Preparation of cobalt-modified magnetite and its magnetic properties
YANG Xi-yun(��ϲ��)1, GONG Zhu-qing(������)1, LIU Feng-liang(������)2, HUANG Jian(�� ��)2
(1. School of Metallurgical Science and Engineering, Central South University,Changsha 410083, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: Magnetite was modified by reaction with alkaline solution containing Co2+ and Fe2+ to obtain a cobalt ferrite layer on the surface of particles. The influences of modification conditions on the properties were investigated. The as-prepared particles were characterized by X-ray diffraction(XRD) and transmission electron microscope(TEM). The results show that pH value influences the particles composition directly, the desirable CoFe2O4 is obtained as pH value is 12. The coercivity of particles increases with the increase of cobalt content, and the cobalt efficiency reaches a maximum value at cobalt content of 2.71%(mass fraction). With cobalt modification, the magnetite particles have the similar lattice constant and structure to that without cobalt modification, and the squareness ratio is almost 0.5. The increase of the coercivity is attributed to the uniaxial magnetic anisotropy and magnetocrystalline anisotropy of cobalt-ferrite itself.
Key words: magnetite; cobalt; magnetic property CLC number: TQ586.1; O482.54
Document code: A
1 INTRODUCTION
Acicular magnetite has a number of novel properties and performances, its high magnetic and thermal properties, good electric conduction and black color make it a new type of materials promising in magnetic recording field[1-4]. With the development of high recording density application such as video and audio tape, instrumentatic tapes and flexible disks, it is needed to increase the coercivity of magnetite particles further. For this purpose, an effective method is to deposit at or near the surface of magnetite particles[5]. Doping with cobalt on the surface is thought to form an epitaxial layer of cobalt-ferrite, CoFe2O4, on the surface of oxide particles, which results in increase in the coercivity and magnetocrystalline anisotropy and these in turn greatly improve the recording density[6-8]. This technique is widely used to produce cobalt modified ��- Fe2O3 particles[9-11]. However, most of cobalt-modified magnetite is still dependent on importation. Some reports[1, 12] about cobalt-doped magnetite only gave a simple process, which lacked completely satisfactory explanation of the origin of coercivity of cobalt surface-modified iron oxide and this is one of the most intriguing of the unsolved problems in the field of recording materials, so there exists theoretical and practical significance on study and exploitation of cobalt-modified magnetite. In this paper, the influences of process variables such as pH, cobalt content, reaction time in the precipitation reaction have been investigated in detail, the reason of enhancement of coercivity is clarified.
2 EXPERIMENTAL
In this study, Fe3O4 was used as the precursor, whose average specific surface area is 25.06m2/g, coercivity(Hc) and saturation magnetization(��s) are 28.48kA/m and 75A��m2/kg, respectively. CoSO4��7H2O and FeSO4��7H2O were used as sources of Co2+ and Fe2+ ions in the aqueous solution, and the concentration of FeSO4��7H2O was 5g/L. NaOH was used as precipitant. The Fe3O4 particles were suspended in the metal ions solution for 20min with nitrogen blowing in. To convert the sulfates into hydroxides of cobalt and iron, NaOH solution was added drop likewise so as to get the pH value of 9, 10, 11 and 12. Then the suspension was heated at 90�� for 2, 4, 6h, respectively. The resultant particles were filtered, washed with water and dried. The phase composition and lattice constant of the particles were characterized by XRD. The morphologies were observed by TEM. The magnetic properties were measured by a vibrating sample magnetometer under a magnetic field of 400kA/m. The difference between the coercivity of the core particles and final product(��Hc) was determined. Specific surface area(SSA) was determined by BET method. Cobalt content in the particles was measured by means of chemical analysis.
