Two-step carbon modification of NaTi2(PO4)3 with improved sodium storage performance for Na-ion batteries
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2018���10��
�������ߣ������� Ѧϼ �ﵩ ���ܹ� ��С�� �źغ� TANG You-gen(���и�)
����ҳ�룺2320 - 2331
Key words��two-step carbon modification; NaTi2(PO4)3; sodium storage performance; Na-ion batteries
Abstract: NASICON-type structured NaTi2(PO4)3 has been regarded as a promising anode material for non-aqueous and aqueous Na-ion batteries, whereas its sodium storage performance was greatly restricted by its inherent inferior electronic conductivity. In the present work, a two-step carbon modification method using prefabricated carbon spheres as support and phenolic resin as carbon source was proposed to prepare advanced NaTi2(PO4)3/C. The as-prepared composite with carbon spheres displayed a much higher reversible capacity (126.7 mA��h/g vs 106.7 mA��h/g at 0.5C) than the control sample without carbon spheres. Superior rate capability with discharge capacities of 115.1, 95.5, 80.8 mAh/g at 1C, 10C, 20C, respectively and long-term cycling stability with capacity retention of 92.4% after 1000 cycles at 5C were also observed. Owing to the designing of two-step carbon modification, although the as-prepared sample shows much smaller surface area, it possesses much better conductive network and more uniform particle distribution, resulting in higher electronic conductivity and faster ionic conductivity, thereby superior sodium storage ability at high rate.
Cite this article as: XUE Xia, SUN Dan, ZENG Xian-guang, HUANG Xiao-bing, ZHANG He-he, TANG You-gen, WANG Hai-yan. Two-step carbon modification of NaTi2(PO4)3 with improved sodium storage performance for Na-ion batteries [J]. Journal of Central South University, 2018, 25(10): 2320�C2331. DOI: https://doi.org/10.1007/ s11771-018-3916-3.

J. Cent. South Univ. (2018) 25: 2320-2331
DOI: https://doi.org/10.1007/s11771-018-3916-3
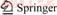
XUE Xia(Ѧϼ)1, SUN Dan(�ﵩ)1, ZENG Xian-guang(���ܹ�)2, HUANG Xiao-bing(��С��)3,ZHANG He-he(�źغ�)1, TANG You-gen(���и�)1, WANG Hai-yan(������)1
1. Hunan Provincial Key Laboratory of Chemical Power Sources, Hunan Provincial Key Laboratory of Efficient and Clean Utilization of Manganese Resources, College of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. Material Corrosion and Protection Key Laboratory of Sichuan Province, Zigong 643000, China;
3. College of Chemistry and Chemical Engineering, Hunan University of Arts and Science,Changde 415000, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: NASICON-type structured NaTi2(PO4)3 has been regarded as a promising anode material for non-aqueous and aqueous Na-ion batteries, whereas its sodium storage performance was greatly restricted by its inherent inferior electronic conductivity. In the present work, a two-step carbon modification method using prefabricated carbon spheres as support and phenolic resin as carbon source was proposed to prepare advanced NaTi2(PO4)3/C. The as-prepared composite with carbon spheres displayed a much higher reversible capacity (126.7 mA��h/g vs 106.7 mA��h/g at 0.5C) than the control sample without carbon spheres. Superior rate capability with discharge capacities of 115.1, 95.5, 80.8 mAh/g at 1C, 10C, 20C, respectively and long-term cycling stability with capacity retention of 92.4% after 1000 cycles at 5C were also observed. Owing to the designing of two-step carbon modification, although the as-prepared sample shows much smaller surface area, it possesses much better conductive network and more uniform particle distribution, resulting in higher electronic conductivity and faster ionic conductivity, thereby superior sodium storage ability at high rate.
Key words: two-step carbon modification; NaTi2(PO4)3; sodium storage performance; Na-ion batteries
Cite this article as: XUE Xia, SUN Dan, ZENG Xian-guang, HUANG Xiao-bing, ZHANG He-he, TANG You-gen, WANG Hai-yan. Two-step carbon modification of NaTi2(PO4)3 with improved sodium storage performance for Na-ion batteries [J]. Journal of Central South University, 2018, 25(10): 2320�C2331. DOI: https://doi.org/10.1007/ s11771-018-3916-3.
1 Introduction
Solar, wind, tide energy and many other renewable energy sources have become excellent candidates to alleviate the energy issues resulted from the rapid growth of economy and population. However, these energy sources are largely limited by area, seasons and time, which make it an urgent priority to develop safe and cost-effective electric storage systems to alleviate this problem [1]. Lithium-ion batteries (LIBs) have been widely used in the portable electronic devices and have shown promising applications in electric vehicles and large scale energy storage systems mainly because of their high energy density and outstanding cycling performance [2]. Nevertheless, taking the cost into consideration, the relatively rare reserve and high price of Li are the bottleneck to realizing the large-scale application [3]. Therefore, Na-ions batteries (NIBs), have become a promising substitute to meet the energy storage demands because of the natural abundance and much cheaper price of Na [4]. As sodium and lithium are in the same main group, they have similar physicochemical properties, therefore, NIBs have the same principle as LIBs.
Because of the larger ionic radius of sodium ions, most anode materials in LIBs exhibit inferior sodium storage performance [5]. The anode materials for NIBs can be mainly divided into five types: 1) carbon based materials; 2) metal oxides and sulfides; 3) alloy-based materials; 4) titanium- based composites; 5) organic compounds [6]. Graphite, the most successfully commercialized anode material for LIBs, displays poor sodium storage performance due to the mismatching of the graphite interlayer distance (d002=0.334 nm) to the large sodium ions [7, 8]. Although metal oxides [9, 10] and sulfides [11�C13] along with alloy-based materials [14�C16] have high capacity, they still face a number of issues such as poor initial Coulombic efficiency and inferior cycling stability. Titanium- based compounds [17�C19], for examples, TiO2 [20�C24], LiTi2(PO4)3 [25], NaTi2(PO4)3 [26] have drawn much attention owing to its low cost and superior cycling stability.
