���������ͭ����pHֵ����������������˾����ڰ��⵰�Ͱ�����ǵ�Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���2��
�������ߣ����حZ ������ ������ ���� ��ΰ�� ��ѧ�� �����
����ҳ�룺406 - 412
�ؼ��ʣ����⵰�ף���������������˾�����ͭ�����������pHֵ
Key words��extracellular protein; Acidithiobacillus ferrooxidans; chalcopyrite; bioleaching; pH value
ժ Ҫ���Ƚ��о����������ͭ����pHֵ����������������˾����ڰ��⵰�Ͱ�����ǵ�Ӱ�졣ʵ������������pHֵΪ1.0��1.5��2.0��2.5ʱ����ϸ����������Ӧ�ڡ����������ں��ȶ��ڣ�����������������ڵİ��⵰�����Ȱ���������࣬�෴��������Һ�е�����ϸ�����ڵİ��⵰�����Ȱ���������٣�����ϸ����Ҫͨ�����ڰ�����Ƕ����ǰ��⵰�����ֿ���߽�������Ũ�ȺͲ�����pH�Ȳ���������Һ����������ϸ�����ڵİ�����ǺͰ��⵰������Ҫ���������Һ��ȶ����ǿ����Խ���������Ũ�ȡ������ϸ����Ҫ��ͨ�����ڸ������������Կ����������������⵰�������������ϵͳ������-��ԭ��Ӧ������Ҫ���á�
Abstract: The effect of pH values on the extracellular protein and polysaccharide secretions of Acidithiobacillus ferrooxidans was comparatively investigated in different phases of bacterial growth during chalcopyrite bioleaching. The results indicate that the extracellular protein is always more than the extracellular polysaccharide secreted by attached cells on the chalcopyrite, on the contrary, and is always less than the extracellular polysaccharide secreted by free cells in the solution at bacterial adaptive phase, logarithmic phase and stationary phase whenever pH value is at 1.0, 1.5, 2.0 or 2.5; free cells are mainly through the secretion of extracellular polysaccharide rather than the extracellular protein to fight against disadvantageous solution environment, such as high concentration of metal ions and unsuitable pH solution; both amounts of polysaccharide and protein secreted by attached cells are mainly positively related to the solution acidity rather than the total concentration of soluble metal ions. The experimental results imply that bacteria are mainly through secreting more extracellular polysaccharide to fight against disadvantageous environment and the extracellular protein perhaps plays an important role in oxidation-reduction reactions in the bioleaching system.
Trans. Nonferrous Met. Soc. China 27(2017) 406-412
Zhao-jing YU1,2, Run-lan YU1,2, A-juan LIU1,2, Jing LIU1,2, Wei-min ZENG1,2, Xue-duan LIU1,2, Guan-zhou QIU1,2
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Biometallurgy of Ministry of Education, Central South University, Changsha 410083, China
Received 21 December 2015; accepted 8 September 2016
Abstract: The effect of pH values on the extracellular protein and polysaccharide secretions of Acidithiobacillus ferrooxidans was comparatively investigated in different phases of bacterial growth during chalcopyrite bioleaching. The results indicate that the extracellular protein is always more than the extracellular polysaccharide secreted by attached cells on the chalcopyrite, on the contrary, and is always less than the extracellular polysaccharide secreted by free cells in the solution at bacterial adaptive phase, logarithmic phase and stationary phase whenever pH value is at 1.0, 1.5, 2.0 or 2.5; free cells are mainly through the secretion of extracellular polysaccharide rather than the extracellular protein to fight against disadvantageous solution environment, such as high concentration of metal ions and unsuitable pH solution; both amounts of polysaccharide and protein secreted by attached cells are mainly positively related to the solution acidity rather than the total concentration of soluble metal ions. The experimental results imply that bacteria are mainly through secreting more extracellular polysaccharide to fight against disadvantageous environment and the extracellular protein perhaps plays an important role in oxidation-reduction reactions in the bioleaching system.
