��ͭ����������������������ڿ������ĸ���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2016���2��
�������ߣ����ü� �ܵ� ��ѧ�� ������ ���� ������ ���� ����� ��ΰ��
����ҳ�룺544 - 550
�ؼ��ʣ���ͭ�������������������ȡ�������������������
Key words��chalcopyrite; ferric iron; extracellular polymeric substance; extraction; sonication; bioleaching
ժ Ҫ��Ϊ̽����ͭ�������������������������ϵ��������ڿ������ĸ��������ó��������Ⱥ���������ȡ���������������������������48 ��C�����³�����һ����Ч�ķ�����ɨ��羵������ɫɢX��������(EDX)�����������������ͭ�������ڴ����ѷ�Ͱ��ݣ��������������������Щ�ѷ�Ͱ����С��о��������̻�ͭ������������Ͱ�������ĺ����仯�������������������ĺ����ڽ���ǰ10 dѸ��������֮��ά����һ���ȶ���ˮƽ�������������������ʱ����ӳ������ӣ��������ڽ������ڡ�
Abstract: In order to investigate the enrichment of ferric iron bound by extracellular polymeric substance (EPS) on the mineral surface during bioleaching of chalcopyrite, several methods including sonication, heating and vortexing were used and sonication at 48 ��C was shown as a good way to extract ferric iron. Scanning electron microscope (SEM) and energy dispersive X-ray spectrometer (EDX) analysis showed that lots of cracks and pits can be found on the chalcopyrite surface after bioleaching and that iron oxide was filled in these cracks and pits. The variations of contents of ferric iron and EPS on the chalcopyrite surface were investigated. The results indicated that the content of EPS increased rapidly in the first 10 d and then maintained at a stable level, while ferric iron content increased all the time, especially in the later stage of bioleaching.
Trans. Nonferrous Met. Soc. China 26(2016) 544-550
Tang-jian PENG1, Dan ZHOU1, Xue-duan LIU1,2, Run-lan YU1,2, Tao JIANG1,2, Guo-hua GU1,2, Miao CHEN3, Guan-zhou QIU1,2, Wei-min ZENG1,2,3
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Biometallurgy, Ministry of Education, Central South University, Changsha 410083, China;
3. CSIRO Process Science and Engineering, Box 312, Clayton South, Victoria 3169, Australia
Received 24 March 2015; accepted 15 September 2015
Abstract: In order to investigate the enrichment of ferric iron bound by extracellular polymeric substance (EPS) on the mineral surface during bioleaching of chalcopyrite, several methods including sonication, heating and vortexing were used and sonication at 48 ��C was shown as a good way to extract ferric iron. Scanning electron microscope (SEM) and energy dispersive X-ray spectrometer (EDX) analysis showed that lots of cracks and pits can be found on the chalcopyrite surface after bioleaching and that iron oxide was filled in these cracks and pits. The variations of contents of ferric iron and EPS on the chalcopyrite surface were investigated. The results indicated that the content of EPS increased rapidly in the first 10 d and then maintained at a stable level, while ferric iron content increased all the time, especially in the later stage of bioleaching.
Key words: chalcopyrite; ferric iron; extracellular polymeric substance; extraction; sonication; bioleaching
1 Introduction
Chalcopyrite is the most refractory and abundant copper sulphide mineral in the world. Traditionally, this mineral has been treated by pyrometallurgical methods to obtain copper [1]. Due to high emission of polluted gas mainly as SO2 and high capital costs, the research for a hydrometallurgical process as an alternative to conventional smelting has been accelerated. Bioleaching of chalcopyrite is a copper extraction process with low capital and operation costs, and environmental benefits [2]. At present, more attention has been paid to chalcopyrite bioleaching with moderately thermophilic microorganisms [3]. This is because using moderate thermophiles compared with mesophiles to bioleach chalcopyrite can greatly improve the leaching reaction kinetics, hence, the total copper extraction is higher [4].