NaTi2(PO4)3, as a typical Na-super ionic conductor (NASICON) structure compound, possesses highly covalent 3D open-framework structure and high ionic conductivity, which make it a promising electrode material for NIBs [19, 27]. It can offer a capacity of 133 mA��h/g in theory with two Na ions intercalation/deintercalation, and present a well-defined redox plateau at ~2.1 V (vs Na+/Na). Therefore, NaTi2(PO4)3 can be used as anode material for aqueous sodium-ion batteries (ASIBs) [28�C30]. However, the low intrinsic electronic conductivity is a big problem, resulting in a low specific capacity and inferior rate performance [31, 32]. Two general strategies have been widely adopted to overcome this drawback [33�C35]: 1) decreasing the particle size to reduce the ionic/electronic transporting path, thus increasing the kinetics of lithium/sodium storage;2) carbon coating to form a well conductive network and protect NaTi2(PO4)3 from the erosion of electrolyte and alleviate the crystal expansion during rapid charge-discharge process. In our previous work, we synthesized the NaTi2(PO4)3 nanoparticles embedded in carbon matrix and constructed the NaTi2(PO4)3/C//LiMn2O4 ALIBs system, which displayed outstanding cycling stability with 95% capacity retention over 3600 cycles at 1500 mA/g [26]. The intercalated porous NaTi2(PO4)3 spheres delivered a high specific capacity of 134 mA��h/g at 1C as well as superior cycling performance with 81 mA��h/g remaining after 1000 cycles at a super- high current density of 50C [36].
In this work, NaTi2(PO4)3/C composites were synthesized by a facile reflux and sintering process, and carbon spheres (synthesized by glucose through hydrothermal method) and phenolic resin were used as two kinds of carbon sources to modify the NaTi2(PO4)3 nanoparticles. Although the morphology was not well controlled, this two-step carbon modification leaded to more competitive conductive network on NaTi2(PO4)3 nanoparticles. As verified, the as-prepared sample possesses much higher electronic conductivity and ionic conductivity. Accordingly, it displayed excellent rate performance with a high reversible capacity 126.7 and 80.8 mA��h/g at 0.5C and 20C, respectively, as well as superior cycling stability with capacity retention of 92.4% after 1000 cycles at 5C.
2 Experimental section
2.1 Materials synthesis
2.1.1 Preparation of carbon spheres
The carbon spheres were prepared according to the previous reports [37, 38]. Firstly, 0.3 mol of glucose was dissolved in 30 mL of deionized water under stirring by magnetic for 10 min. Then the solution was poured into Teflon-lined autoclave and reacted at 180 ��C for 10 h. After that, the precipitates were filtered and washed by deionized water and absolute alcohol for several times, then dried for standby application.
2.1.2 Preparation of NaTi2(PO4)3
First of all, 0.3 g of the prepared carbon spheres was added into 20 mL of absolute alcohol and treated by ultrasonic for 90 min, then the mixed solution with 1.6726 g of tetrabutyl titanate,0.2098 g of CH3COONa and 10 mL of absolute alcohol was dropped into the above solution under magnetic stirring. After that, 0.887 g of H3PO4 in 10 mL absolute alcohol was added and the turbid solution was refluxed at 80 ��C for 5 h. Finally, the solvent was evaporated at that temperature and brown powder was obtained.
2.1.3 Carbon coating with phenolic resin
Phenolic resin was used as carbon source to coat the brown powder. First, 0.5 g of the obtained brown powder was treated in 30 mL of absolute alcohol by ultrasonic for 90 min, then 0.3 g of phenolic resin with 10 mL of absolute alcohol was added. After that, the mixed solution was stirred at 60 ��C until the solvent was evaporated. The collected precursor was ground by agate mortar, then it was placed in tube furnace and sintered at 550 ��C for 2 h first and then 700 ��C for 6 h under Ar flow. The as-prepared sample by two-step carbon modification was marked as C-NTP-C.
For comparison, the control sample was also synthesized by the similar method without the addition of carbon spheres, which was marked as NTP-C. For fair comparison, we optimized the experiment to achieve the similar carbon content in final sample by adding more phenolic resin.
As it has been found that certain kind of carbons in some cases may have sodium storage capacity contribution within the voltage range of 1.0�C3.0 V [39�C41], pure carbon (PC) only with carbon spheres and phenolic resin were also synthesized in the same way for electrochemical test.
2.2 Characterizations
X-ray diffraction (XRD) measurements were carried out by X-ray diffractometer (Dandong China, TD-3500) with Cu K�� radiation (��=0.15418 nm), scan step length of 0.02�� and scanning range of 0�C80��. Transmission electron microscope (TEM) and high resolution TEM (HRTEM) images were obtained by field emission high resolution transmission electron microscopy (Titan G2 60�C300). Scanning electron microscope (SEM) images were obtained through JSM-6390LV (JEOL). Thermogravimetric analysis (TGA) was carried out on a STA 449C. The Brunauer-Emmett- Teller (BET) specific surface area was measured by SSA-4200. Raman spectra were operated on the LabRAM HR800 (HORIBA Jobin YvonS.A.S) spectrometer. X-ray photoelectron spectroscopy (XPS) was performed on a K-Alpha 1063 spectrometer, and the XPS fitting was performed using XPSpeak41 software. The electronic conductivity was obtained through four-point probe measurement (Guangzhou 4 Probes Tech, RTS-9).
2.3 Electrochemical measurements
The electrochemical tests were characterized in a CR2016-type coin cell. The electrodes were made up of active material, acetylene black and polyvinylidene fluoride (PVDF) with a weight ratio of 8:1:1. Firstly, a slurry was obtained by mixing the materials in N-methyl-2-pyrrolidone (NMP), then it was coated on a Cu foil. After dried in vacuum oven at 80 ��C overnight, the coated foil was cut into small disks with a diameter of 12 mm. The mass loading of the electrode was 1.3 to 1.8 mg/cm2. Coin cell was assembled in an Ar-filled Mikrouna glovebox with NaTi2(PO4)3 active material as cathode, sodium metal as the counter electrode, 1 mol/L NaClO4 in 95:5 V/V propylene carbonate/fluoroethylene carbonate as the electrolyte, and glass fiber as the separator. Cyclic voltammetry (CV) test was performed with a desired scan rate of 0.2 mV/s at room temperature through the CHI 660e electrochemical station (Shanghai Chenhua, China). Galvanostatic charge�C discharge (GCD) experiments were carried out within the voltage range of 1.5�C3.0 V (versus Na+/Na) by Neware battery testing system (CT-3008W) at a constant temperature of 30 ��C. Electrochemical impedance spectroscopy (EIS) was obtained in the frequency range of 100 kHz to 10 mHz through CHI 660e electrochemical station (Shanghai Chenhua, China).
