����ӵ�ظ�����������Li3-2xMgxV2(PO4)3/C�ĺϳɼ���绯ѧ����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2015���6��
�������ߣ�����÷ ������ ���� ��Ȩ�� ��Ȳ� ����־
����ҳ�룺1978 - 1985
Key words��lithium-ion batteries; lithium vanadium phosphate; diffusion coefficient; sol-gel method
ժ Ҫ�������ܽ�-�������Ʊ�Li3-2xMgxV2(PO4)3/C (x=0��0.01��0.03��0.05)���ϲ��ϣ���ͨ��X��������(XRD)��ɨ��羵(SEM)�͵绯ѧ���ԵȲ����ֶζԺϳɲ��Ͻ��б�����XRD�����������Li3V2(PO4)3���λ��������Mg2+����û�����Ըı�Li3V2(PO4)3����ĵ�б�ṹ����þ����Li3V2(PO4)3�ľ����������δ����Li3V2(PO4)3�ľ������������þ���Ӹ��ϲ���Li3-2xMgxV2(PO4)3/C (x=0.01��0.03��0.05)�ĵ绯ѧ���ܾ�����Li3V2(PO4)3/C���ϲ��ϣ����У�Li2.94Mg0.03V2(PO4)3/C ���������в����о�����ߵ���������õ�ѭ�����ܡ���Li3V2(PO4)3/C��Li2.94Mg0.03V2(PO4)3/C���������ɢϵ�����绯ѧ���ܽ��жԱȷ��������������Li+��þ����Li3V2(PO4)3�����еĿ�����ɢ����Mg2+���λ�IJ�����ɣ�Li+�ڻ��Բ����еĿ�����ɢʹþ����Li3V2(PO4)3���Ͼ��������ĵ绯ѧ���ܡ�
Abstract: The Li3-2xMgxV2(PO4)3/C (x=0, 0.01, 0.03 and 0.05) composites were prepared by a sol-gel method and characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM) and electrochemical measurements. The XRD results reveal that a small amount of Mg2+ doping into Li sites does not significantly change the monoclinic structure of Li3V2(PO4)3, but Mg-doped Li3V2(PO4)3 has larger cell volume than the pristine Li3V2(PO4)3. All Mg-doped composites display better electrochemical performance than the pristine one, and Li2.94Mg0.03V2(PO4)3/C composite exhibits the highest capacity and the best cycle performance among all above-mentioned composites. The analysis of Li+ diffusion coefficients in Li3V2(PO4)3/C and Li2.94Mg0.03V2(PO4)3/C indicates that rapid Li+ diffusion results from the doping of Mg2+ and the rapid Li+ diffusion is responsible for the better electrochemical performance of Mg-doped Li3V2(PO4)3/C composite cathode materials.
Trans. Nonferrous Met. Soc. China 25(2015) 1978-1985
Wu-mei YIN1, Ting-ting ZHANG1, Qing ZHU2, Quan-qi CHEN1,2, Gu-cai LI3, Ling-zhi ZHANG2
1. School of Chemistry, Xiangtan University, Xiangtan 411105, China;
2. College of Chemistry and Bioengineering, Guilin University of Technology, Guilin 541004, China;
3. College of Chemistry and Chemical Engineering, Hunan Institute of Engineering, Xiangtan 411104, China
Received 8 July 2014; accepted 26 October 2014
Abstract: The Li3-2xMgxV2(PO4)3/C (x=0, 0.01, 0.03 and 0.05) composites were prepared by a sol-gel method and characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM) and electrochemical measurements. The XRD results reveal that a small amount of Mg2+ doping into Li sites does not significantly change the monoclinic structure of Li3V2(PO4)3, but Mg-doped Li3V2(PO4)3 has larger cell volume than the pristine Li3V2(PO4)3. All Mg-doped composites display better electrochemical performance than the pristine one, and Li2.94Mg0.03V2(PO4)3/C composite exhibits the highest capacity and the best cycle performance among all above-mentioned composites. The analysis of Li+ diffusion coefficients in Li3V2(PO4)3/C and Li2.94Mg0.03V2(PO4)3/C indicates that rapid Li+ diffusion results from the doping of Mg2+ and the rapid Li+ diffusion is responsible for the better electrochemical performance of Mg-doped Li3V2(PO4)3/C composite cathode materials.
