Oxidation and roasting characteristics of artificial magnetite pellets
来源期刊:中南大学学报(英文版)2016年第11期
论文作者:张汉泉 路漫漫 付金涛
文章页码:2999 - 3005
Key words:artificial magnetite; pellets; oxidation degree; roasting temperature
Abstract: Compared with natural magnetite concentrate, artificial magnetite with more lattice defects and higher activity tends to be oxidized. And the artificial magnetite pellet at the temperature of 400 °C has the oxidation degree approaching to natural magnetite concentrate pellet fired at 1000 °C. Besides, two kinds of pellets displayed quite different roasting characteristics. When preheated at the same temperature for the same period of time, natural magnetite concentrate pellet and artificial magnetite concentrate pellet need to be roasted at the temperature of 1100 °C and 1250 °C, respectively, for 25 min to reach the compressive strength of 3000 N per pellet. When roasted at the same temperature of 1200 °C, natural magnetite pellet and artificial magnetite pellet need to be roasted for 15 min and 30 min, respectively, to reach the compressive strength over 3000 N per pellet. It can be seen from the test that artificial magnetite pellet has a faster oxidation, resulting in the high porosity in the produced pellet, and it requires a roasting process at higher temperature for a longer time to reach the desired compressive strength for industrial production.
J. Cent. South Univ. (2016) 23: 2999-3005
DOI: 10.1007/s11771-016-3363-y
ZHANG Han-quan(张汉泉), LU Man-man(路漫漫), FU Jin-tao(付金涛)
School of Resources and Civil Engineering, Wuhan Institute of Technology, Wuhan 430073, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: Compared with natural magnetite concentrate, artificial magnetite with more lattice defects and higher activity tends to be oxidized. And the artificial magnetite pellet at the temperature of 400 °C has the oxidation degree approaching to natural magnetite concentrate pellet fired at 1000 °C. Besides, two kinds of pellets displayed quite different roasting characteristics. When preheated at the same temperature for the same period of time, natural magnetite concentrate pellet and artificial magnetite concentrate pellet need to be roasted at the temperature of 1100 °C and 1250 °C, respectively, for 25 min to reach the compressive strength of 3000 N per pellet. When roasted at the same temperature of 1200 °C, natural magnetite pellet and artificial magnetite pellet need to be roasted for 15 min and 30 min, respectively, to reach the compressive strength over 3000 N per pellet. It can be seen from the test that artificial magnetite pellet has a faster oxidation, resulting in the high porosity in the produced pellet, and it requires a roasting process at higher temperature for a longer time to reach the desired compressive strength for industrial production.
Key words: artificial magnetite; pellets; oxidation degree; roasting temperature
1 Introduction
Magnetite concentrates are widely used to produce hematite pellets for ironmaking. During firing, green pellets are heated and exposed to oxygen-containing gas (using a grate-kiln process in many cases). Firing oxidizes magnetite to hematite. Pellet strength relies on the physical bonds formed between oxidized particles; fine outgrowths form on the oxidized particle surfaces, favoring formation of bonds between particles [1]. Artificial magnetite, with principal chemical composition of Fe3O4, is usually obtained after the iron oxide ore, like hematite(Fe2O3), siderite (FeCO3), limonite (mFe2O3・nH2O ), treated by process of magnetizing roasting followed by low-intensity magnetic separation (LIMS), which results in the incomplete lattice structure in most of artificial magnetite and quick formation of solid solution. And more lattice defects of artificial magnetite lead to its surfactivity and reactivity higher than natural one, resulting in a higher reaction rate for artificial magnetite [2-3]. It is such difference between artificial and natural magnetite concentrates in terms of magnetism, surface physical and chemical properties, which makes their pellets quite different from each other in the process of oxidizing roasting. For this reason, studies on the properties of pellets of artificial and natural magnetite concentrate in oxidization and roasting are of great importance for improving equipment structure, setting proper thermotechnical parameters, so as to increase pellet quality and decrease energy consumption [4-5]. The discussion on the difference between artificial magnetite pellet and natural magnetite pellet in the oxidation roasting was presented in this paper. This difference made a significant impact on roasting properties of two kind of magnetite pellet, as well as reveals design of parameters of induration equipment of the two kinds of pellet, and providing right selections of thermal operation model.