3 RESULTS AND DISCUSSION
3.1 Effect of pH value
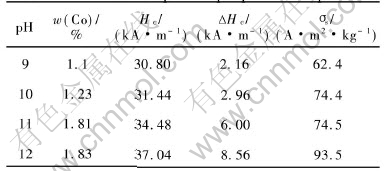
The XRD patterns of the Fe3O4 precursor and products at different pH values are given in Fig.1 and Fig.2, respectively. At pH value of 9, the product yields undesired impurity in addition to desired CoFe2O4 and precursor. When the pH value is 12, all Co is precipitated, resulting in the formation of pure CoFe2O4. These changes are reflected in the magnetic properties and cobalt content of the product as shown in Table 1. With the increase of pH value from 9 to 12, the cobalt content varies by about 0.8%, the coercivity of the product increases from 30.8 to 37.04kA/m, and ��s increases from 62.4 to 88.5A��m2/kg. This may be attributed to the increase of CoFe2O4 content in the product.
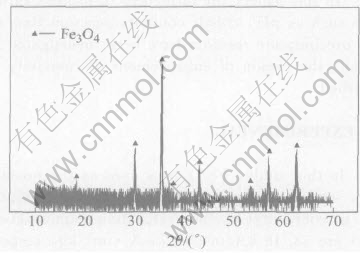
Fig.1 XRD pattern of precursor of Fe3O4
3.2 Effect of cobalt content
At pH value of 12, different amounts of cobalt were added into the solution. The effects of cobalt content on the magnetic properties of the product
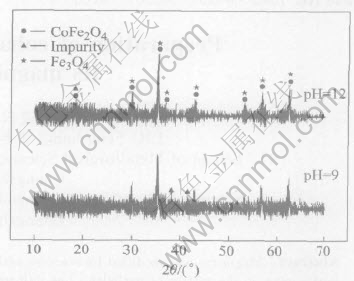
Fig.2 XRD patterns of Co-modified Fe3O4
Table 1 Effects of pH on properties of product
are shown in Table 2. The formation of cobalt ferrite on the surface of Fe3O4 results in the increase of coercivity. The incorporation of 2.71%(mass fraction) Co results in the increase of coercivity by 56% over than that of the precursor. Further increasing cobalt content to 5.03%(mass fraction), the coercivity shows no obvious increase.
Fig.3 shows the correlation between cobalt efficiency(Hc divided by the mass fraction of cobalt in the particles) and cobalt content. The cobalt efficiency reaches the peak at cobalt content of 2.71%, and then decreases to level of about 3.22. The result can be explained by the following reasons. One is that nonferromagnetic materials such as Co2O3��nH2O are expected to be present, although XRD shows only CoFe2O4. The second is that the product becomes loosely, which is verified with the larger SSA shown in Table 2. The results
Table 2 Effects of cobalt content on properties of product
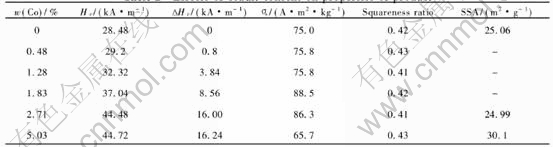
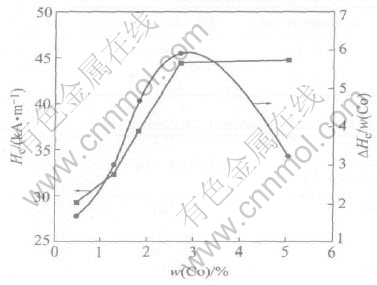
Fig.3 Influence of Co content on cobalt efficiency
agree well with the results in the Ref.[13]. The third is that demagnetization field of inner core Fe3O4 decreases with increasing cobalt content, which will be discussed in later section.
3.3 Effect of reaction time
The effect of reaction time on the product is shown in Table 3. The coercivity increases with increasing reaction time, because the crystalline is more regular and integrated with the time increasing. However, the saturation magnetization decreases when the reaction time is 6h. It is probable that Co2+ is unstable and oxidized to Co3+. XPS can detect the presence of Co2O3��nH2O in CoFe-��-Fe2O3[14].
Table 3 Effects of reaction time on product

3.4 Effect of axial ratio of Fe3O4
The effects of axial ratio of Fe3O4 on the product are shown in Table 4. High axial ratio favors uniform distribution of cobalt and results in the increase of coercivity and saturation magnetization.