Key words: lithium-ion batteries; lithium vanadium phosphate; diffusion coefficient; sol-gel method
1 Introduction
The lithium-ion battery has been recognized as one of the most promising energy systems because of its high efficiency of energy conversion, high energy density and long cycle life, and its electrochemical performance is mainly dependent on cathode and anode materials. Recently, monoclinic Li3V2(PO4)3 cathode material has received extensive attention due to its high theoretical capacity, high operating voltage and excellent thermostability [1-3]. However, the low electronic conductivity (2��10-8 S/cm) [2] of Li3V2(PO4)3 degrades its electrochemical performance and limits its large-scale applications, especially in electric vehicles (EVs) and hybrid electric vehicles (HEVs). Numerous reports have confirmed that the addition of conductive carbon materials to Li3V2(PO4)3 can greatly enhance the electronic conductivity of the composite and hence to improve the electrochemical performance of Li3V2(PO4)3 [3-6]. However, the addition of conductive carbon materials cannot improve the intrinsic electronic conductivity of the electroactive materials and plays adverse roles in the enhancement of the density of the electrode materials due to the low density of the conductive carbon materials, which is unfavorable for the improvement of energy density. Compared with the addition of conductive carbon, the improvement of intrinsic conductivity of the electroactive materials is a more effective way to improve the electrochemical performance and energy density. It was reported that the intrinsic electronic conductivity of LiFePO4, which was much lower than that of Li3V2(PO4)3, was increased by 8 orders magnitude by partial substitution of Li+ with high-valent cations such as Zr4+ [7], Nb5+ [7], Mg2+ [7] and Cr3+ [8], and those doped LiFePO4 samples exhibited excellent electrochemical performance. However, to our knowledge, the doping of high valent cations except Ca2+ [9] in Li sites of Li3V2(PO4)3 has not been explored. Based on the consideration that the cations in lattice are easily replaced by other cations with similar radii and Mg2+ has a radius of 0.066 nm, similar to that of Li+ (r=0.068 nm), the monoclinic Mg-doped Li3-2xMgxV2(PO4)3 samples may be readily synthesized and the samples potentially exhibit better electro- chemical performance than the pristine Li3V2(PO4)3. In this work, Li3-2xMgxV2(PO4)3/C composites were prepared by a sol-gel method and the effects of Mg2+ content on the physical and electrochemical properties of Li3-2xMgxV2(PO4)3/C (x=0, 0.01, 0.03 and 0.05) composites were investigated.
2 Experimental
All the Li3-2xMgxV2(PO4)3/C (x=0, 0.01, 0.03 and 0.05) composites were prepared by a sol-gel method, which is similar to the previous reports [10,11]. The typical procedure of preparation of Li3-2xMgxV2(PO4)3/C is as follows: a certain amount of V2O5 powder was dissolved in 10% (volume fraction) hydrogen peroxide solution to form a clear brown solution, then citric acid with equivalent mole to V2O5 and stoichiometric amounts of NH4H2PO4, CH3COOLi��2H2 O and Mg(NO3)2��6H2O were added to the above brown solution with vigorous stirring. The mixture solution was aged with continuous stirring at 80 ��C for 10 h in thermostatic bath to obtain a dark green gel. The obtained gel was dried in a vacuum oven at 120 ��C, pelletized and calcined at 300 ��C for 4 h in a tubular furnace with the following argon gas to get a fluffy black product. The resulted product was reground and pelletized again, and sintered at 800 ��C for 8 h in argon atmosphere at a ramp rate of 2 ��C/min, and then cooled to room temperature to yield a black Li3-2xMgxV2(PO4)3/C composite.
The crystal structure of the resulted composites was analyzed by a Rigaku 2500 X-ray diffractometer (XRD) with Cu K�� radiation (��=0.15418 nm). The carbon content of the samples was determined by a carbon- sulfur analyzer (Mlti EA2000). The surface morphology of the samples was observed by a JSM-5600LV scanning electron microscope (SEM).
The electrochemical tests of Li3-2xMgxV2(PO4)3/C composites were carried out using 2032 coin cells. The cathodes were prepared by pasting the mixture of 80% Li3-2xMgxV2(PO4)3/C composite, 10% acetylene black and 10% polyvinylidene fluoride (PVDF) (mass fraction) onto an aluminum foil current collector. The coin cell was assembled in an argon-filled glove box using Li foil as the anode, 1 mol/L LiPF6 in a mixture of ethylene carbonate (EC) and dimethyl carbonate (DMC) (1:1, volume ratio) as the electrolyte, and Celgard 2400 as the separator. The coin cells were galvanostatically cycled at different current densities (1C=133 mA/g) in the voltage range of 3.0-4.3 V at 25 ��C using a NEWARE battery testing system. The specific capacity and current density were calculated based on the mass of the active materials. The measurements of electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV) were conducted on an IM6ex electrochemistry workstation (Zahner Elektrick, German).
3 Results and discussion
All the final products are black and contain about 3.6% carbon (mass fraction), which was determined by a carbon-sulfur analyzer. The XRD patterns of the final products are presented in Fig. 1. All diffraction peaks of the samples can be indexed by a well-defined monoclinic phase, which is consistent with monoclinic Li3V2(PO4)3 (PDF: 01-074-3236). The absence of diffraction peaks of impurity suggests that the final products are composites of a monoclinic phase and amorphous carbon, implying that a small amount of Mg2+ dopant does not significantly affect the monoclinic structure of Li3V2(PO4)3. The composites are denoted as Li3V2(PO4)3/ C, Li2.98Mg0.01V2(PO4)3/C, Li2.94Mg0.03V2(PO4)3/C and Li2.9Mg0.05V2(PO4)3/C, respectively, and the cell parameters of the corresponding compounds are listed in Table 1.
