���õͱ����Ʊ�������ǰ����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2013���8��
�������ߣ������� ����ΰ ������
����ҳ�룺2422 - 2430
�ؼ��ʣ��������������壻���Բ��ϣ����������ͱ���
Key words��nickel ferrite; ferrite; magnetic materials; co-precipitation; low grade nickel matte
ժ Ҫ���Եͱ���Ϊԭ�ϣ����ò����ι��������ϳɿ���ϸС��������ǰ����NiFe2(C2O4)3��6H2O��NiCl2-FeCl2-(NH4)2C2O4-H2O��ϵ������ѧ�о�������Ni2+��Fe2+��������ѹ�����pHֵΪ2�� ��Ni2+��Fe2+���Ӿ��н�ǿ��������á��������о��Ļ����ϣ�������������Գ����ʺ�ǰ�������ȵ�Ӱ�졣�����������ѹ���������Ϊ��ҺpH=2����Ӧ�¶�Ϊ45 ��C��(NH4)2C2O4������Ϊ����ֵ��1.2����PEG400������Ϊ3%���ڴ������£�Ni2+��Fe2+�ij����ʴ�99.8%������ǰ���������Ϊ1~2 um��XRD��TG-DTA��������������ǰ����Ϊ�����û������壬��Ӧ������������ԭ���ȡ����
Abstract: Fine nickel ferrite precursors NiFe2(C2O4)3��6H2O were obtained via co-precipitation method with low grade nickel matte as the raw material. Thermodynamic analysis of NiCl2-FeCl2-(NH4)2C2O4-H2O system for precipitation identified that the theoretical optimum co-precipitation pH value is 2, and has strong complexation with Ni2+ and Fe2+ ions. Based on these theoretical considerations, the effects of parameters on the precipitation rates and precursors size were investigated systematically. The results show that the optimum co-precipitation conditions are pH=2, temperature 45 ��C, 1.2 times theoretical amount of (NH4)2C2O4 dosage and 3% PEG400 addition. Under these conditions, the precipitation rates of Ni2+ and Fe2+ are both over 99.8%, with the precursors size of 1-2 um. Furthermore, X-ray diffraction (XRD) and thermogravimetry-differential thermal analysis (TG-DTA) demonstrate that the precursors are single-phase solid solution, wherein the nickel/iron atoms are replaced by the iron/nickel atoms reciprocally.
Trans. Nonferrous Met. Soc. China 23(2013) 2422-2430
Li-hua HE1, Zhong-wei ZHAO1, You-xin ZHANG1,2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Jinchun Group Co., Ltd., Jinchang 737104, China
Received 23 July 2012; accepted 8 May 2013
Abstract: Fine nickel ferrite precursors NiFe2(C2O4)3��6H2O were obtained via co-precipitation method with low grade nickel matte as the raw material. Thermodynamic analysis of NiCl2-FeCl2-(NH4)2C2O4-H2O system for precipitation identified that the theoretical optimum co-precipitation pH value is 2, and  has strong complexation with Ni2+ and Fe2+ ions. Based on these theoretical considerations, the effects of parameters on the precipitation rates and precursors size were investigated systematically. The results show that the optimum co-precipitation conditions are pH=2, temperature 45 ��C, 1.2 times theoretical amount of (NH4)2C2O4 dosage and 3% PEG400 addition. Under these conditions, the precipitation rates of Ni2+ and Fe2+ are both over 99.8%, with the precursors size of 1-2 um. Furthermore, X-ray diffraction (XRD) and thermogravimetry-differential thermal analysis (TG-DTA) demonstrate that the precursors are single-phase solid solution, wherein the nickel/iron atoms are replaced by the iron/nickel atoms reciprocally.
has strong complexation with Ni2+ and Fe2+ ions. Based on these theoretical considerations, the effects of parameters on the precipitation rates and precursors size were investigated systematically. The results show that the optimum co-precipitation conditions are pH=2, temperature 45 ��C, 1.2 times theoretical amount of (NH4)2C2O4 dosage and 3% PEG400 addition. Under these conditions, the precipitation rates of Ni2+ and Fe2+ are both over 99.8%, with the precursors size of 1-2 um. Furthermore, X-ray diffraction (XRD) and thermogravimetry-differential thermal analysis (TG-DTA) demonstrate that the precursors are single-phase solid solution, wherein the nickel/iron atoms are replaced by the iron/nickel atoms reciprocally.