Key words: extracellular protein; Acidithiobacillus ferrooxidans; chalcopyrite; bioleaching; pH value
1 Introduction
It is well known that bacteria can secret extracellular polymeric substances (EPSs), which consist of polysaccharides, proteins and lipids [1,2], and mediate the attachment of microorganisms to sulphide surfaces, gradually form a biofilm on the mineral surfaces during bioleaching [3-7]. The EPS plays an important role in the dissolution of sulfide minerals via an ��EPS contact-leaching mechanism�� [8]. But the EPS interface-action mechanism is sophisticated and has not been fully understood so far.
Some works have indicated that the presence of EPS layer on the cell is an important factor for bacterial adhesion to sulfide minerals. A decrease of attachment of Acidithiobacillus ferrooxidans to minerals is produced by the deficiency of EPS, which can recover mostly when the EPS is re-added into the EPS-free cells. The extent of cell adhesion to chalcopyrite increases when EPS and Fe3+ are added, and decreases when Fe2+ is added, which imply that the electrostatic interaction plays a main role in initial adhesion between bacteria and minerals [9,10]. EPS properties are changed with mineral types and bacterial types. The attachment of four strains of Acidithiobacillus ferrooxidans to pyrite, chalcopyrite, galena, sphalerite or quartz was found to be mineral- selective, and the chemical analyses of EPS of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans and Leptospirillum ferrooxidans indicated neutral sugars, fatty acids and uronic acids [10,11]. EPS properties also changed with the bacterial cultures. In contrast to cells of Acidithiobacillus ferrooxidans grown on sulphur, cells grown on pyrite or iron (II) sulphate incorporate uronic acids and ferric ion in their EPS [10,12]. Compared with Fe3+ ions, Cu2+ ions can stimulate bacteria to secret more EPS, especially, EPS produced by Acidithiobacillus ferrooxidans rapidly increases when the concentration of Cu2+ ions is more than bacterial tolerable concentration [13]. The polysaccharide components in EPS layer are variable with leaching time during bioleaching chalcopyrite [14].
The pH value plays an important role in the bioleaching of sulphide minerals [15]. The effect of pH value on the extracellular polysaccharide secreted by Acidithiobacillus ferrooxidans was investigated in different phases of bacterial growth during chalcopyrite bioleaching in our previous works [16]. It was found that extracellular polysaccharide secretion from the cells attached to chalcopyrite is more efficient than that from the free cells in the bioleaching solution whenever pH value is at 1.0, 1.5, 2.0 or 2.5. The extracellular polysaccharide secretion of the free cells at the logarithmic phase of bacterial growth is mainly related to the total concentration of soluble metal ions in the bioleaching solution. However, the extracellular polysaccharide secretion of the attached cells is mainly dependent on the pH value of the bacterial culture.
The extracellular protein is one of key components of EPS, but EPS components and properties are basically characterized through analyzing the extracellular polysaccharides during bioleaching in previous EPS works mentioned above, there are a few researches about the change of extracellular protein in EPS during bioleaching. At the same time, bacteria maybe display different behaviors between secreting extracellular protein and secreting extracellular polysaccharide during chalcopyrite bioleaching.
The aim of the current work was to examine the influence of controlled pH values on the extracellular protein secreted by Acidithiobacillus ferrooxidans (ATCC23270), and to study the difference between extracellular protein and polysaccharide secretions of Acidithiobacillus ferrooxidans under the conditions of different pH values during chalcopyrite bioleaching.
2 Experimental
2.1 Microorganisms and mass cultivation
Acidithiobacillus ferrooxidans ATCC23270 were obtained from Key Laboratory of Biometallurgy of Ministry of Education, China. The microorganisms were cultivated and activated in a modified 9K medium with initial pH 2.0 at 30 ��C in an orbital shaker. The mass bacteria were harvested by centrifugation at 12000g for 20 min. The modified 9K medium consisted of the following compounds: 3.0 g/L (NH4)2SO4, 2.1 g/L Na2SO4, 0.5 g/L MgSO4��7H2O, 0.1 g/L KCl, 0.05 g/L K2HPO4, 0.01 g/L Ca(NO3)2 and 30 g/L FeSO4��7H2O.
