J. Cent. South Univ. Technol. (2010) 17: 979-984
DOI: 10.1007/s11771-010-0587-0
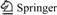
Combination of chlorine and magnetic ion exchange resin for drinking water treatment of algae
HAN Zhi-gang(��־��)1, 2, CHEN Wei(����)1, LI Lei(����)1, CAO Zhe(�܆�)1
1. College of Environmental Science and Engineering, Hohai University, Nanjing 210098, China;
2. Department of Environment and Municipal of Urban Planning and Design Institute,
Tsinghua University, Beijing 100084, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: The effectiveness of a magnetic ion exchange resin (MIEX) for the treatment of Hongze Lake water in China was evaluated. The kinetics of natural organic matter (NOM) removal at various MIEX doses and contact time, multiple-loading experiments, impacts of MIEX prior to coagulation on coagulant demands and the effectiveness of combination of MIEX, pre-chlorination and coagulation were investigated. Kinetic experimental results show that more than 80% UV254 and 67% dissolved organic carbon (DOC) from raw water can be removed by the use of MIEX alone. 94% sulfate, 69% nitrate and 98% bromide removals are obtained after the first use of MIEX in multiple-loading experiments. It is suggested that MIEX can be loaded up to 1 250 bed volume (BV, volume ratio of tested water to resin) or more without saturation when regarding organics removal as a target. MIEX can remove organics to a greater extend than coagulation and lower the coagulant demand when combining with coagulation. Chlorination experimental results show that MIEX can remove 57% chlorine demand and 77% trihalomethane formation potential (THMFP) for raw water. Pre-chlorination followed by MIEX and coagulation can give additional organic and THMFP removals. The results suggest that MIEX provides a new method to solve the problem algae reproduction.
Key words: drinking water treatment; magnetic ion exchange resin; natural organic matter; pre-chlorination; disinfection by- products; algae
1 Introduction
In recent years, algae outbreak has received much attention in drinking water treatment. Algae cells and algae-derived organic matter (AOM) are the main components of natural organic matter (NOM), which can be responsible for microbial re-growth in the distribution network and lead to the formation of disinfection by- products (DBPs) by reacting with chemical disinfectants [1-3]. Many studies demonstrated that a relatively large part of NOM could not be removed by conventional process [4-5]. Hence, many alternative technologies for NOM removal were considered. Among all of the alternative technologies, ion exchange technologies were received significant attention.
Magnetic ion exchange resin (MIEX) is a new ion exchange resin jointly developed by the Australian Water Quality Centre, CSIRO and Orica Australian, specifically for the removal of NOM in drinking water treatment. The small particle size of the resin, around 180 ?m, provides a high surface area, allowing rapid adsorption of NOM. The magnetic part of the resin allows the resin to agglomerate into larger settling particles, which shows a recovery rate of more than 99.9%. Any resin carried over is removed in downstream processes [5-6].
MIEX shows the effectiveness for the removal of NOM and hence reduces the formation of DBPs. MIEX can remove 25%-90% dissolved organic carbon (DOC) and 55%-95% UV254 from different raw waters [7-11]. Many investigations show that MIEX has the potential to remove more DOC and UV254 than coagulation, and can remove NOM with a wider range of relative molecular mass (RMM) and organic acid fractions than coagulation. The combination of MIEX and coagulation shows little additional NOM removal, and the combined process can reduce the coagulant demand by 40%-60% compared with coagulation alone when regarding NOM and turbidity removal as targets [5-6, 8-10]. Some researches show that MIEX is more effective for removing NOM from water with a high specific UV254 absorbance (SUVA, the ratio of UV absorbance at 254 nm to DOC times 100) ��3.0 L/(mg?m) DOC, which is characterized by hydrophobic NOM and low ionic strength, while MIEX is also shown to be able to remove hydrophilic NOM [7, 10-11]. The NOM fractions analysis of RMM shows that very large RMM organics (��5 000), which can be removed by coagulation, cannot be removed by MIEX, and RMM organics with RMM between 500 and 1 500 can be well removed by MIEX [4-5, 12]. MIEX treatment and coagulation are complementary treatment processes. A further benefit of MIEX is that it can remove bromide [7-8, 11-13]. JOHNSON and SINGER [11] showed that bromide removal by MIEX increased with decreasing alkalinity and initial bromide concentration. It is found that MIEX pretreatment improves the permeability of the membranes (UF and MF) and reduces the fouling of the membranes [14-15]. MIEX pretreatment can prevent the pore-blockage of powdered activated carbon (PAC). MIEX and PAC treatments are also complementary treatment processes [16].