Ferric iron plays a very important role during bioleaching of chalcopyrite. It has been reported that bioleaching of chalcopyrite includes several steps, which starts with the dissolution of mineral sulphides by protons and ferric iron presented in the bioleaching medium [5,6]. This step liberates metals in their ionic forms and produces hydrogen sulphide. Hydrogen sulphide is transformed by the oxidation with Fe(III) and subsequent dissociation and dimerization in the polysulphide. As a result of the combined actions of protons and ferric iron, elemental sulphur is produced. The reactions of this mechanism are described as follows:
CuFeS2+Fe3++H+��Cu2++Fe2++H2Sn (n��2) (1)
0.5H2Sn+Fe3+��0.125S8+Fe2++H+ (2)
The oxidation of sulphur to sulphuric acid is mainly completed by the sulphur-oxidizing microorganisms.  et al [7] recognized that ferric iron could also oxidize elemental sulphur to sulphuric acid (Eq. (3)), and the oxidation rate increased with the increase of dissolved oxygen in the solution. Therefore, they believed that ferric iron was the main oxidant in the bioleaching system (Eq. (3)).
et al [7] recognized that ferric iron could also oxidize elemental sulphur to sulphuric acid (Eq. (3)), and the oxidation rate increased with the increase of dissolved oxygen in the solution. Therefore, they believed that ferric iron was the main oxidant in the bioleaching system (Eq. (3)).
CuFeS2+4Fe3++3O2+2H2O��Cu2++5Fe2++2H2SO4 (3)
It has been pointed out that extracellular polymeric substance (EPS) also plays a pivotal role during bioleaching of chalcopyrite [8,9]. EPS is generated by bioleaching microorganisms and mediate the attachment of microorganisms to the mineral surface. After the initial attachment of cells, the biofilm develops within a few days, covering the mineral surface with cells embedded in a continuous EPS layer [10,11]. Generally, the EPS consists of polysaccharides as well as some proteins, nucleic acids, lipids and humic substances, and the composition varies with the mineral surface chemistry, temperature, pH, leachate solution, the microbial species and growth substrates [12-14].
Ferric iron on the mineral surface is reported to be concentrated by complexation through uronic acids or other residues of EPS [15,16]. This would create an environment of net positive charges which may assist in attachment of cells to the negatively charged minerals. The EPS containing bound ferric iron therefore plays an active role in the dissolution of sulphide minerals by creating a reaction space for the dissolution process to take place [17]. To date, little is known about conditions within this reaction space, including the pH, oxidation reduction potential (ORP), and especially the ferric iron that is critical for the metal-winning. Although there have been some reports about the extraction of EPS from the ore surface during bioleaching, few studies have been conducted to extract the EPS-bound ferric iron, and little information is available with respect to the variation of EPS and the bound iron (III) during bioleaching [15]. To this end, several methods (sonication, heating and vortexing) were designed to extract iron(III) bound with EPS from the mineral surface. The chalcopyrite surface during bioleaching was analyzed by SEM-EDX to detect the enrichment of ferric iron. Finally, the variations of the contents of EPS and ferric iron on the mineral surface during bioleaching were investigated.
2 Experimental
2.1 Microorganisms enrichment
Acid mine drainages (AMD) samples from several chalcopyrite mines in China were collected. The samples were mixed and then inoculated into the culture medium for the enrichment of moderate thermophiles. The medium used for the enrichment consisted of the following compounds: (NH4)2SO4 3.0 g/L, Na2SO4 2.1 g/L, MgSO4��7H2O 0.5 g/L, K2HPO4 0.05 g/L, KCl 0.1 g/L, Ca(NO3)2 0.01 g/L. Chalcopyrite concentrate (50 g/L) was added as energy source. The samples were enriched at 48 ��C and pH 2.0. After adaptation to chalcopyrite concentrate for more than 4 years, this moderately thermophilic culture showed excellent performance of chalcopyrite dissolution. Previous studies have shown that Acidithiobacillus caldus and Leptospirillum ferriphilum were the dominant microorganisms in the culture [18,19].
