���Ҷ�����ʮ����������廯臨Լ��Ե����п��Zn-Ni�Ͻ�ʴ��Эͬ��������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���1��
�������ߣ�Hany M. ABD EL-LATEEF Mahmoud ELROUBY
����ҳ�룺259 - 274
�ؼ��ʣ�ЭͬЧӦ����ʴ��п-���Ͻ𣻼��Ե��
Key words��synergistic effect; corrosion inhibition; Zn-Ni alloy; alkaline batteries
ժ Ҫ��ͨ������λ����/��������(PDP)�͵绯ѧ�迹��(EIS)�����۲���Ҷ���-400(PEG-400)��ʮ����������廯�(CTMAB)��п��Zn-Ni�Ͻ���ZnO���͵�8 mol/L KOH��Һ�и�ʴ��Эͬ�������á��绯ѧ�о�֤ʵ��PEG-400��CTMAB��������п����Ͻ�ĸ�ʴ����ЭͬЧӦ���о����֣�250 mg/L CTMAB + 250 mg/L PEG-400��ϻ�ʴ���Ļ�ʴЧ��Զ���ڵ�һ��ʴ��500 mg/L PEG-400��500 mg/L CTMAB�Ļ�ʴЧ������ʴʵ��ǰ��ֱ����Ʒ����ɨ��羵(SEM)�۲죬֤����ϻ�ʴ������ЭͬЧӦ����ˣ��ڼ�����Һ�м������о��Ļ�ʴ��������Ե�������ķŵ�Ч�ʺ����������õ绯ѧ������������������õ�����ԡ�
Abstract: The synergistic inhibition effect of poly(ethylene glycol)-400 (PEG-400) and cetyltrimethylammonium bromide (CTMAB) on the corrosion of Zn and Zn-Ni alloys in 8 mol/L KOH solution saturated with ZnO was observed by potentiodynamic anodic/cathodic polarization (PDP), and electrochemical impedance spectroscopy (EIS) measurements. The electrochemical studies confirmed that there was a synergism between PEG-400 and CTMAB on corrosion inhibition of Zn and its alloys. Corrosion inhibition efficiency of the mixed inhibitors, 250 mg/L CTMAB + 250 mg/L PEG-400, was found to be much higher than that of the single inhibitor, 500 mg/L PEG-400 or 500 mg/L CTMAB. Scanning electron microscopic (SEM) investigations before and after the corrosion inhibition process emphasize the synergistic effect of the mixed inhibitors. Accordingly, it was found that the addition of the investigated inhibitors to the alkaline solution enhanced the discharge and capacity of the alkaline battery anodes. The obtained electrochemical data exhibited a good correlation with the computational one.
Trans. Nonferrous Met. Soc. China 30(2020) 259-274
Hany M. ABD EL-LATEEF1,2, Mahmoud ELROUBY1
1. Department of Chemistry, Faculty of Science, Sohag University, Sohag 82524, Egypt;
2. Department of Chemistry, College of Science, King Faisal University, P. O. Box 380 Al Hofuf 31982 Al-Hassa, Saudi Arabia
Received 24 March 2019; accepted 15 July 2019
Abstract: The synergistic inhibition effect of poly(ethylene glycol)-400 (PEG-400) and cetyltrimethylammonium bromide (CTMAB) on the corrosion of Zn and Zn-Ni alloys in 8 mol/L KOH solution saturated with ZnO was observed by potentiodynamic anodic/cathodic polarization (PDP), and electrochemical impedance spectroscopy (EIS) measurements. The electrochemical studies confirmed that there was a synergism between PEG-400 and CTMAB on corrosion inhibition of Zn and its alloys. Corrosion inhibition efficiency of the mixed inhibitors, 250 mg/L CTMAB + 250 mg/L PEG-400, was found to be much higher than that of the single inhibitor, 500 mg/L PEG-400 or 500 mg/L CTMAB. Scanning electron microscopic (SEM) investigations before and after the corrosion inhibition process emphasize the synergistic effect of the mixed inhibitors. Accordingly, it was found that the addition of the investigated inhibitors to the alkaline solution enhanced the discharge and capacity of the alkaline battery anodes. The obtained electrochemical data exhibited a good correlation with the computational one.