Key words: nickel ferrite; ferrite; magnetic materials; co-precipitation; low grade nickel matte
1 Introduction
Spinel nickel ferrite (NiFe2O4) is one of the most promising magnetic materials. It has been widely used in the aspects of magnetic recording media and magnetic fluids for the storage and/or retrieval of information, magnetic resonance imaging enhancement, catalysis, magnetically guided drug delivery, sensors and pigments due to its magnetic and electrical properties with chemical and thermal stabilities [1-3]. Conventionally, NiFe2O4 powders are prepared by solid-state reaction method at high temperatures with a repeated grinding [4-6]. However, this approach has some conspicuous disadvantages such as larger particle size, broader particle size distribution and impurities. In contrast, wet chemical preparation routes such as co-precipitation[7,8], sol-gel [9-11], hydrothermal[12,13] and microemulsion method [14,15], have an indisputable advantage over solid-state reactions in achieving better homogeneity, in which the starting compounds were mixed on molecular level. Especially the co-precipitation method, as a commercially feasible process, has attracted much attention recently.
Remarkably, in the traditional processes, the preparation of NiFe2O4 always uses high-purity metal salts as the raw materials, and it undoubtedly increases production costs. Therefore, finding a simple and cost-effective route to prepare NiFe2O4 is quite attractive. In recent years, some research works have focused on the direct synthesis technologies by using the smelting slag or metallic ores as the raw materials [16,17]. For instance, AHMED [18] synthesized a manganese ferrite from non-standard raw materials using ceramic technique. Coincidentally, RASHAD [19] also prepared a manganese ferrite by using low-grade manganese ore. Compared with the traditional processes, these direct methods have many significant advantages such as low cost, high resource utilization and friendly environment. And for the preparation of nickel ferrite NiFe2O4, it just needs Ni and Fe two resources. Fortunately, low-grade nickel matte, as an intermediate product of nickel smelting processes, mainly contains three elements, i.e. Ni, Fe and S [20,21]. If Ni and Fe in the nickel matte are dissolved into solution, then it is possible to synthesize NiFe2O4 via precipitation method.
So, the aim of the present work is the synthesis of NiFe2O4 precursors from low-grade nickel matte via co-precipitation method. The study focused on the effects of process parameters such as pH of the solution, reaction temperature, the addition of ammonium oxalate and surfactant on the synthesis of NiFe2O4 precursors. Moreover, the morphology and crystal structure of the produced powders were also examined.
2 Experimental
The precursors of spinel nickel ferrite were prepared by the oxalate coprecipitation method after leaching low grade nickel matte with FeCl3 agent. Low grade nickel matte samples were provided by Jinchuan Group Co., Ltd., China, and its total chemical composition is listed in Table 1. Firstly, nickel matte sample was ground to <106 ��m, and dissolved under the conditions of reaction temperature of 85 ��C, reaction time of 2 h, FeCl3 dosage of 1.2 times to theoretical value and HCl initial concentration of 1 mol/L, corresponding with the Ni extraction rate of 97.5%. Secondly, pyrite was added to the leaching solution to convert the excess Fe3+ into Fe2+. During this process, the impurities such as Cu2+ and Pb2+ were removed simultaneously. Thirdly, after filtering the solution, and nickel chloride was added to the obtained filtrate for correction the Fe:Ni molar ratio to 2:1, and the chemical composition of the final solution is listed in Table 2. Finally, co-precipitation processes were operated under different pH values, reaction temperatures and polyethyleneglycol 400 (PEG 400) addition by using ammonium oxalate as the precipitant. Then the precipitated solution was filtered, washed several times with deionized water, and dried in a vacuum oven at 80 ��C for 10 h.