2.2 Mineral preparation and characteristics
Chalcopyrite, a floatation concentrate was obtained from Mengzhi Bainiu Mine in Yunnan Province, China. The chalcopyrite was prepared and characterized as described in Ref. [16]. The chalcopyrite ore sample was crushed, sieved, and the size fraction of with size ��75 ��m was used in the experiments. The XRD analysis showed that the mineral sample mainly consisted of 61.7% CuFeS2, 29.7% (Zn0.825Fe0.175)S, 3.7% PbSO4, and 1.1% PbCuAsS3. The chemical element analysis showed that the mineral was composed of 21.46% Cu, 35.39% S, 23.90% Fe, 8.40% Zn, 5.02% Pb, 0.08% Mg, 0.18% Ca, 0.20% Al, 0.088% Mn, and 5.28% Sn.
2.3 Bioleaching chalcopyrite under conditions of different controlled pH values
The bioleaching experiments were carried out in 500 mL Erlenmeyer flasks containing 300 mL modified 9K media without FeSO4��7H2O, and 2% chalcopyrite added as energy sources as described in Ref. [16]. Before bioleaching, these flasks were set four initial pH gradients for 1.0, 1.5, 2.0 and 2.5, respectively. All the Erlenmeyer flasks and their media were sterilized in a pressure steam sterilizer at 121 ��C for 20 min. Then, activated Acidithiobacillus ferrooxidans were inoculated into these flasks respectively at the density of about 5��107 cell/mL. All the bioleaching experiments were carried out in an orbital shaker, shaking at 180 r/min and 30 ��C. In order to maintain its pH value constant in every flask during the bioleaching experiment, its pH value in every flask was carefully adjusted and controlled into its initial pH value by adding sulfuric acid (50%) or sodium hydroxide (2 mol/L) at eight o��clock and twenty o��clock every day, the total time of adjusting pH twice every day is about 1 h. The evaporated water was compensated with distilled water. The concentrations of Cu2+, total iron, pH, redox potential and cell density were analyzed at regular intervals. Each set of the experiments was carried out in triplicate.
2.4 EPS extraction
The EPS of Acidithiobacillus ferrooxidans was extracted at the adaptive phase, the logarithmic phase, the maximum cells density phase and the decline phase according to bacterial growth curves in bioleaching chalcopyrite, respectively. When cells grew up to a definite stage, ore residue was separated in natural after laying the flask aside 2 h quietly. The EPS of free microorganisms in the leached solution was extracted through sonication method associated with centrifugation [9]. The EPS of the attached microorganisms on the leached ore residue was extracted by repeated vortex and elution [14].
The leached solution was treated by centrifuging at 10000g for 20 min to collect the free cells. Collected cells were washed and centrifuged two times using sterile iron-free 9K media with pH 2.0, and then these cells were re-suspended in 10 mL sterile iron-free 9K media, shaking at 30 ��C for 1 h. The sample was put in ice bath, ultrasonicating in JY92-II ultrasonator for 9 min (ultrasonic power of 80 W and work interval of 2 s) to strip the EPS from the free microorganisms, then centrifuged with 12000g at 4 ��C for 10 min (J-E Avanti centrifuge, Beckman Coulter Inc.). The extracted EPS samples of free cells were stored at -20 ��C until analysis [1].
6 g ore sample, 1.5 g glass beads with a diameter of 0.2 mm and 10 mL sterile water were shaken together on a vortexer (250 r/min) for 10 min. After this, the mixture was centrifuged at 3000g for 2 min to separate the ore residue. The ore residue was put back to continue vortex and elution until the microorganism in the supernatant cannot be seen from the microscope. Generally speaking, the procedure was repeated five times. The treated ore residue mentioned above was re-suspended in 10 mL sterile water, heated in water bath at 75 ��C for 30 min to stripe the cells on the ore completely. After being cooled, it was centrifuged at 8000g for 5 min. All of the supernatants were collected into centrifuge tube, and then centrifuged with 12000g at 4 ��C for 10 min to extract the EPS of the attached microorganisms. The EPS was also stored at -20 ��C until analysis.
