基于FeO-SiO2-Al2O3渣型的废旧铝壳锂离子电池还原熔炼回收有价金属
来源期刊:中国有色金属学报(英文版)2017年第2期
论文作者:任国兴 肖松文 谢美求 潘炳 陈坚 王奉刚 夏星
文章页码:450 - 456
关键词:锂离子电池;还原熔炼;钴;镍;铜渣;铁橄榄石
Key words:lithium ion battery; smelting reduction; cobalt; nickel; copper slag; fayalite
摘 要:提出一种基于FeO-SiO2-Al2O3渣型废旧铝壳锂离子电池还原熔炼回收有价金属的新工艺,该工艺仅采用铜渣作造渣剂。研究表明:在造渣剂用量为铝壳锂离子电池质量的4.0倍、熔炼温度1723 K、熔炼时间30 min条件下,钴、镍、铜的回收率最高,分别为98.83%、98.39%和93.57%;还原熔炼合理的渣型组成为m(FeO):m(SiO2)=0.58:1~1.03:1,Al2O3含量为17.19%~21.52%;熔炼产出合金主要由Fe-Co-Cu-Ni固溶体相和冰铜相构成,产出炉渣的主要矿物成分为铁橄榄石和铁铝尖晶石,铜在渣中损失的主要机制是板条状铁橄榄石对冰铜和金属铜的机械夹杂。
Abstract: A novel smelting reduction process based on FeO-SiO2-Al2O3 slag system for spent lithium ion batteries with Al cans was developed, while using copper slag as the only slag former. The feasibility of the process and the mechanism of copper loss in slag were investigated. 98.83% Co, 98.39% Ni and 93.57% Cu were recovered under the optimum conditions of slag former/battery mass ratio of 4.0:1, smelting temperature of 1723 K, and smelting mass ratio of time of 30 min. The FeO-SiO2-Al2O3 slag system for the smelting process is appropriate under the conditions of m(FeO):m(SiO2)=0.58:1-1.03:1, and 17.19%-21.52% Al2O3 content. The obtained alloy was mainly composed of Fe-Co-Cu-Ni solid solution including small amounts of matte. The obtained slag mainly consisted of fayalite and hercynite. Meanwhile, the mechanism of copper loss is the mechanical entrainment from strip-like fayalite particles in the main form of copper sulfide and metallic copper.
Trans. Nonferrous Met. Soc. China 27(2017) 450-456
Guo-xing REN, Song-wen XIAO, Mei-qiu XIE, Bing PAN, Jian CHEN, Feng-gang WANG, Xing XIA
Changsha Research Institute of Mining and Metallurgy Co., Ltd, Changsha 410012, China
Received 3 December 2015; accepted 30 August 2016
Abstract: A novel smelting reduction process based on FeO-SiO2-Al2O3 slag system for spent lithium ion batteries with Al cans was developed, while using copper slag as the only slag former. The feasibility of the process and the mechanism of copper loss in slag were investigated. 98.83% Co, 98.39% Ni and 93.57% Cu were recovered under the optimum conditions of slag former/battery mass ratio of 4.0:1, smelting temperature of 1723 K, and smelting mass ratio of time of 30 min. The FeO-SiO2-Al2O3 slag system for the smelting process is appropriate under the conditions of m(FeO):m(SiO2)=0.58:1-1.03:1, and 17.19%-21.52% Al2O3 content. The obtained alloy was mainly composed of Fe-Co-Cu-Ni solid solution including small amounts of matte. The obtained slag mainly consisted of fayalite and hercynite. Meanwhile, the mechanism of copper loss is the mechanical entrainment from strip-like fayalite particles in the main form of copper sulfide and metallic copper.
Key words: lithium ion battery; smelting reduction; cobalt; nickel; copper slag; fayalite
1 Introduction
Lithium ion batteries (LIBs) are widely used in mobile phones, laptops and other electronic products. Large quantities of end-of-life LIBs are generated with the tremendous growth of waste electronic products. LIBs contain not only large amounts of valuable metals such as cobalt, nickel and copper, but also hazardous waste [1-3]. If disposing improperly, a huge valuable resource will be wasted and result in serious environment burden. Therefore, the development of the advanced recycling technology of spent LIBs is necessary.
