Deep purification of As(V) in drinking water by silica gel loaded with FeOOH and MnO2
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2021���6��
�������ߣ���ͮ ��ΰ ʯͨɼ ���� ����
����ҳ�룺1692 - 1706
Key words��As(V) removal; adsorption; drinking water; composite material
Abstract: For deep purification of As(V) from drinking water by adsorption, two adsorbents S-FeOOH and S-MnO2 were successfully synthesized by loading FeOOH and MnO2 nanoparticles onto silica gel in situ. Characterization of the adsorbents implied that S-FeOOH and S-MnO2 with large particle size (diameter of 150-250 ��m) still had high specific surface areas (357.0 and 334.6 m2/g) due to their specific amorphous and porous structure. In batch experiments, the influences of pH, contact time, adsorbent dosage, and temperature on the adsorption were investigated. Comparing with other adsorbents reported, the synthesized adsorbents in this study, especially S-FeOOH, showed good performance for As(V) removal in a wide pH (2-12) and temperature (25-65 ��C) range. The residual As(V) concentration after S-FeOOH treatment was around 0.01 mg/L, which met the drinking water standard. The adsorption process followed the pseudo-second-order kinetic model, and the adsorption equilibrium was reached within 5 min. The equilibrium adsorption data of S-FeOOH can be well fitted by the Langmuir isotherm, while that of S-MnO2 followed Freundlich model, which indicated their different adsorption mechanisms. The results show that S-FeOOH is superior to S-MnO2 in eliminating As(V), and S-FeOOH could be used as a promising adsorbent for the deep purification of As(V) in drinking water.
Cite this article as: SHI Tong-shan, JIANG Feng, WANG Pan, YUE Tong, SUN Wei. Deep purification of As(V) in drinking water by silica gel loaded with FeOOH and MnO2 [J]. Journal of Central South University, 2021, 28(6): 1692-1706. DOI: https://doi.org/10.1007/s11771-021-4727-5.