2.5 Chemical analysis
Cell numbers were estimated by microscopic enumeration. The total concentration of the proteins of EPS was determined using BCA Protein Assay Kit (Shanghai Tiangen Corporation, China) and using BSA as protein standard solution through measuring the absorbance at 562 nm by UV-9200 spectrophotometer. The total concentration of the polysaccharides of EPS was determined using phenol-sulfuric acid method through measuring the absorbance at 490 nm by UV-9200 spectrophotometer [17]. Total soluble iron and Cu2+ ions in bioleaching solution were determined by BSH9-D atomic absorption spectrometer. 2-keto-3- deoxyoctonate (KDO) is part of the cell membrane in gram negative bacteria, which can be used as a marker for bacterial membrane break, and the detection of DNA concentration could give more information about the break extent of all bacteria during EPS extraction. Contamination caused by damaging cells as possible was checked by analysis for 2-keto-3-deoxyoctonate (KDO) [18] and DNA in EPS sample [14].

Fig. 1 Effect of pH value on contents of extracellular proteins secreted by free and attached bacteria during chalcopyrite bioleaching
3 Results and discussion
3.1 Effect of pH value on bacterial protein secretions during chalcopyrite bioleaching
Figure 1 shows the changes of the extracellular protein contents in EPSs secreted by free and attached bacteria with bacterial growth phases during chalcopyrite bioleaching under different conditions of controlled pH values. Obviously, whenever pH value is at 1.0, 1.5, 2.0 or 2.5, the protein content in EPS produced by attached cells is basically much more than that produced by free cells in the solution at bacterial adaptive phase, logarithmic phase and stationary phase. The free bacteria only produce small amount of extracellular protein. The extracellular protein contents per 1010 cells basically do not vary with bacterial growth phase at pH 1.0 and 2.5, but there is a maximal peak of extracellular protein content at logarithmic phase, pH 1.5 and 2.0, respectively. The extracellular protein content at logarithmic phase is about 0.2, 4.8, 0.7 and 0.08 mg per 1010 cells at pH 1.0, 1.5, 2.0 and 2.5, respectively. As the total concentrations of the copper ions plus iron ions in the logarithmic growth phase on day 10 of bioleaching are 0.857, 1.095, 0.979 and 0.642 g/L at pH 1.0, 1.5, 2.0 and 2.5, respectively, the extracellular protein content in EPS secreted by the free cells in the logarithmic growth phase is mainly related to the total concentration of soluble metal ions at pH 1.5, 2.0 and 2.5, except at pH 1.0. For the attached cells, the amount of DNA extracted from the ore residue was less than 0.2 mg/g, and the KDO content in extracted EPS was in the range of 0.03�C0.08 mg/g. So, the low amounts of DNA and KDO in extracted EPS indicated the negligible contamination of intracellular substances in the collected EPS. The changes in extracellular protein contents per 1010 cells of attached cells at bacterial adaptive phase, logarithmic phase and stationary phase with controlled pH values show the pattern as the following order: pH 1.0 >pH 1.5 > pH 2.0 > pH 2.5, and the protein content per 1010 cells basically decreases in proper order of bacterial adaptive phase, logarithmic phase, stationary phase and decline phase at 1.0 1.5 and 2.5, except at 2.0. There is an obviously maximal peak of extracellular protein at logarithmic phase and pH 2.0. The experimental results indicate that the extracellular protein secreted by attached cells is mainly positively related to the solution acidity.