2 Material properties and testing method
Properties of pellet feeds are shown in Tables 1, 2 and 3. Table 1 shows that natural magnetite grading 64.34% TFe with low content of harmful elements of S and P, can be regarded as a superior iron concentrate. In contrast, artificial magnetite grading 61.13%TFe with a little amount of gangue (SiO2), of small particle size, is difficult to be used in sintering for agglomeration. Table 2 indicates that the bentonite with a higher content of montmorillonite and good water absorbency, is a good binder for pelletization.
It can be seen from Table 3, the average particle size of natural magnetite concentrate and the number- weighted average size for artificial one are all around 0.026 mm. As for the size distribution, artificial magnetite concentrate predominantly falls in coarse-size fraction and fine-size fraction, as compared to natural one, which is beneficial for pelletization [6-7]. Furthermore, with the similar average particle size, artificial magnetite concentrate has a specific surface area of 2720.4 cm2/g, compared with only 1115.9 cm2/g for natural one, indicating that artificial magnetite has rougher surface structure with more inner pores, thus tending to be pelletized [8]. It is found that natural magnetite concentrate with higher iron grade and higher density than artificial one, has a ballability index of only 0.41, being the medium-rank material for balling. In contrast, artificial one with a ballability index of 1.16 can be regarded as excellent raw material for pelletization [9].
Table 1 Chemical compositions of pellet feed
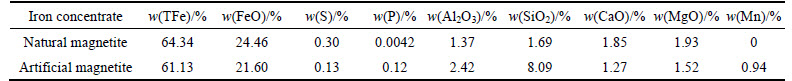
Table 2 Physical properties of bentonite
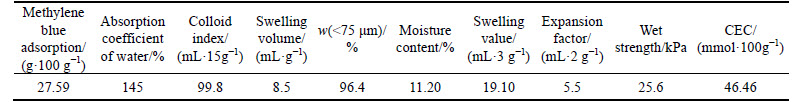
Table 3 Physical properties of pellet feed

The variance in the green ball growth is attributed to the different wettability of raw materials. For raw materials of good wettability, water will form a film closely adhering to the surface of pellet. And the liquid bridge between two pellets can be formed after a collision with another pellet, resulting in coalescence between the two pellets due to surface tension, which can promote the pellet growth. However, raw materials with bad wettability will result in the liquid film loosely on the pellet surface and less liquid bridge formed under collision, leading to less chance for pellet agglomeration. It can be seen from Table 4, Figs. 1 and 2, natural magnetite has a contact angle of 43.86° while artificial one has a contact angle of 35.11° by using distilled water as wetting liquid. It means that artificial magnetite has hydrophilic surface with more surfactivity to form more liquid film during the pelletizing process, tending to adhere more iron concentrate particles, thus beneficial for pelletization process.
The heat of wetting is measured with SETARAM C80 micro calorimeter. The effect of environmental temperature fluctuations is deleted with a twin calorimeter. The thermal signals produced by the system are amplified by amplification system. It can be seen that heat of wetting is negative for two kinds of magnetite, indicating that a heat-releasing process existed in their wetting process. The comparison between these two kinds of magnetite shows a bigger difference in their wetting properties. The heat of wetting of natural magnetite is -0.092 J/g, while -0.548 J/g for artificial one. In another words, lunder the same wetting condition, artificial magnetite tends to be wetted with more heat released, which is in agreement with the measurement of the contact angle [6].