J. Cent. South Univ. (2021) 28: 1692-1706
DOI: https://doi.org/10.1007/s11771-021-4727-5
SHI Tong-shan(ʯͨɼ)1, 2, JIANG Feng(����)1, 2, WANG Pan(����)1, 2, YUE Tong(��ͮ)1, 2, SUN Wei(��ΰ)1, 2
1. School of Resource Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Hunan Province for Clean and Efficient Utilization of Strategic Calcium-containing Mineral Resources, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Abstract: For deep purification of As(V) from drinking water by adsorption, two adsorbents S-FeOOH and S-MnO2 were successfully synthesized by loading FeOOH and MnO2 nanoparticles onto silica gel in situ. Characterization of the adsorbents implied that S-FeOOH and S-MnO2 with large particle size (diameter of 150-250 ��m) still had high specific surface areas (357.0 and 334.6 m2/g) due to their specific amorphous and porous structure. In batch experiments, the influences of pH, contact time, adsorbent dosage, and temperature on the adsorption were investigated. Comparing with other adsorbents reported, the synthesized adsorbents in this study, especially S-FeOOH, showed good performance for As(V) removal in a wide pH (2-12) and temperature (25-65 ��C) range. The residual As(V) concentration after S-FeOOH treatment was around 0.01 mg/L, which met the drinking water standard. The adsorption process followed the pseudo-second-order kinetic model, and the adsorption equilibrium was reached within 5 min. The equilibrium adsorption data of S-FeOOH can be well fitted by the Langmuir isotherm, while that of S-MnO2 followed Freundlich model, which indicated their different adsorption mechanisms. The results show that S-FeOOH is superior to S-MnO2 in eliminating As(V), and S-FeOOH could be used as a promising adsorbent for the deep purification of As(V) in drinking water.
Key words: As(V) removal; adsorption; drinking water; composite material
Cite this article as: SHI Tong-shan, JIANG Feng, WANG Pan, YUE Tong, SUN Wei. Deep purification of As(V) in drinking water by silica gel loaded with FeOOH and MnO2 [J]. Journal of Central South University, 2021, 28(6): 1692-1706. DOI: https://doi.org/10.1007/s11771-021-4727-5.
1 Introduction
Medically, arsenic is considered to be one of the most toxic and harmful pollution elements to human health. Long-term intake of low-concentration arsenic contaminated water may lead to kidney, liver and skin cancer [1]. So, we must strictly control the content of arsenic in water to protect the water environment and ensure the safety of drinking water. A much more stringent limit (10 ��g/L) provided by the World Health Organization (WHO) in 1993 has been adopted as the drinking water standard to minimize the harmful effects of arsenic on health [2]. However, with the development of social economy and industry, arsenic pollution in water has become one of the important environmental and social problems faced by many countries in the world; the major route of arsenic getting in the water has changed from the natural geological process to the human industrial process, due to a large amount of arsenious solid waste, waste water and smoke produced in the industry process, especially the mineral processing and smelting of ore resources. Harmless treatment of arsenic-bearing waste water has become a key issue of global concern. Therefore, the study of arsenic removal technology in drinking water is of great significance for protecting the ecological environment and people��s health.
In a typical aquatic environment, arsenic is primarily present in inorganic forms with arsenite (As(III)) and arsenate (As(V)) [3], and As(V) is more prevalent in oxygenated surface water while As(III) is found in anaerobic groundwater. As(III) is more toxic and harder to remove from aqueous environment than As(V). Therefore, As(III) is normally oxidized to As(V) in arsenic removal process, after which arsenic can be removed more easily using various treatment methods, such as adsorption [4], precipitation [5], ion exchange [6], reverse osmosis [7], and electro dialysis [8]. Adsorption is considered to be a simple, economical, and efficient method to remove arsenic pollution in aqueous solution [9-14]. However, the need to adjust the solution pH, the relatively low adsorption capacity, and the potential dissolution of adsorbents during the adsorption process limits the removal efficiency of arsenic from the aqueous solution. It is urgent to develop novel arsenic adsorbents to overcome these limitations, especially in the deep purification of drinking water.
It was widely reported that various materials, such as activated carbon, waste biomass sorbents, organic polymers, zeolite, clay, and more selective metal oxides, have been used as the adsorbents for arsenic removal [15-20]. The results of many studies revealed that the cost-effective and easily available iron/manganese (hydr)oxides were promising adsorbent materials, because they had strong affinities towards arsenic and high selectivity to arsenic in the adsorption process [21-26]. Therefore, more researches recently focused on removing arsenic with iron/manganese (hydr)oxides or based materials as adsorbents. In these effective adsorbents, iron (hydr)oxides include amorphous iron hydroxide, ferrihydrite, hematite, and goethite [27-30]; manganese oxides include nanostructured manganese oxide, alone or as iron-manganese binary oxide [31-33], are well-known for their ability to removal arsenic from aqueous system.
In order to increase the adsorption capacity, the iron/manganese (hydr)oxide materials were prepared with a micrometer scale, even nanoscale to extend the adsorption surface area. However, these fine particles tended to aggregate together due to their high surface activity energy, which decreased the effective adsorption area [14]. In addition, these fine adsorbent particles were hard to separate from aqueous solution in the recycle of adsorbents. By contrast, loading the adsorbents on the surface of large particle carriers leads the adsorbents to have better dispersibility and usability. Furthermore, the carriers may alter the adsorption performance of the loaded adsorbents through the electrostatic interaction or chemical bonding. Therefore, it is significative to develop the high-efficiency carrier material for improving arsenic removal performance of the iron/manganese (hydr)oxides adsorbents.
In this study, silica gel, which is widely used, cheap, easy to obtain, and has very good porosity and high active adsorption, was chosen as the carrier of FeOOH and MnO2 adsorbents for arsenic removal from drinking water. The adsorbents, S-FeOOH and S-MnO2 were synthesized through self-assembly and embedding FeOOH and MnO2 nanoparticles in silica gel in situ. These novel materials have been characterized and then studied for As(V) adsorption from the aqueous solution under different experimental conditions in detail, such as solution pH, contact time, amount of adsorbent and temperature.
2 Material and methods
2.1 Materials
Sodium arsenate (Na3AsO4) was purchased from Jinjinle Chemical Co., Ltd., China. The arsenic stock solution with concentration of 1 g/L was prepared with deionized water. The arsenic working solution was also prepared by diluting the arsenic solution with deionized water in subsequent experiments. Chemicals, which were analytical grade and used without further purification, such as sulfuric acid (H2SO4), hydrochloric acid (HCl), sodium hydroxide (NaOH), sodium silicate (Na2SiO3), ferric chloride (FeCl3), potassium permanganate (KMnO4), and manganese sulfate monohydrate (MnSO4H2O), were purchased from Sinopharm Chemical Reagent Co., Ltd. Deionized water was solvent in all solutions.
2.2 Adsorbents preparation
The adsorbents (S-FeOOH and S-MnO2) were prepared according to Ref. [34], which could modify the mechanical properties of the adsorbents produced. In brief, when Si(OH)4 prepared by reaction of sodium silicate solution with hydrochloric acid contacted with metal compounds, silica-metal surface complexes between Si and metal ions formed. The complexation could enhance the binding of ultrafine metal particles and hence increase the physical strength of the adsorbents produced. The Si/Fe and Si/Mn molar ratios of the adsorbents prepared in this experiment both were1:1.
2.2.1 Preparation of S-FeOOH
S-FeOOH was mainly produced by the following two reactions:
Na2SiO3+H2O+2HCl=Si(OH)4+2NaCl (1)
FeCl3+3NaOH=FeOOH��+H2O+3NaCl (2)
Firstly, mixing 100 mL sodium silicate solution (1 mol/L) with 300 mL sodium hydroxide solution (1 mol/L) in a stirred vessel. The 100 mL ferric salt solution (1 mol/L) was then added in the vessel under vigorously stirring conditions to precipitate gelatinous Si-Fe complexes. The pH of the precipitation process was controlled to the range of 3-10, which was adjusted with 5% HCl or 5% NaOH, and the final pH of the precipitation reaction should be maintained between 6.5 and 7.5, which ensured that the adsorbents were obtained under neutral conditions. After formation, the gelatinous complexed Si-Fe slurry went through the following four processing steps: continuously stirred for 2 h, allowed by age for 4 h, washed with deionized water, and dewatered by filtration or centrifugation. Then, the dewatered solids were granulated to the required size and shape by extrusion or other methods (In this study, spherical particles with the high specific surface area were chosen) and dried at temperature between 120 and 250 ��C for several hours to get final S-FeOOH.
2.2.2 Preparation of S-MnO2
The preparation principle of S-MnO2 was the same as S-FeOOH, which was prepared by Eq. (1) and the following chemical equation:
2KMnO4+3MnSO4+2H2O=5MnO2��+K2SO4+2H2SO4 (3)
Similarly, the preparation process remained, and just the ferric chloride solution (FeCl3) to the 100 mL potassium permanganate (0.4 mol/L) and 100 mL manganese sulfate monohydrate (0.6 mol/L) to precipitate gelatinous Si-Mn complexes changed.
2.3 Batch adsorption tests
The optimum amount of adsorbent and contact time to reach adsorption equilibrium was determined by batch experiments. Each dosage was tested in five 100 mL flasks with 50 mL of 40 mg/L As(V) solution. Under the action of magnetic stirrer, the mixtures of five flasks were stirred for 1, 3, 5, 7, and 10 min at 150 r/min, respectively. Experiments to determine the effect of solution pH on As(V) removal were performed by adding 5 g of S-FeOOH/ S-MnO2 into a serial of 100 mL flasks containing 50 mL of 40 mg/L As(V) solution. The pH was adjusted to desired values with 5% HCl and 5% NaOH. For these experiments, the initial pH was varied within the range 2-12 and the solution was stirred at room temperature at 150 r/min for 5 min. And the temperature experiments were carried out in an magnetic stirring water bath at 25, 35, 45, 55 and 65 ��C, respectively. At each temperature, 5 g of adsorbents and 50 mL As (V) solution with 40 mg/L concentration were stirred together in a 100 mL flask, stirred at pH of 6.0-7.5 for 5 min.
The kinetics of As(V) adsorption was studied by batch experiments with varying contact times. Herein, 5 g S-FeOOH/S-MnO2 was thoroughly mixed with 50 mL of 40 mg/L As(V) solution in 100 mL flasks. The mixtures were withdrawn at a period of time between 1 and 10 min and filtered. Then, the sample solution of residual As(V) concentration was analyzed at different contact time.
Adsorption isotherms studies were conducted, 5 g S-FeOOH/S-MnO2 and 50 mL As(V) solution with a concentration range of 10-1000 mg/L were added into 100 mL flasks at 25 ��C. The mixtures were carried out in magnetic stirring water bath for 5 min. And the specific amount of arsenic adsorbed was calculated from the following equation:
 (4)
(4)
where Qe (mg/g) is the equilibrium adsorption capacity; C0 and Ce (mg/L) are the initial and equilibrium concentration of the adsorbate in solution, respectively; V is the volume (mL) of the arsenic solution; m is the mass (mg) of adsorbent used in the experiments.
2.4 Characterization methods
The morphologies and structural features were analyzed using XRD (Parnacre X-ray diffractometer), EDS (EDS300 DME), and SEM (FEI Magellan 400L XHR field emission scanning electron microscope) techniques. Specific surface area and the micropore characteristics were determined from the adsorption isotherms for liquid nitrogen at 77.3 K using a BET surface analyzer (Quantachrome NovaWin instruments. version 11.02). The sample solution was filtered with 0.45 ��m membrane to analyze the residual As(V) concentration with an inductively coupled plasma mass spectrometry equipment (ICP�CMS: SPECTRO-Blue II). All samples were analyzed within one day of collection.
3 Results and discussion
3.1 Adsorbents characterization
The XRD patterns of S-FeOOH and S-MnO2 in Figure 1 show that there is a cladding peak at 2�� of 15��-30�� and no obvious crystalline peak at other angles. The phenomenon indicates that the FeOOH and MnO2 formed on silica gel with amorphous structure, without fixed crystal form. This is because the presence of silicates prevents their crystal growth. As the adsorbents, the crystalline forms of FeOOH and MnO2 were not required, because the specific surface area of the crystal is much smaller than that of amorphous or low-crystalline form at the same particle size[34]. In addition, the existence of FeOOH and MnO2 did not change the inherent properties of silica gel, so these two composites could be applied in all fields of silica gel application.