Fig. 2 Effect of pH value on extracellular protein and polysaccharide contents in EPSs secreted by free bacteria during chalcopyrite bioleaching
3.2 Difference between extracellular protein and polysaccharide secretions during chalcopyrite bioleaching at different pH values
Figure 2 shows the differences between the extracellular protein and polysaccharide contents in EPSs secreted by free bacteria (solution bacteria) at different growth phases during chalcopyrite bioleaching under the conditions of different pH values. The extracellular protein content is basically less than the extracellular polysaccharide content secreted by free bacteria at bacterial adaptive phase, logarithmic phase and stationary phase, respectively. The changes of the bacterial extracellular protein and polysaccharide contents per 1010 cells with bacterial growth phase show similar change law at pH 1.5 and 2.0, except at pH 1.0 and 2.5 which are unsuitable environments for bacterial growth. The change in bacterial extracellular polysaccharide contents per 1010 cells at logarithmic phase with pH value shows the pattern as pH 1.5 >pH 2.0 > pH 1.0> pH 2.5. As the total concentrations of the copper ions plus iron ions in the logarithmic growth phase on day 10 of bioleaching are 1.095, 0.979, 0.857, and 0.642 g/L at pH 1.5, 2.0, 1.0, and 2.5, respectively, the bacterial extracellular polysaccharide content per 1010 cells secreted by the free cells at the logarithmic growth phase is positively related to the total concentration of metal ions in the bioleaching solution. The change in bacterial extracellular polysaccharide contents per 1010 cells at adaptive phase with pH value shows the pattern as pH 1.0 > pH 2.5 �� pH 1.5 > pH 2.0. As this is just the order of the unsuitable pH environment, free cells are through secreting more extracellular polysaccharides to fight against disadvantageous pH solution environments at the adaptive phase. According to the discussion mentioned in Section 3.1, the extracellular protein content in EPS secreted by the free cells in the logarithmic growth phase is mainly related to not only the total concentration of metal ions but also the solution pH value in the bioleaching solution. At adaptive phase, bacterial extracellular protein contents per 1010 cells at pH 1.0, 1.5, 2.0 and 2.5 are very small, and the change in bacterial extracellular protein content per 1010 cells with pH value is also different from that in bacterial extracellular polysaccharide content per 1010 cells with pH value. So, the experimental results imply that free cells are mainly through the secretion of extracellular polysaccharide rather than the extracellular protein to fight against disadvantageous solution environment, such as high concentration of metal ions and unsuitable pH solution.
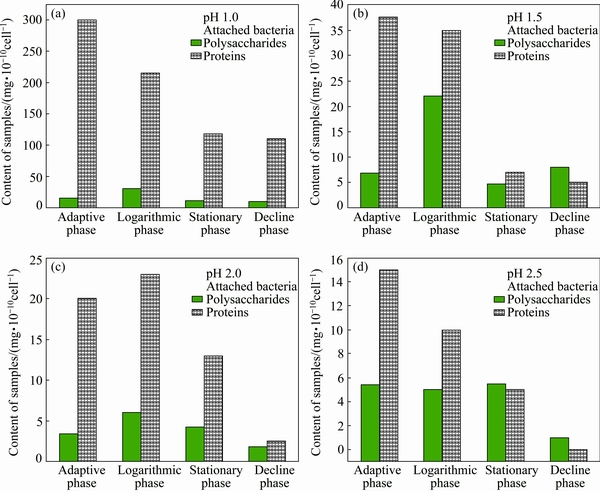
Fig. 3 Effect of pH value on extracellular protein and polysaccharide contents in EPSs secreted by attached bacteria during chalcopyrite bioleaching
Figure 3 shows the differences between the extracellular protein and polysaccharide contents in EPSs secreted by attached bacteria at different cell growth phases of bioleaching chalcopyrite under the conditions of different pH values. The polysaccharide and protein contents per 1010 cells in EPSs of attached cells in various growth phases all decrease with the increase in pH value as the following order: pH 1.0 > pH 1.5 > pH 2.0 > pH 2.5. The special phenomenon at pH 2.5 perhaps results from the hydrolysis precipitation of Fe3+ ions on the surface of chalcopyrite, which hinders the bacterial activity and bioleaching. The total concentrations of the copper ions plus iron ions in the logarithmic growth phase on day 10 of bioleaching are 1.095, 0.979, 0.857 and 0.642 g/L at pH 1.5, 2.0, 1.0 and 2.5, respectively. So, both polysaccharide and protein secreted by attached cells are mainly positively related to the solution acidity rather than the total concentration of solution metal ions. This result also indicates that EPS biofilm only binds up a very small amount of metal ions, which is in agreement with our previous works [13]. Interestingly, it is found that the extracellular protein is always more than the extracellular polysaccharide secreted by attached cells at bacterial adaptive phase, logarithmic phase and stationary phase; on the contrary, the extracellular protein is always less than the extracellular polysaccharide secreted by free cells at bacterial adaptive phase, logarithmic phase and stationary phase. KINZLER et al [12] thought that EPS can bind up more Fe3+ ions on the surface of sulfide mineral and it is helpful for bioleaching. But current experiments imply that extracellular protein perhaps plays a more important role in oxidation-reduction reactions in EPS biofilm.