At present, the recycling of spent LIBs predominantly aimed at the recovery of Co, Ni and Cu. The recycling technologies can be divided into hydrometallurgy and pyrometallurgy [4,5]. Most published literatures concerned the hydrometallurgical treatment of spent LIBs, which can produce high quality products. To find suitable operating conditions for recovering valuable metals, these published works mainly focused on the investigations of leaching [1-3,6-8] and leachate purification [9,10]. However, these works were widely carried out by manual dismantling or laboratory scale mechanical dismantling, without considering the risk of explosion for LIBs during the industrial process. Thus, the industrial Recupyl and Toxco processes are performed under protective atmosphere and cryogenic condition, respectively, which obviously increases the investment and operation cost [5]. Additionally, the hydrometallurgical process is suitable only for treatment of the specific type of batteries [4].
In pyrometallurgical process, spent LIBs and their scraps are subjected to high temperature smelting. Co and Ni contained in cathode material can be deoxidized to metallic Co and Ni by metallic aluminum or graphite which is plenty present in LIBs [11-13], and the reactions can be expressed as
2LiCoO2+2Al=Li2O+2Co+Al2O3 (1)
2LiCo1-xNixO2+3C=Li2O+(1-x)/2Co+x/2Ni+3CO(g) (2)
4LiCo1-xNixO2+Al=2Li2O+(1-x)/4Co+x/4Ni+2Al2O3 (3)
The metallic Cu of batteries keeps metallic during the reduction process. This allows the valuable metals such as Co, Ni and Cu to preferentially concentrate into an alloy phase, while the metallic Al of batteries is slagged in the form of Al2O3, and the C from batteries is evaporated as the form of carbon monoxide. Although Al cannot be recovered, a large amount of energy is produced by the above reaction, which is advantage to decrease the energy consumption [11]. Currently, the typical pyrometallurgical technologies mainly include the roasting-smelting process of Xstrata [14] and the VAL’EAS process of Umicore [11]. Compared with the hydrometallurgical process, pyrometallurgical process is flexible in feed materials and higher throughput.
The pyrometallurgical processes of spent LIBs widely employ the slag system CaO-SiO2-Al2O3, in which CaO along with SiO2 is chosen as slag former [11,15-17], while Al2O3 in slag mainly comes from the spent LIBs. Compared with other types of batteries, the content of Al-containing spent LIBs with Al cans is very high, more than 30%, which is disadvantage to economically recover the valuable metals through the smelting process. In order to improve the economic benefit of this process effectively, batteries need to be fed together with heterogenite in the VAL’EAS process [11]. The heterogenite supplies not only the slag former SiO2, but also extra valuable metals such as Co and Cu. However, the application of the process is limited due to the scarce of the heterogenite ore in China and the complexity of the feeding.
In this study, a novel slag system FeO-SiO2-Al2O3 is proposed for the aim of recovering valuable metals from spent LIBs with Al cans by a simple and economical method. In the method, waste copper slag was used as the only flux. As we all know, the copper slag mainly consists of fayalite, and the total content of FeO and SiO2 is more than 70%. So, the copper slag can supply the slag-maker of FeO and SiO2 simultaneously for the smelting process based on the FeO-SiO2-Al2O3 slag. Additionally, KIM et al [18] have already used waste copper slag as the major slag-maker for metals recovery of spent printed circuit boards by a smelting process [18]. For Umicore, it is easy to switch the CaO-SiO2-Al2O3 slag system to the FeO-SiO2-Al2O3 slag system without changing any their existing recycling facility. Moreover, this switch is also very economical due to the cheaper price of copper slag than that of heterogenite. The feasibility of the new method and the mechanism of copper loss in slag were explored in detail.
2 Experimental
2.1 Materials and reagents
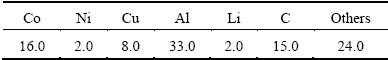
In this study, spent LIBs with sizes of 60 mm × 43 mm × 5 mm were collected from market. Table 1 presents the chemical composition of the spent LIBs used in this study. As can be seen from Table 1, the spent LIBs mainly consist of Co, Ni, Cu, Al and C. Co and Ni exist as the form of lithium mixed oxides (LiCo1-xNixO2) in the cathode. Cu and Al of the current collectors are in the form of a metal, while C of the anode is graphite.
Table 1 Chemical composition of LIBs used in this work (mass fraction, %)
Copper slag was generated in electric furnace cleaning process of a copper smelting plant, which was chosen as the slag former, and then milled into powders of -3 mm. Table 2 shows the chemical composition of the copper slag. According to the XRD pattern and the SEM analysis of the copper slag given in Figs. 1 and 2, respectively, it was found that the copper slag mainly consists of strip-like fayalite (Fe2SiO4), magnetite (Fe3O4) and matte (copper-iron sulfide). And the magnetite densely dispersed in the fayalite.