Figure 1 XRD patterns of S-FeOOH and S-MnO2
The morphologies of S-FeOOH and S-MnO2 were confirmed by the SEM and the results in Figure 2. It could be seen that the S-FeOOH and S-MnO2 were a spherical particle colloidal material with the diameter of 150-250 ��m, and FeOOH and MnO2 were evenly distributed on the surface of silica gel. From the EDS analysis in Figures 2(c)-(f) and Table 1, the results showed that there were iron and manganese on the surface and inner of silica gel. In other words, the effective combination of FeOOH and MnO2 with silica gel was further confirmed. Sometimes, EDS analysis needed to spray gold to enhance the conductivity of the sample, so the Pt peak appeared in the figure. Additionally, from Table 1, the content of manganese on silica gel was higher than that of iron, which indicated that the binding ability of MnO2 on silica gel could be better than that of FeOOH.
In order to compare the changes of specific surface area and pore size of silica gel before and after loading metal oxide materials, BET/BJH tests were carried out on pure silica gel, S-FeOOH, and S-MnO2. The results are shown in Figure 3 and Table 2. Figures 3(a)-(c) show that the adsorption isotherms at the relative pressure (p/p0) 0.03-0.10 of the three materials were straight lines with the great coefficient of correlation, indicating that they were multi-molecular layer adsorption, which meets the BET equation. The specific surface areas (Table 2) of pure silica gel, S-FeOOH and S-MnO2 were 339.6, 357.0 and 334.6 m2/g, respectively. In addition, the specific surface area of the prepared absorbents S-FeOOH and S-MnO2 were significantly greater than that of the pure FeOOH (261 m2/g) or MnO2 (117 m2/g) [35], which increased the contact probability of functional adsorption sites on the surface of FeOOH and MnO2 nanoparticles with As(V). Figure 3(d) shows the pore size distribution curves of the adsorbents based on the nitrogen equilibrium adsorption isotherm at 77.3 K. It shows that the pure silica gel has relatively uniform mesoporous with pore diameter at around 10 nm, while the both adsorbents have some macropores (40-80 nm). Because FeOOH/MnO2 particles were loaded on the pore and surface of silica gel, the surface of silica gel became rougher, the specific surface area of silica gel was enlarged and the pore size decreased.

Figure 2 SEM images of S-FeOOH (a) and S-MnO2 (b) and inner and surface EDS analysis of S-FeOOH and S-MnO2 (c-f)
Table 1 Inner and surface metal content of S-FeOOH and S-MnO2 by EDS

3.2 Effects of dosage and contact time
The effects of dosage and contact time were important parameters for arsenic removed by adsorption. In Figure 4(a), the equilibrated removal efficiency of As(V) for S-FeOOH rose from 70% to 95% with increasing dosage of S-FeOOH from 1 to 3 g, ascribing to the increase of adsorption sites and surface area. Further increasing the dosage, the promotion of As(V) removal efficiency slowed down due to the shortage of adsorbate in the solution [36]. Hence, the equilibrium removal efficiency of As(V)
was over 99.9% at the S-FeOOH dosage more than 5 g. Comparing with S-FeOOH, the S-MnO2 had a worse performance for As(V) removal at the same dosage. The maximum removal efficiency of As(V) was 93%-97% when the adsorbent dosage increased to 5 and 7 g. The residual concentrations of As(V) with the optimal absorbents dosage of 5 g at different adsorption time are shown in Figure 4(c). It could be seen that the adsorption equilibrium of As(V) on S-FeOOH and S-MnO2 occurred at around the contact time of 5 min. The equilibrium residual concentration of As(V) with S-FeOOH was 0.01 mg/L, meeting the drinking water standard (0.01 mg/L) of As(V), while it was still 1.04 mg/L for S-MnO2. Comparing with other adsorbents which has been studied, the time required for these two adsorbents to reach adsorption equilibrium was shorter [37-39], indicating the remarkable affinity of the prepared adsorbents with As(V) in aqueous solution.

Figure 3 Nitrogen adsorption-desorption isotherms (a-c) and corresponding pore-size distribution curves (d) for pure silica gel, S-FeOOH and S-MnO2
Table 2 Textural properties of pure silica gel, S-FeOOH, and S-MnO2 adsorbents

3.3 Effect of pH
In Figure 5, different from S-MnO2, the removal efficiency of As(V) for S-FeOOH was around 98% with pH range from 2 to 12. In other words, the As(V) adsorption capacity of S-FeOOH hardly depends on pH. Such excellent adsorption performance, stabilized and high-efficiency arsenic adsorption capacity in a wide pH range, had not been reported on the previous studies. Additionally, it indicated that the main As(V) removal mechanism of S-FeOOH is the ligand interchange between arsenic and iron oxyhydroxide, rather than the electrostatic attraction [40]. Since the pHpzc of amorphous FeOOH was about 8.0 [41], S-FeOOH had positive charge when pH<8.0. So the As(V) was removed by S-FeOOH through electrostatic adsorption under the alkaline condition, and S-FeOOH still had a certain adsorption capacity, which indicated that S-FeOOH interacted with As(V) through the specific adsorption of coordination complexation that As(V) was adsorbed to S-FeOOH surface to form an inner-sphere complex with S-FeOOH surface. Therefore, it could be concluded that in the whole process of adsorbing As(V) adsorption on S-FeOOH, besides electrostatic adsorption, ligand exchange adsorption was also involved.