Enhanced coagulation is shown to remove organic matter effectively. Hence, trihalomethane formation potential (THMFP) and haloacetic acid formation potential (HAAFP) are reduced [17]. But high coagulant dose required in enhanced coagulation process contributes to the high alum or iron content in water distribution system and high cost. Pre-chlorination process is a conventional and widely applied pre-oxidation technique for algae removal, but chlorination of algal-laden water can produce DBPs. Although published studies and installed water treatment plant (WTP) of MIEX show the benefit of using MIEX for removing NOM and reducing DBPs, few researches were carried out on the effectiveness of combination of MIEX and pre-chlorination for the treatment of algal-laden water, which is usually treated by pre-chlorination and enhanced coagulation.
Increasing pollution of Hongze Lake located in Huaian, Jiangsu Province, China, causes severe rise in the concentration of organic and inorganic compounds. Especially, the raw water is subject to elevated algae counts during summer months, which introduces process problems. The aim of this work was to evaluate the removal effectiveness of NOM and DBPs by MIEX on the algal-laden lake water. Besides, effects of MIEX on coagulation of turbidity and combination of MIEX and chlorine were also investigated.
2 Experimental
2.1 Raw water
Raw water was collected from Hongze Lake, China. Its main characteristics are listed in Table 1.
2.2 Experimental approach
General experimental approach of the combination of pre-chlorination and MIEX treatments is shown in Fig.1. Preliminary coagulation experiments were conducted to determine the optimal poly-aluminum chloride (PAC) dose for turbidity removal of raw water. Raw water was treated by MIEX to determine an optimal MIEX dose and mixing time for UV254 removal. Then, raw water was batch-treated at this optimal MIEX dose and mixing time. Water produced by MIEX jar tests was coagulated by PAC to determine an optimal PAC dose for turbidity removal, which was compared with the optimal PAC dose for raw water.
Table 1 Main characteristics of raw water

Fig.1 General experimental approach of combination of pre- chlorination and MIEX
In this experiment, pre-chlorination of raw water was performed before MIEX and PAC treatment to investigate the effectiveness of combination of pre-chlorination and MIEX processes. THMFP was then determined following chlorination to show the effect of MIEX process on the formation of DBPs.
2.3 MIEX treatment
In the first phase of this work, kinetic tests were conducted. In kinetic tests, raw water was placed on a jar test mixer (Shenzhen Zhongrun Co., China), dosed with MIEX (2, 6, 8, and 10 mL/L), and mixed at 200 r/min for 40 min. Samples were withdrawn at determined time intervals (5, 10, 15, 20, 25, 30, 35, and 40 min) for UV254 measurements.
Multiple-loading jar tests were conducted to estimate the performance of a continuously operated MIEX process based on successive jar tests with the same resin being used 10 times without regeneration. The resin dose and contact time for multiple-loading jar tests were 8 mL/L and 15 min selected from kinetic tests. The multiple-loading jar tests represented a loading of bed volume (BV, volume ratio of treated water to resin) 125-1 250. Samples were transferred to a tank for subsequent coagulation experiments and also taken for UV254, DOC, sulfate (SO42-), nitrate (NO3-), bromide (Br-), chlorination and THMFP measurements.
2.4 Coagulation treatment
PAC solution was used to coagulate raw water and MIEX-treated water (1 250 BV composite sample). The optimal PAC dose was defined as the PAC dose beyond which DOC and UV254 were no longer appreciably removed with increasing PAC dose, provided that turbidity was less than 2 NTU. After coagulation, samples from the supernatant were taken for UV254, DOC, turbidity, chlorination and THMFP measurements.
2.5 Pre-chlorination
A 5% NaOCl solution was used for all the chlorination experiments. Chlorine dose and reaction time were determined from raw water characteristics, such as the quantity of algae and TOC. According to a water plant, of which raw water was from Hongze Lake, chlorine dose and chlorination time in pre-chlorination process were 2 mg/L and 30 min, respectively. The dose and time were also chosen in this experiment. After pre-chlorination, chlorinated water was treated with MIEX and PAC by the same methods as mentioned above. Then, samples were taken for UV254, DOC, turbidity, chlorination and THMFP measurements.