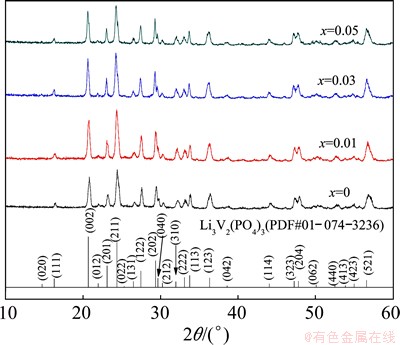
Fig. 1 XRD patterns of Li3-2xMgxV2(PO4)3/C composites
Table 1 Lattice parameters of Li3-2xMgxV2(PO4)3 compounds
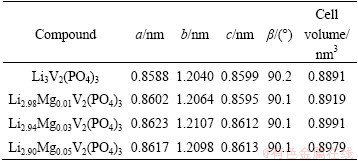
The results reveal that the cell parameters slightly increase with the addition of Mg. The cell volumes of Mg-doped Li3V2(PO4)3 compounds are somewhat larger than that of the pristine one and similar to the variation trends of that of Na-doped, K-doped and Ca-doped Li3V2(PO4)3 samples [9,12-14], and the partial substitution of monovalent Li+ (r=0.068 nm) by bivalent Mg2+ (r=0.066 nm) with approximate radius is responsible for the variation. The larger cell volume could provide larger channel for Li+ transport and facilitate rapid diffusion of Li+ in the active materials, which is beneficial to improving the electrochemical performance of Li3-2xMgxV2(PO4)3/C composites. Furthermore, the partial replacement of monovalent Li+ by bivalent Mg2+ produces vacancies in Li sites [7,15], which may be favorable to facilitating the transport of electrons and Li+, possibly resulting in much better electrochemical performance of the electroactive material.
The SEM images of Li3-2xMgxV2(PO4)3/C composites are shown in Fig. 2. It can be clearly seen that the composites display similar morphologies, indicating that the introduction of small amounts of Mg2+ into Li sites does not affect the morphologies of Li3V2(PO4)3/C composites.
To evaluate the effects of partial substitution of Li+ by Mg2+ on the electrochemical mechanism of Li3V2(PO4)3, the cyclic voltammetry tests at a scan rate of 0.05 mV/s in the potential range of 3.0-4.3 V were carried out on the Mg-doped and pristine composites, and the cyclic voltammograms (CVs) are presented in Fig. 3. All the Li3-2xMgxV2(PO4)3/C composites exhibit similar shape of the CV curves including three anodic peaks (denoted as a1, a2 and a3) and three cathodic peaks (labeled as c1, c2 and c3), suggesting that the dopant of Mg2+ in Li sites does not alter the mechanism of the insertion/extraction of Li+ into/from Li3V2(PO4)3 during the discharge-charge processes. However, compared with the pristine Li3V2(PO4)3/C sample, the Mg-doped samples display lower anodic peak potentials and higher cathodic peak potentials, which implies that the doped samples exhibit smaller polarization than the pristine one during the charge-discharge processes. The detailed peak potentials of the samples are listed in Table 2. The potential differences between the anodic peaks and the corresponding cathodic peaks of doped Li3V2(PO4)3 samples are lower than those of pristine Li3V2(PO4)3, and the potential difference of Li2.94Mg0.03V2(PO4)3/C is the smallest among all the samples, suggesting that Li2.94Mg0.03V2(PO4)3/C may have the best electrochemical performance.