3 Results and discussion
3.1 Materials characterizations
The crystal structures of C-NTP-C and NTP-C composites were identified by XRD patterns as shown in Figure 1(a). As seen clearly, all diffraction peaks in two samples can be well indexed to the rhombohedral structured NaTi2(PO4)3 with the R-3c space group (PDF#33-1296), which also implies a high purity of the as-prepared NaTi2(PO4)3 (NTP) composites and the carbon spheres and phenolic resin have no effect on the crystal structure of NaTi2(PO4)3. The crystal lattice parameters of C-NTP-C are a=0.8471 nm, b=2.1587 nm, which are in good agreement with those reported in literature [26, 31, 32]. The NTP-C with broad peaks implies smaller grain size, which is consistent with the larger BET surface area in the following [42]. It should be noted that we added more phenolic resin as carbon source for the synthesis of NTP-C to achieve similar carbon content in the final samples. The sharp peaks of C-NTP-C indicate improved crystallinity due to the incorporation of carbon spheres, which could be helpful for long-lived cycling stability. Thermogravimetric analysis (TGA) was performed to determine the carbon contents of the prepared composites. According to the profiles in Figure 1(b), the carbon contents in C-NTP-C and NTP-C are 18.9 wt% and 18.5 wt%, respectively.
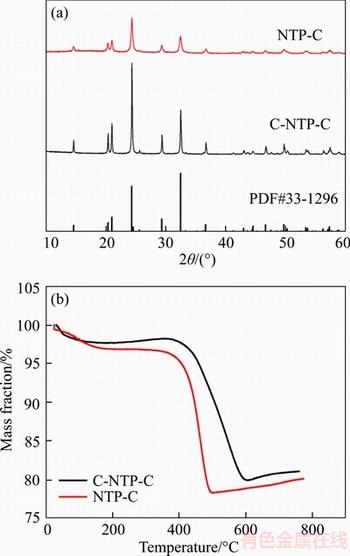
Figure 1 XRD patterns (a) and TG curves (b) of C-NTP-C and NTP-C
The SEM images of C-NTP-C and NTP-C composites are displayed in Figures 2(a) and (b). Obviously, both samples show the particle morphology, which consists of primary nanoparticles. Although the primary particle size of C-NTP-C seems a little larger than that of NTP-C, its distribution is more uniform. Note that some nubs are formed by the aggregation of primary nanoparticles. Individual particles with size range from 20 to 100 nm are clearly seen for C-NTP-C composites in TEM image (Figure 2(c)). From the HRTEM images (Figures 2(d)�C(f)), it can be seen that the pure NTPs and carbon materials are cross distributed, and on the boundary region the carbon layer is around 3 nm. As marked in Figures 2(d) and (f), the inter-planar spacing of 0.36 mm is corresponding to the d-spacing of the (113) plane of NTP [27]. From Figure 2(e), we can see that the relatively big particle is formed by a carbon layer outside and many NTPs and carbon materials are wrapped inside. The conductive network constructed by two-step modification may be beneficial to fast transmission for electrons. The lattice fringe of carbon in Figure 2(f) indicates good electronic conductivity of carbon layer.
The N2 adsorption-desorption isotherms of C-NTP-C and NTP-C are displayed in Figure 3. Obviously, the isotherms with a distinct hysteresis loop belong to a typical of IV isotherm, indicating the existence of mesoporous [43, 44]. The Brunauer-Emmett-Teller (BET) surface areas of C-NTP-C and NTP-C are 46.43 m2/g and 110.87 m2/g, respectively. In this work, to achieve the similar carbon content for both samples, much more phenolic resin was added for NTP-C, which would result in higher surface area. From the inset Barrett-Joyner-Halenda (BJH) pore size distribution plot, both samples show similar mesoporous structures in a size range of 3�C9 nm.
Surface element analysis for the as-prepared composites is conducted by X-ray photoelectron spectroscopy (XPS). As displayed in Figures 4(a) and (b), Ti, P, O and C elements can be observed in both C-NTP-C and NTP-C survey spectra. According to the peak ratio of O1s and C1s, the O/C ratio in C-NTP-C and NTP-C is 5.01 and 1.11, respectively. The high-resolution C1s XPS spectra of C-NTP-C and NTP-C can be deconvoluted into four different signals. The peak at ~284.8 eV is attributed to the C=C bond, and the peaks at ~286.0 eV, ~287.2 eV and ~289.2 eV are corresponding to C��O, C=O and O��C=O bonds, respectively [45�C47]. The ratios of oxygen- containing functional groups in two samples are different. The C��O/C=C peak area ratio of C-NTP-C is 0.486 (NTP-C 0.404), which indicates that there may be some oxygen-containing functional groups in the carbon spheres, therefore, the interaction forces between pure NTP and carbon materials can be enhanced through the carbon spheres, resulting in higher charge transfer between carbon layer and NTP. Additionally, carbon spheres with oxygen-containing functional groups may be able to coordinate with titanium ions and that may be one of the reasons for the uniform particle size distribution of C-NTP-C. Moreover, the existence of O heteroatom could create some extrinsic defects and active sites, which could be helpful for rapid Na+ absorption and diffusion in the coated carbon layer [45].

Figure 2 SEM images of C-NTP-C (a) and NTP-C (b), TEM (c) and HRTEM images (d, e, f) of C-NTP-C
3.2 Electrochemical properties
The as-prepared samples were tested in sodium-ion batteries. Figure 5(a) shows the cyclic voltammetry curves at a scan rate of 0.2 mV/s within a voltage range of 1.5 to 3.0 V. As can be seen, both composites have a pair of redox peaks located at ~2.06 V (red.) and ~2.26 V (ox.), respectively, indicating a high reversibility of Na+ insertion/extraction in NaTi2(PO4)3. For comparison, C-NTP-C shows sharper oxidation/reduction peaks and smaller peak difference, implying faster Na+ extraction/insertion kinetics. This may be attributed to the more uniform particle size distribution and higher electronic conductivity in C-NTP-C composites. Figure 5(b) shows EIS profiles of the two samples. Each profile is composed by a depressed semicircle in high-to-middle frequency region and an inclined line at low frequency, indicating dual control of charge transfer and sodium ions diffusion in the electrochemical process [34, 48]. Generally, the diameter of the depressed semicircle is attributed to the charge-transfer resistance (Rct) and the inclined line is related to the solid state diffusion of ions in the active materials [25]. Apparently, the much smaller diameter of the depressed semicircle of C-NTP-C indicates lower Rct. As it has been reported before, the Rct is the main reason of the voltage drop for the kinetics limitation at high current density, thereby resulting in the deterioration of electrochemical performance [42, 49]. The C-NTP-C with much lower Rct may display improved electrochemical performance.