Table 2 Chemical composition of copper slag (mass fraction, %)
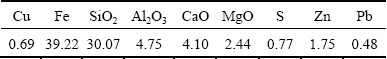
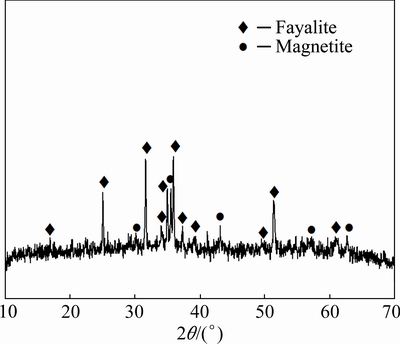
Fig. 1 XRD pattern of copper slag
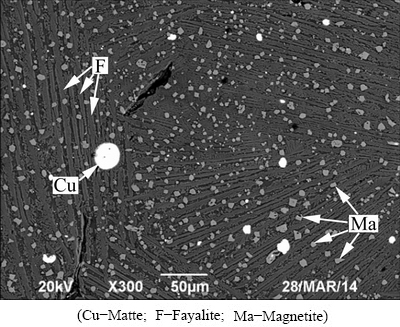
Fig. 2 SEM image of copper slag
2.2 Smelting test process
Spent Li-ion batteries were sheared into several small parts, and then mixed with the slag former according to the certain mass ratios of copper slag to battery. The mixed specimens were held in an alumina crucible with a cover, and smelted in an electrical melting furnace (box type) with MoSi2 alloy heating rods. After specimens were heated to the temperature of 1723 K, held for the pre-set time of 30 min and cooled down naturally to room temperature, the crucible was withdrawn from the furnace. The products were manually separated into alloy and slag, and then ground into powders. The effect of slag former addition on the recoveries of valuable metals (Co, Ni and Cu) in alloys was investigated.
2.3 Analytical methods
The contents of Co, Ni, Cu and Fe in the alloy and FeO, SiO2, Al2O3, CaO, MgO in the obtained slag were analyzed by chemical methods. The contents of Co, Ni and Cu in the obtained slag were analyzed by atomic absorption spectroscopy (AAS, TAS-990 Super, Beijing Purkinje General). The recovery of valuable metals was calculated according to the mass balance principle. Copper chemical phases in the obtained slag were analyzed by chemical methods. The phase compositions of the copper slag obtained were determined by X-ray diffraction (XRD, D8 advance, Bruker). The microstructure of the obtained slag was analyzed by scanning electron microscope (SEM) equipped with EDX qualitative analysis (SEM-EDS, JSM-6490LV, JEOL).
3 Results and discussion
3.1 Influence of slag former addition on chemical composition of slag and recovery of valuable metals
In order to investigate the effect of slag former addition (the mass ratio of copper slag to spent LIBs) on the chemical composition of obtained slags and recovery of valuable metals, a series of smelting experiments were performed from 4.0:1 to 10.0:1 (mass ratio). Conditions of the smelting process were as follows: smelting temperature 1723 K and time 30 min. The results are shown in Table 3 and Fig. 3, respectively.
As shown in Table 3, FeO, SiO2 and Al2O3 were the main compositions for obtained slags. Furthermore, it is feasible to describe the slags by the simplified system FeO-SiO2-Al2O3. From Table 3, it is also found that the Cu content decreased from 0.69% of the copper slag to 0.26% of the obtained slag by the reduction smelting process. This means that Cu contained in the copper slag was also recovered.
As can be seen from Fig. 3, the FeO/SiO2 mass ratio of the obtained slags increased from 0.58:1 to 1.38:1 as the slag former addition increased from 4.0:1 to 10.0:1. Cu recovery decreased significantly, with slag former addition increasing, while Ni and Co recoveries were not obvious. Cu recovery decreased from 93.57% to 71.46%, when the slag former addition increased from 4.0:1 to 10.0:1. For the addition range of the slag former investigated in this study, the most appropriate slag former addition was 4.0:1.