Figure 4 Effects of dosage and contact time for As(V) adsorption efficiency on S-FeOOH (a) and S-MnO2 (b) at (25��0.5) ��C, residual concentration of As(V) in treated water (c)

Figure 5 Effect of pH on As (V) adsorption onto S-FeOOH and S-MnO2 at (25��0.5) ��C
For S-MnO2, the removal efficiency of As(V) was gradually decreased from 97.80% to 85.00% as the pH increased from 2 to 10. When the pH further increased to 12, the removal efficiency slumped to 25%. The variety of the above adsorption capacity of S-MnO2 to As(V) was attributed to the adsorption of As(V) on the surface of S-MnO2 belonging to electrostatic adsorption [42]. The high adsorption of As(V) under acidic and neutral conditions was due to the fact that the positively charged adsorbent surface sites were easily occupied by the negatively charged arsenate species as well as the lower concentration of OH- could less compete for active sites on the adsorbent. With alkalinity increasing, the positive charge of S-MnO2 surfaces gradually became negatively charged, the arsenate gradually dissociated and formed strong electrostatic repulsion between OH- and arsenate. Hence a sharply decrease of adsorption was seen over pH=10. Accordingly, the As(V) removal by S-MnO2 was more evidently dependent on pH than that by S-FeOOH and the greatest adsorption occurring under acidic conditions and decreased with higher pH.
3.4 Effect of temperature
Figure 6 depicts the adsorption efficiency of S-FeOOH and S-MnO2 to As(V) at different ambient temperatures (25-65 ��C). Whether S-FeOOH or S-MnO2, the removal efficiency of As(V) remained approximately constant at each temperature. The adsorption efficiency of S-FeOOH to As(V) was maintained at 100%, which was obviously higher than that of S-MnO2 (93.83%). This was consistent with the above results that S-FeOOH was better than S-MnO2 to the adsorption of As(V) under pH>2 conditions. Apparently, the adsorption properties of the two adsorbents were not affected by the temperature range set in this experiment, implying that these two adsorbents were relatively excellent heat-resistant adsorption materials.

Figure 6 Effect of temperature on As(V) adsorption onto S-FeOOH and S-MnO2 at 25-65 ��C
3.5 Adsorption kinetics
The study of adsorption kinetics was very important for the description of adsorption behavior of adsorbents. So, the pseudo-first-order kinetic model and the pseudo-second-order kinetic model were used to investigate the adsorption rate of As(V) adsorption on S-FeOOH and S-MnO2 and evaluate the dominant adsorption mechanism. Their linear forms were as follows:
 (5)
(5)
 (6)
(6)
where Qt (mg/g) is the adsorption capacity of the adsorbent at time t (min); K1 (min-1) and K2 (g/(mg��min)) are the adsorption rate constants of the pseudo-first-order adsorption and pseudo-second-order adsorption, respectively.
The kinetics simulation and kinetics parameters of As(V) adsorption on S-FeOOH and S-MnO2 are shown in Figure 7 and Table 3, respectively. As being indicated from R2 values in Table 3, the pseudo-second-order kinetic model gave a higher R2 value for the As(V) removal by S-FeOOH and S-MnO2(R2>0.999), and the Qe,cal value of the pseudo-first-order kinetic model was close to the Qe,exp, which further confirmed that the pseudo-second-order kinetic model fitted the kinetics data well. The k value indicated the relative adsorption rate of metal ions, which was relative to the diffusion rate of metal ions from the bulk solution to adsorbents [43]. The K values in Table 3 showed KS-MnO2��KS-FeOOH, indicating that As(V) is adsorbed by S-MnO2 and S-FeOOH at equal rates, which is consistent with the experiment result of contact time and dosage (they both only take 5 min to reach equilibrium adsorption). It could be concluded that the rate limiting step is chemisorption involving valence forces through the chemisorption involving valence forces through the sharing or exchange of electrons between adsorbents and As(V) [44, 45], indicating the dominant mechanism of chemisorptions involved in As(V) adsorption.Table 4 compares the adsorption rate of S-FeOOH with other similar reports. The K value represented the relative adsorption rate of metal ions on adsorbent. It is obvious that the K value of S-FeOOH is close to 21.92, much larger than other adsorption materials, and it takes only 5 min for the adsorption of As(V) on S-FeOOH to reach equilibrium which could be concluded from Figure 4. It is also inferred that S-FeOOH is more attractive to As(V) than other adsorbents in Table 4.

Figure 7 Pseudo-first-order kinetics model (a) and pseudo-second-order kinetics model (b) for adsorption of As(V) on S-FeOOH and S-MnO2, respectively
Table 3 Kinetics parameters for adsorption of As(V) by S-FeOOH and S-MnO2

Table 4 Comparison of As(V) adsorption rate for some related adsorbent materials

3.6 Adsorption isotherms
To obtain more information about the As(V) adsorption on S-FeOOH and S-MnO2, the isotherm models of Langmuir and Freundlich were used to describe the adsorption equilibrium data. The Langmuir isotherm model assumes that monolayer adsorption occuring on a homogeneous surface. When monolayer adsorption is completely saturated, there is no interaction between adsorbates. On the contrary, the Freundlich isotherm model describes the multilayer adsorption on a heterogeneous surface on which the adsorbates are interactive [47-50].
Langmuir and Freundlich isotherm equations are given as follows:
 (7)
(7)
 (8)
(8)
where Qmax (mg/g) and Qe (mg/L) are the maximum adsorption capacity and the mass of As(V) adsorbed at equilibrium, respectively; Ce (mg/L) is the As(V) equilibrium concentration; Kl (L/mg) and Kf (mg/g) are Langmuir constant and Freundlich constant, which indicate the attraction between As(V) and the adsorbents.
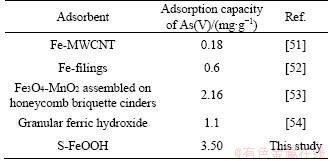
A non-linear regression was applied to obtaining all Langmuir and Freundlich isotherm parameters presented in Table 5. From Figure 8, the As(V) adsorption on S-FeOOH, Langmuir model (R2>0.946) had a better regression coefficient than Freundlich model (R2>0.943), which was similar to the result obtained by ZHANG et al [33]. And the adsorption data of S-MnO2 could be described well by the Freundlich model (R2>0.988) rather than Langmuir model (R2>0.964). In isotherm study, the saturated adsorption capacity (Qmax) was the practical maximum of S-FeOOH. However, the BET surface area analysis had determined that S-MnO2 had a lower specific surface area (334.6m2/g) than that of S-FeOOH (357 m2/g). And the pHpzc of SMnO2 was 4.8, which was lower than that of S-FeOOH (8.0). The surface charge of S-MnO2 was more negative at lower pH, and there was an electrostatic repulsion S-MnO2 and arsenate. So, the above two reasons could be eliminated. According to the isotherm adsorption results, it was known that S-FeOOH was monomolecular layer adsorption and S-MnO2 was multimolecular layer adsorption, which may explain why S-MnO2 had the larger adsorption capacity. It was obvious that Kf>Kl from Table 5, indicating that S-MnO2 might have a higher affinity for As(V) than S-FeOOH. In fact, the above result was not consistent with Figure 4(c), which may be due to the fact that the hypothesis of Langmuir model was far from the reality, and the information obtained had a certain amount of error. In addition, the adsorption capacity of S-FeOOH with other similar reports is compared in Table 6. It could be clearly obtained that S-FeOOH had a higher adsorption capacity than other adsorbents. Combined with the previous analysis, it is indicated that S-FeOOH was a promising adsorbent for the treatment of low concentration arsenic contaminated drinking water.
Table 5 Isotherm parameters for As(V) adsorption on S-FeOOH and S-MnO2