2.2 Mineral components
The chalcopyrite sample was collected from Dabaoshan in Guangdong Province, China, with diameters of the particles less than 75 ��m. The mineral sample was analyzed by X-ray diffraction (XRD) and mainly consisted of 98% chalcopyrite and 2% silicate (mass fraction).
2.3 Bioleaching experiments
Bioleaching experiments were carried out in a 3 L glass cylindrical reactor with a mechanical stirrer operated at 120 r/min. 1950 mL medium, 0.4 g/L yeast extract and 120 g chalcopyrite (6% pulp density) were added into the reactor, and 50 mL seed culture (no soluble copper) was inoculated to get a cell density of 1��106 cell/mL. Air was blown into the base of the reactor at an approximate rate of 360 mL/min. The experiments were performed at 48 ��C and pH 2.0. Evaporation loss was compensated by distilled water. The ORP and the concentrations of Cu2+, ferrous iron and total iron in the solution were analyzed every two days, while the pH and ORP of the solution were measured each day. These parameters were determined as described in Ref. [19]. Furthermore, ore residue was analyzed at the end of bioleaching by XRD.
Attached cells were removed from the mineral surface as previously reported in Ref. [11]. Briefly, the leaching solution was centrifuged at 2000 r/min for 2 min, then the obtained pellets were re-suspended and voretexed, and centrifuged at 2000 r/min again. These steps were repeated thrice. A final solution was obtained and centrifuged at 12000 r/min for 10 min to collect the sessile microorganisms. Cells were counted by a counting chamber under an optical microscope.
2.4 Methods for extraction of iron on mineral surface
It has been reported that sonication can remove ions from solid materials. Two main mechanisms may contribute to the process: acoustic streaming which can aid the mass transport of species away from the solid-liquid interface and cavitation which can cause cleaning of the surface [20]. In our previous study [19], vortexing was used to collect attached microorganisms during bioleaching. We found that with increasing the vortexing speed and time, some metal ions would release into solution. Also, vortexing has been employed for the removal of harmful contents from waste samples [21]. Another method generally involved in this process is heating [20]. Therefore, in this experiment, sonication, vortexing and heating were used to extract ferric iron from the ore surface during bioleaching of chalcopyrite. The detail is shown as follows.
After bioleaching of chalcopyrite for 20 d, the solution was settled for 1.5 h and then the supernatant was decanted. The thickened sludge was centrifuged at 2000 r/min and 4 ��C for 4 min and 4 g pellets were collected and resuspended with 10 mL H2SO4 at pH 2.0. For extraction of ferric iron from the mineral surface, the ore residue was treated by five methods including sonication at 48 ��C for 15 min, sonication at 24 ��C for 15 min, heating at 70 ��C for 30 min, vortexing with 1 g glass beads (with diameter about 0.2 mm) on a vortexer for 15 min, heating for 20 min and then vortexing for 10 min. The control experiment without any treatment was carried out. Then, the treated residue and the control were centrifuged at 2000 r/min and 4 ��C for 2 min. The solution was used to determine the concentrations of total iron, ferric and ferrous iron. These experiments were carried out in triplicate.
In order to detect the chalcopyrite surface, the chalcopyrite ore was ground to a slice from a cube and then immobilized in the bioleaching solution. After being bioleached for 20 d, the chalcopyrite slice was removed from the bioleaching solution and then washed by diluted sulphuric acid (pH 2.0) and exposed in the air for 2 h for drying. After that, the slice surface was analyzed by SEM and EDX, which were performed on an FEI Quanta 400 field emission, environmental scanning electron microscope (ESEM) under high vacuum conditions. Secondary electron imaging was performed at a beam energy of 15 kV and a probe current of 140 to 145 pA. EDX was performed at the beam energy of 15 kV and a probe current of approximately 800 pA. The components of the mineral sample and ore residue were analyzed by X-ray diffraction (XRD) (Panalytical, X'pert Pro, Netherlands) with Cu K�� radiation (40 kV, 35 mA).