Table 1 Chemical composition of low-grade nickel matte (mass fraction, %)

Table 2 Chemical composition of final solution (g��L-1)

The chemical compositions of the raw materials and solutions were carried out via atomic absorption spectroscopy (WFX-120, Thermo Electron Corporation), the particle morphology was observed by SEM (JXA-8800R, JEOL Ltd.), the crystal structures of the samples were characterized by X-ray diffraction (XRD) with Cu K�� radiation (TTR III, Rigaku Corporation) at a speed of 4 (��)/min, and the TG-DTA curves of the samples were determined by SDTQ600 (TA Instrument Corporation) at a heating rate of 10 ��C/min in air atmosphere with the flow rate of 100 mL/min.
3 Thermodynamic analysis
To get early insights into the properties and behaviors of the solution and find optimal conditions for precipitation, thermodynamic analysis of the NiCl2- FeCl2-(NH4)2C2O4-H2O system was studied firstly. In this system, 34 ionic species were assumed to exist in the solution: H+, OH-, Cl-, HCl,  ,
, 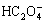 , H2C2O4(aq), NH3,
, H2C2O4(aq), NH3, 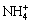 , Ni2+, Ni(OH)+, Ni(OH)2(aq),
, Ni2+, Ni(OH)+, Ni(OH)2(aq),  , NiC2O4(aq),
, NiC2O4(aq),  ,
,  , Ni(NH3)2+,
, Ni(NH3)2+, 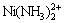 ,
,  ,
,  ,
,  ,
,  , NiCl+, Fe2+, Fe(OH)+, Fe(OH)2(aq),
, NiCl+, Fe2+, Fe(OH)+, Fe(OH)2(aq),  ,
, 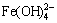 , FeC2O4(aq),
, FeC2O4(aq),  ,
,  , Fe(NH3)2+,
, Fe(NH3)2+,  , FeCl+, and 4 species as insoluble compounds were assumed to exist: NiC2O4(s), Ni(OH)2(s), FeC2O4(s), Fe(OH)2(s). This inventory leads to 37 equilibriums listed in Tables 3 and 4, in which equilibrium constant values were taken from Ref. [22].
, FeCl+, and 4 species as insoluble compounds were assumed to exist: NiC2O4(s), Ni(OH)2(s), FeC2O4(s), Fe(OH)2(s). This inventory leads to 37 equilibriums listed in Tables 3 and 4, in which equilibrium constant values were taken from Ref. [22].
For each equilibrium state, the concentration of each ion existing in the solution should not only satisfy its equilibrium reactions, but also the mass balance equations. In this work, the calculation model based on the Newton�CRaphson iteration theory was carried out by using Microsoft Excel.
Generally, the oxalate dosage and solution pH value may play significant roles in the process of oxalate co-precipitation. Figure 1 shows the effect of oxalate concentration on Ni2+ and Fe2+ co-precipitation. It can be seen that the concentration of nickel decreases in the pH range of 0-1.5, yet increases from pH 1.5-5, and stays flat at pH 5-8 under a certain oxalate concentration. The theoretical optimum pH value is 1-2, corresponding with the [Ni]T value of 10-4 mol/L. In addition, it is worthy to note that if the pH value is lower than the theoretical optimum value, the higher oxalate concentration in solution, the lower total nickel concentration will be, and the opposite situation happens when the operated pH value is larger than the theoretical optimum. The tendency of Fe2+ in lg[Fe]T vs pH (Fig. 1(b)) is similar to that of Ni2+ in lg[Ni]T vs pH (Fig. 1(a)), and the theoretical optimum pH for Fe2+ precipitation is 2-3 with the [Fe]T value of 10-3.5mol/L.