Fig. 2 SEM images of Li3-2xMgxV2(PO4)3/C
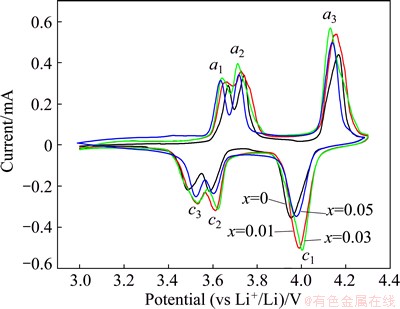
Fig. 3 Cyclic voltammograms of Li3-2xMgxV2(PO4)3/C composites at scan rate of 0.05 mV/s in potential range of 3.0-4.3 V at 25 ��C
Table 2 Potentials for CV peaks of Li3-2xMgxV2(PO4)3/C composites
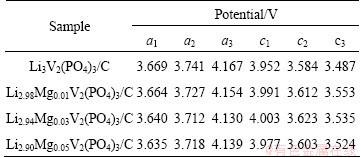

Fig. 4 Nyquist plots of Li3-2xMgxV2(PO4)3/C composites initially charged to 4.3 V (Equivalent circuit used for curve fitting is shown in inset)
Figure 4 shows the impedance behavior of Li3-2xMgxV2(PO4)3/C composites which are initially charged to 4.3 V, and all the composites display typical Nyquist characteristics. It can be seen that all the Nyquist plots are comprised of an intercept in the high frequency region, a depressed semicircle in the high-middle frequency region, and an inclined line in the low frequency region. The intercept impedance on the Z�� real axis represents ohmic resistance (Rs), which is the total resistance of the electrolyte, electrodes and contacts of the cell. The semicircle indicates the charge transfer process on the electrode interface, demonstrating the lithium transfer rate parameters as well as the capacitance of the electrolyte/SEI (solid electrolyte interface) double-layer. The inclined line depicts the Warburg impedance (ZW), which relates with the diffusion of Li+ in Li3-2xMgxV2(PO4)3 active particles. The constant phase element (CPE) stands for the double-layer capacitance.
The equivalent circuit shown in the inset of Fig. 4 was used to fit the plots, and the fitted results are listed in Table 3. The charge transfer resistance (Rct) of all Mg-doped Li3V2(PO4)3/C composites is lower than that of pristine Li3V2(PO4)3/C. The smaller Rct is favorable for rapid electrochemical reaction and may result in better electrochemical performance, and the smallest Rct for Li2.94Mg0.03V2(PO4)3/C indicates that Li2.94Mg0.03V2(PO4)3/C has the best electrochemical performance among the four composites.
Table 3 Fitting results for Nyquist plots of Li3-2xMgxV2(PO4)3/C composites

Figure 5 shows the initial charge-discharge profiles and the cycle performance of Li3-2xMgxV2(PO4)3/C composites (x=0, 0.01, 0.03 and 0.05) at 0.1C (13.3 mA/g) in the voltage range of 3.0-4.3 V at 25 ��C. Figure 5(a) demonstrates that all the Li3-2xMgxV2(PO4)3/C composites exhibit three charge voltage plateaus and three discharge ones, which implies that there exist three two-phase reactions in the electrochemical intercalation/ extraction of lithium in all the composites during the charge-discharge processes, which is in accordance with the results of CVs in Fig. 3.
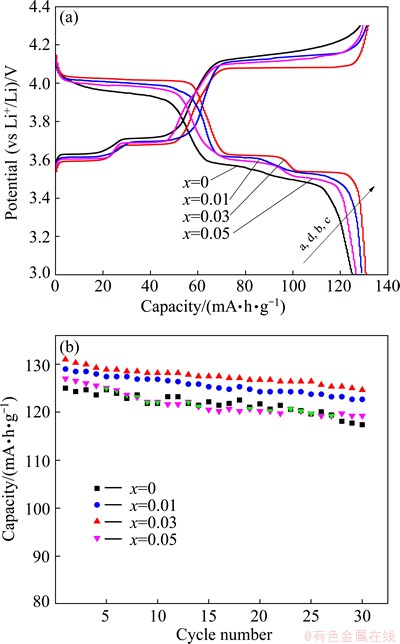
Fig. 5 Initial charge/discharge profiles (a) and cycle performance (b) of Li3-2xMgxV2(PO4)3/C composites at 0.1C in voltage range of 3.0-4.3 V at 25 ��C
Compared with the pristine Li3V2(PO4)3/C composite, the doped Li3V2(PO4)3/C composites display lower charge voltage plateaus and higher discharge ones, suggesting that the Mg2+ doping in Li sites favors decreasing the polarization during the charge and discharge processes. Among the above-mentioned doped samples, Li2.94Mg0.03V2(PO4)3/C shows the lowest charge voltage plateaus and the highest discharge ones with the highest charge and discharge capacities. By comparison with the initial discharge capacity of 125 mA��h/g for pristine Li3V2(PO4)3/C, the doped composites exhibit higher initial discharge capacity. As shown in Fig. 5(b), the initial discharge capacities for Li2.98Mg0.01V2(PO4)3/C, Li2.94Mg0.03V2(PO4)3/C and Li2.90Mg0.05V2(PO4)3/C are 129, 131 and 127 mA��h/g, respectively. After 30 cycles, the corresponding discharge capacities are 122, 125 and 119 mA��h/g, with the capacity retention rates of 94.6%, 95.4% and 93.7%, respectively, which are somewhat higher than that of 93.5% for the pristine Li3V2(PO4)3/C. The capacity of the Mg-doped Li2.94Mg0.03V2(PO4)3/C is much higher than that of Ca-doped Li2.96Ca0.02V2- (PO4)3/C (93 mA��h/g) [9] and K-doped Li2.85K0.15V2- (PO4)3/C (95 mA��h/g) [14], and the cyclability of Li2.94Mg0.03V2(PO4)3/C is also higher than those of Ca-doped and K-doped Li3V2(PO4)3/C samples, indicating that Mg2+ may be more suitable than Ca2+ and K+ to substitute Li+ of Li3V2(PO4)3 to improve the electrochemical performance of Li3V2(PO4)3.