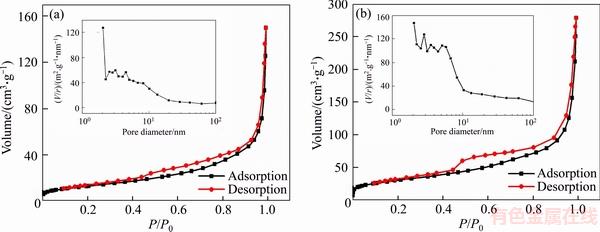
Figure 3 N2 adsorption-desorption isotherm of C-NTP-C (a) and NTP-C (b) (the figures inset are Barrett-Joyner- Halenda (BJH) pore size distribution plot of C-NTP-C and NTP-C)

Figure 4 Survey XPS spectra of C-NTP-C (a) and NTP-C (b) and C1s high-resolution XPS spectra of C-NTP-C (c) and NTP-C (d)

Figure 5 CV curves (a) of C-NTP-C and NTP-C at 0.2 mV/s, Nyquist plots (b) of C-NTP-C and NTP-C
Figure 6(a) gives a comparison of the rate performance of the two composites at various rates range from 1C to 20C (1C=150 mA/g in this work) within the voltage range of 1.5�C3.0 V (vs Na+/Na). As can be seen, the C-NTP-C delivers a much higher capacity at all rates than NTP-C. The discharge capacities of C-NTP-C are 115.1, 95.5, 80.8 mA��h /g at 1C, 10C, 20C, respectively, while those of the NTP-C are 81.3, 74.4, 63.2 mA��h/g. It is worth pointing out that the discharge capacity of C-NTP-C at 1C is about 33.8 mA��h/g higher than that of NTP-C. After the rate performance test, when the C rate goes back to 1C, the discharge capacity can go back to the initial value, which shows that the C-NTP-C electrode has good reversibility and can withstand ultra-fast charge/ discharge process. The rate performance of C-NTP-C here can be comparable to those recently reported in references, such as carbon nanotube supported NaTi2(PO4)3 (101.7 mA��h/g at 0.5C and 84.2 mA��h /g at 10C) [44] and porous NaTi2(PO4)3/C plate (110 mA��h /g at 1C and 85 mA��h /g at 10C) [50]. The splendid rate performance may be attributed to the uniform particle size distribution and the good conductive network constructed by our two-step carbon modification method. The charge/discharge curves of C-NTP-C at different rates are displayed in Figure 6(b), from which a typical voltage plateau around 2.1 V correspond to the redox reaction of Ti4+/Ti3+ is observed. With the increase of current density, the overpotential also increases, but the voltage plateau still remains apparently, suggesting outstanding electrochemical stability of C-NTP-C electrode. The four-point probe method was further used to evaluate the electron-transporting performance of samples. As determined, electronic conductivities of C-NTP-C and NTP-C are 3.33��10�C2 S/cm and 5.56��10�C3 S/cm, respectively. Obviously, with the carbon spheres support, the electronic conductivity has been significantly improved. The much higher electronic conductivity should be the main factor for superior rate performance.
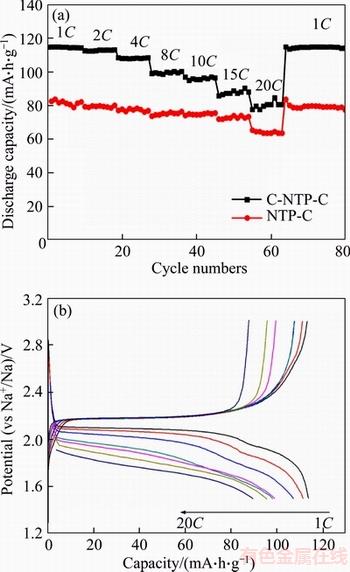
Figure 6 Rate performance (a) of C-NTP-C and NTP-C and charge/discharge curves (b) of C-NTP-C at different rates
Cycling stability is also a vital parameter in the practical application of electrode materials.Figures 7(a)�C(c) displays the cycling performance of the two samples at 0.5C, 1C and 5C. As seen, both samples show good cycling stability. The discharge capacity of C-NTP-C is much higher than that of NTP-C, which is 126.7 mA��h/g and 112.8 mA��h /g at 0.5C and 1C, respectively. However, the discharge capacity of NTP-C is only 106.7 mA��h/g and 79.9 mA��h/g at the same current density. This may be attributed to the uniform particle distribution and decreased charge-transfer resistance by our two-step carbon modification. As far as we know, the discharge capacity of C-NTP-C at 0.5C (95.2 % of theoretical capacity) is quite higher than those in the previous reports [44, 51, 52]. The galvanostatic charge�Cdischarge experiment of pure carbon was also carried out. From Figure 7(b), the capacity delivered by pure carbon is very low between 1.5 and 3.0 V, which could be ignored. Figure 7(c) shows the long-term cycling stability of C-NTP-C at 5C. An initial discharge capacity of 115.2 mA��h /g is delivered and 106.5 mA��h /g is maintained after 1000 cycles with capacity retention of 92.4%. Note that the Coulombic efficiency at 5C is almost close to 100%. The charge/discharge profiles of C-NTP-C at 5C at different cycles in Figure 7(d) indicate good voltage plateau. The good cycling performance and high reversible discharge capacity should be attributed to the conductive network constructed through our two-step carbon modification method.