Table 3 Equilibrium reactions for precipitation system at 25 ��C

Table 4 Mass balance equations for precipitation system at 25 ��C


Fig. 1 Effect of oxalate concentrations on Ni2+ (a) and Fe2+ (b) precipitation
These results can be explained by the effect of complexation, i.e. the oxalate existing mainly in the form of H2C2O4 and/or  in strong acid aqueous medium and
in strong acid aqueous medium and  in weak acid and alkaline solution. So, for the pH<2, higher oxalate concentration in the solution can provide more
in weak acid and alkaline solution. So, for the pH<2, higher oxalate concentration in the solution can provide more  ions, which is beneficial for Ni2+ and Fe2+ co-precipitation. But in alkaline solution, excess
ions, which is beneficial for Ni2+ and Fe2+ co-precipitation. But in alkaline solution, excess  will strongly coordinate with Ni2+ and Fe2+, causing the obtained precipitate redissolution, which in turn leads to higher concentrations of [Ni]T and [Fe]T in solution. Actually, the coordination effect between Ni2+ and Fe2+ with
will strongly coordinate with Ni2+ and Fe2+, causing the obtained precipitate redissolution, which in turn leads to higher concentrations of [Ni]T and [Fe]T in solution. Actually, the coordination effect between Ni2+ and Fe2+ with  can clearly be seen from Fig. 2. For Ni2+, when pH value ranges from 0 to 8, nickel in solution mainly exists in the forms of Ni2+, NiC2O4(aq),
can clearly be seen from Fig. 2. For Ni2+, when pH value ranges from 0 to 8, nickel in solution mainly exists in the forms of Ni2+, NiC2O4(aq),  and
and 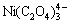 , and these for iron are Fe2+, FeC2O4(aq),
, and these for iron are Fe2+, FeC2O4(aq),  and
and  in order.
in order.
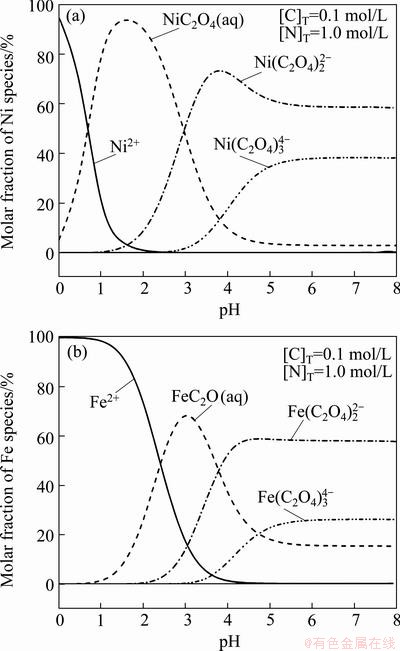
Fig. 2 Distribution of species containing Ni2+ (a) and Fe2+ (b) ions
Theoretically, in order to have a high Ni2+ and Fe2+ co-precipitation rate, it is essential to control the pH value precisely during the experiment.
4 Co-precipitation aspects
4.1 Effect of pH value
Based on the results of thermodynamic analysis, it is seen that the pH value has a major impact on the process of precipitation. Here, the precipitation experiments were carried out at pH 1, 1.5, 2, 2.5 and 3, respectively, with the reaction temperature of 55 ��C, 1.2 times theoretical amount of (NH4)2C2O4 and aging time of 30 min. The effect of pH value on the Ni2+ and Fe2+ precipitation rate is shown in Fig. 3.
It can be seen that the precipitation rate of Ni is kept around 100%, and that of Fe gradually increases from 94.1% to 99.5% in the pH range of 1-2, while both decrease with the pH value increasing from 2 to 3. It is because that the concentration of free oxalate in these experiments is about 0.2 mol/L, and the theoretical total concentrations of Ni and Fe are 10-4 and 10-1.75-10-3 mol/L under the condition of pH 1-2 (Fig. 1), respectively. It is hard for the Fe2+ ions to precipitate completely, especially at lower pH value. When the pH value is higher than 2, the complexation between Ni2+/Fe2+ and  is strong, which causes higher nickel and iron concentration in solution, resulting in a lower precipitation rate.
is strong, which causes higher nickel and iron concentration in solution, resulting in a lower precipitation rate.

Fig. 3 Effect of pH on precipitation rates of Ni2+ and Fe2+
Obviously, these experimental results are consistent with the information in the thermodynamic analysis. On the other hand, it also verifies the correctness of the theoretical analysis. In order to obtain NiFe2O4 powders in stoichiometric proportion, the optimum pH value is chosen to be 2.0. Under this condition, the precipitation rates of Ni2+ and Fe2+ are close.