2.5 Extraction of EPS and ferric iron during bioleaching
During bioleaching of chalcopyrite, the EPS and ferric iron on the mineral surface were extracted on the 1st, 5th, 10th, 15th and 20th day. The extraction methods of EPS are described in detail as follows: the bioleaching solution was settled for 1.5 h, and then the supernatant was decanted. The thickened sludge was centrifuged at 2000 r/min and 4 ��C for 4 min and 4 g pellet was re-suspended in Milli Q water. 1 g glass bead with a diameter of 0.2 mm was added into the re-suspension and shaken on a vortexer (0-1400 r/min) for 10 min. Then, 10 mmol/L tris-HCl (pH 7.0), 1 ��mol/L N- dodecyl-N, N-dimethyl-3-ammonio-1-propane-sulfonate and 1 mmol/L ethylene glycol tetraacetic acid (EGTA) were added into the solution at 4 ��C. Following extraction, all samples were centrifuged and filtered through 0.2 ��m filters to collect the soluble fractions. The extracted samples were stored at -20 ��C until analysis [10]. Contamination by the membrane fragments of damaged cells, possibly caused by vortexing, was checked by detection of 2-keto-3-deoxyoctonate (KDO) and DNA. KDO is part of the lipopolysaccharide (LPS) in Gram-negative bacteria, and it can be used as a marker for their outer membrane compound contamination. Since the Gram-positive bacteria have stronger cell wall structure than Gram-negative bacteria, they are hard to be broken by vortexing in this experiment. Furthermore, the detection of DNA concentration could offer more information to investigate the breaking extent of all bacteria and archaea. Therefore, low levels of both DNA and KDO indicate that the EPS extracted was not contaminated by significant amounts of intracellular materials [11].
Crude EPS was analyzed for nitrogen, uronic acids and protein spectrophotometrically [11]. Qualitative and quantitative estimations of fatty acids and sugar monomers were achieved by gas�Cliquid chromatography. Experimental procedures have been described in Ref. [10].
3 Results and discussion
3.1 Bioleaching of chalcopyrite by moderate thermophiles
Bioleaching of chalcopyrite by moderately thermophilic microorganisms was carried out in the stirred tank reactor for 20 d. Figure 1(a) shows the copper concentration in solution as a function of bioleaching time. The copper concentration increased quickly from the very beginning without lag phase, this was because the mixed culture has been acclimated for more than 4 years [18]. On the 16th day, the soluble copper achieved up to 12.18 g/L. After that, the chalcopyrite dissolution rate became very slow. On the 20th day, the soluble copper was 12.66 g/L, and the final copper extraction percentage achieved was 61.7%.
The concentration of iron in solution showed different variation trends compared with copper concentration (Fig. 1(a)). Before the 8th day, the concentrations of ferrous and ferric iron both increased. After that, the concentration of ferric iron decreased. At that time, the pH value was between 1.4 and 1.7 and the ORP was between 400 and 470 mV. When most of the copper was leached out (after 16 d), the concentration of ferrous iron began to decrease and the concentration of ferric iron began to increase, which corresponded to an increase of ORP (up to 623 mV on day 20). FOWLER and CRUNDWELL [17] reported that iron played a key role in the bioleaching of chalcopyrite. Ferric iron dissolved chalcopyrite to generate copper and ferrous iron. Ferrous iron as the energy source of bioleaching microorganisms was oxidized to regenerate ferric iron. In the final stage of bioleaching, the dissolution of chalcopyrite was hindered, and the concentration of ferric iron increased when the surface of chalcopyrite was covered with passivation substances formed as jarosite [18]. In the meanwhile, ferrous iron was oxidized continuously by microorganisms and reached a low concentration in the end.