2.6 Analytical methods
A spectrophotometer (Hach Co., USA) was used to measure UV254 absorbance. DOC measurement was performed by the high temperature combustion method using a MultiN/C2100 TOC analyzer. Turbidity and pH were measured by a 2100P turbidity meter (Hach Co., USA) and pH meter (Hach Co., USA), respectively. Concentrations of sulfate, nitrate and bromide were analyzed on a ZIC-2 ion chromatograph system (Qingdao Puren Co., China). A chlorine pocket colorimeter (Hach Co., USA) was used to measure chlorine residuals.
Chlorine demand was measured under uniform formation conditions (UFC) [18]. The conditions were as follows: 1 mg/L free chlorine residual, pH 8, and incubation in the dark at 20 �� for 24 h. After determining the chlorine demand, water samples were chlorinated by excessive chlorine and stored in refrigerator (4 ��) for 14 d. Then, the samples were taken for THMs analysis. Chloroform (CHCl3), dichlorobromomethane (CHCl2Br), chlorodibromo- methane (CHClBr2) and bromoform (CHBr3) were measured by a gas chromatopraph (Shimadzu Co., Japan) with electron capture detector (ECD).
3 Results and discussion
3.1 Removal of organics and anions with MIEX
UV254 removal at various MIEX doses is depicted in Fig.2. UV254 decreases with increasing MIEX dose and contact time. UV254 removal of up to 85.5% is achieved at the maximum dose (10 mL/L) and contact time of 35 min. The use of a relatively high MIEX dose (8 mL/L) and a relatively short contact time (15 min) shows that 80% UV254 removal is achievable compared with 38% UV254 removal for a low MIEX dose of 2 mL/L. The majority of UV254 removal occurs in the first 15 min and the removal rate increases by less than 3% when MIEX dose increases from 8 to 10 mL/L. According to the kinetic test results, 8 mL/L and 15 min are chosen as the optimal dose and time in the following experiments, respectively.
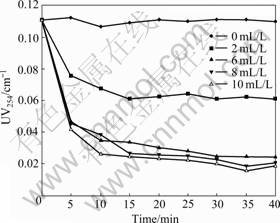
Fig.2 Impact of MIEX dose and contact time on UV254 removal
Fig.3 illustrates removal efficiencies UV254, DOC, sulfate, nitrate and bromide in multiple-loading jar tests. UV254 removal efficiency remains around 72% consistently after consecutive uses of MIEX with a drop from 81% after the first use of the resin to 70% after cycles of 10. A similar pattern is seen for DOC removal efficiency with a slight drop in the removal efficiency of DOC from 67% after the first use to 51% after cycles of 10. The removal behaviors of sulfate, nitrate and bromide
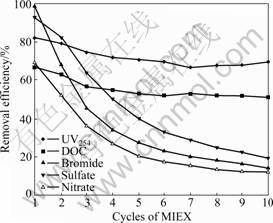
Fig.3 Impact of MIEX cycles on organics and inorganic compounds removal efficiencies
are different. The removal efficiencies of sulfate, nitrate and bromide are initially 94%, 69% and 98%, and quickly drop to only 19%, 12% and 14%, respectively.
It is demonstrated that MIEX can be loaded up to 1 250 BV or more without saturation with target organics removal achieved. This is advantageous in terms of the amount and the frequency of resin regeneration required. Based on the results, MIEX can remove sulfate, nitrate and bromide effectively, but it cannot be loaded up to more than 300 BV. Different behaviors of organics and anions removals in multiple-loading jar tests indicate that MIEX can provide more exchange sites for organics than for anions. The removal of bromide by MIEX is significant as brominated DBPs are believed to be harmful to public health. It is also suggested that anion compounds may compete with organics for exchange sites on the MIEX resin [9]. The conductivity of Hongze Lake water is 48 mS/m, which shows a high anionic strength. But it does not influence the removal of organics obviously, which does not correspond very well to the results in Refs.[9, 11]. Some researches show that algal-laden water contains high concentrations of algogenic matters and most of these compounds tend to be uncharged and have no affinity for the exchange onto the resin [4, 19]. However, the dominant algae in Hongze Lake water are chlorophyta and the algal-derived extra-cellular organic matters (EOM) of them are most acids being typical of charged small RMM matters that have strong affinity with MIEX. Humic and fulvic acids, which can be removed easily by MIEX, are also the main parts of NOM in raw water. So, MIEX can remove most of NOMs which can be removed from this water.