Figure 8 Adsorption isotherms for different concentrations of As(V) with S-FeOOH and S-MnO2
Table 6 Comparison of As(V) adsorption capacity for some related adsorbent materials
3.7 Adsorption thermodynamics
Through the study of adsorption thermodynamics, it could be understood the driving process of adsorption reaction, the degree of adsorption, and the effects of various factors on adsorption. So, S-FeOOH with better adsorption performance was selected for thermodynamic study. And the thermodynamic parameters of adsorption involve enthalpy (H), entropy (S), and Gibbs free energy (G).
These parameters can be obtained from the following equations:
 (9)
(9)
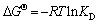 (10)
(10)
 (11)
(11)
 (12)
(12)
where KD is equilibrium adsorption partition coefficient, which is reliable to S-FeOOH when Ce<40 mg/L; R (8.314 J/(mol��K)) is gas constant; T (K) is temperature; ��G��, ��H��, and ��S�� are standard Gibbs free energy ��G (kJ/mol), standard enthalpy change, and standard entropy change, respectively.
The ��G parameter was calculated as -2.31, -3.62, -6.53 and -10.29 kJ/mol for 298, 308, 318, and 328 K in Table 7, respectively. In the temperatures range, the negative ��G indicated that the adsorption process was spontaneous. The absolute value of ��G increased with the increase of temperature. It was concluded that the net driving force and the degree of spontaneity gradually increased in adsorption process. The reasonable explanation was that the pore size on adsorbent surface increased or adsorbent surface was activated with the increasing temperature [55, 56]. As shown in Figure 9, the KD -1/T plot was used to determine ��H and ��S. These parameters were found to be 73.97 and 225.71 kJ/mol, respectively. ��H>0 indicated that the adsorption process was endothermic, which was confirmed by the increased KD values with increasing temperature. ��S>0 indicated that the adsorption reaction makes the whole system confusing. S-FeOOH adsorbed As(V) in water, which makes the degree of freedom of the whole system decrease and leads to the decrease of entropy. In adsorption process, some molecules would be separated from the system; the ��S of the system was the sum of the increase of entropy due to As(V) adsorption on S-FeOOH and the decrease of entropy of S-FeOOH surface analytical molecules.
Table 7 Thermodynamics parameters of S-FeOOH


Figure 9 lnKD-T-1 plot used in determination of thermodynamic parameters for adsorption of As(V) onto S-FeOOH
In this study, the value of entropy increase was greater than the value of entropy decrease in adsorption process, so the value of ��S was positive value [57].
3.8 Regeneration and wastewater treatment
In order to reduce the cost and recycled utilization of adsorbent, desorption experiments of S-FeOOH were conducted by mixing the As(V)-loaded S-FeOOH with different pH. On the premise that the structure of S-FeOOH was not destroyed, the result of desorption is shown in Figure 10.
In Figure 10, the desorption efficiency increased with the increase of pH, but decreased suddenly under strong alkali due to the fact that S-FeOOH was easily corroded under strong alkali. The desorption efficiency reached its maximum when pH was 10, but the desorption efficiency was also not ideal, only about 32.13%.

Figure 10 Desorption efficiency of S-FeOOH
So, another method was adopted to maximize utilization and minimize costs. It was generally known that FeOOH was soluble in strong acid, while silica gel was acid resistant. After mixing S-FeOOH which has been saturated for As(V) adsorption and 0.2 mol/L H2SO4 at the mass ratio of 1:30, the mixture was stirred at a constant speed (200 r/min). After 0.5 h, FeOOH was completely dissolved from silica gel and As(V) was desorbed simultaneously. Then, the filtered silica gel was cleaned by deionized water and dried at 60 ��C. FeOOH was loaded on silica gel by the same preparation method as new adsorbent which would be used in next round and their ability of As(V) treatment did not decrease. However, there was a high concentration of As(V) in the acid washing wastewater, which needed to be further treated to prevent secondary pollution. Solidification landfill was a common treatment method for arsenic contaminated wastewater. Based on many literatures, the lime, cement and ferrous sulfate were used as solidified agent and the solidification was conducted by mixing sludge, lime, cement, and ferrous sulfate with the mass ratio of 5:0.7:1:2 [57, 58].
The specific steps are shown in Figure 11. First, because the acid washing wastewater was arsenic-high iron wastewater (mainly Fe3+) and ferric iron was easy to hydrolyze, NaClO solution (VNaClO:Vwater=1:400) was added to promote its hydrolysis to form FeOOH colloid and adsorb arsenic to form sludge. Second, a certain amount of sludge and deionized water twice the weight of the sludge were mixed and stirred fully to the paste,adding the solidified agent prepared according to the proportion. Third, adding cement and a small amount of deionized water to the mixture of step 2 with stirring. After the sludge was fully mixed, it was poured into the mold, sealed, and placed in a cool and ventilated place for natural maintenance and air drying.