Figure 6 depicts the initial charge-discharge profiles and the cycle performance of Li3-2xMgxV2- (PO4)3/C composites (x=0, 0.01, 0.03 and 0.05) at 1C (133 mA/g) in the voltage range of 3.0-4.3 V at 25 ��C. As observed in Fig. 6(a), the initial discharge capacities of Li3V2(PO4)3/C, Li2.98Mg0.01V2(PO4)3/C, Li2.94Mg0.03V2- (PO4)3/C and Li2.90Mg0.05V2(PO4)3/C are 117, 124, 127 and 121 mA��h/g, respectively, which are smaller than those of the corresponding composites at 0.1C, resulting from higher polarization at higher current density.
Among all the composites, Li2.94Mg0.03V2(PO4)3/C delivers the highest discharge capacity and the smallest voltage difference between charge and discharge voltage plateaus, suggesting that Li2.94Mg0.03V2(PO4)3/C has the lowest electrochemical polarization and results in better reversibility during the charge-discharge processes. The discharge capacity of Li2.94Mg0.03V2(PO4)3/C at 1C is much higher than those of Li2.96Ca0.02V2(PO4)3/C (80 mA��h/g) [9], Li2.9Na0.1V2(PO4)3/C (120.57 mA��h/g at 0.5C) [13] and Li3(V0.9Mg0.1)2(PO4)3/C (121 mA��h/g at 0.5C) [16], suggesting that Li2.94Mg0.03V2(PO4)3/C exhibits higher capacity and better rate capability than Li2.96Ca0.02V2(PO4)3/C, and Mg2+ doping in Li sites is more favorable than doping in V sites to improve the electrochemical performance, which might be caused by different amount of vacancies in Li sites for Li2.94Mg0.03V2(PO4)3/C and Li3(V0.9Mg0.1)2(PO4)3/C composites. Figure 6(b) illustrates that the four composites still keep good cycle performance at higher current density of 1C, which may be attributed to the excellent structural stability of the pristine and Mg-doped monoclinic Li3V2(PO4)3, further revealing that the doping of Mg2+ in Li sites does not affect the stable monoclinic structure of Li3V2(PO4)3. Li2.94Mg0.03V2- (PO4)3/C displays the highest discharge capacity and the best cyclability among the four composites, and exhibits capacity retention of 96.8% and still remains a capacity of 123 mA��h/g at 1C after 30 cycles, while the capacity retention rates of Li3V2(PO4)3/C, Li2.98Mg0.01V2(PO4)3/C and Li2.90Mg0.05V2(PO4)3/C are 95.9%, 95.6% and 93.2%, respectively. The above results indicate that the doping of Mg2+ in Li sites favors improving the capacity and cycle performance of Li3V2(PO4)3.
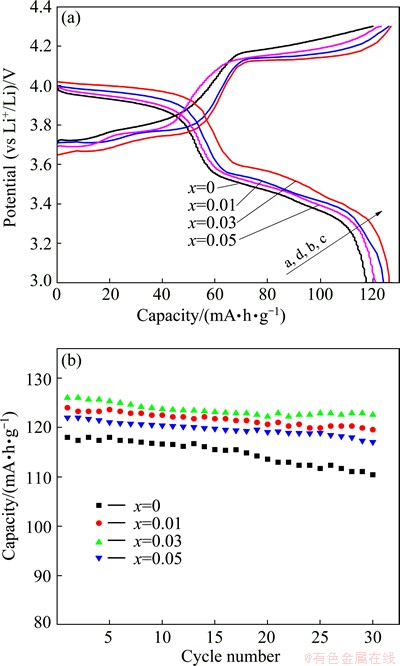
Fig. 6 Initial charge/discharge profiles (a) and cycle performance (b) of Li3-2xMgxV2(PO4)3/C composites at 1C in voltage range of 3.0-4.3 V at 25 ��C
In order to investigate the effect of Mg2+ doping on the diffusion of Li+ in the electroactive Li3-2xMgxV2(PO4)3 particles, the comparison of Li+ diffusion coefficient, DLi, in Li3V2(PO4)3/C and Li2.94Mg0.03V2(PO4)3/C was carried out. The diffusion coefficient of Li+ was determined by a CV method and DLi was calculated according to the classical Randles- Sevcik equation [17-20]:
 (1)
(1)
where Ip, n, A, 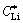 and v are the peak current (mA), the charge-transfer number, the electrode area (cm2), the bulk concentration of Li+ in the electrode (mol/cm3), and the potential scan rate (mV/s), respectively.
and v are the peak current (mA), the charge-transfer number, the electrode area (cm2), the bulk concentration of Li+ in the electrode (mol/cm3), and the potential scan rate (mV/s), respectively.