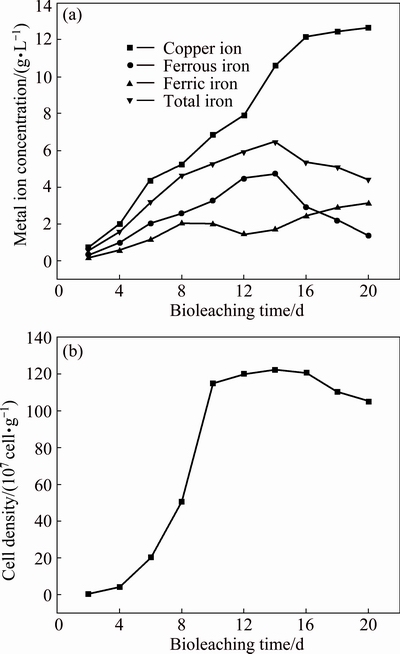
Fig. 1 Variations of copper ion, ferrous iron, ferric iron and total iron concentrations (a) and attached cell density (b) as function of time during bioleaching of chalcopyrite by moderate thermophiles
As can be seen from Fig. 1(b), the number of attached cells increased in the first 10 d with short lag phase and then kept stable at around 1.2��109 cell/g of ore residue from the 12th to 16th day. During that time, copper extraction rate increased in a large scale. After 16 d, when most of copper was leached out, the sessile biomass decreased mainly due to the diminishing of some essential nutrients (ferrous iron and sulphur).
At the end of bioleaching, the content of ore residue was 41 g/L, and the XRD analysis showed that the component of ore residue mainly included jarosite (44%) and chalcopyrite (50%). The content of element sulphur was very low, less than 4%. During bioleaching of chalcopyrite, temperature is a very important factor for the precipitation of iron compounds like jarosite, and their precipitates increase as temperature rises. Thus, in moderate thermophilic environments (like 48 ��C), more jarosite is readily formed than in mesophilic environments [22].
3.2 Methods for extraction of iron bound with EPS
The ore residue after being bioleached for 20 d was treated in five different ways to extract iron bound with EPS. Table 1 shows the results of extracted iron from mineral surface. It can be seen that the treatment by ultrasonic at 48 ��C can obtain a maximum content of iron. The contents of total iron and ferric iron were 70.5 mg/g and 42.3 mg/g, respectively. Sonication has been proved to be an effective technique to facilitate the removal and isolation of a variety of substances, such as fluoride (F-), cyanide (CN-) and sodium (Na+) ions, lead and methylmercury [20,23]. However, to the best of our knowledge, few studies have been conducted to extract EPS-bound iron on the mineral surface by ultrasonic methods [24]. For confirming that the iron was extracted from the mineral surface, but not from chalcopyrite or jarosite, the components of ore residue before and after treatment were analyzed by XRD. The result showed that compared with the ore residue without treatment, there was no obvious difference in the component of ore residue after treatment by ultrasonic at 48 ��C. Therefore, it is suggested that the treatment by ultrasonic at 48 ��C can effectively extract iron from the mineral surface.
According to the maximum content of ferric iron extracted, the global concentration of the EPS-bound ferric iron after being bioleached for 20 d was about 1.7 g/L. Considering that ferric iron was filled in the imperfections of mineral surface (see results of SEM and EDX), the local concentration at the mineral/cell interface would be much higher. This indicates that lots of ferric iron is concentrated on the mineral surface, which provides experimental evidence to support the ��indirect contact mechanism�� of bioleaching [6]. In the indirect contact mechanism, it is supposed that the bioleaching microorganisms prefer to attaching onto the mineral surface and producing EPS. A reaction zone as extracellular polymeric layer between the cell wall and the chalcopyrite is created. In this zone, ferric iron is concentrated to dissolve chalcopyrite. The dissolution of chalcopyrite would release the energy source such as ferrous iron for growth of microorganisms.
For better understanding the enrichment of ferric iron on the mineral surface, the chalcopyrite slices (98% CuFeS2) with and without bioleaching were analyzed by SEM and EDX (Fig. 2). Lots of imperfections (cracks and pits) can be found on the chalcopyrite surface after bioleaching. The analysis of the chalcopyrite surface by EDX showed that iron oxide was filled in these cracks and pits. This further indicated that ferric iron was enriched on the mineral surface during bioleaching. The iron oxide mainly originated from the reaction of ferric iron and oxygen when the chalcopyrite slice was exposed to air in the experiment.