4 Conclusions
1) The extracellular protein is always more than the extracellular polysaccharide secreted by attached cells on the chalcopyrite, on the contrary, is always less than the extracellular polysaccharide secreted by free cells in the solution at bacterial adaptive phase, logarithmic phase and stationary phase whenever is at 1.0, 1.5, 2.0 or 2.5.
2) Free cells are mainly through the secretion of extracellular polysaccharide rather than the extracellular protein to fight against disadvantageous solution environment, such as high concentration of metal ions and unsuitable pH solution.
3) Both polysaccharide and protein secreted by attached cells are mainly positively related to the solution acidity rather than the total concentration of soluble metal ions.
4) The extracellular protein perhaps plays a more important role in oxidation-reduction reactions in EPS biofilm.
References
[1] GEHRKE T, TELEGDI J, THIERRY D, SAND W. Importance of extracellular polymeric substances from Thiobacillus ferrooxidans for bioleaching [J]. Applied and Environmental Microbiology, 1998, 64(7): 2743-2747.
[2] FU Jian-Hua, QIU Guan-zhou, HU Yue-hua. The role of EPS of Thiobacillus ferrooxidans during bioleaching [J]. Acta Laser Biology Sinica, 2004,13(1): 62-64. (in Chinese)
[3] CRUNDWELL F. The formation of biofilms of iron-oxidising bacteria on pyrite [J]. Minerals Engineering, 1996, 9(10): 1081-1089.
[4] NIKOLOV L, KARAMANEV D, MAMATARKOVA V. Properties of the biofilm of Thiobacillus ferrooxidans formed in rotating biological contactor [J]. Biochemical Engineering Journal, 2002, 12(1): 43-48.
[5] FLEMMING H C, WINGENDER J. The biofilm matrix [J]. Nature Reviews: Microbiology, 2010, 8: 623-633.
[6] WANGNER M, NATALIA P, IVLEVA A, HAISCH C, NIESSNER R, HORN H. Combined use of confocal laser scanning microscopy (CLSM) and Raman microscopy (RM): Investigations on EPS �C Matrix [J]. Water Research, 2009, 43: 63-76.
[7] GARNY K, NEU T R, HORN H, VOLKE F, MANZ B. Combined application of 13C NMR spectroscopy and confocal lasers canning microscopy��Investigation on biofilm structure and physico- chemical properties [J]. Chemical Engineering Science, 2010, 65(16): 4691-4700.
[8] SAND W, GEHRKE H. Extracellular polymeric substances mediate bioleaching/biocorrosion via interfacial processes involving iron (��) ions and acidophilic bacteria [J]. Research in Microbiology, 2006, 157(1): 49-56.
[9] YU Run-lan, OU Yang, TAN Jian-xi, WU Fa-deng, SUN Jing, MIAO Lei, ZHONG Dai-li. Effect of EPS on adhesion of Acidithiobacillus ferrooxidans on chalcopyrite and pyrite mineral surfaces [J]. Transactions of Nonferrous Metals Society of China��2011, 21(2): 407-412.
[10] HARNEIT K, GOKSEL A, KOCK D, KLOCK J H, GEHRKE T, SAND W. Adhesion to metal sulfide surfaces by cells of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans and Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2006, 83(1-4): 245-254.
[11] ALBERTO S, OMAR T. Extracellular polymeric bacterial coverages as minimal area surfaces [J]. Journal of Colloid and Interface Science, 2006, 304(2): 554-557.