Figure 11 Flowchart of S-FeOOH regeneration and wastewater treatment
By comparing the arsenic content of the sludge before solidification and the leaching solution after solidification, it could be found that the As(V) in the solidified sludge leaching solution greatly decreased (6 ug/L), and the solidification rate was 99.6%, which ensured that the solidified sludge met the landfill requirements.
4 Conclusions
1) In this study, S-FeOOH and S-MnO2 are synthesized by metal (hydr)oxides formed in situ on silica gel as the adsorbents to adsorb As(V).
2) S-FeOOH and S-MnO2 both show excellent adsorption properties towards As(V). The adsorption is efficient, because the adsorption equilibrium could be achieved within 5 min. The adsorption capacities of S-FeOOH and S-MnO2 for As(V) remain stable in a wide pH range (2�C12), especially S-FeOOH, which is a limitation in the study of arsenic adsorption.And the adsorption performance is not easily affected by the solution temperature (25-65 ��C). In addition, there was no secondary pollution in adsorption process and the solid-liquid separation was easy to achieve.
3) From the practical point of view, S-FeOOH is more likely to be the promising adsorbent for the treatment of arsenic-contaminated natural water than S-MnO2, especially in the field of lower concentration, because S-FeOOH is more stable and efficient than S-MnO2 in As(V) adsorption.
Contributors
The overarching research goals were developed by YUE Tong and SUN Wei. SHI Tong-shan and JIANG Feng provided the test plan and test data, and the measured data were analyzed by SHI Tong-shan and WANG Pan. The initial draft of the manuscript was written by SHI Tong-shan and YUE Tong. All authors replied to reviewers�� comments and revised the final version.
Conflict of interest
SHI Tong-shan, JIANG Feng, WANG Pan, YUE Tong, and SUN Wei declare that they have no conflict of interest.
References
[1] BODDU V M, ABBURI K, TALBOTT J L, SMITHA E D, HASSCH R. Removal of arsenic (III) and arsenic (V) from aqueous medium using chitosan-coated biosorbent [J]. Water Research, 2008, 42(3): 633-642. DOI: 10.1016/j.watres.2007. 08.014.
[2] LUO Xu-biao, WANG Cheng-cheng, LUO Sheng-lian, DONG Rui-zhi, TU Xin-man, ZENG Gui-sheng. Adsorption of As (III) and As (V) from water using magnetite Fe3O4-reduced graphite oxide-MnO2 nanocomposites [J]. The Chemical Engineering Journal, 2012, 187(2): 45-52. DOI: 10.1016/j.cej.2012.01.073.
[3] CHEN Bo, ZHU Zhi-liang, MA Jie, QIN Yan-ling. Surfactant assisted Ce-Fe mixed oxide decorated multiwalled carbon nanotubes and their arsenic adsorption performance [J]. Journal of Materials Chemistry A, 2013, 1(37): 11355-11367. DOI: 10.1039/c3ta11827d.
[4] MARTINSON C A, REDDY K J. Adsorption of arsenic(III) and arsenic(V) by cupric oxide nanoparticles [J]. Journal of Colloid & Interface Science, 2009, 336(2): 406-411. DOI: 10.1016/j.jcis.2009.04.075.
[5] NISHIMURA T, UMETSU Y. Oxidative precipitation of arsenic (III) with manganese (II) and iron(II) in dilute acidic solution by ozone [J]. Hydrometallurgy, 2001, 62(2): 83-92. DOI: 10.1016/S0304-386X(01)00188-8.
[6] KIM J, BENJAMIN M M. Modeling a novel ion exchange process for arsenic and nitrate removal [J]. Water Research, 2004, 38(8): 2053-2062. DOI:10.1016/j.watres.2004.01.012.
[7] ABEJON A, GAREA A, LRABIEN A. Arsenic removal from drinking water by reverse osmosis: Minimization of costs and energy consumption [J]. Separation and Purification Technology, 2015, 144: 46-53. DOI: 10.1016/j.seppur. 2015.02.017.
[8] MENDOZA R, KAN C C, CHUANG S S, PINGUL-ONG S M B, DALIDA M L P, WAN Meng-wei. Feasibility studies on arsenic removal from aqueous solutions by electrodialysis [J]. Journal of Environmental Science & Health A, 2014, 49(5): 545-554. DOI: 10.1080/10934529.2014.859035.
[9] KUMAR A S K, JIANGS J. Chitosan-functionalized graphene oxide: A novel adsorbent an efficient adsorption of arsenic from aqueous solution [J]. Journal of Environmental Chemical Engineering, 2016, 4(2): 1698-1713. DOI: 10.1016/j.jece. 2016.02.035.
[10] SULLIVAN C, TYRER M, CHEESEMAN C R, GRAHAM N J D. Disposal of water treatment wastes containing arsenic��A review [J]. Science of the Total Environment, 2010, 408(8): 1770-1778. DOI: 10.1016/j.scitotenv.2010.01. 010.
[11] NGAH W S W, ENDUD C S, MAYANAR R. Removal of copper(II) ions from aqueous solution onto chitosan and cross-linked chitosan beads [J]. Reactive & Functional Polymers, 2002, 50(2):181-190. DOI: 10.1016/S1381-5148(01)00113-4.
[12] GUPTA K, BASU T, GHOSH U C. Sorption Characteristics of Arsenic(V) for removal from water using agglomerated nanostructure iron(III)-zirconium(IV) bimetal mixed oxide [J]. Journal of Chemical & Engineering Data, 2009, 54(8): 2222-2228. DOI:10.1021/je900282m.
[13] BISSEN M, FRIMMEL F H. Arsenic��A review. Part II: Oxidation of arsenic and its removal in water treatment [J]. Acta Hydrochimica et Hydrobiologica, 2003, 31(2): 97-107. DOI:10.1002/aheh.200300485.
[14] BUDIMIROVIC D, VELICKOVIC Z S, DJOKIC V R, MILOSAVLJEVIC M, MARKOVSKI J, LEVIC S, MARINKOVIC A D. Efficient As(V) removal by ��-FeOOH and ��-FeOOH/��-MnO2 embedded PEG-6-arm functionalized multiwall carbon nanotubes [J]. Chemical Engineering Research & Design, 2017, 119: 75-86. DOI:10.1016/j.cherd.2017.01.010.
[15] HUAN Ming, ZHANG Shu-juan, PAN Bing-cai, ZHANG Wei-ming, LV Lu, ZHANG Quan-xing. Heavy metal removal from water/wastewater by nanosized metal oxides: A review [J]. Journal of Hazardous Materials, 2012, 211-212: 317-331. DOI:10.1016/j.jhazmat.2011.10.016.
[16] PANDEY P K, CHOUBEY S, VERMA Y, PANDEY M, CHANDRASHEKHAR K. Biosorptive removal of arsenic from drinking water [J]. Bioresource Technology, 2009, 100(2): 634-637. DOI: 10.1016/j.biortech.2008.07.063.