During the charge/discharge process, because of the repetitive volume expansion/shrinkage caused by Na ions insertion/extraction, structure deterioration, electrode crushing and active material detachment from conductive substrate are regarded as the main factors for capacity fading [53, 54]. Bearing this in mind, we examined the surface morphology and structure change of the C-NTP-C electrodes after different cycles (3, 250 and 1000) at 5C. As shown in Figure 8(d), the XRD patterns of the electrodes after different cycles remain almost the same except for some change in peak intensity, and no new impurity peaks are observed, revealing excellent structural stability. The surface morphology evolution of C-NTP-C electrodes was also investigated by SEM (Figures 8(a)�C(c)). As can be seen, the surface microstructures of the electrode remain very intact even it was after 1000 cycles when compared to that after 3 cycles. It is known that inhomogenous carbon coating with limited charge transfer could cause charge accumulation on the electrode surface, resulting in active material falling off from the electrode, thereby leading to poor electrochemical performance [55]. In this work, using the two-step carbon modification method, good conductive network could be constructed and some NTPs could be fully wrapped inside the carbon layers. Therefore, the crush of electrode and detachment of active material from the conductive substrate could be well controlled and superior rate capacity and cycling stability could be obtained.

Figure 7 Cycling stability of as-prepared composites at 0.5C (a) and 1C (b) (1C=150 mA/g) in voltage range from 1.5 to 3.0 V; (c) Cycling stability and Coulombic efficiency of C-NTP-C at 5C; (d) Charge/discharge curves of C-NTP-C at 5C after different cycles
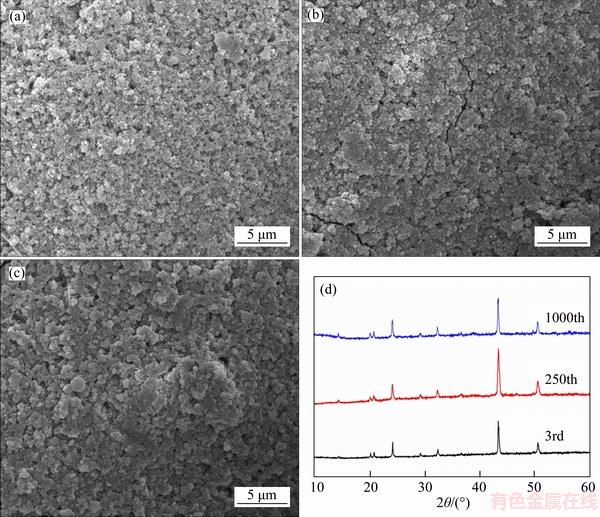
Figure 8 SEM images of C-NTP-C electrode after 3 cycles (a), 250 cycles (b) and 1000 cycles (c) at 5C (1C=150 mA/g); (d) XRD patterns of C-NTP-C electrodes after different cycles at 5C (Before disassembling, each cell was charged to 2.1 V and then kept at that voltage for 2 h)
4 Conclusions
1) The NaTi2(PO4)3/C composites by two-step carbon modification showed better particle distribution and improved electronic conductivity. More importantly, the EIS results indicated that this sample had much smaller charge-transfer resistance.
2) The electrochemical performances of NaTi2(PO4)3/C composites were greatly improved in comparison with that without carbon spheres support. It exhibited a quite high discharge capacity of 126.7 mA��h/g at 0.5C and superior capacity retention of 92.4% after 1000 cycles at 5C. Excellent rate capability was also demonstrated by the discharge capacity of 115.1 mA��h/g at 1C and 80.8 mA��h /g at 20C.
3) This two-step carbon modification method could be extended to other kinds of electrode material for achieving high rate performance.
References
[1] DUNN B, KAMATH H, TARASCON J M. Electrical energy storage for the grid: A battery of choices [J]. Science, 2011, 334(6058): 928�C935. DOI:10.1126/science.1212741.
[2] ARMAND M, TARASCON J M. Building better batteries [J]. Nature, 2008, 451(7179): 652. DOI:10.1038/451652a.
[3] KUNDU D, TALAIE E, DUFFORT V, NAZAR L F. The emerging chemistry of sodium ion batteries for electrochemical energy storage [J]. Angewandte Chemie International Edtion in English, 2015, 54(11): 3431�C3448. DOI:10.1002/anie.201410376.
[4] SLATER M D, KIM D, LEE E, JOHNSON C S. Sodium-ion batteries [J]. Advanced Functional Materials, 2013, 23(8): 947�C958. DOI:10.1002/adfm.201200691.
[5] KANG Hong-yan, LIU Yong-chang, CAO Kang-zhe, ZHAO Yan, JIAO Li-fang, WANG Yi-jing, YUAN Hua-tang. Update on anode materials for Na-ion batteries [J]. Journal of Materials Chemistry A, 2015, 3(35): 17899�C17913. DOI: 10.1039/C5TA03181H.
[6] YABUUCHI N, KUBOTA K, DAHBI M, KOMABA S. Research development on sodium-ion batteries [J]. Chemical Reviews, 2014, 114(23): 11636�C11682. DOI:10.1021/ cr500192f.
[7] LIU Yuan-yue, MERINOV B V, GODDARD W A. Origin of low sodium capacity in graphite and generally weak substrate binding of Na and Mg among alkali and alkaline earth metals [J]. Proceedings of the National Academy of Sciences of the United States of America, 2016, 113(14): 3735�C3739. DOI:10.1073/pnas.1602473113.
[8] DOEFF M M, MA Yan-ping, VISCO S J, DEJONGHE L C. Electrochemical insertion of sodium into carbon [J]. Journal of The Electrochemical Society, 1993, 140(12): 169�C170. DOI:10.1149/1.2221153.
[9] JIAN Ze-lang, ZHAO Bin, LIU Pan, LI Fu-jun, ZHENG Ming-bo, CHEN Ming-wei, SHI Yi, ZHOU Hao-shen. Fe2O3 nanocrystals anchored onto graphene nanosheets as the anode material for low-cost sodium-ion batteries [J]. Chemical Communications, 2014, 50(10): 1215�C1217 DOI: 10.1039/c3cc47977c.
[10] XIA Wei-wei, XU Feng, ZHU Chong-yang, XIN Huo-lin, XU Qing-yu, SUN Ping-ping, SUN Li-tao. Probing microstructure and phase evolution of ��-MoO3 nanobelts for sodium-ion batteries by in situ transmission electron microscopy [J]. Nano Energy, 2016, 27: 447�C456. https://doi.org/10.1016/j.nanoen.2016.07.017.
[11] QU Bai-hua, MA Chu-ze, JI Ge, XU Chao-he, XU Jing, MENG Y S, WANG Tai-hong, LEE J Y. Layered SnS2-reduced graphene oxide composite��A high-capacity, high-rate, and long-cycle life sodium-ion battery anode material [J]. Advanced Materials, 2014, 26(23): 3854�C3859. DOI:10.1002/adma.201306314.