Table 1 Contents of total iron, ferric iron and ferrous iron per gram of ore residue after being bioleached for 20 d (mg/g)

3.3 Extraction and analysis of ferric iron and EPS on mineral surface during bioleaching
The content of DNA extracted from the ore residue was less than 0.25 mg/g, and the KDO content in the extracted EPS was in the range of 0.03-0.08 mg/g. The low contents of DNA and KDO in the extracted EPS indicated negligible contamination by intracellular substances in the collected EPS [10].
Table 2 shows the components and contents of EPS and ferric iron extracted from the ore residues on the 1st, 5th, 10th, 15th and 20th day during bioleaching of chalcopyrite. As can be seen, the collected EPS was primarily composed of sugars and fatty acids and the content of protein was relatively low. The composition of the EPS was similar to that of previous studies [10]. The content of extracted EPS varied according to the sampling time. From the 1st day to the 10th day, the content of extracted EPS increased rapidly to 59.7 mg/g. During the first 10 d of bioleaching, the increase of EPS benefits the enrichment of ferric iron on the mineral surface, and the content of ferric iron increased from 4 to 19 mg/g, which would oxidize chalcopyrite to release energy source for microbial growth. Thus, during this time, the cell density in the solution increased and reached the logarithmic phase. From the 10th day up to the end of bioleaching, although the cell density in the bioleaching began to decrease, the total content of extracted EPS still maintained at a steady level and the final content of EPS extracted was 69.6 mg/g. This indicated that once the EPS was produced, it was difficult to be eliminated from the bioleaching system.

Fig. 2 SEM images and EDX patterns of chalcopyrite surface with and without bio-leaching
Table 2 Components and contents of EPS and ferric iron in ore residue during bioleaching of chalcopyrite (��b.d.�� means below detection limit)

It has been reported that for some Acidithiobacillus ferrooxidans strains, the uronic acids within the EPS combine with ferric iron at a molar ratio of about 2:1. This causes the cells to have a net positive charge, therefore, enhancing cell attachment to the negatively charged substrates [15]. However, in this study, the molar ratio of uronic acids to ferric iron was lower than 2:1 and rather varied in different bioleaching periods. Two main reasons account for the inconsistency. Firstly, complexation of ferric ion varied between strains, and because we used a microbial consortium in this study, the ratio would be quite different from that of a pure culture. Secondly, there may be some other mechanisms for the enrichment of ferric iron. Sugars like glucose, fatty acids, amino acids, or other negatively charged groups, could also contribute to the binding of ferric iron [15,25]. The content of uronic acids in the EPS kept at a relatively stable level after the 10th day, while the content of ferric iron increased as bioleaching continued. Especially in the later stage of bioleaching, the content of ferric iron enriched on the mineral surface increased from 19 mg/g (on the 10th day) to 45 mg/g (on the 20th day). This may be because during this period, large amounts of EPS and jarosite were covered on the mineral surface to form a passivation layer, which would block the diffusion of metal ions to the outside of the layer. However, in the micro-space on the mineral surface, the dissolution of chalcopyrite still continued, and the metal ions like ferric ion were released and accumulated.
4 Conclusions
1) A mixed culture of moderately thermophilic microorganisms was used to bioleach chalcopyrite in a stirred tank reactor, and showed good performance on copper extraction.
2) Sonication at 48 ��C is effective for extraction of the EPS-bound iron.
3) A large amount of ferric iron was enriched on the surface of mineral, and in particular, in the cracks and pits.
4) The variations of EPS and the ferric iron bound by EPS showed different trends and could be relevant to the leaching of chalcopyrite.
References
[1] PAN Hao-dan, YANG Hong-ying, TONG Lin-lin, ZHONG Cong-bin, ZHAO Yu-shan. Control method of chalcopyrite passivation in bioleaching [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(9): 2255-2260.
[2] XIA Jin-lan, YANG Yi, HE Huan, LIANG Chang-li, ZHAO Xiao-juan, ZHENG Lei, MA Chen-yan, ZHAO Yi-dong, NIE Zhen-yuan, QIU Guan-zhou. Investigation of the sulfur speciation during chalcopyrite leaching by moderate thermophile Sulfobacillus thermosulfidooxidans [J]. International Journal of Mineral Processing, 2010, 94(1-2): 52-57.