Table 4 Surface wettability of pellet feeds [10]
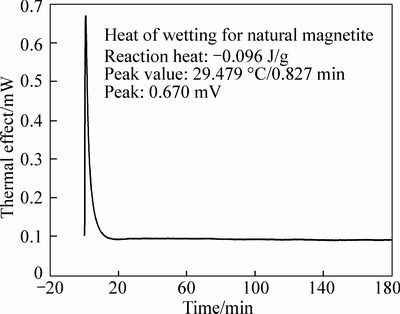
Fig. 1 Wetting heat of natural magnetite concentrate
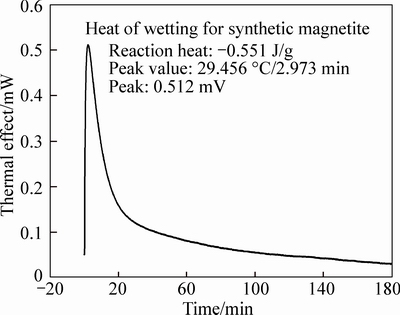
Fig. 2 Wetting heat of artificial magnetite concentrate
The process adopted in the experiment consists of grinding, blending, mixing, pelletizing, drying and roasting. The laboratory disc pelletizer used in the experiments is 800 mm in disc diameter and 210 mm in lip height, rotated at an adjustable speed of 18-28 r/min with an adjustable inclination of 41°-45° to the horizontal plane. After that, those green pellets were manually screened with pellets in the size fraction from 10 to 12 mm as qualified, which was subjected to the following pre-heating and roasting process in a horizontal tube furnace after being dried, for testing the impact of roasting temperature and time on the roasting characteristics of pellet. Finally, compressive strength test was conducted for 10 pellets, with the average value obtained as the compressive strength of the pellet product.
The porosity of fired pellet was calculated by equation:
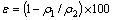 (1)
(1)
where ε is the porosity of fired pellets (%); ρ1 is apparent density (g/cm3); ρ2 is true density (g/cm3).
3 Oxidation characteristics of artificial magnetite pellet
Previous tests have identified the optimal condition for pelletization with natural and artificial magnetite as follows: adding bentonite at an amount of 2.5%, disc pelletizer with an inclination of 45° to the horizontal plane, water addition of 9% and pelletizing time controlled at 18 min. The produced green pellets were subjected to the roasting process at the temperatures of 400 °C, 600 °C, 800 °C, 1000 °C and 1200 °C, respectively, for 15 min, 20 min, 25 min, 30 min and 35 min, for investigating the oxidation degree of two kinds of magnetite concentrate ( i.e. Fe2O3-to-FeO ratio).
The oxidation of magnetite iron ore pellets is an important issue for several reasons. The oxidizing reaction is highly exothermic and releases a major part of the total energy required in sintering. Oxidation of magnetite leads to produce strong bonds in contact points, which reduces the generation of dust when pellets are loaded into the kiln [11]. Magnetite goes through the following two stages during the oxidation starting at 200 °C and completing around 1000 °C, generating α-Fe2O3 after the complete oxidation.
1st stage: 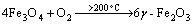 (2)
(2)
At this stage, the chemical action prevails without any crystal transformation occurring (both Fe3O4 and γ-Fe2O3 are cubic crystal). Only Fe3O4 was transformed to γ-Fe2O3, (i.e. magnetic hematite was generated). However, γ-Fe2O3 is not the stable phase.
2nd stage: 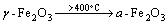 (3)
(3)
Due to γ-Fe2O3 phase being unstable, crystals tend to rearrange themselves at an elevated temperature, with some oxygen ions penetrating through the surface by direct diffusion, proceeding into the second stage of oxidization.