Table 3 Chemical compositions of obtained slags
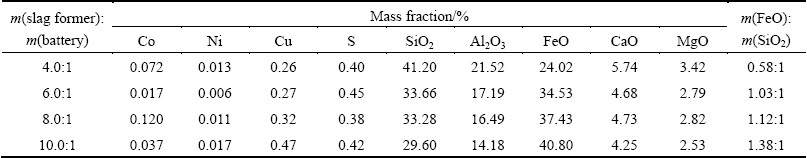
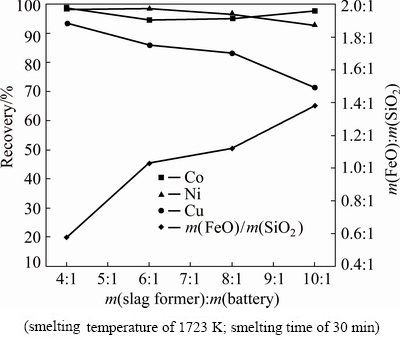
Fig. 3 Effect of slag former addition on recovery of valuable metals and FeO/SiO2 mass ratio of slags obtained

Fig. 4 pictures of alloy (a) and slag (b) produced in optimum conditional experiment
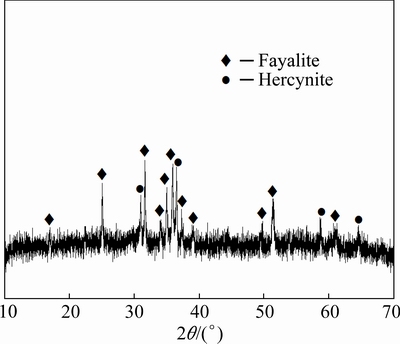
Fig. 5 XRD pattern of slag under optimum experiment
Figure 4 shows the pictures of the slag and alloy under the condition of the mass ratio of slag former addition to battery of 4.0:1. It can be seen that the alloy-slag separation was very well. The major phases in the slag were fayalite (Fe2SiO4) and hercynite (FeAl2O4) by XRD as shown in Fig. 5. This observation is in agreement with the results of PARK et al [19]. The FeO in slags was from the reduction of magnetite by aluminum or graphite from spent LIBs according to the following reactions [20]:
3(Fe3O4)+2[Al]=9(FeO)+(Al2O3) (4)
(Fe3O4)+C=3(FeO)+CO(g) (5)
Figure 6 shows the SEM-EDS analysis of the alloy under the optimum experiment. It reveals that the alloy was mainly composed of Fe-Co-Cu-Ni solid solution, including small amounts of matte. The S contained matte particles was from the waste copper slag, while the Fe in alloys and matte came from the further reduction of iron protoxide [21,22]. The reactions are as follows:
2[Al]+3(FeO)=(Al2O3)+3[Fe] (6)
C+(FeO)=[Fe]+CO(g) (7)
3.2 Adequate composition of slag system
Physico-chemical property of slag, especially liquidus temperature and viscosity, is the key factor that influences the efficiency of the high-temperature extractive metallurgical operations [23,24]. According to Table 3, the chemistries of these slags can be described by the simplified system FeO-SiO2-Al2O3 based on the major components of slags. The FeO-SiO2-Al2O3 is one of the important fundamental slag systems in the copper industry [19], which has been systematically investigated by some authors recently [19,25]. According to the results of PARK et al [19] and CHEN et al [25], the viscosity values of slags are very low with the FeO/SiO2 mass ratio ranging from 0.58:1 to 1.38:1, which implies that the slag-alloy separation should be very well at 1723 K. Their studies also show that the melting temperature of slag will increase sharply when the FeO/SiO2 mass ratio is less than 0.58:1. However, the recovery of valuable metals, especially Cu, decreases, when the FeO/SiO2 mass ratio reaches 1.03:1. Moreover, the slag is appropriate under the condition of about m(FeO):m(SiO2)=0.58:1-1.03:1, and 17.19%-21.52% Al2O3 in order to obtain the higher recovery of valuable metals.