4.2 Effect of (NH4)2C2O4 dosage
Figure 4 shows the influence of the amount of ammonium oxalate addition on the precipitation rates of Ni2+ and Fe2+ under the condition of 55 ��C, pH value of 2.0 and aging time of 30 min. As observed from Fig. 4, with the increasing ammonium oxalate dosage, the precipitation rate of Ni2+ keeps constant (about 100%), and that of Fe2+ improves from 94.2% to 99.8%, and then gradually stabilizes at 99.8% when the quantity of (NH4)2C2O4 is over 1.2 times the theoretical value. This can be attributed to the difference of solubility product constant. More specifically, the solubility product constant of NiC2O4(s) (Ksp=10-9.4) is smaller than that of FeC2O4(s) (Ksp=10-6.5), as listed in Table 1, and the Ni2+ ion will be preferentially precipitated when the (NH4)2C2O4 precipitant is added into the solution. Therefore, in order to ensure the Ni2+ and Fe2+ ions homogeneous precipitation, the ammonium oxalate addition must be controlled to a certain range.

Fig. 4 Effect of ammonium oxalate addition on precipitation rates of Ni2+ and Fe2+
Furthermore, not only the precipitation rate, but also the particle size of the precursors can be influenced by the ammonium oxalate addition. The more ammonium oxalate is added, the smaller size of the precursors will be (Fig. 5). This can be attributed to the complexation of the excess  with Ni2+ and Fe2+ in solution. On one hand, the complexation can avoid the grain growth too fast; On the other hand, it is helpful to refine the grain size and alleviate the particle agglomeration.
with Ni2+ and Fe2+ in solution. On one hand, the complexation can avoid the grain growth too fast; On the other hand, it is helpful to refine the grain size and alleviate the particle agglomeration.
Taking both the precipitation rates and particle size into consideration, the dosage of (NH4)2C2O4 is chosen to be 1.2-1.3 times the theoretical value in this work.
4.3 Effect of precipitation temperature
Unlike pH value and ammonium oxalate addition, the temperature in the process of co-precipitation has no obvious effect on the precipitation rates of Ni2+ and Fe2+, both of which are about 99.8% in the experimental temperature range of 35-75 ��C, as listed in Table 5. Nevertheless, the particle size and distribution of the precursors are strongly influenced by the reaction temperature. According to the results presented in Fig. 6, fine and well-crystallized precursors powders are obtained at a lower temperature, and the size of precursors increases obviously with the rising temperature, accompanying with serious aggregation phenomenon. In this study, the optimum reaction temperature can be chosen as 20-45 ��C.
4.4 Effect of PEG400 addition
As well as temperature, the effect of surfactant PEG 400 addition on the precipitate rates of Ni2+ and Fe2+ is puny (Table 6), and these results are similar with those of other research. In this work, both the precipitate rates of Ni2+ and Fe2+ are over 99.8%.
In fact, the main purpose of surfactant addition is to modify the size and distribution of the particles in the process of precipitation [23,24], rather than to improve the precipitation rate. As is well known, appropriate amount of surfactant addition is beneficial for obtaining smaller particles. As shown in Fig. 7, the precursors are severely agglomerated without PEG400, and the average particle size of precursors decreases with increasing PEG400 dosage. In this study, when the PEG400 dosage is 1% (mass fraction), the particle size is about 5 ��m; as the PEG400 addition increases to 4%, the particle size is decreased to 1-2 ��m.