Table 4 Effects of axial ratio of Fe3O4 on product

3.5 Post-treatment of product
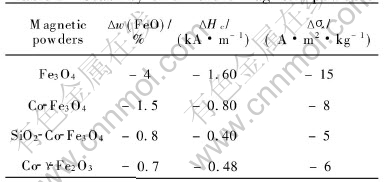
Magnetite is unstable in chemical composition due to the oxidation of Fe2+, these in turn affect the stability of magnetic properties. As a result, the magnetic recording media may suffer from magnetic accommodation and print-through effects. Therefore, it is necessary to improve the stability of cobalt-modified magnetite. Coating an SiO2 layer is an effective method to increase the stability of oxide[15]. In order to confirm the behavior of SiO2, the particles with and without SiO2-treatment are exposed to air for 20 days at 50��. The results are shown in Table 5. The particles with SiO2-treatment is more stable than that without treatment, which shows similar stability to CoFe-��-Fe2O3.
Table 5 Stability of different magnetic powders
3.6 Analysis of effect of cobalt modification on magnetic properties of magnetite
The major aim of cobalt modification is to increase the coercivity of the powder. There are some interpretations on the origin of enhancement of coercivity, which mainly comes from the following three aspects[7, 10, 16]: 1) magnetocrystalline anisotropy of cobalt-ferrite, 2) shape anisotropy, 3) magnetic induced anisotropy.
Table 2 shows that the coercivity increases with increasing cobalt content. This indicates that the magnetocrystalline anisotropy of cobalt-ferrite plays an important role in the enhancement of coercivity.
Fig.4 and Fig.5 show the TEM photographs of Fe3O4 and cobalt-modified Fe3O4 particles,respectively. It is clear that the photographs are almost the same except for the larger short axis in Fig.5. So the shape anisotropy of these particles would not be different. The lattice constants of the two particles shown in Table 6 are also the same. These results mean that the shape anisotropy has little effect on the increase of coercivity.
The larger the saturation magnetization of inner core Fe3O4, the greater the demagnetization field[7]. Different saturation magnetization can be obtained by controlling the content of FeO in Fe3O4[17]. The effect of magnetic properties of inner core Fe3O4 on the coercivity enhancement is shown in Table 7. It is seen that the coercivity enhancement is dependent on the value of saturation magnetization. The squareness ratio of the particles before and after modification shown in Table 6 is about 0.5. This indicates that the particles ap-
Table 6 Properties of Fe3O4 with and without cobalt modification

Table 7 Effect of magnetic properties of Fe3O4 on enhancement of coercivity
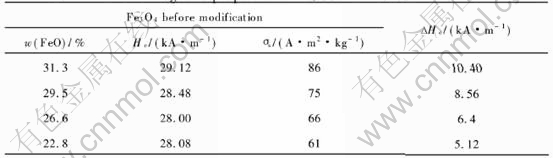

Fig.4 TEM photograph of Fe3O4

Fig.5 TEM photograph of cobalt-modified Fe3O4
pear to have uniaxial magnetic anisotropy, which results from migration of Co2+ ions to the preferred sites alignment with the direction of magnetic field of the core Fe3O4. So the uniaxial magnetic anisotropy induced by inner core Fe3O4 leads to the enhancement of coercivity. The cobalt-ferrite layer thickness increases with increasing cobalt content, and the demagnetization field decreases with the thickness very sharply. That is to say, the demagnetization field decreases with increasing cobalt content. As a result, demagnetization field is equal to zero and can��t induce uniaxial magnetic anisotropy while cobalt content gets to some amount. Thus we can explain why there exists a maximum value of cobalt efficiency in Fig.3.
4 CONCLUSIONS
1) Cobalt-modified magnetite is prepared by reaction with alkaline solution containing Co2+ and Fe2+ at 90��, the optimum conditions are reaction time of 4h, cobalt content of 2.71% and pH value of 12.
2) The pH value influences the composition of the product directly, the desired CoFe2O4 is obtained at pH 12. The formation of CoFe2O4 on the surface of magnetite results in the increase of coercivity. The coercivity increases with increasing cobalt content, and the cobalt efficiency reaches a maximum value at cobalt content of 2.71%. The increase of the coercivity is attributed to the uniaxial magnetic anisotropy induced by inner core Fe3O4 and magnetocrystalline anisotropy of cobalt-ferrite itself.