As can be seen from Fig. 3 to Fig. 7, FeO content in both two kinds of magnetite pellet decreases with increasing roasting temperature or prolonging roasting time. The oxidation degree of artificial magnetite pellet was up to over 80% at 400 °C, approaching to natural one at 1000 °C. However, the oxidation degree of natural one begins to decrease at 1200 °C, which is possibly attributed to the existing Fe2O3 decomposed to Fe3O4 under neutral or weak oxidizing atmosphere. It also indicates that oxidation degree of natural magnetite increased with the elevated temperature. While artificial magnetite reached 90% of oxidation degree at 600 °C, then little change in the oxidation degree was observed with the temperature change, in which there was something with the properties of artificial magnetite. Lots of researches have shown that crystallinity of artificial magnetite is lower than natural one, which is attributed to no occurrence of complete grain growth during artificial magnetite formation [12]. More lattice defects of artificial magnetite led to their surfactivity and reactivity higher than natural one. The incomplete lattice structure of artificial magnetite also led to a faster formation of solid solution by oxidation. Consequently, γ-Fe2O3 was generated at 200 °C. In contrast, natural magnetite started oxidizing only at temperature up to 400 °C, and the reaction rate was slower than artificial one due to its weak reactivity [3].
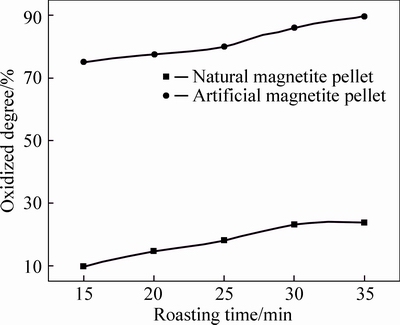
Fig. 3 Oxidation degree of pellets at 400 °C
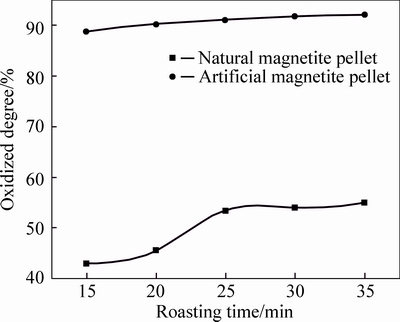
Fig. 4 Oxidation degree of pellets at 600 °
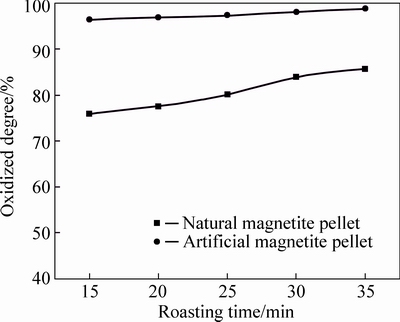
Fig. 5 Oxidation degree of pellets at 800 °C
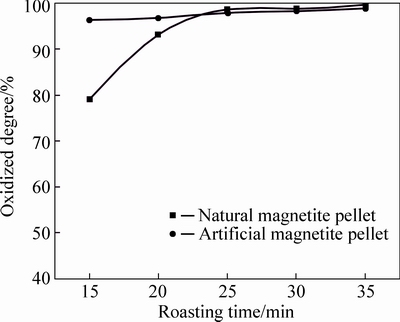
Fig. 6 Oxidation degree of pellets at 1000 °C
It can be concluded that with artificial magnetite as feed for pelletizing, most part of oxidization process can be completed with preheating at lower temperature, but higher temperature is required for roasting process than natural magnetite for meeting the qualified strength for industry application.
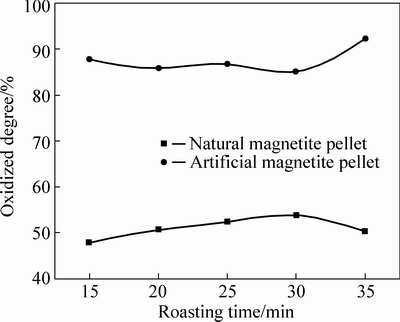
Fig. 7 Oxidation degree of pellets at 1200 °C
3 Roasting characteristics of artificial magnetite pellet
With the optimal condition for pelletization with natural and artificial magnetites as follows: adding bentonite as binder at an amount of 2.5%, disc pelletizer with an inclination of 45° to the horizontal plane, water addition of 9% and pelletizing time controlled at 18 min (including 2 min for nucleus formation, 12 min for nucleus growth and 4 min for green ball consolidation). Then, 10 green pellets with qualified grain size (10-12 mm) after drying were delivered for roasting test.