Fig. 5 SEM images of precursors obtained at different ammonium oxalate addition
Table 5 Effect of reaction temperature on precipitation rates of Ni2+ and Fe2+

Table 6 Effect of PEG400 addition on precipitation rates of Ni2+ and Fe2+

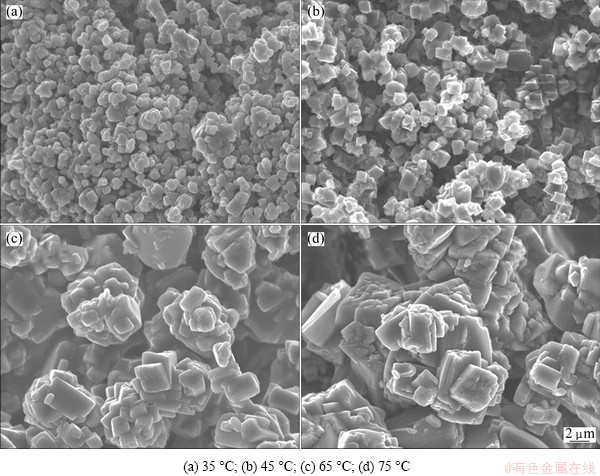
Fig. 6 SEM images of precursors obtained at different temperatures

Fig. 7 SEM images of precursors obtained with different PEG400 addition
4.5 Identification of Fe-Ni oxalate precursors
Generally, the properties of the NiFe2O4 product mainly depend on the morphology, purity and mixing degree of the precursors. Figure 10 shows the XRD patterns of the precursors obtained under the conditions of pH value of 2, temperature of 45 ��C, (NH4)2C2O4 dosage 1.2 times of stoichiometric value, and 3% PEG400 addition. Figure 8 shows the X-ray diffraction pattern of the prepared precursors. It can be seen that the XRD peaks of the precursors are sharp, which means that the precursors are well-crystallized. However, these peaks do not match those of NiC2O4��2H2O (JCPDS#25- 0581) or FeC2O4��2H2O (JCPDS#22-0355) exactly. By further studying the structure of the precursors, it is observed that the lattice parameters of the precursors lie between those of FeC2O4��2H2O and NiC2O4��2H2O phasess (Table 7). Therefore, it is speculated that the nickel/iron atoms replaced or substituted for the iron/nickel atoms reciprocally in the process of co-precipitation, with a substitutional solid solution being formed. It means that NiC2O4��2H2O and FeC2O4��2H2O are not formed independently and separately, but homogeneously.

Fig. 8 XRD pattern of precursor
Table 7 Lattice parameters of FeC2O4��2H2O, NiC2O4��2H2O and precursors

In order to confirm the above hypothesis, a comparative experiment was designed. First, NiC2O4��2H2O and FeC2O4��2H2O were prepared respectively via precipitation method by using NiCl2��6H2O, FeCl2��4H2O and (NH4)2C2O4 as the raw materials under the same conditions. A mechanical mixture was obtained by mixing the prepared NiC2O4��2H2O and FeC2O4��2H2O samples in a planetary ball mill at a speed of 400 r/min for 2 h. TG-DTA curves of different samples are illustrated in Fig. 9. It can obviously be seen that the difference between DTA curves belongs to different samples. For NiC2O4��2H2O, FeC2O4��2H2O and co-precipitation precursors, there is only one exothermic peak, and the exothermic peak temperature of co-precipitation precursor (283.82 ��C) is between those of FeC2O4��2H2O (242.71 ��C) and NiC2O4��2H2O (341.33 ��C). But for the mechanical mixed sample, there are two exothermic peaks, corresponding to 236.33 and 339.03 ��C, respectively. Comparing with the exothermic peak temperatures of NiC2O4��2H2O and FeC2O4��2H2O, it can be observed that the exothermic peaks of the mixture sample are matched with those of NiC2O4��2H2O and FeC2O4��2H2O samples, respectively. Thus, taking all these into consideration, it naturally comes to a conclusion that the precursor obtained from co-precipitation process is solid solution, and it is consistent with the result of XRD analysis. Furthermore, for the as-obtained precursor, the total water mass loss is 20.05% in the temperature range of 30-225 ��C (Fig. 9(d)), and it corresponds closely to the theoretical mass loss (19.9%) of mixture of NiC2O4��2H2O+ 2(FeC2O4��2H2O). Therefore, the chemical formula of the prepared precursors can be simply described as NiFe2(C2O4)3��6H2O.
Fig. 9 TG-DTA curves of different samples
Final NiFe2O4 product is obtained by calcining the precursors at 700 ��C for 4 h, and its SEM image and XRD pattern are illustrated in Fig. 10. The results indicate that the prepared NiFe2O4 product has a single and pure phase, and the size of the product is about 300 nm. The characteristics of NiFe2O4 product will be reported in the subsequent paper.

Fig. 10 SEM image (a) and XRD pattern (b) of final NiFe2O4 product after being calcined at 700 ��C for 4 h
5 Conclusions
1) Well-crystallized and fine NiFe2(C2O4)3��6H2O precursors for NiFe2O4 preparation were successfully synthesized via co-precipitation method by using the low-grade nickel matte as raw material.