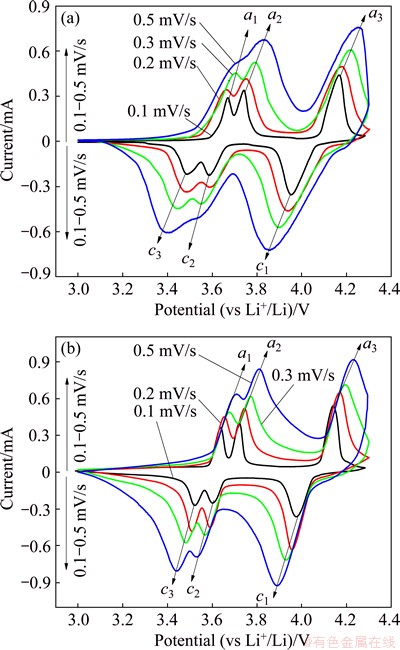
Fig. 7 Cyclic voltammograms of Li3V2(PO4)3/C (a) and Li2.94Mg0.03V2(PO4)3/C (b) composites at different scan rates in potential range of 3.0-4.3 V
Figures 7(a) and (b) show the CVs of Li3V2(PO4)3/C and Li2.94Mg0.03V2(PO4)3/C at different scan rates. It is obvious that the height and area of redox peaks increase with increasing the potential scan rates. The peak area divided by the scan rate should be equal to the total charge and discharge capacity of the same electrode based on the assumption of no polarization. Additionally, the anodic peaks shift to higher potentials and the corresponding cathodic peaks shift to lower potentials when the potential scan rate increases, implying that polarization becomes stronger due to the increase of scan rates. Although the polarization increases with increasing the potential scan rates, the shapes of CVs of Li2.94Mg0.03V2(PO4)3/C keep almost unchanged even at high scan rate of 0.5 mV/s, while two anodic peaks (a1 and a2) and two cathodic peaks (c2 and c3) of Li3V2(PO4)3/C emerge into one anodic peak and one cathodic peak, respectively, indicating that Li2.94Mg0.03V2(PO4)3/C displays smaller polarization and better reversibility than Li3V2(PO4)3/C.
As shown in Figs. 8(a) and (b), the peak current (Ip) has a linear relationship with the square root of scan rate (v1/2), revealing that it is reasonable to calculate the diffusion coefficient of Li+, DLi, by Eq. (1). Based on the slope of Ip versus v1/2 and Eq. (1), the diffusion coefficients of Li+ for Li3V2(PO4)3/C resulting from a2, a3, c1 and c3 are 3.7��10-11, 3.4��10-11, 4.9��10-11 and 4.4��10-11 cm2/s, respectively, while the corresponding diffusion coefficients of Li+ for Li2.94Mg0.03V2(PO4)3/C are 6.6��10-11, 5.2��10-11, 9.0��10-11 and 1.0��10-10 cm2/s, respectively. It is obvious that the Li+ diffusion coefficients of Li2.94Mg0.03V2(PO4)3/C are at least 1.5 times of the corresponding DLi of Li3V2(PO4)3/C, suggesting that Li2.94Mg0.03V2(PO4)3/C is more favorable to rapid diffusion of Li+ and the rapid diffusion of Li+ may result in better electrochemical performance of the electroactive materials. The difference of Li3V2(PO4)3/C and Li2.94Mg0.03V2(PO4)3/C in Li+ diffusion coefficients results from Mg2+ doping into Li sites. The Mg2+ doping plays key roles in improving Li+ diffusion coefficients. On one hand, the substitution of monovalent Li+ by bivalent Mg2+ produces vacancies in Li sites, which is beneficial to facilitating the transport of electrons and Li+, resulting in the improved rapid Li+ diffusion. On the other hand, Mg2+ doping enlarges the cell volume of the pristine Li3V2(PO4)3 and provides larger channel for Li+ transport in the electroactive materials, leading to the improved rapid Li+ diffusion.