[3] WANG Yu-guang, SU Li-jun, ZENG Wei-min, QIU Guan-zhou, WAN Li-li, CHEN Xin-hua, ZHOU Hong-bo. Optimization of copper extraction for bioleaching of complex Cu-polymetallic concentrate by moderate thermophiles [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(4): 1161-1170.
[4] BEHRAD V A. A comparison of bioleaching ability of mesophilic and moderately thermophilic culture on copper bioleaching from flotation concentrate and smelter dust [J]. International Journal of Mineral Processing, 2011, 101(1-4): 94-99.
[5] ROHWERDER T, GEHRKE T, KINZLER K, SAND W. Bioleaching review. Part A: Progress in bioleaching: Fundamentals and mechanisms of bacterial metal sulfide oxidation [J]. Applied Microbiology and Biotechnology, 2003, 63(3): 239-248.
[6] SAND W, GEHRKE T. Extracellular polymeric substances mediate bioleaching/biocorrosion via interfacial processes involving iron(III) ions and acidophilic bacteria [J]. Research in Microbiology, 2006, 157(1): 49-56.
[7]  BALLESTER A. Leaching of chalcopyrite with ferric ion. Part II: Effect of redox potential [J]. Hydrometallurgy, 2008, 93(3-4): 88-96.
BALLESTER A. Leaching of chalcopyrite with ferric ion. Part II: Effect of redox potential [J]. Hydrometallurgy, 2008, 93(3-4): 88-96.
[8] VERA M, SCHIPPERS A, SAND W. Progress in bioleaching: Fundamentals and mechanisms of bacterial metal sulfide oxidation��Part A [J]. Applied Microbiology and Biotechnology, 2013, 97(17): 7529-7541.
[9] YU Run-lan, LIU Jing, CHEN An, ZHONG Dai-li, LI Qian, QIN Wen-qing, QIU Guan-zhou, GU Guo-hua. Interaction mechanism of Cu2+, Fe3+ ions and extracellular polymeric substances during bioleaching chalcopyrite by Acidithiobacillus ferrooxidans ATCC2370 [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(1): 231-236.
[10] GEHRKE T, TELEGDI J, THIERRY D, SAND W. Importance of extracellular polymeric substances from Thiobacillus ferrooxidans for bioleaching [J]. Applied and Environmental Microbiology, 1998, 64(7): 2743-2747.
[11] ZENG Wei-min, TAN Su-nee, CHEN Miao, QIU Guan-zhou. Detection and analysis of attached microorganisms on the mineral surface during bioleaching of pure chalcopyrite with moderate thermophiles [J]. Hydrometallurgy, 2011, 106(1-2): 46-50.
[12] BAZAKA K, CRAWFORD R, NAZARENKO E, IVANOVA E. Bacterial adhesion [M]. Netherlands: Springer, 2011: 213-226.
[13] ZHANG R Y, BELLENBERG S, CASTRO L, NEU T R, SAND W, VERA M. Colonization and biofilm formation of the extremely acidophilic archaeon Ferroplasma acidiphilum [J]. Hydrometallurgy, 2014, 150(0): 245-252.
[14] HE Zhi-guo, YANG Yan-ping, ZHOU Shan, HU Yue-hua, ZHONG Hui. Effect of pyrite, elemental sulfur and ferrous ions on EPS production by metal sulfide bioleaching microbes [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(4): 1171-1178.
[15] KINZLER K, GEHRKE T, TELEGDI J, SAND W. Bioleaching��A result of interfacial processes caused by extracellular polymeric substances (EPS) [J]. Hydrometallurgy, 2003, 71(1-2): 83-88.
[16] YU Run-lan, TAN Jian-xi, GU Guo-hua, HU Yue-hua, QIU Guan-zhou. Mechanism of bioleaching chalcopyrite by Acidithiobacillus ferrooxidans in agar-simulated extracellular polymeric substances media [J]. Journal of Central South University of Technology, 2010, 17(1): 56-61.