After drying process, artificial and natural magnetite pellets were subjected to 5-min preheating at the temperature of 500 °C, 600 °C and 700 °C, respectively, followed by 25-min roasting process at 1100 °C, 1150 °C, 1200 °C, 1250 °C and 1300 °C, respectively. The compressive strength of roasted pellets at different temperature is shown in Fig. 8.
As it can be seen in Fig. 8, the compressive strength increases with the elevated temperature for two kinds of magnetite. When the natural magnetite pellet was roasted at the temperature lower than 1100 °C, its compressive strength reached over 3000 N/pellet. In contrast, for the compressive strength up to 2500 N per pellet, the artificial magnetite pellet needs to be roasted at the temperature over 1150 °C. The roasting temperature for natural magnetite pellet was lower than artificial magnetite pellet, owing to the high content of completely crystallized Fe3O4 in the natural magnetite, which was transferred to Fe2O3 after oxidation during the roasting process, resulting in a large amount of heat released [11].
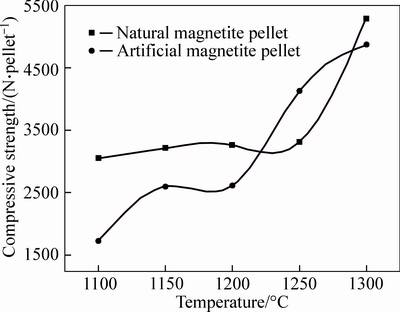
Fig. 8 Pellet compressive strength at different roasting temperatures
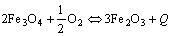 (4)
(4)
where Q is equal to 260 kJ/mol
Furthermore, the magnetite in the pellet together with gangue minerals like SiO2, is prone to generate low-melting-point compound, thus increasing the liquid compound in the pellet. Trace of liquid phase will make pellet volume decrease, resulting in a compact structure. And liquid phase will also make grains of Fe3O4 and Fe2O3 in the pellet dissolve and re-crystallize. The occurrence of liquid phase is favor for the diffusion of solid particles, promoting the grain growth, thus accelerating the solidification of solid phase in pellet, resulting in the pellet strength improved.
After being dried, natural and artificial magnetite pellets were subjected to 5-min preheating at 500 °C, followed by 5-min preheating at 600 °C and 5-min preheating at 700 °C, then roasting process at 1200 °C for 15 min, 20 min, 25 min, 30 min and 35 min, respectively. The compressive strength of roasted pellets after different roasting time is shown in Fig. 9.
It can be seen that at the same roasting temperature (1200 °C), the compressive strength of two kinds of magnetite increased with prolonging the roasting time, but in a lower rate, indicating that the roasting time has not as much impact as roasting temperature on the pellet strength. Natural magnetite pellet obviously required shorter roasting time than artificial magnetite pellet, with 15-min roasting to reach compressive strength up to 3000 N/pellet, but 30 min required for the artificial magnetite pellet to reach the same strength. Prolonging the roasting time would intensify the diffusion among particles, and promote a further oxidization and heat releasing, resulting in more active Fe2O3, which benefits the growth and intergrowth of crystals [13].