3.2 Combination of MIEX and PAC
Effects of MIEX pre-treatment on coagulation were evaluated in terms of turbidity, DOC and UV254 removal. Turbidity of the coagulated water treated with and without MIEX is shown in Fig.4. As shown in Fig.4, PAC demands, to achieve turbidity level of 2 NTU with
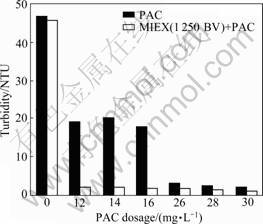
Fig.4 Impact of coagulation on turbidity for water treated with and without MIEX
and without MIEX pretreatment, are 14 and 30 mg/L, respectively. MIEX pre-treatment allows a coagulation dose reduction of 60% depending on turbidity removal. This is consistent with many other studies.
Furthermore, the use of MIEX exhibits a greater NOM removal efficiency than coagulation alone (Fig.5). When MIEX is employed alone, approximately 78% UV254 absorbing material is removed and 65% DOC is removed. When an optimal PAC dose is added, the UV254 and DOC removal efficiencies are improved to 86% and 80%, respectively. The RMM distribution results indicate that MIEX-coagulation process removes a broad range of NOM, while PAC coagulation preferentially removes the highest apparent RMM NOM fractions [5-6, 10]. Low RMM fractions composed of hydrophilic compounds are the most recalcitrant fractions to be treated by coagulation [20]. Recalcitrant fractions are also shown to impact the effectiveness of coagulation and increase coagulant demands. It is also shown that MIEX appears to be more suitable for removing middle and low RMM fractions of NOM and less effective for the high RMM fractions [5, 10]. So, the combination of MIEX and PAC
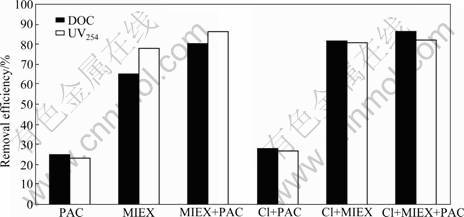
Fig.5 DOC and UV254 removal efficiencies with different treatments options
can improve NOM removals, which also leads to PAC demand reduction.
It is observed that in MIEX-coagulation trials the flocs formed are bigger and easier to settle than those in coagulation trails. PAC demand reduction is also significant for chemical sludge disposal and associated environmental impacts.
3.3 Pre-chlorination and disinfection by-products
Pre-chlorination is considered as an effective method to aid the coagulation of algae-rich water. The effect of pre-chlorination on NOM removal is also shown in Fig.5. Chlorine has distinct effect on the surface structure of algae cell, which leads to the release of cellular organic compounds [21]. Therefore, combined treatment by pre-chlorination and coagulation is effective for removing NOM. The removal efficiencies of DOC and UV254 with combined treatment are increased by 2.6% and 3.5%, respectively. The combination of MIEX and pre-chlorination increases DOC removal efficiency by 16.2% as compared with MIEX alone, but UV254 removal efficiency is not increased in combined treatment of MIEX and pre-chlorination. Similar phenomenon is found in combined treatment of pre-chlorination, MIEX and coagulation. After pre-chlorination, DOC removal efficiency is increased by 6%, whereas UV254 removal efficiency is reduced by 4%. A possible explanation for this phenomenon is that during the chlorination reactions, some high RMM DOC fractions that cannot be characterized by using UV254 detection are converted to low RMM organics, which can be easily removed with MIEX.
Chlorine demand was estimated by UFC method, which provided an indication on the level of NOM contributing to the formation of DBPs in water samples. Fig.6 presents the chlorine demands of raw water and water treated with MIEX and PAC. Treatment with MIEX reduces chlorine demand of the raw water to a greater extent than coagulation. Coagulation decreases 13% of the chlorine demand, whereas MIEX decreases 57% of the chlorine demand. Coagulation of MIEX-treated water shows 3% additional chlorine demand reduction. More organics removal will lead to more chlorine demand reduction. The results of chlorine demand are consistent with organics removal.