[17] FOWLER T A, CRUNDWELL F K. Leaching of zinc sulfide by Thiobacillus ferrooxidans: Experiments with a controlled redox potential indicate no direct bacterial mechanism [J]. Applied and Environmental Microbiology, 1998, 64(10): 3570-3575.
[18] ZHOU Hong-bo, ZENG Wei-min, YANG Zhi-feng, XIE Ying-jian, QIU Guan-zhou. Bioleaching of chalcopyrite concentrate by a moderately thermophilic culture in a stirred tank reactor [J]. Bioresource Technology, 2009, 100(2): 515-520.
[19] ZENG Wei-min, QIU Guan-zhou, ZHOU Hong-bo, PENG Juan-hua, CHEN Miao, TAN Su-nee, CHAO Wei-liang, LIU Xue-duan, ZHANG Yan-sheng. Community structure and dynamics of the free and attached microorganisms during moderately thermophilic bioleaching of chalcopyrite concentrate [J]. Bioresource Technology, 2010, 101(18): 7068-7075.
[20] SATERLAY A J, HONG Q, COMPTON R G, CLARKSON J. Ultrasonically enhanced leaching: Removal and destruction of cyanide and other ions from used carbon cathodes [J]. Ultrasonics Sonochemistry, 2000, 7(1): 1-6.
[21] MAURER A, FRIMMEL F H, EITEL W. Comparative assessment of accelerated leaching methods for on-Site verification testing of special waste [J]. Field Analytical Chemistry and Technology, 1998, 2(4): 195-203.
[22] BEVILAQUA D, LEITE A L L C, GARCIA O, TUOVINEN O H. Oxidation of chalcopyrite by Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans in shake flasks [J]. Process Biochemistry, 2002, 38(4): 587-592.
[23] SATERLAY A J, WILKINS S J, COMPTON R G. Towards greener disposal of waste cathode ray tubes ultrasonically enhanced lead leaching [J]. Green Chemistry, 2001, 3(4): 149-155.
[24] YU Run-lan, OU Yang, TAN Jian-xi, WU Fa-deng, SUN Jing, MIAO Lei, ZHONG Dai-li. Effect of EPS on adhesion of Acidithiobacillus ferrooxidans on chalcopyrite and pyrite mineral surfaces [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(2): 407-412.
[25] SAND W, GEHRKE T, JOZSA P G, SCHIPPERS A. (Bio) chemistry of bacterial leaching��Direct vs. indirect bioleaching [J]. Hydrometallurgy, 2001, 59(2-3): 159-175.
���ü�1���� ��1����ѧ��1,2��������1,2���� ��1,2��������1,2���� ��3�������1,2����ΰ��1,2,3
1. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083��
2. ���ϴ�ѧ ����ұ��������ص�ʵ���ң���ɳ 410083��
3. CSIRO Process Science and Engineering, Box 312, Clayton South, Victoria 3169, Australia
ժ Ҫ��Ϊ̽����ͭ�������������������������ϵ��������ڿ������ĸ��������ó��������Ⱥ���������ȡ���������������������������48 ��C�����³�����һ����Ч�ķ�����ɨ��羵������ɫɢX��������(EDX)�����������������ͭ�������ڴ����ѷ�Ͱ��ݣ��������������������Щ�ѷ�Ͱ����С��о��������̻�ͭ������������Ͱ�������ĺ����仯�������������������ĺ����ڽ���ǰ10 dѸ��������֮��ά����һ���ȶ���ˮƽ�������������������ʱ����ӳ������ӣ��������ڽ������ڡ�
�ؼ��ʣ���ͭ�������������������ȡ�������������������
(Edited by Wei-ping CHEN)
Foundation item: Project (31200382) supported by the National Natural Science Foundation of China; Project (2013FJ4068) supported by the Planned Science and Technology Project of Hunan Province, China; Project supported by Australia CSIRO OCE Science Leader Grant
Corresponding author: Wei-min ZENG; Tel: +86-731-88879212; E-mail: zengweimin1024@sina.com
DOI: 10.1016/S1003-6326(16)64143-2