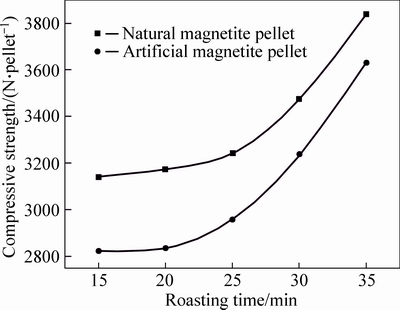
Fig. 9 Pellet compressive strength of different roasting time
4 Roasting characteristics of pellet of mixed magnetite concentrate
A mixed feed material with natural magnetite and artificial magnetite in the ratio of 4:1 was prepared for the pelletization test (previous research showed two kinds of magnetite in a ratio less than 4:1 would result in a sharp dropdown in the cracking temperature of green pellet), with bentonite dosage of 2.5%, disc pelletizer with an inclination of 45° to the horizontal plane, water addition of 9% and pelletizing time controlled at 18 min (including 2 min for nucleus formation, 12 min for nucleus growth and 4 min for green ball consolidation). Then, 10 green pellets with qualified grain size after drying were delivered for roasting test.
After being dried, pellets made of such mixed magnetite were subjected to 5-min preheating at 500 °C, 600 °C and 700 °C, followed by 25-min roasting at 1100 °C, 1150 °C, 1200 °C, 1250 °C and 1300 °C, respectively. The compressive strength of roasted pellets at different temperature is shown in Fig. 10. It can be seen that the compressive strength of obtained pellet increases with the rising temperature, partly because the liquid phase compound inside the pellet increased and filled into the inner voids of pellet, resulting in a more compacted pellet with complete structure. It showed that the compressive strength of pellet of mixed magnetite was over 3050 N per pellet at 1100 °C [14].
In another test, such mixed magnetite pellets, after being dried, were preheated for 5 min at 500 °C, 600 °C and 700 °C, followed by roasting process at 1200 °C for 15 min, 20 min, 25 min, 30 min and 35 min, respectively, resulting in the different compressive strength for the obtained roasted pellets (shown in Fig. 11). As it can be seen in Fig. 11, prolonging the roasting process led to an increase in the compressive strength of pellet, because it enhanced the diffusion among particles, also promoted the further oxidation of magnetite and more heat releasing. More reactive Fe2O3 generated is beneficial for the crystal growth and interconnection with each other. Results also showed the produced pellet had better compressive strength (up to 3140 N/pellet) after 15-min roasting.
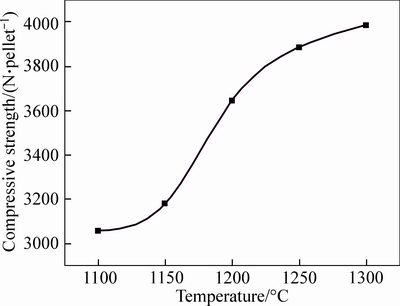
Fig. 10 Pellet compressive strength at different roasting temperatures
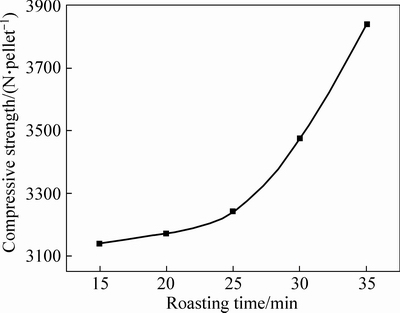
Fig. 11 Pellet compressive strength in roasting time
5 Mineral phase of produced magnetite pellet
Different magnetite pellets after 25-min roasting at 1200 °C were taken for mineral phase study. The microstructures are shown in Figs. 12, 13 and 14. After the process of oxidizing roasting, the magnetite pellet is predominantly composed of hematite (H), with trace of residual magnetite (M). Due to the artificial magnetite pellet with lower grade than natural magnetite pellet, around 8% of siliceous component (Q) can be observed in these figures. The secondary hematite in the natural magnetite pellet has a better crystallinity. Those closer to the center of pellet are larger in grain size. A large amount of closed voids and some debris of silicate minerals like quartz, feldspar, pyroxene were distributed among the hematite particles. In the mixed magnetite pellet, the hematite crystal closer to the center of pellet is in larger particle size. The magnetite in the pellet occurs more frequently in the center than along the margin, with the content around 7%, principally in the form of irregular granules.