2) Under the conditions of solution pH=2 and 1.2 times theoretical amount of (NH4)2C2O4 addition, both the precipitation rates of Ni2+ and Fe2+ are higher than 99.8%. The obtained precursor is substitutional solid solution, wherein the nickel/iron atoms replace or substitute for the iron/nickel atoms reciprocally, indicating that the iron and nickel are mixed on molecular level, which is beneficial for synthesizing stoichiometric NiFe2O4 in the subsequent heat treating process.
3) Pure and well-crystallized NiFe2O4 powders are obtained after being calcined at 700 ��C for 4 h, and the particle size of the NiFe2O4 powders is about 300 nm.
References
[1] PILENI M P. Magnetic fluids: Fabrication, magnetic properties, and organization of nanocrystals [J]. Advanced Functional Materials, 2001, 11(5): 323-336.
[2] SUGIMOTO M. The past, present, and future of ferrites [J]. Journal of the American Ceramic Society, 1999, 82(2): 269-280.
[3] SONG Q, ZHANG Z J. Shape control and associated magnetic properties of spinel cobalt ferrite nanocrystals [J]. Journal of the American Chemical Society, 2004, 126(19): 6164-6168.
[4] YANG H M, ZHANG X C, AO W Q, QIU G Z. Formation of NiFe2O4 nanoparticles by mechanochemical reaction [J]. Materials Research Bulletin, 2004, 39(6): 833-837.
[5] MUROI M, STREET R, McCORMICK P G, AMIGHIAN J. Magnetic properties of ultrafine MnFe2O4 powders prepared by mechanochemical processing [J]. Physical Review B, 2001, 63(18): 184414-184421.
[6] CEYLAN A, OZCAN S, Ni C, ISMAT S S. Solid state reaction synthesis of NiFe2O4 nanoparticles [J]. Journal of Magnetism and Magnetic Materials, 2008, 320(6): 857-863.
[7] CEYLAN A, OZCAN S, NI C, ISMAT S S. Investigation of nickel ferrite formation in a binary Fe (III)-Ni (II) hydroxide precipitate containing H2O with or without Li2O doping [J]. Journal of Alloys and Compounds, 2009, 486(1-2): 824-829.
[8] DOH S G, KIM E B, LEE B H, OH J H. Characteristics and synthesis of Cu-Ni ferrite nanopowders by coprecipitation method with ultrasound irradiation [J]. Journal of Magnetism and Magnetic Materials, 2004, 272-276: 2238-2240.
[9] CHEN D H, HE X R. Synthesis of nickel ferrite nanoparticles by sol-gel method [J]. Materials Research Bulletin, 2001, 36(7): 1369-1377.
[10] CHATTERJEE A, DAS D, PRADHAN S K, CHAKRAVORTY D. Synthesis of nanocrystalline nickel-zinc ferrite by the sol-gel method [J]. Journal of Magnetism and Magnetic Materials, 1993, 127(1): 214-218.
[11] GEORGE M, MARY J A, NAIR S S, JOY P A, ANANTHARAMAN M R. Finite size effects on the structural and magnetic properties of sol�Cgel synthesized NiFe2O4 powders [J]. Journal of Magnetism and Magnetic Materials, 2006, 302(1): 190-195.
[12] HUO J Z, WEI M Z. Characterization and magnetic properties of nanocrystalline nickel ferrite synthesized by hydrothermal method [J]. Materials Letters, 2009, 63(13): 1183-1184.
[13] ZHOU J, MA J F, SUN C, XIE L J, ZHAO Z Q, TIAN H, WANG Y G, TAO J T, ZHU X Y. Low temperature synthesis of NiFe2O4 by a hydrothermal method [J]. Journal of the American Ceramic Society, 2005, 88(12): 3535-3537.
[14] FANG J, SHAMA N, TUNG L D, SHIN E Y, O��CONNOR C J, STOKES K L. Ultrafine NiFe2O4 powder fabricated from reverse microemulsion process [J]. Journal of Applied Physics, 2003, 93(10): 7483-7485.
[15] MISRA R D K, KALE A, SRIVASTAVA R S, SENKOV O N. Synthesis of nanocrystalline nickel and zinc ferrites by microemulsion technique [J]. Materials Science and Technology, 2003, 19(6): 826-830.