[12] HU Zhe, WANG Li-xiu, ZHANG Kai, WANG Jian-bin, CHENG Fang-yi, TAO Zhan-liang, CHEN Jun. MoS2 nanoflowers with expanded interlayers as high-performance anodes for sodium-ion batteries [J]. Angewandte Chemie International Edtion in English, 2014, 126(47): 12794�C12798. DOI: 10.1002/anie.201407898.
[13] SUN Dan, YE De-lai, LIU Ping, TANG You-gen, GUO Jun, WANG Lian-zhou, WANG Hai-yan. MoS2/graphene nanosheets from commercial bulky MoS2 and graphite as anode materials for high rate sodium-ion batteries [J]. Advanced Energy Materials, 2017, 1702383�C1702394. DOI:10.1002/aenm.201702383.
[14] LIU Yong-chang, ZHANG Ning, JIAO Li-fang, TAO Zhan-liang, CHEN Jun. Ultrasmall Sn nanoparticles embedded in carbon as high-performance anode for sodium-ion batteries [J]. Advanced Functional Materials, 2015, 25(2): 214�C220. DOI: 10.1002/adfm.201402943.
[15] DARWICHE A, MARINO C, SOUGRATI M T, FRAISSE B, STIEVANO L, MONCONDUIT L. Better cycling performances of bulk Sb in Na-ion batteries compared to Li-ion systems: An unexpected electrochemical mechanism [J]. Journal of the American Chemical Society, 2012, 135(27): 20805�C20811. DOI:10.1021/ja310347x.
[16] QIAN Jiang-feng, WU Xian-yong, CAO Yu-liang, AI Xin-ping, YANG Han-xi. High capacity and rate capability of amorphous phosphorus for sodium ion batteries [J]. Angewandte Chemie International Edtion in English, 2013, 52(17): 4633�C4636. DOI: 10.1002/anie.201209689.
[17] XU Yang, LOTFABAD E M, WANG Huan-lei, FARBOD B, XU Zhan-wei, KOHANDEHGHAN A, MITLIN D. Nanocrystalline anatase TiO2: A new anode material for rechargeable sodium ion batteries[J]. Chemical Communications, 2013, 49(79): 8973�C8975. DOI:10.1039/ c3cc45254a.
[18] YU Peng-fei, LI Chi-lin, GUO Xiang-xin. Sodium storage and pseudocapacitive charge in textured Li4Ti5O12 thin films [J]. Journal of Physical Chemistry C, 2014, 118(20): 10616�C10624. DOI:10.1021/jp5010693.
[19] PAN Hui-lin, LU Xia, YU Xi-qian, HU Yong-sheng, LI Hong, YANG Xiao-Qing, CHEN Li-quan. Sodium storage and transport properties in layered Na2Ti3O7 for room-temperature sodium-ion batteries [J]. Advanced Energy Materials, 2014, 3(9): 1186�C1194. DOI:10.1002/aenm. 201300139.
[20] HE Han-na, ZHANG Qi, WANG Hai-yan, ZHANG He-he, LI Jia-dong, PENG Zhi-guang, TANG You-gen, SHAO Min-hua. Defect-rich TiO2-�� nanocrystals confined in a mooncake-shaped porous carbon matrix as an advanced Na ion battery anode [J]. Journal of Power Sources, 2017, 354: 179�C188. https://doi.org/10.1016/j.jpowsour.2017.04.035.
[21] HE Han-na, WANG Hai-yan, SUN Dan, SHAO Min-hua, HUANG Xiao-bing, TANG You-gen. N-doped rutile TiO2/C with significantly enhanced Na storage capacity for Na-ion batteries [J]. Electrochimica Acta, 2017, 236: 43�C52. DOI:10.1016/j.electacta.2017.03.104.
[22] HE Han-na, SUN Dan, ZHANG Qi, FANG Fu, TANG You-gen, GUO Jun, SHAO Min-hua, WANG Hai-yan. Iron doped cauliflower-like rutile TiO2 with superior sodium storage properties [J]. ACS Applied Materials & Interfaces, 2017, 9(7): 6093�C6103. DOI:10.1021/acsami.6b15516.
[23] HE Han-na, GAN Qing-meng, WANG Hai-yan Wang, XU Gui-liang, ZHANG Xiao-yi, HUANG Dan, FU Fang, TANG You-gen, AMINE K, SHAO Min-hua. Structure-dependent performance of TiO2/C as anode material for Na-ion batteries [J]. Nano Energy, 2018, 44: 217�C227. DOI:10.1016/j. nanoen.2017.11.077.
[24] ZHANG Qi, HE Han-na, HUANG Xiao-bing, YAN Jun, TANG You-gen, WANG Hai-yan. TiO2@C nanosheets with highly exposed (001) facets as a high-capacity anode for Na-ion batteries [J]. Chemical Engineering Journal, 2018, 332: 57�C65. DOI:10.1016/j.cej.2017.09.044.
[25] SUN Dan, XUE Xia, TANG You-gen, JING Yan, HUANG Bin, REN Yu, YAO Yan, WANG Hai-yan, CAO Guo-zhong. High-rate LiTi2(PO4)3@N-C composite via bi-nitrogen sources doping [J]. ACS Applied Materials & Interfaces, 2015, 7(51): 28337�C28345. DOI:10.1021/acsami.5b08697.
[26] SUN Dan, JIN Guan-hua, TANG You-gen, ZHANG Rui, XUE Xia, HUANG Xiao-bing, CHU Hai-liang, WANG Haiyan. NaTi2(PO4)3 nanoparticles embedded in carbon matrix as long-lived anode for aqueous lithium ion battery [J]. Journal of The Electrochemical Society, 2016, 163(7): A1388�CA1393. DOI:10.1149/2.1181607jes.
[27] PANG Gang, NIE Ping, YUAN Chang-zhou, SHEN Lai-fa, ZHANG Xiao-gang, LI Hong-sen, ZHANG Cun-liang. Mesoporous NaTi2(PO4)3/CMK-3 nanohybrid as anode for long-life Na-ion batteries [J]. Journal of Materials Chemistry A, 2014, 2(48): 20659�C20666. DOI:10.1039/c4ta04732j.