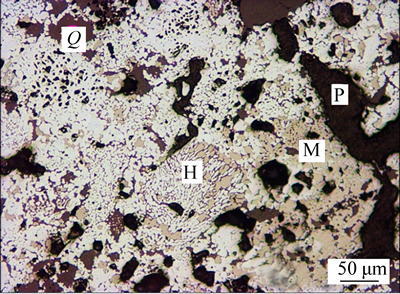
Fig. 12 Mineral phases of fired artificial magnetite pellets under optical microscope
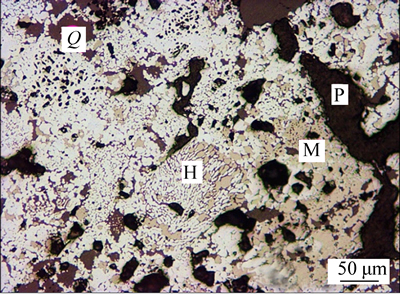
Fig. 13 Mineral phases of fired natural magnetite pellets under optical microscope

Fig. 14 Mineral phases of mixed magnetite pellets under optical microscope
In terms of inner pores (P), natural magnetite pellet with more closed pores has around 10% porosity. The irregular voids and pore passage principally distributed within the inter particle voids of hematite, magnetite and silicate. And the artificial magnetite pellet with poor consolidation of liquid phase inside has 20% porosity, with pores in form of irregular shape and distributed unevenly [15]. However, the mixed magnetite pellet has 16% porosity, with relatively good pore formation, mostly in irregular form and some in round or near olive shape. Inner pores are predominantly closed with an uneven distribution. Pores and pore passage are more frequently distributed in the inner band of pellet than in outer band, mostly in larger grain size and less likely to be closed [16].
6 Conclusions
1) It is found that the artificial magnetite pellet is different from natural one in terms of oxidization characteristics. Artificial magnetite pellet can reach 80% of oxidation degree after roasting at the temperature of 400 °C, which approximates natural magnetite pellet at 1000 °C. That means that artificial magnetite pellet has a higher oxidation degree than natural under the same oxidizing condition. Consequently, artificial magnetite pellet for pelletizaiton can complete most oxidizing reaction by being preheated at lower temperature.
2) After 25-min roasting at 1100 °C, the produced natural magnetite pellet can have compressive strength up to 3000 N/pellet, while artificial magnetite pellet needs to be roasted at the temperature over 1250 °C for reaching the same strength. Furthermore, at the same roasting temperature of 1200 °C, it takes 15 min for natural magnetite pellet, but 30 min for artificial magnetite pellet to reach the compressive strength of 3000 N/pellet. It is concluded that artificial magnetite pellet due to fast oxidation, requires the elevated temperature and prolonged time for roasting process to reach the expected compressive strength.
3) The mineral phase microstructure of pellet made of different kind of magnetite revealed that natural magnetite pellet has around 10% porosity, with pores mostly closed and irregular pore passage distributed within the interparticle voids of hematite, magnetite and silicate. In contrast, the artificial magnetite pellet with poor consolidation of liquid phase inside, has 20% porosity with pores in form of irregular shape and distributed unevenly. However, the mixed magnetite pellet has 16% porosity with pores in relatively good formation, mostly in irregular form and some in round or near olive shape. Inner pores are predominantly closed, with a uneven distribution. Pores and pore passage are more frequently distributed in the inner band of pellet than in outer band, mostly in larger grain size and less likely to be closed.
References
[1] CHO H J, TANG M, PISTORIUS P C. Magnetite particle size distribution and pellet oxidation [J]. Metallurgical and Materials Transactions B, 2014, 45B: 1213-1220.