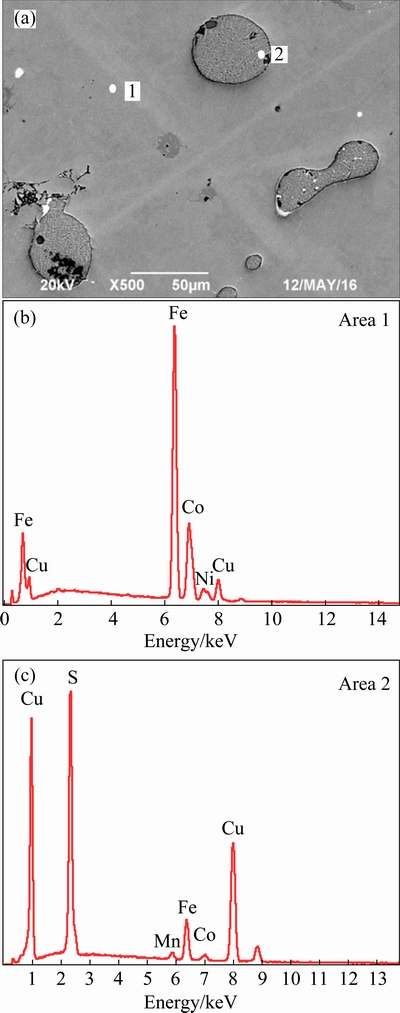
Fig. 6 SEM image (a) and corresponding EDS results (b, c) of alloy under optimum experiment
3.3 Mechanism of copper loss
An increase of the slag former addition decreased obviously the recovery of Cu. In order to analyze the mechanism of copper loss in slag, the copper phase composition in the slags is analyzed and shown in Table 4. It is noted that the contents of metallic copper and copper sulfide were up to 4.0 times higher than those of cuprous oxide (Cu2O). Moreover, the Cu2O content was very low, less than 0.024%, indicating that the loss of Cu2O is negligible. This means that the main form of the copper loss was copper sulfide and metallic copper.
Table 4 Results of copper phase compositions of slags obtained
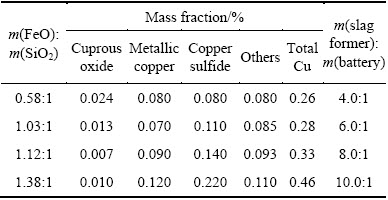
Figure 7 shows the microstructures of slag at the different FeO/SiO2 ratios. As can be seen in Fig. 7, the results of microstructures are particularly similar to the observation by ZHAI et al [26] as well as the main phase compositions. Matte (copper-iron sulfide) was seen easily, which was identical to the results of chemical phase analysis. However, the metallic copper is found hardly, this may be explained that metallic copper is included in the matte.
The fayalite particles were very fine discontinuous in shape at m(FeO):m(SiO2)=0.58:1 (Fig. 7(a)), and the size of matte particles was less than 10 μm. With the increase of FeO/SiO2 mass ratio, the sizes of matte and fayalite increased significantly. It can be inferred that the growth rate of the fayalite particles increased as the FeO/SiO2 mass ratio in slag increased during the cooling process. When the FeO/SiO2 mass ratio reached 1.38:1, the fayalite particles grew into strip-like in shape. Meanwhile, the larger matte particles were embedded into the interstice of the strip-like fayalite. And the size of matte particles was more than 100 μm. It can be explained that there is not enough time for matte particles to settle away from the slag with m(FeO):m(SiO2)=1.38:1, while the fayalite particles have grew into strip-like in shape and prevented the settlement of the matte particles. It can be concluded that the major mechanism of copper loss is the mechanical entrainment from the strip-like fayalite in the main form of copper sulfide and metallic copper.

Fig. 7 SEM images of obtained slag at different FeO/SiO2 mass ratios (Cu-Matte; F-Fayalite; H-Hercynite)
4 Conclusions
1) Under conditions of slag former/battery mass ratio of 4.0:1, smelting temperature of 1723 K, and smelting temperature of 30 min, 98.83% Co, 98.39% Ni and 93.57% Cu were recovered and the content of valuable metals was below 0.26%.
2) The FeO-SiO2-Al2O3 slag system for the smelting process was appropriate under the conditions of about m(FeO)/m(SiO2)=0.58:1-1.03:1, and 17.19%- 21.52% Al2O3.
3) The recovery of Cu decreased significantly as the slag former addition increased. The obtained slag mainly consisted of fayalite and hercynite. Meanwhile, the mechanism of copper loss in slags is the mechanical entrainment from strip-like fayalite particles in the main form of copper sulfide and metallic copper.
Acknowledgments
The authors are grateful to the State-Owned Enterprise Electric Vehicle Industry Alliance for financial support of the project JS-211 “Technology for Li-ion battery recycling”. Additionally, the authors acknowledge also Dr. Yang LIU and Dr. You-qi FAN, for their important advices.