3) The chemical and magnetic stability are improved by coating with a layer SiO2.
REFERENCES
[1]Masacchika H, Hiroyuki W, Takeshi M. Magnetic Particles and Process for Production Thereof [P]. Japan EP0808801, 1997-11-26.
[2]Bate G. Magnetic recording materials since 1975 [J]. Journal of Magnetism and Magnetic Materials, 1991, 100: 413-424.
[3]Koji N, Kouji K, Seiji S. Processes for Producing Hydrated Iron Oxide and Ferromagnetic Iron Oxide [P]. Japan EP0857693, 1998-08-12.
[4]ZHU Yi-hua, WU Qiu-fang. Synthesis of magnetite nanoparticles by precipitation with forced mixing [J]. Journal of Nanoparticle Research, 1999, 1: 393-396.
[5]MAO Xing-yu. The formation of cobalt modified ultra-fine iron particles [J]. Journal of Jimei University(Natural Science), 2000, 5(2): 17-20.(in Chinese)
[6]Blagev A V, Hirz S J. Effect of surface properties on cobalt modification of iron [J]. IEEE Transactions on Magnetics, 1992, 28(5): 2382-2385.
[7]ZHANG Shu-guang, GAO Zheng-sheng, JIANG Shou-ting, et al. An investigation of anisotropy of cobalt-surface-modified iron oxide magnetic particles [J]. IEEE Transactions on Magnetics, 1990, 26(3): 1148-1153.
[8]Schwab E, Veitch R J, Auweter. Cobalt-modified iron oxides with improved aging stability [J]. Journal of Magnetism and Magnetic Materials, 1993, 120: 43-47.
[9]Jeschke J C. Precipitation Process for Preparing Acicular Magnetic Metal Oxide Particles [P]. USP3243375, 1996-3-29.
[10]YUAN Wei, JIN Xin, XIE Jin. Study on the preparation of cobalt modified ��-Fe2O3 magnetic powder [J]. Fine Chemicals, 1997, 14(1): 29-32.(in Chinese)
[11]Sesigur H, Acma E, Addemir O, et al. The preparation of cobalt-modified magnetic iron oxide [J]. Materials Research Bulletin,1996, 31(12): 1581-1586.
[12]HAN D H, WANG J P, LUO H L. Crystallite size effect on saturation magnetization of fine ferromagnetic particles [J]. Journal of Magnetism and Magnetic Materials, 1994, 136: 176-182.
[13]Radovenchik V M, Ivanenko E I. Oxidation of ferrous ions with air oxygen for preparation of magnetite suspension review [J]. Ekotekhnologii i Resursosberezhenie, 2001(5): 37-48.(in Russian)
[14]WANG Qi-xiang, PAN Hai-bin, SONG Bao-zhen, et al. Surface properties of inorganic coated Fe2O3 by XPS [J]. Journal of Inorganic Materials, 2002, 17(4): 782-785. (in Chinese)
[15]CUI Ai-li, WANG Ting-jie, JIN Yong, et al. Mechanism and structure analysis of TiO2 surface coated with SiO2 and Al2O3 [J]. Chemical Journal of Chinese Universities, 1998, 19(11): 1727-1729.(in Chinese)
[16]Orth T, Moller M, Pelzl J, et al. Characterisation of the anisotropy behavior of different cobalt modified ��-Fe2O3 tapes [J]. Journal of Magnetism and Magnetic Materials, 1995, 145: 243-254.
[17]Meisen U, Kathrein H. The influence of particle size,shape and particle size distribution on properties of magnetites for the production of toners [J]. Journal of Imaging Science and Technology, 2000, 44(6): 508-513.
(Edited by YUAN Sai-qian)
Received date: 2004-05-08; Accepted date: 2004-10-20
Correspondence: YANG Xi-yun, PhD candidate; Tel: +86-731-8830752; E-mail: yxy7412@mail.csu.edu.cn[