[17] JACUKOWICZ-SOBALA I, OCINSKI D, KOCIOLEK-BALAWEJDER E. Iron and aluminium oxides containing industrial wastes as adsorbents of heavy metals: Application possibilities and limitations [J]. Waste Management & Research, 2015, 33(7): 612-629. DOI: 10.1177/0734242X 15584841.
[18] KYZAS G Z, MATIS K A. Nanoadsorbents for pollutants removal: A review [J]. Journal of Molecular Liquids, 2015, 203: 159-168. DOI:10.1016/j.molliq.2015.01.004.
[19] KURZ E E C, LUNOG V T, HELLRIEGEL U. Iron-based subsurface arsenic removal (SAR): Results of a long-term pilot-scale test in Vietnam [J]. Water Research, 2020, 181: 115929. DOI: 10.1016/j.watres.2020.115929.
[20] GILES D E, MOHAPATRA M, ISSA T B, ANAND S, SINGH P. Iron and aluminium based adsorption strategies for removing arsenic from water [J]. Journal of Environmental Management, 2011, 92(12): 3011-3022. DOI: 10.1016/ j.jenvman.2011.07.018.
[21] DIXIT S, HERIN J G. Comparison of arsenic(V) and arsenic(III) sorption onto iron oxide minerals:Implications for arsenic mobility [J]. Environmental Science & Technology, 2003, 37(18): 4182-4189. DOI:10.1021/es030309t.
[22] DAVRANCHE M, BOLLINGER J C. Heavy metals desorption from synthesized and natural iron and manganese oxyhydroxides:Effect of reductive conditions [J]. Journal of Colloid & Interface Science, 2000, 227(2): 531-539. DOI: 0.1006/jcis.2000.6904.
[23] OCINSKI D, JACUKOWICZ-SOBALA I, MAZUR P, RACZYK J. Water treatment residuals containing iron and manganese oxides for arsenic removal from water- Characterization of physicochemical properties and adsorption studies [J]. Chemical Engineering Journal, 2016, 294: 210-221. DOI:10.1016/j.cej.2016.02.111.
[24] MANGWAND C, SUHAIMI S N A, LIU J T, DHENGE R M, ALBADARIN A B. Design, production and characterisation of granular adsorbent material for arsenic removal from contaminated wastewater [J]. Chemical Engineering Research and Design, 2016, 110: 70-81. DOI: 10.1016/j.cherd.2016. 04.004.
[25] JANG M, MIN S H, KIM T H, PARK J K. Removal of arsenite and arsenateusing hydrous ferric oxideincorporated into naturallyoccurring porous diatomite [J]. Environmental Science & Technology, 2006, 40(5): 1636-1643. DOI: 10.1021/es051501t.
[26] MAHLER J, PERSSON I. Rapid adsorption of arsenic from aqueous solution by ferrihydrite-coated sand and granular ferric hydroxide [J]. Applied Geochemistry, 2013, 37: 179-189. DOI: 10.1016/j.apgeochem.2013.07.025.
[27] ZHANG Man, DAI Min, XIA Ling, SONG Shao-xian. Comparison of arsenic adsorption on goethite and amorphous ferric oxyhydroxide in water [J]. Water Air & Soil Pollution, 2017, 228(11): 427. DOI: 10.1007/s11270-017-3602-3.
[28] ANTELO J, ARCE F, FIOL S. Arsenate and phosphate adsorption on ferrihydrite nanoparticles. Synergetic interaction with calcium ions [J]. Chemical Geology, 2015, 410: 53-62. DOI: 10.1016/j.chemgeo.2015.06.011.
[29] WANG Sheng-sen, GAO Bin, ZIMMERMAN A R, LI Yun-cong, MA Le-na, HARRIS W G, MIGLIACCIO K W. Removal of arsenic by magnetic biochar prepared from pinewood and natural hematite [J]. Bioresource Technology, 2015, 175: 391-395. DOI: 10.1016/j.biortech.2014.10.104.
[30] DENG Ying-xuan, LI Yong-tao, LI Xiao-jing, SUN Yang, MA Jie, LEI Mei, WENG Li-ping. Influence of calcium and phosphate on pH dependency of arsenite and arsenate adsorption to goethite [J]. Chemosphere, 2018, 199(5): 617-624. DOI:10.1016/j.chemosphere.2018.02.018.
[31] THANH D N, SINGH M, ULBRICH P, STEPANEKB F, STRNADOVA N. As (V) removal from aqueous media using ��-MnO2 nanorods-impregnated laterite composite adsorbents [J]. Materials Research Bulletin, 2012, 47(1): 42-50. DOI: 10.1016/j.materresbull.2011.10.004.
[32] ZHANG Gao-sheng, QU Jiu-hui, LIU Hui-juan. Removal mechanism of As(III) by a novel Fe-Mn binary oxide adsorbent:Oxidation and sorption [J]. Environmental Science & Technology, 2007, 41(13): 4613-4619. DOI: 10.1021/es063010u.
[33] ZHANG Gao-sheng, LIU Hui-juan, LIU Rui-ping, QU Jiu-hui. Adsorption behavior and mechanism of arsenate at Fe-Mn binary oxide/water interface [J]. Journal of Hazardous Materials, 2009, 168(2, 3): 820-825. DOI: 10.1016/ j.jhazmat.2009.02.137.
[34] ZENG L. A method for preparing silica-containing iron(III) oxide adsorbents for arsenic removal [J]. Water Research, 2003, 37(18): 4351-4358. DOI: 10.1016/S0043-1354(03) 00402-0.
[35] XU Wei, LAN Hua-chun, WANG Hong-jie, LIU Hong-ming, QU Jiu-hui. Comparing the adsorption behaviors of Cd, Cu and Pb from water onto Fe-Mn binary oxide, MnO2 and FeOOH [J]. Frontiers of Environmental Science & Engineering, 2015, 9(3): 385-393. DOI: 10.1007/s11783-014-0648-y.
[36] HAN Tian-li, ZHANG Xiao-man, FU Xiang-qian, LIU Jin-yun. Facile synthesis of chitosan nanoparticle-modified MnO2 nanoflakes for ultrafast adsorption of Pb(II) from aqueous solution [J]. Water Science and Technology-Water Supply, 2017, 17(1): 32-38. DOI:10.2166/ws.2016.109.
[37] ESCUDERO C, FIOL N, VILLAESCUSA I, BOLLINGER J C. Arsenic removal by a waste metal (hydr)oxide entrapped into calcium alginate beads [J]. Journal of Hazardous Materials, 2009, 164(2, 3): 533-541. DOI: 10.1016/ j.jhazmat.2008.08.042.
[38] LENOBLE V, LACLAUTRE C, SERPAUD B, DELUCHAT V, BOLLINGER J C. As(V) retention and As(III) simultaneous oxidation and removal on a MnO2-loaded polystyrene resin [J]. Science of the Total Environment, 2004, 326(1-3): 197-207. DOI: 10.1016/j.scitotenv.2003.12.012.
[39] LENOBLE V, BOURAS O, DELUCHAT V, SERPAUD B, BOLLINGER J C. Arsenic adsorption onto pillared clays and iron oxides [J]. Journal of Colloid & Interface Science, 2002, 255(1): 52-58. DOI: 10.1006/jcis.2002.8646.
[40] ARCIBAR-OROZCO J A, JOSUE D B, RIOS-HURTADO J C, RANGEL-MENDEZ J R. Influence of iron content, surface area and charge distribution in the arsenic removal by activated carbons [J]. Chemical Engineering Journal, 2014, 249: 201-209. DOI: 10.1016/j.cej.2014.03.096.