References
[1] LI Jin-hui, LI Xin-hai, ZHANG Yun-he, HU Qi-yang, WANG Zhi-xing, ZHOU You-yuan, FU Fang-ming. Study of spent battery material leaching process [J]. Transactions of Nonferrous Metals Society of China, 2012, 22: 2274-2281.
[2] ZHU Shu-guang, HE Wen-zhi, LI Guang-ming, ZHOU Xu, ZHANG Xiao-jun, HUANG Ju-wen. Recovery of Co and Li from spent lithium-ion batteries by combination method of acid leaching and chemical precipitation [J]. Transactions of Nonferrous Metals Society of China, 2012, 22: 2274-2281.
[3] KU H, JUNG Y, JO M, PARK S, KIM S, YANG D, RHEE K, AN E M, SOHN J, KWON K. Recycling of spent lithium-ion battery cathode materials by ammoniacal leaching [J]. Journal of Hazardous Materials, 2016, 313: 138-146.
[4] AL-THYABAT S, NAKAMURA T, SHIBATA E, IIZUKA A. Adaptation of minerals processing operations for lithium-ion (LiBs) and nickel metal hydride (NiMH) batteries recycling: Critical review [J]. Minerals Engineering, 2013, 45: 4-17.
[5] VADENBO C O. Prospective environmental assessment of lithium recovery in battery recycling [D]. Zuiich: Institute for Environmental Decisious, 2009: 19-38.
[6] LI L, ZHAI L, ZHANG X, LU J, CHEN R, WU F, AMINE K. Recovery of valuable metals from spent lithium-ion batteries by ultrasonic-assisted leaching process [J]. Journal of Power Sources, 2014, 262: 380-385.
[7]  M, LAUCOURNET R, BILLY E. Hydrometallurgical process for the recovery of high value metals from spent lithium nickel cobalt aluminum oxide based lithium-ion batteries [J]. Journal of Power Sources, 2014, 247: 551-555.
M, LAUCOURNET R, BILLY E. Hydrometallurgical process for the recovery of high value metals from spent lithium nickel cobalt aluminum oxide based lithium-ion batteries [J]. Journal of Power Sources, 2014, 247: 551-555.
[8] CHEN X, CHEN Y, ZHOU T, LIU D, HU H, FAN S. Hydrometallurgical recovery of metal values from sulfuric acid leaching liquor of spent lithium-ion batteries [J]. Waste Management, 2015, 38: 349-356.
[9] JOO S H, SHIN D J, OH C H, WANG J P, SENANAYAKE G, SHIN S M. Selective extraction and separation of nickel from cobalt, manganese and lithium in pre-treated leach liquors of ternary cathode material of spent lithium-ion batteries using synergism caused by Versatic 10 acid and LIX 84-I [J]. Hydrometallurgy, 2016, 159: 65-74.
[10] CHEN Liang, TANG Xin-cun, ZHANG Yang, QU Yi, WANG Zhi-min. Separation and recovery of Ni, Co and Mn from spent lithium-ion batteries [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(146): 1192-1198. (in Chinese)
[11] CHERET D,  S. Battery recycling: European Patent, 1589121 [P]. 2005-10-26.
S. Battery recycling: European Patent, 1589121 [P]. 2005-10-26.
[12] GARCIA E M, LINS V D F C,  H A, MATENCIO T, DOMINGUES R Z, SANTOS J A F D. The anode environmentally friendly for water electrolysis based in LiCoO2 recycled from spent lithium-ion batteries [J]. International Journal of Hydrogen Energy, 2012, 37(22): 16795-16799.
H A, MATENCIO T, DOMINGUES R Z, SANTOS J A F D. The anode environmentally friendly for water electrolysis based in LiCoO2 recycled from spent lithium-ion batteries [J]. International Journal of Hydrogen Energy, 2012, 37(22): 16795-16799.
[13] SIRET C, LOPEZ I, DAMME G V. Umicore and salt closing the battery loop-recycling lowers environmental impacts: The case of Li-ion batteries [R]. Fort Lauderdale: The 12th Battery Materials Recycling Seminar and Exhibit, 2008.
[14] BARTSCH E. Do not expose to heat or dispose of in fire! [EB/OL]. http://www.xps.ca/EN/Media/XPS%20Bulletin/XPS%20Bulletin%20Issur%201%20-%Fall%202008.pdf
[15] GEORGI-MASCHLER T, FRIEDRICH B, WEYHE R, HEEGN H, RUTZ M. Development of a recycling process for Li-ion batteries [J]. Journal of Power Sources, 2012, 207: 173-182.