Fig.6 also illustrates the impact of pre-chlorination on the chlorine demand. Pre-chlorinated water consumes less chlorine than water without pre-chlorination. Chlorine demand of raw water can be reduced from 7.7 to 6.0 mg/L by the treatment with pre-chlorination. The combination of pre-chlorination and coagulation decreases chlorine demand by 27% compared with that treated with coagulation alone. The combination of pre-chlorination and MIEX decreases chlorine demand by only about 3% compared with that without pre-chlorination. The results indicate that MIEX can remove the compounds that consume more chlorine than other portions of NOM, which leads to the reduction of DBPs formation potential.
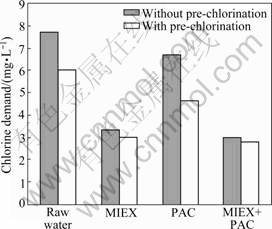
Fig.6 Chlorine demands for water with different treatment options
The concentrations of DBPs and THMFP in different water samples under UFC test are presented in Table 2.
The formation and species of DBPs depend on water quality parameters. THMFP of raw water is composed of 20.8% CHCl3, 61.1% CHCl2Br, 9.5% CHClBr2 and 8.6% CHBr3, which indicates that most NOMs in raw water are the precursors of CHCl2Br and CHCl3. Low bromide concentration of raw water also contributes to less CHBr3 and CHClBr2 formation than CHCl2Br and CHCl3 formation. As shown in Table 2,
Table 2 Concentrations of DBPs and THMFP in different water samples with different treatments (��g/L)
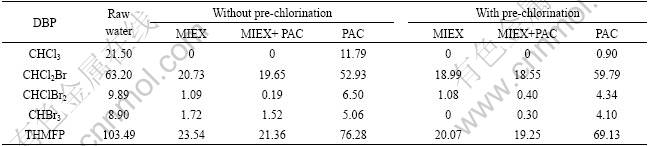
coagulation reduces THMFP by 26%, while MIEX removes 77% THMFP for raw water. Coagulation of MIEX-treated water provides only 2% additional THMFP removal efficiency. Similar change tendency of THMFP is shown in Table 2 when pre-chlorination is used. It is also found that MIEX preferentially removes CHCl3, while pre-chlorination preferentially increases removal efficiencies of CHBr3 and CHClBr2. Removal efficiency of THMFP can be improved by only 2% when MIEX is combined with pre-chlorination, which indicates that MIEX can remove DBPs precursors effectively.
4 Conclusions
(1) MIEX can remove organics and anions effectively at a low dose and short contact time. However, MIEX provides more exchange sites for organics than for anions.
(2) MIEX pre-treatment can reduce coagulant demand and increase organics and turbidity removals. The combination of MIEX and coagulation is also significant for environmental impacts and cost savings.
(3) Chlorination experimental results show that MIEX can remove the compounds that consume more chlorine than other portion of NOM effectively, hence reducing DBPs. Pre-chlorination only improves removal efficiencies of organics and THMFP slightly when it is combined with MIEX, which indicates that it is unnecessary to use pre-chlorination in the application of MIEX for Hongze Lake water treatment.
(4) The findings of this research suggest that MIEX technology represents another tool for the potential use by water plant to treat algal-laden water, which is dominated by chlorophyta.
Acknowledgement
The authors would like to thank ORICA Watercare for providing MIEX resin and discussion.
References
[1] GRAHAM N J D, WARDLAW V E, PERRY R, JIANG J Q. The significance of algae as trihalomethane precursors [J]. Water Science and Technology, 1998, 37(2): 83-89.
[2] LEENHEER J A. Comprehensive assessment of precursors, diagenesis, and reactivity to water treatment of dissolved and colloidal organic matter [J]. Water Supply, 2004, 4(4): 1-9.
[3] PLUMMER J D, EDZWALD J K. Effect of ozone on algae as precursors for trihalomethane and haloacetic acid production [J]. Environment Science and Technology, 2001, 35(18): 3661-3668.
[4] ALLPIKE B P, HEITZ A, JOLL C A, KAGI R I. Size exclusion chromatography to characterize DOC removal in drinking water treatment [J]. Environment Science and Technology, 2005, 39(7): 2334-2342.