[41] SAMARGHANDI M R, YANG J K, GIAHI O, SHIRZAD-SIBONI M. Photocatalytic reduction of hexavalent chromium with illuminated amorphous FeOOH [J]. Environmental Technology, 2015, 36(9-12): 1132-1140. DOI: 10.1080/ 09593330.2014.982718.
[42] PI Kun-fu, XIE Xian-jun, MA Teng, SU Chun-li, WANG Yan-xin. Arsenic immobilization by in-situ iron coating for managed aquifer rehabilitation [J]. Water Research, 2020, 181: 115859. DOI: 10.1016/j.watres.2020.115859.
[43] WANG Dai-zhang, XIN Jiang, RAO Wei, HE Ji-zheng. Kinetics of soil cadmium desorption under simulated acid rain [J]. Ecological Complexity, 2009, 6(4): 432-437. DOI: 10.1016/j.ecocom.2009.03.010.
[44] JOSE L, ALVAREZ-CRUZ S, ESPERANZA G H. Effect of the mole ratio of Mn/Fe composites on arsenic(V) adsorption [J]. The Science of the total environment, 2019, 668: 47-55. DOI: 10.1016/j.scitotenv.2019.02.234.
[45] XIONG Ying, TONG Qiang, SHAN Wei-jun, XING Zhi-qiang, WANG Yue-jiao, WEN Si-qi, LOU Zhen-ning. Arsenic transformation and adsorption by iron hydroxide/manganese dioxide doped straw activated carbon [J]. Applied Surface ence, 2017, 416: 618-627. DOI: 10.1016/j.apsusc. 2017.04.145.
[46] FU Dan-dan, HE Zhuo-qun, SU Shan-shan, XU Bin, LIU Yu-ling, ZHAO Ya-ping. Fabrication of ��-FeOOH decorated graphene oxide-carbon nanotubes aerogel and its application in adsorption of arsenic species [J]. Journal of Colloid & Interface Science, 2017, 505: 105-114. DOI: 10.1016/j.jcis. 2017.05.091.
[47] YASAR A A, NURAN D A. Adsorption of chromium on chitosan: Optimization, kinetics and thermodynamics [J]. Chemical Engineering Journal, 2009, 151(1-3): 188-194. DOI:10.1016/j.cej.2009.02.010.
[48] ZHOU Ying, LIANG Chun-yan, YU Jin-gang, JIANG Xin-yu. Adsorption properties of a novel 3D graphene/MgO composite for heavy metal ions [J]. Journal of Central South University, 2019, 26(4): 813-823. DOI:10.1007/s11771-019-4051-5.
[49] TIAN Qing-hua, WANG Xiao-yang, MAO Fang-fang. Absorption performance of DMSA modified Fe3O4@SiO2 core/shell magnetic nanocomposite for Pb2+ removal [J]. Journal of Central South University, 2019, 25(4): 709-718. DOI:10.1007/s11771-018-3775-y.
[50] ANDJELKOVIC I, STANKOVIC D M, NESIC J, KRSTIC J. Fe doped Tio2 prepared by microwave-assisted hydrothermal process for removal of As(III) and As(V) from water [J]. Industrial & Engineering Chemistry Research, 2014, 53(27): 10841-10848. DOI:10.1021/ie500849r.
[51] ADDO N S, WTIRA S. Removal of trace arsenic to meet drinking water standards using iron oxide coated multiwall carbon nanotubes [J]. Journal of Chemical & Engineering Data, 2011, 56(5): 2077-2083. DOI:10.1021/je1010664.
[52] LI Yi, ZHANG Fu-shen, XIU Fu-rong. Arsenic (V) removal from aqueous system using adsorbent developed from a high iron-containing fly ash [J]. Science of the Total Environment, 2009, 407(21): 5780-5786. DOI: 10.1016/j.scitotenv.2009. 07.017.
[53] ZHU Jin, BAIG S A, SHENG Tian-tian, LOU Zi-mo, WANG Zhuo-xing, XU Xin-hua. Fe3O4 and MnO2 assembled on honeycomb briquette cinders (HBC) for arsenic removal from aqueous solutions [J]. Journal of Hazardous Materials, 2015, 286: 220-228. DOI:10.1016/j.jhazmat.2015.01.004.
[54] HAO Lin-lin, LIU Meng-zhu, WANG Nan-nan, LI Gui-ju. A critical review on arsenic removal from water using iron-based adsorbents [J]. RSC Advances, 2018, 8(69): 39545-39560. DOI:10.1039/c8ra08512a.
[55] GANESAN P, LAKSHMI J, SOZHAN G. Removal of manganese from water by electrocoagulation: Adsorption, kinetics and thermodynamic studies [J]. The Canadian Journal of Chemical Engineering, 2013, 91(3): 448-458. DOI: 10.1002/cjce.21709.
[56] SONG Pei-pei, YANG Zhao-hui, ZENG Guang-ming, YANG Xia, XU Hai-yin, HUANG Jing, WANG Li-ke. Optimization, kinetics, isotherms, and thermodynamics studies of antimony removal in electrocoagulation process [J]. Water Air & Soil Pollution, 2015, 226(11): 3801-3809. DOI:10.1007/s11270-015-2615-z.
[57] HUI Xu, MIAO Jian-dong, CHEN Ping, ZHAN Liang-tong, WANG Yu-ze. Chemical and geotechnical properties of solidified/stabilized MSWI fly ash disposed at a landfill in China [J]. Engineering Geology, 2019, 255: 59-68. DOI: 10.1016/ j.enggeo.2019.04.019.
[58] XIAO Kai, MA Jun, HU Xue-tao, DONG Yi, TIAN Yu. Influence of Curing condition on properties of solidified clay in landfill leachate environment [J]. Environmental Progress, 2019, 38(5): 147-155. DOI:10.1002/ep.13149.
(Edited by FANG Jing-hua)
���ĵ���
�轺����FeOOH��MnO2������ˮ��As(V)����ȴ���
ժҪ��Ϊ���о�������������ˮ��As(V)����Ⱦ���������ԭλ���ɷ�����FeOOH��MnO2�������Ӹ��ص��轺�ϣ��ɹ��غϳ�������������(S-FeOOH��S-MnO2)���������ı���������S-FeOOH��S-MnO2���нϴ�Ŀ����ߴ�(����150~250 ��m)������������ķǾ��Ͷ�ṹ��ʹ����нϸߵıȱ����(357.0��334.6 m2/g)���ڼ�Ъʵ���У��о���pH���Ӵ�ʱ�䡢�������������¶ȶ��������������ܵ�Ӱ�졣��ʵ������������������ȣ����о��ϳɵ����������ر���S-FeOOH���ڽϿ���pH(2~12)���¶�(25~65 ��C)��Χ�ڣ������ֳ����õ�As(V)ȥ�����ܡ��ڱ�S-FeOOH����������ˮ�У�As(V)�IJ���Ũ��ԼΪ0.01 mg/L���ﵽ������ˮ��������������ѭα��������ѧģ�ͣ���5 min�������ﵽƽ�⡣S-FeOOH��ƽ���������ݿ�ͨ��Langmuir��������ϣ���S-MnO2������������ѭFreundlichģ�ͣ�˵�����Ǿ��в�ͬ���������������������S-FeOOH��ȥ��As(V)��������S-MnO2����S-FeOOH����Ϊһ���з�չǰ������������������ˮ��As(V)����Ⱦ�����
�ؼ��ʣ�As(V)��ȥ��������������ˮ�����ϲ���
Foundation item: Projects(2019YFC0408305, 2018YFC1901601) supported by the National Key Scientific Research of China; Project(2018CX036) supported by the Innovation-Driven Plan of Central South University, China; Project(2018TP1002) supported by the Key Laboratory of Hunan Province for Clean and Efficient Utilization of Strategic Calcium-Containing Mineral Resources, China
Received date: 2020-06-28; Accepted date: 2020-10-16
Corresponding author: YUE Tong, PhD, Associate Professor; Tel: +86-13607430125; E-mail: yuetong@csu.edu.cn; ORCID: https://orcid.org/0000-0002-3330-3468; SUN Wei, PhD, Professor; Tel: +86-13507310692; E-mail: sunmenghu@csu.edu.cn; ORCID: 0000-0002-0204-4520