[12] KINZLER K, GEHRKE T, TELEGDI J, SAND W. Bioleaching��A result of interfacial processes caused by extracellular polymeric substances (EPS) [J]. Hydrometallurgy, 2003, 71(1-2): 83-88.
[13] YU Run-lan, LIU Jing, CHEN An, ZHONG Dai-li, LI Qian, QIN Wen-qing, QIU Guan-zhou, GU Guo-hua. Interaction mechanism of Cu2+, Fe2+ ions and extracellular polymeric substances during bioleaching chalcopyrite by Acidithiobacillus ferroxidans ATCC2370 [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(1): 231-234.
[14] ZENG Wei-min, QIU Guan-zhou, ZHOU Hong-bo, LIU Xue-duan, CHEN Miao, CHAO Wei-liang, ZHANG Cheng-gui, PENG Juan-hua. Characterization of extracellular polymeric substances extracted during the bioleaching of chalcopyrite concentrate [J]. Hydrometallurgy, 2010, 100(3-4): 177-180.
[15] YU Run-lan, SHI Li-juan, GU Guo-hua, ZHOU Dan, YOU Long, CHEN Miao, QIU Guan-zhou, ZENG Wei-min. The shift of microbial community under the adjustment of initial and processing pH during bioleaching of chalcopyrite concentrate by moderate thermophiles [J]. Bioresource Technology, 2014, 162(1): 300-307.
[16] YU Run-lan, LIU Jing, TAN Jian-xi, ZENG Wei-min, SHI Li-juan, GU Guo-hua, QIN Wen-qing, QIU Guan-zhou. Influence of controlled pH on extracellular polysaccharides secreted by Acidithiobacillus ferrooxidans during bioleaching chalcopyrite [J]. International Journal of Minerals, Metallurgy and Materials, 2014, 21(4): 311-316.
[17] YU Run-lan, ZHONG Dai-li, MIAO Lei, QIU Guan-zhou, GU Guo-hua. Relationship and effect of redox potential, jarosites and extrcellular polymeric substances in bioleaching chalcopyrite by Acidithiobacillus ferroxidans [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(7): 1634-1640.
[18] KARKHANIS Y D, ZELTNER J Y, JACKSON J J, CARLO D J. A new and improved microassay to determine 2-keto-3-deoxyoctonate in lipopolysaccharide of gram-negative bacteria [J]. Analytical Biochemistry, 1978, 85: 595-597.
���حZ1,2��������1,2��������1,2���� ��1,2����ΰ��1,2����ѧ��1,2�������1,2
1. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083��
2. ���ϴ�ѧ ����ұ��������ص�ʵ���ң���ɳ 410083
ժ Ҫ���Ƚ��о����������ͭ����pHֵ����������������˾����ڰ��⵰�Ͱ�����ǵ�Ӱ�졣ʵ������������pHֵΪ1.0��1.5��2.0��2.5ʱ����ϸ����������Ӧ�ڡ����������ں��ȶ��ڣ�����������������ڵİ��⵰�����Ȱ���������࣬�෴��������Һ�е�����ϸ�����ڵİ��⵰�����Ȱ���������٣�����ϸ����Ҫͨ�����ڰ�����Ƕ����ǰ��⵰�����ֿ���߽�������Ũ�ȺͲ�����pH�Ȳ���������Һ����������ϸ�����ڵİ�����ǺͰ��⵰������Ҫ���������Һ��ȶ����ǿ����Խ���������Ũ�ȡ������ϸ����Ҫ��ͨ�����ڸ������������Կ����������������⵰�������������ϵͳ������-��ԭ��Ӧ������Ҫ���á�
�ؼ��ʣ����⵰�ף���������������˾�����ͭ�����������pHֵ
(Edited by Wei-ping CHEN)
Foundation item: Project (31200382) supported by the National Natural Science Foundation of China
Corresponding author: Run-lan YU; Tel: +86-731-88877472; E-mail: YRL715@sina.com
DOI: 10.1016/S1003-6326(17)60046-3