[5] FEARING D A, BANKS J, GUYETAND S, MONFORT E C, WILSON D, HILLIS P, CAMPBELL A T, PARSONS S A. Combination of ferric and MIEX? for the treatment of a humic rich water [J]. Water Research, 2004, 38(10): 2551-2558
[6] CHEN Wei, HAN Zhi-gang, LIU Cheng, XIE Cheng-ta, ZHOU Hua. Study on MIEX technology for controlling drinking water disinfection by-products [J]. China Water & Waster Water, 2009, 25(6): 14-18. (in Chinese)
[7] BOYER T H, SINGER P C. Bench-scale testing of a magnetic ion exchange resin for removal of disinfection by-product precursors [J]. Water Research, 2005, 39(7): 1265-1276.
[8] BOYER T H, SINGER P C. A pilot-scale evaluation of magnetic ion exchange treatment for removal of natural organic material and inorganic anions [J]. Water Research, 2006, 40(15): 2865-2876.
[9] KITIS M, HARMAN B I, YIGIT N O, BEYHAN M, NGUYEN H, ADAMS B. The removal of natural organic matter from selected Turkish source waters using magnetic ion exchange resin (MIEX?) [J]. Reactive and Functional Polymers, 2007, 67(12): 1495-1504.
[10] SINGER P C, BILYK K. Enhanced coagulation using a magnetic ion exchange resin [J]. Water Research, 2002, 36(16): 4009-4022.
[11] JOHNSON C J, SINGER P C. Impact of a magnetic ion exchange resin on ozone demand and bromate formation during drinking water treatment [J]. Water Research, 2004, 38(17): 3738-3750.
[12] MERGEN M R D, JEFFERSON B, PARSONS S A, JARVIS P. Magnetic ion-exchange resin treatment: Impact of water type and resin use [J]. Water Research, 2008, 42(8): 1977-1988.
[13] HUMBERT H, GALLARD H, SUTY H, CROUE J P. Performance of selected anion exchange resins for the treatment of a high DOC content surface water [J]. Water Research, 2005, 39(9): 1699-1708.
[14] HUMBERT H, GALLARD H, JACQUEMET V, CROUE J P. Combination of coagulation and ion exchange for the reduction of UF fouling properties of a high DOC content surface water [J]. Water Research, 2007, 41(17): 3803-3811.
[15] FABRIS R, LEE E K, CHOW C W K, CHEN V, DRIKAS M. Pre-treatments to reduce fouling of low pressure micro-filtration (MF) membranes [J]. Journal of Membrane Science, 2007, 289(1/2): 231-240.
[16] HUMBERT H, GALLARD H, SUTY H, CROUE J P. Natural organic matter (NOM) and pesticides removal using a combination of ion exchange resin and powdered activated carbon (PAC) [J]. Water Research, 2008, 42(6/7): 1635-1643.
[17] LIANG L, SINGER P C. Factors influencing the formation and relative distribution of haloacetic acids and trihalomethanes in drinking water [J]. Environment Science and Technology, 2003, 37(13): 2920-2929.
[18] SUMMERS R S, HOOPER S M, SHUKAIR H M, SOLARIK G, OWEN D. Assessing DBP yield: Uniform formation conditions [J]. Journal of the American Water Works Association, 1996, 88(6): 80-93.
[19] HER N, AMY G, PARK H R, SONG M. Characterizing algogenic organic matter (AOM) and evaluating associated NF membrane fouling [J]. Water Research, 2004, 38(6): 1427-1438.
[20] BOLTO B, DIXON D, ELDRIDGE R, KING S. Cationic polymer and clay or metal oxide combinations for natural organic matter removal [J]. Water Research, 2001, 35(11): 2669-2676.
[21] SUKENIK A, TELTCH B, WACHS A W, SHELEF G, NIR I, LEYANON D. Effect of oxidants on micro-algal flocculation [J]. Water Research, 1987, 21(5): 533-539.
(Edited by CHEN Wei-ping)
Foundation item: Project(2008ZX07421-002) supported by the Key National Science and Technology Project of China; Project(50638020) supported by the National Natural Science Foundation of China
Received date: 2009-12-30; Accepted date: 2010-03-29
Corresponding author: CHEN Wei, PhD, Professor; Tel: +86-25-83787618; E-mail: cw5826@hhu.edu.cn