[16] AHMED Y M Z, EWAIS E M M, ZAKI Z I. In situ synthesis of high density magnetic ferrite spinel (MgFe2O4) compacts using a mixture of conventional raw materials and waste iron oxide [J]. Journal of Alloys and Compounds, 2010, 489(1): 269-274.
[17] HESSIEN M M, RASHAD M M, HASSAN M S, EL-BARAWY K. Synthesis and magnetic properties of strontium hexaferrite from celestite ore [J]. Journal of Alloys and Compounds, 2009, 476(1-2) 373-378.
[18] AHMED Y M Z. Synthesis of manganese ferrite from non-standard raw materials using ceramic technique [J]. Ceramics International, 2010, 36(3): 969-977.
[19] RASHAD M M. Synthesis and magnetic properties of manganese ferrite from low grade manganese ore [J]. Materials Science and Engineering B, 2006, 127(2-3): 123-129.
[20] KELLOGG H H. Thermochemistry of nickel-matte converting [J]. Canadian Metallurgical Quarterly, 1987, 26(4): 285-298.
[21] DUCRET A, RANKIN W. Liquidus temperatures and viscosities of FeO-Fe2O3-SiO2-CaO-MgO slags at compositions relevant to nickel matte smelting [J]. Scandinavian Journal of Metallurgy, 2002, 31(1): 59-67.
[22] SPEIGHT J G. Lange��s handbook of chemistry, 6th Edition [M]. New York: McGRAW-HILL, 2005.
[23] WANG Jin, LIU Xiao-min, YANG Hui. Synthesis and electrochemical properties of highly dispersed Li4Ti5O12 nanocrystalline for lithium secondary batteries [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(3): 613-620.
[24] ZOU Min-min, AI Deng-jun, LIU Kai-yu. Template synthesis of MnO2/CNT nanocomposite and its application in rechargeable lithium batteries [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(9): 2010-2014.
������1������ΰ1��������1,2
1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. ���Źɷ�����˾����� 737104
ժ Ҫ���Եͱ���Ϊԭ�ϣ����ò����ι��������ϳɿ���ϸС��������ǰ����NiFe2(C2O4)3��6H2O��NiCl2-FeCl2-(NH4)2C2O4-H2O��ϵ������ѧ�о�������Ni2+��Fe2+��������ѹ�����pHֵΪ2�� ��Ni2+��Fe2+���Ӿ��н�ǿ��������á��������о��Ļ����ϣ�������������Գ����ʺ�ǰ�������ȵ�Ӱ�졣�����������ѹ���������Ϊ��ҺpH=2����Ӧ�¶�Ϊ45 ��C��(NH4)2C2O4������Ϊ����ֵ��1.2����PEG400������Ϊ3%���ڴ������£�Ni2+��Fe2+�ij����ʴ�99.8%������ǰ���������Ϊ1~2 um��XRD��TG-DTA��������������ǰ����Ϊ�����û������壬��Ӧ������������ԭ���ȡ����
��Ni2+��Fe2+���Ӿ��н�ǿ��������á��������о��Ļ����ϣ�������������Գ����ʺ�ǰ�������ȵ�Ӱ�졣�����������ѹ���������Ϊ��ҺpH=2����Ӧ�¶�Ϊ45 ��C��(NH4)2C2O4������Ϊ����ֵ��1.2����PEG400������Ϊ3%���ڴ������£�Ni2+��Fe2+�ij����ʴ�99.8%������ǰ���������Ϊ1~2 um��XRD��TG-DTA��������������ǰ����Ϊ�����û������壬��Ӧ������������ԭ���ȡ����
�ؼ��ʣ��������������壻���Բ��ϣ����������ͱ���
(Edited by Jing-hua FANG)
Foundation item: Project (2012BAB10B04) supported by National Key Technology R&D Program of China; Project supported by Hunan Provincial Innovation Foundation for Postgraduate, China
Corresponding author: Zhong-wei ZHAO; Tel: +86-731-88830476; E-mail: zhaozw@csu.edu.cn
DOI: 10.1016/S1003-6326(13)62750-8