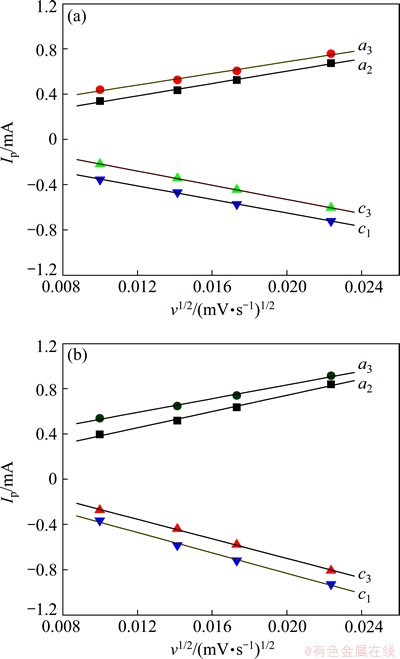
Fig. 8 Relationship between peak current (Ip) and square root of scan rate (v1/2) for CVs of Li3V2(PO4)3/C (a) and Li2.94Mg0.03V2(PO4)3/C (b)
4 Conclusions
1) Li3-2xMgxV2(PO4)3/C (x=0, 0.01, 0.03 and 0.05) composites were prepared by a sol-gel method and the composites are comprised of a single monoclinic phase of pristine Li3V2(PO4)3 or Mg-doped Li3V2(PO4)3 and amorphous carbon.
2) The cell volume of Mg-doped Li3V2(PO4)3 is somewhat larger than that of pristine Li3V2(PO4)3.
3) The electrochemical performances of all Mg-doped Li3V2(PO4)3/C are better than those of pristine Li3V2(PO4)3/C. Li2.94Mg0.03V2(PO4)3/C exhibits the best electrochemical performance among the four composites, and the capacities of Li2.94Mg0.03V2(PO4)3/C at 0.1C and 1C are 131 and 127 mA��h/g, respectively, which are higher than those of 125 mA��h/g (at 0.1C) and 117 mA��h/g (at 1C) for Li3V2(PO4)3/C. Furthermore, Li2.94Mg0.03V2(PO4)3/C displays better cycle performance at both 0.1C and 1C than Li3V2(PO4)3/C.
4) The comparative investigation on Li+ diffusion coefficients of Li3V2(PO4)3/C and Li2.94Mg0.03V2(PO4)3/C indicates that the partial substitution of monovalent Li+ by a suitable amount of bivalent Mg2+ is favorable to improving the diffusion rate of Li+ and results in high capacity and better cycle performance.
5) The results suggest that the partial substitution of Li+ by high valence of ions with similar radius may be an effective way to improve the electrochemical performance of other cathode and anode electrode materials for lithium-ion batteries.
References
[1] SAIDI M Y, BARKER J, HUANG H, SWOYER J L, ADAMSON G. Electrochemical properties of lithium vanadium phosphate as a cathode material for lithium-ion batteries [J]. Electrochemical and Solid State Letters A, 2002, 5(7): 149-151.
[2] YIN S C, GRONDEY H, STROBEL P, ANNE M, NAZAR L F. Electrochemical property: Structure relationships in monoclinic Li3-yV2(PO4)3 [J]. Journal of the American Chemical Society, 2003, 125(34): 10402-10411.
[3] RUI X H, YAN Q Y, SKYLLAS-KAZACOS M, LIM T M. Li3V2(PO4)3 cathode materials for lithium-ion batteries: A review [J]. Journal of Power Sources, 2014, 258: 19-38.
[4] HUAN H, YIN S C, KERR T, TAYLOR N, NAZAR L F. Nanostructured composites a high capacity, fast rate Li3V2(PO4)3/ carbon cathode for rechargeable lithium batteries [J]. Advanced Materials, 2002, 14(21): 1525-1528.
[5] LIU Guo-cong, LIU You-nian, LIU Su-qin. Grinding�Csol�Cgel synthesis and electrochemical performance of mesoporous Li3V2(PO4)3 cathode materials [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(2): 439-444.
[6] XIANG Wei, TANG Yan, WANG Yan-ying, ZHONG Ben-he, FANG Wei-mao, LIU Heng, GUO Xiong-dong. Influence of pH value and chelating reagent on performance of Li3V2(PO4)3/C cathode material [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(5): 1395-1402.
[7] CHUNG S Y, BLOKING J T, CHIANG Y M. Electronically conductive phospho-olivines as lithium storage electrodes [J]. Nature Materials, 2002, 1(2): 123-128.
[8] SHI S, LIU L, OUYANG C, WANG D S, WANG Z, CHEN L, HUANG X. Enhancement of electronic conductivity of LiFePO4 by Cr doping and its identification by first-principles calculations [J]. Physical Review B, 2003, 68(19): 195108.
[9] SUN C W, RAJASEKHARA S, DONG Y Z, GOODENOUGH J B. Hydrothermal synthesis and electrochemical properties of Li3V2(PO4)3/C-based composites for lithium-ion batteries [J]. ACS Applied Materials & Interfaces, 2011, 3(9): 3772-3776.
[10] CHEN Q Q, WANG J M, TANG Z, HE W C, SHAO H B, ZHANG J Q. Electrochemical performance of the carbon coated Li3V2(PO4)3 cathode material synthesized by a sol�Cgel method [J]. Electrochimica Acta, 2007, 52(16): 5251-5257.
