technology Press, 1988: 162-164. (in Chinese)
[17] WANG Qiang. Experiment research of strengthen silver reclaim in galenit and sphalerite [D]. Shenyang: College of Resource and Civil Engineering, Northeastern University, 2004: 20-31. (in Chinese)
[18] SHEN W Z, FORNASIERO D, RALSTON J. Flotation of sphalerite and pyrite in the presence of sodium sulfite [J]. International journal of Mineral Processing, 2001, 63(1): 17-28.
[19] LASKOWSKI J S, LIU Q, ZHAN Y. Sphalerite activation: Flotation and electrokinetic studies [J]. Minerals Engineering, 1997(8): 787- 802.
(Edited by DENG Lü-xiang)
Foundation item: Project(50874117) supported by the National Natural Science Foundation of China
Received date: 2011-06-20; Accepted date: 2011-08-31
Corresponding author: SUN Wei, Professor, PhD; Tel: +86-13507310692; E-mail: sunmenghu@126.com