[28] HOU Zhi-guo, LI Xiao-na, LIANG Jian-wen, ZHU Young-chun, QIAN Yi-tai. An aqueous rechargeable sodium ion battery based on a NaMnO2�CNaTi2(PO4)3 hybrid system for stationary energy storage [J]. Journal of Materials Chemistry A, 2015, 3(4): 1400�C1404. DOI:10.1039/ c4ta06018k.
[29] ZHAO Bai-dan, WANG Qiu-yue, ZHANG Sen, DENG Chao. Self-assembled wafer-like porous NaTi2(PO4)3 decorated with hierarchical carbon as a high-rate anode for aqueous rechargeable sodium batteries [J]. Journal of Materials Chemistry A, 2015, 3(22): 12089�C12096. DOI:10.1039/ c5ta02568k.
[30] HE Zhang-xing, JIANG Ying-qiao, ZHU Jing, LI Yue-hua, JIANG Zhen, ZHOU Hui-zhu, MENG Wei, WANG Li, DAI Lei. Boosting the performance of LiTi2(PO4)3/C anode for aqueous lithium ion battery by Sn doping on Ti sites [J]. Journal of Alloy and Compounds, 2018, 731: 32�C38. DOI:10.1016/j.jallcom.2017.10.038.
[31] PANG Gang, YUAN Chang-zhou, NIE Ping, DING Bing, ZHU Jia-jia, ZHANG Xiao-gang. Synthesis of NASICON- type structured NaTi2(PO4)3-graphene nanocomposite as an anode for aqueous rechargeable Na-ion batteries [J]. Nanoscale, 2014, 6(12): 6328�C6334. DOI:10.1039/ c3nr06730k.
[32] LI Zheng, YOUNG D, XIANG Kai, CARTER W C, CHIANG Yet-ming. Towards high power high energy aqueous sodium-ion batteries: The NaTi2(PO4)3/Na0.44MnO2 System [J]. Advanced Energy Materials, 2012, 3(3): 290�C294. DOI:10.1002/aenm.201200598.
[33] WANG Cong, GUO Zi-yang, SHEN Wei, XU Qun-jie, LIU Hai-mei, WANG Yong-gang. B-doped carbon coating improves the electrochemical performance of electrode materials for Li-ion batteries [J]. Advanced Functional Materials, 2014, 24(35): 5511�C5521. DOI:10.1002/ adfm.201401006.
[34] HU Zhe, ZHANG Kai, GAO Hai-yan, DUAN Wen-chao, CHENG Fang-yi, LIANG Jing, CHEN Jun. Li2MnSiO4@C nanocomposite as a high-capacity cathode material for Li-ion batteries [J]. Journal of Materials Chemistry A, 2013, 1(40): 12650�C12656. DOI:10.1039/c3ta12106b.
[35] XIAO Zheng-wei, ZHANG Ying-jie, HU Guo-rong. Effect of different carbon precursors on properties of LiFePO4/C [J]. Journal of Central South University, 2015, 22(12): 4507�C4514. DOI:10.1007/s11771-015-2999-3.
[36] GENG Hong-bo, YANG Jun, YU Hong, LI Cheng-chao, DONG Xiao-chen. Carbon intercalated porous NaTi2(PO4)3 spheres as high-rate and ultralong-life anodes for rechargeable sodium-ion batteries [J]. Materials Chemistry Frontiers, 2017, 1(7): 1435�C1440. DOI:10.1039/ c7qm00048k.
[37] SEVILLA M, FUERTES A B. Chemical and structural properties of carbonaceous products obtained by hydrothermal carbonization of saccharides [J]. Chemistry-A European Journal, 2009, 15(16): 4195�C4203. DOI:10.1002/ chem.200802097.
[38] SUN Xiao-ming, LI Ya-dong. Colloidal carbon spheres and their core/shell structures with noble-metal nanoparticles [J]. Angewandte Chemie International Edtion in English, 2004, 43(5): 597�C601. DOI:10.1002/anie.200352386.
[39] ROH H K, KIM H K, KIM M S, KIM D H, CHUNG K Y, ROH K C, KIM K B. In situ synthesis of chemically bonded NaTi2(PO4)3/rGO 2D nanocomposite for high-rate sodium-ion batteries [J]. Nano Research, 2016, 9: 1844�C1855. DOI:10.1007/s12274-016-1077-y.
[40] ZHU Qing, NAN Bo, SHI Yang, ZHU Ying-gang, WU Si-si, HE Li-qing, DENG Yong-hong, WANG Li-ping, CHEN Quan-qi, LU Zhou-guang. Na3V2(PO4)3/C nanofiber bifunction as anode and cathode materials for sodium-ion batteries [J]. Journal of Solid State Electrochemistry, 2017, 21: 2985�C2995. DOI:10.1007/s10008-017-3627-y.
[41] WU Shao-fei, WANG Wei-xin, LI Min-chan, CAO Lu-jie, LYU Fu-cong, YANG Ming-yang, WANG Zhen-yu, SHI Yang, NAN Bo, YU Si-cen, SUN Zhi-fang, LIU Yao, LU Zhou-guang. Highly durable organic electrode for sodium- ion batteries via a stabilized ��-C radical intermediate [J]. Nature Communication, 2016, 7: 13318. DOI: 10.1038/ ncomms13318.
[42] LU Peng, HUANG Xiao-bing, REN Yu-rong, DING Jian-ning, WANG Hai-yan, ZHOU Shi-biao, CHEN Yuan-dao, DING Xiang. Na+ and Zr4+ co-doped Li4Ti5O12 as anode materials with superior electrochemical performance for lithium ion batteries [J]. RSC Advances, 2016, 6(93): 90455�C90461. DOI:10.1039/c6ra16717a.
[43] CHEN Jun-song, TAN Yi-ling, LI Chang-ming, CHEAH Yan-ling, LUAN De-yan, MADHAVI S, BOEY F Y C, ARCHER L A, LOU Xiong-wen. Constructing hierarchical spheres from large ultrathin anatase TiO2 Nanosheets with nearly 100% exposed (001) facets for fast reversible lithium storage [J]. Journal of the American Chemical Society, 2010, 132(17): 6124�C6130. DOI:10.1021/ja100102y.
[44] XU G B, YANG L W, WEI X L, DING J W, ZHONG J X, CHU P K. Hierarchical porous nanocomposite architectures from multi-wall carbon nanotube threaded mesoporous NaTi2(PO4)3 nanocrystals for high-performance sodium electrodes [J]. Journal of Power Sources, 2016, 327: 580�C590. DOI:10.1016/j.jpowsour.2016.07.089.