[2] PANG Jian-ming, GUO Pei-min, ZHAO Pei. Reduction of 1-3 mm iron ore by CO on fluidized bed [J]. Journal of Iron and Steel Research, International, 2011, 18(3): 1-5.
[3] PANGEY B K, SHARMA T. Reducing agents and double-layered iron ore pellets [J]. International Journal of Mineral Processing, 2000, 59: 295-304.
[4] JIANG Chang-wei, FU Ju-ying, LI Si-dao, YAO Jun-feng. Study on the Induration property of Aoshan magnetite concentrates pellets [J]. Journal of Central South University of Technology: Natural Science, 1998, 29(5): 435-437. (in Chinese)
[5] FU Ju-ying, LI Yun-tao, JIANG Chang-wei, WANG Li-guang. Oxidation kinetics of magnetite concentrate pellets [J]. Journal of Central South University: Natural Science, 2004, 35(6): 950-954. (in Chinese)
[6] FORSMO S P E, SAMSKOG P O, BJORKMAN B M T. A study on plasticity and compression strength in wet iron green pellets related to real process variations in raw material fineness [J]. Powder Technology, 2008, 181: 321-330.
[7] LIU Shu, LU Man-man, FU Jin-tao, ZOU Peng, ZHANG Han-quan. Influences of content of iron materials with partical size of -0.025 mm on ballability of magnetite concentrates [J]. Sintering and Pelletizing, 2014, 39(5): 31-34.
[8] ZHANG Han-quan, WANG Feng-ling. Regulation of mineral composition and phase transformation in limonite magnetic roasting process [J]. Journal of Iron and Steel Research, 2014, 26(7): 8-11. (in Chinese)
[9] FU Ju-ying, JIANG Tao, ZHU De-qing. Principle of sintering and pelletizing [M]. Changsha: Central South University of Technology Press, 1996. (in Chinese)
[10] ZHANG Han-quan, WANG Feng-ling. Analysis of surface wettability of synthetic magnetite [J]. Journal of Wuhan University of Technology: Mater, 2014, 29(4): 679-683.
[11] FORSMOA S P E, FORSMOA S E, SAMSKOG P O, BJ RKMAN B M T. Mechanisms in oxidation and sintering of magnetite iron ore green pellets [J]. Powder Technology, 2008, 183: 247-259.
RKMAN B M T. Mechanisms in oxidation and sintering of magnetite iron ore green pellets [J]. Powder Technology, 2008, 183: 247-259.
[12] HUANG Xiao-yan, GUO Shan-shan, LI Huai-hua, et al. Research on magnetic properties and magnetic separation behavior of natural and artificial magnetite [J]. Mining and Metallurgical Engineering, 2012, 32(2): 30-33. (in Chinese)
[13] ESTHER L, JAVIER H, JOSE A P. Steam-iron process: Influence of steam on the kinetics of iron oxide reduction [J]. International Journal of Hydrogen Energy, 2011, 36: 13425-13434.
[14] FAN Jian-jun, QIU Guan-zhou, JIANG Tao, SHEN Hui-ting. Roasting properties of pellets with iron concentrate of complex mineral composition [J]. Journal of Iron and Steel Research International. 2011, 18(7): 1-7.
[15] BUIYAN L U, MOUZON J, FORSMO S P E, HEDLUND J. Quantitative image analysis of bubble cavities in iron ore green pellets [J]. Powder Technology, 2011(214): 306-312.
[16] BAI Guo-hua, ZHOU Xiao-qing. Study on pelletizing of pyrite cinder with magnetite [J]. Iron and Steel, 2009, 44(7): 7-10. (in Chinese)
(Edited by YANG Hua)
Foundation item: Project(51474161) supported by the National Natural Science Foundation of China
Received date: 2015-11-11; Accepted date: 2016-03-17
Corresponding author: ZHANG Han-quan, Professor, PhD; Tel: +86-27-87194821; E-mail:springt@139.com