[11] CHEN Q Q, QIAO X C, WANG Y B, ZHANG T T, PENG C, YIN W M, LIU L. Electrochemical performance of Li3-xNaxV2(PO4)3/C composite cathode materials for lithium ion batteries [J]. Journal of Power Sources, 2012, 201: 267-273.
[12] KUANG Q, ZHAO Y M, LIANG Z Y. Synthesis and electrochemical properties of Na-doped Li3V2(PO4)3 cathode materials for Li-ion batteries [J]. Journal of Power Sources, 2011, 196(23): 10169-10175.
[13] YAN J, YUAN W, XIE H, TANG Z Y, LIU F J, MAO W F, XU Q, ZHANG X H. Preparation and electrochemical performance of Na-doped Li3V2(PO4)3/C cathode material [J]. Journal of Solid State Electrochemistry, 2012, 16(10): 3201-3206.
[14] MATEYSHINA Y G, UVAROV N F. Electrochemical behavior of Li3-xM'xV2-yM''y(PO4)3 (M'=K, M''=Sc, Mg+Ti)/C composite cathode material for lithium-ion batteries [J]. Journal of Power Sources, 2011, 196(3): 1494-1497.
[15] RUAN Yan-li, TANG Zhi-yuan. Effects of Mg2+ doping on structure and electrochemical performance of LiFePO4 [J]. Transactions of Nonferrous Metals Society of China, 2005, 15(9): 1416-1420.
[16] DAI C S, CHEN Z Y, JIN H Z, HU X G. Synthesis and performance of Li3(V1-xMgx)2(PO4)3 cathode materials [J]. Journal of Power Sources, 2010, 195(17): 5775-5779.
[17] BARD A J, FAULKNER L R. Electrochemical methods: Fundametals and applications [M]. New York: John Wiley & Sons, Inc, 2001: 230-231.
[18] DAS S R, MAJUMDER S B, KATIYAR R S. Kinetic analysis of the Li+ ion intercalation behavior of solution derived nano-crystalline lithium manganate thin films [J]. Journal of Power Sources, 2005, 139(1-2): 261-268.
[19] TANG A P, WANG X Y, XU G R, ZHOU Z H, NIE H D. Determination of the chemical diffusion coefficient of lithium in Li3V2(PO4)3 [J]. Materials Letters, 2009, 63(16): 1439-1441.
[20] ZHANG X P, GUO H J, LI X H, WANG Z X, WU L. High tap-density Li3V2(PO4)3/C composite material synthesized by sol spray-drying and post-calcining method [J]. Electrochimica Acta, 2012, 64: 65-70.
����÷1��������1���� ��2����Ȩ��1,2����Ȳ�3������־ 2
1. ��̶��ѧ ��ѧѧԺ����̶ 411105��
2. ����������ѧ ��ѧ�����﹤��ѧԺ������ 541004��
3. ���Ϲ���ѧԺ ��ѧ����ѧԺ����̶ 411104
ժ Ҫ�������ܽ�-�������Ʊ�Li3-2xMgxV2(PO4)3/C (x=0��0.01��0.03��0.05)���ϲ��ϣ���ͨ��X��������(XRD)��ɨ��羵(SEM)�͵绯ѧ���ԵȲ����ֶζԺϳɲ��Ͻ��б�����XRD�����������Li3V2(PO4)3���λ��������Mg2+����û�����Ըı�Li3V2(PO4)3����ĵ�б�ṹ����þ����Li3V2(PO4)3�ľ����������δ����Li3V2(PO4)3�ľ������������þ���Ӹ��ϲ���Li3-2xMgxV2(PO4)3/C (x=0.01��0.03��0.05)�ĵ绯ѧ���ܾ�����Li3V2(PO4)3/C���ϲ��ϣ����У�Li2.94Mg0.03V2(PO4)3/C ���������в����о�����ߵ���������õ�ѭ�����ܡ���Li3V2(PO4)3/C��Li2.94Mg0.03V2(PO4)3/C���������ɢϵ�����绯ѧ���ܽ��жԱȷ��������������Li+��þ����Li3V2(PO4)3�����еĿ�����ɢ����Mg2+���λ�IJ�����ɣ�Li+�ڻ��Բ����еĿ�����ɢʹþ����Li3V2(PO4)3���Ͼ��������ĵ绯ѧ���ܡ�
�ؼ��ʣ�����ӵ�أ����ᷰﮣ���ɢϵ�����ܽ�-������
(Edited by Mu-lan QIN)
Foundation item: Project (12JJ3017) supported by the Natural Science Foundation of Hunan Province, China; Project (2013GXNSFAA019304) supported by the Natural Science Foundation of Guangxi Province; Project (51364007) supported by the National Natural Science Foundation of China
Corresponding author: Quan-qi CHEN; Tel: +86-773-5898551; E-mail: quanqi.chen@glut.edu.cn; quanqi.chen@yahoo.com
DOI: 10.1016/S1003-6326(15)63806-7