[45] SHEN Wei, LI Hui, WANG Cong, LI Zhi-hong, XU Qun-jie, LIU Hai-mei, WANG Yong-gang. Improved electrochemical performance of the Na3V2(PO4)3 cathode by B-doping of the carbon coating layer for sodium-ion batteries [J]. Journal of Materials Chemistry A, 2015, 3(29): 15190�C15201. DOI: 10.1039/c5ta03519h.
[46] WANG Shuang-yin, IYYAMPERUMAL E, ROY A, XUE Yu-hua, YU Ding-shan, DAI Li-ming. Vertically aligned BCN nanotubes as efficient metal-free electrocatalysts for the oxygen reduction reaction: A synergetic effect by co-doping with boron and nitrogen [J]. Angewandte Chemie International Edtion in English, 2011, 123(49): 11960�C11964. DOI:10.1002/anie.201105204.
[47] WANG Hai-bo, MAIYALAGAN Thandavarayan, WANG Xin. Review on recent progress in nitrogen-doped graphene: synthesis, characterization, and its potential applications [J]. ACS Catalysis, 2012, 2(5): 781�C794. DOI:10.1021/ cs200652y.
[48] REN Yu-rong, LU Peng, HUANG Xiao-bing, DING Jian-ning, WANG Hai-yan, ZHOU Shi-biao, CHEN Yuan-dao, DING Xiang. Spherical Li1.95Na0.05FeSiO4/C composite as nanoporous cathode material exhibiting high rate capability [J]. Materials Letters, 2016, 173: 207�C210. DOI:10.1016/j.matlet.2016.03.048.
[49] REN Yu-rong, LU Peng, HUANG Xiao-bing, DING Jian-ning, WANG Hai-yan. Synthesis and high cycle performance of Li2ZnTi3O8/C anode material promoted by asphalt as a carbon precursor [J]. RSC Advances, 2016, 6(55): 49298�C49306. DOI:10.1039/c6ra08698e.
[50] HUANG Zhi-feng, LIU Li, YI Ling-guang, XIAO Wei, LI Min, ZHOU Qian, GUO Guo-xiong, CHEN Xiao-ying, SHU Hong-bo, YANG Xiu-kang. Facile solvothermal synthesis of NaTi2(PO4)3/C porous plates as electrode materials for high-performance sodium ion batteries [J]. Journal of Power Sources, 2016, 325: 474�C481. DOI:10.1016/j.jpowsour. 2016.06.066.
[51] YANG Gong-zheng, SONG Hua-wei, WU Ming-mei, WANG Cheng-xin. Porous NaTi2(PO4)3 nanocubes: A high-rate nonaqueous sodium anode material with more than 10000 cycle life [J]. Journal of Materials Chemistry A, 2015, 3(36): 18718�C18726. DOI:10.1039/c5ta04491j.
[52] WANG Lei, WANG Bo, LIU Gui-jing, LIU Tie-feng, GAO Tian-tian, WANG Dian-long. Carbon nanotube decorated NaTi2(PO4)3/C nanocomposite for a high-rate and low- temperature sodium-ion battery anode [J]. RSC Advances, 2016, 6(74): 70277�C70283. DOI:10.1039/c6ra11042h.
[53] SUN Dan, TANG You-gen, HE Ke-jian, REN Yu., LIU Su-qin, WANG Hai-yan. Long-lived aqueous rechargeable lithium batteries using mesoporous LiTi2(PO4)3@C Anode [J]. Scientific Reports, 2015, 5: 17452. DOI: 10.1038/ srep17452.
[54] SUN Hong-tao, XIN Guo-qing, HU Tao, YU Ming-peng, SHAO Da-li, SUN Xiang, LIAN Jie. High-rate lithiation- induced reactivation of mesoporous hollow spheres for long-lived lithium-ion batteries [J]. Nature Communications, 2014, 5(7): 4526. DOI:10.1038/ncomms5526.
[55] HE Zhang-xing, JIANG Yi-fan, SUN Dan, DAI Lei, WANG Hai-yan. Advanced LiTi2(PO4)3/C anode by incorporation of carbon nanotubes for aqueous lithium-ion batteries [J]. Ionics, 2016, 23(3): 575�C583. DOI:10.1007/s11581-016-1828-5.
(Edited by HE Yun-bin)
���ĵ���
����̼���η����NaTi2(PO4)3�������ϵĴ�������
ժҪ��NASICON�ͽṹ��NaTi2(PO4)3����Ϊ�Ƿ�ˮ��Һ��ˮ��Һ�����ӵ�ؼ���Ӧ��ǰ���ĸ������ϣ������䴢�������ܵ��������ӵ絼�ʵ͵�������Լ�����IJ���Ԥ���Ʊ���̼��ͷ�ȩ��֬��Ϊ̼Դ������зֲ������������绯ѧ���ܡ�ʵ������ʾ��Ŀ����ϵĵ绯ѧ�����������ڵ����÷�ȩ��֬���εIJ��ϡ��ò�����0.5C�����¿������126.7 mA��h/g�ķŵ�����������ұ������ܺ�ѭ���ȶ�������Խ����1C��10C��20C�·ŵ�������ֱ�Ϊ115.1��95.5��80.8 mA��h/g����5C��ѭ��1000�κ����������ʸߴ�92.4%����������̼���η��IJ��ã����ϵ��ӵ絼�ʵõ������������ټ��ϲ��������ֲ��������Լ����Ƶĵ�������Эͬ���ã����ϱ������ܺ�ѭ���ȶ��Ծ��õ����Ը��ơ�
�ؼ��ʣ�����̼���Σ�NaTi2(PO4)3���������ܣ������ӵ��
Foundation item: Projects(21671200, 21571189) supported by the National Natural Science Foundation of China; Projects(2016TP1007, 2017TP1001) supported by the Hunan Provincial Science and Technology Plan Project of China; Project(2017CL17) supported by the Opening Project of Material Corrosion and Protection Key Laboratory of Sichuan Province, China; Project(2016CXS009) supported by Innovation-Driven Project of Central South University, China
Received date: 2017-12-15; Accepted date: 2018-03-21
Corresponding author: WANG Hai-yan, PhD, Associate Professor; Tel: +86�C731�C88830886; E-mail: wanghy419@csu.edu.cn; ORCID: 0000-0003-4206-0215