[16] YUAN Wen-hui, QIU Ding-pan, CHEN Cheng-yan. Co-Cu-Fe alloy recycled from spent lithium ion batteries by reducing smelting process [J]. Material Science & Technology, 2010, 18: 455-458. (in Chinese)
[17] MESKERS C E M,  C, DAMME G V. Green recycling of EEE: Special and precious metal recovery from EEE [C]//TMS 2009 Annual Meeting & Exhibition. San Francisco: Materials Society, 2009: 1131-1136.
C, DAMME G V. Green recycling of EEE: Special and precious metal recovery from EEE [C]//TMS 2009 Annual Meeting & Exhibition. San Francisco: Materials Society, 2009: 1131-1136.
[18] KIM B S, LEE J C, JEONG J, YANG D H, SHIN D, LEE K. A novel process for extracting precious metals from spent mobile phone PCBs and automobile catalysts [J]. Materials Transactions, 2013, 54(6): 1045-1048.
[19] PARK H, PARK S S, SOHN I. The viscous behavior of FeOt-Al2O3-SiO2 copper smelting slags [J]. Metallurgical and Materials Transactions B, 2011, 42: 692-699.
[20] BUSOLIC D, PARADA F, PARRA R, SANCHEZ M, PALACIOS J, HINO M. Recovery of iron from copper flash smelting slags [J]. Mineral Processing & Extractive Metallurgy, 2011, 120: 32-36.
[21] HEO J H, KIM B S, PARK J H. Effect of CaO addition on iron recovery from copper smelting slags by solid carbon [J]. Metallurgical and Materials Transactions B, 2013, 44: 1352-1363.
[22] PARK J H, SONG H S, MIN D J. Reduction behavior of EAF slags containing Cr2O3 using aluminum at 1793 K [J]. ISIJ International, 2004, 44: 790-794.
[23] CHEN M, SREEKANTH R, ZHAO B. Viscosity measurements of ‘‘FeO’’-SiO2 slag in equilibrium with metallic Fe [J]. Metallurgical and Materials Transactions B, 2013, 44: 506-515.
[24] KIMURA H, ENDO S, YAJIMA K, TSUKIHASHI F. Effect of oxygen partial pressure on liquidus for the CaO-SiO2-FeOx system at 1573 K [J]. ISIJ International, 2004, 44: 2040-2045.
[25] CHEN M, RAGHUNATH S, ZHAO B. Viscosity of SiO2-“FeO”-Al2O3 system in equilibrium with metallic Fe [J]. Metallurgical and Materials Transactions B, 2013, 44: 820-827.
[26] ZHAI Xiu-jing, LI Nai-jun, ZHANG Xu, FU Yan, JIANG Lan. Recovery of cobalt from converter slag of Chambishi copper smelter using reduction smelting process [J]. Transactions of Nonferrous Metals Society of China, 2011, 21: 2117-2121.
任国兴,肖松文,谢美求,潘 炳,陈 坚,王奉刚,夏 星
长沙矿冶研究院有限责任公司,长沙 410012
摘 要:提出一种基于FeO-SiO2-Al2O3渣型废旧铝壳锂离子电池还原熔炼回收有价金属的新工艺,该工艺仅采用铜渣作造渣剂。研究表明:在造渣剂用量为铝壳锂离子电池质量的4.0倍、熔炼温度1723 K、熔炼时间30 min条件下,钴、镍、铜的回收率最高,分别为98.83%、98.39%和93.57%;还原熔炼合理的渣型组成为m(FeO):m(SiO2)=0.58:1~1.03:1,Al2O3含量为17.19%~21.52%;熔炼产出合金主要由Fe-Co-Cu-Ni固溶体相和冰铜相构成,产出炉渣的主要矿物成分为铁橄榄石和铁铝尖晶石,铜在渣中损失的主要机制是板条状铁橄榄石对冰铜和金属铜的机械夹杂。
关键词:锂离子电池;还原熔炼;钴;镍;铜渣;铁橄榄石
(Edited by Xiang-qun LI)
Foundation item: Project (JS-211) supported by the State-Owned Enterprise Electric Vehicle Industry Alliance, China
Corresponding author: Song-wen XIAO; Tel: +86-731-88657240; E-mail: swinxiao@126.com
DOI: 10.1016/S1003-6326(17)60051-7