Key words: synergistic effect; corrosion inhibition; Zn-Ni alloy; alkaline batteries
1 Introduction
Zn is vastly utilized as a negative effective substance for batteries (e.g. alkaline Mn batteries) because it has an economic feature and considerable energy capacity [1-4]. Zn has been added to Hg (mercury) in order to diminish the hydrogen gas evolution coming from the self-discharge reactions of Zn and increase the indoor cell impedance [5-9]. The application of Hg in batteries has become an environmental problem and the modern attitude has been focused on promoting Hg-free batteries. As usual, either Zn or Zn-based alloys are utilized as anodes (source of electrons) in the alkaline batteries that are of course subjected to dissolution. The purity of the used Zn is extremely high (99.9 wt.%), which affects the hydrogen evolution reaction and also the total cost. Subsequently, the development of Zn or Zn-based alloys as anodes for the alkaline-batteries has become a necessary demand and also in this regard, the development of Hg-free Zn demonstrates a great significance.
Surfactants have been considered to be promising substitutes for Hg in alkaline-batteries of zinc. Surfactant consists of two parts: the non-polar and polar groups [10]. The non-polar groups get away from the zinc surface while the polar-groups are adsorbed on the metal surface to form a protective film that protects Zn from corrosion [11,12]. The surfactant adsorption on Zn substrate affects not only the anodic reaction (Zn dissolution) but also the cathodic reaction (H2 evolution) in alkaline-batteries. Thus, the surfactants that could prohibit the cathodic branch but have less effect on the anodic branch are considered to be inhibition effective for the zinc corrosion in the alkaline-batteries. Many surfactant compounds have been utilized as inhibitors for the zinc corrosion in the alkaline media. It has been found that surfactant-based polyoxyethylene group is considered to be more efficient than other surfactants [11-13].
It is well recognized that the corrosion of Zn in alkaline media is cathodically under control. Consequently, the rate of the cathodic H2 evolution retards Zn corrosion. Therefore, the best method to minimize the corrosion process is to slow down the hydrogen evolution rate, which can be accomplished by the addition of small quantity of other metals as Pb, Bi, In and /or Al into Zn [14]. In our previous work [15] amounts of Ni (0.5%-10%) were added to zinc to enhance the electrochemical behavior, and extinguish the Zn anodic reaction. Nevertheless, still adding a compound (inhibitor) to the electrolyte solution which is able to decrease the corrosion rate of Zn and its alloy in alkaline-batteries media. Subsequently, the present investigation was devoted to studying the corrosion inhibition of Zn and its alloys with Ni by two environment-friendly surfactants, cetyltrimethyl- ammonium bromide (CTMAB) and polyethylene glycol 400 (PEG-400), which were used individually and simultaneously. PEG-400 and CTMAB were combined as a mixed inhibitor system and their inhibition capacity for the corrosion of Zn and its alloys was compared with that of PEG-400 or CTMAB, individually. Electrochemical techniques such as PDP and EIS were accomplished in order to examine the corrosion inhibition of the picking systems. Moreover, battery discharge performance of the investigated electrodes was studied using the discharge technique. Such computational assessments using the density functional theory (DFT) approach were performed on PEG-400 and CTMAB to specify the relationship between experimental inhibition capacity and their calculated energies.
2 Experimental
2.1 Materials and solutions preparation
The electrolyte solution was 8 mol/L KOH saturated with ZnO, which was used in commercial Zn-based batteries. KOH solution was prepared using doubly distilled water by dissolving the appropriate amount of KOH. The Zn-Ni alloys used in this study were prepared by metal fusion at 1273 K for 24 h. The molten mixture of metals was hardly shaken every 6 h to enhance the homogeneity of the formed Zn alloys. Finally, the alloy melts were quenched in ice as discussed in our previous work [16]. Two Zn-Ni alloys were prepared with the compositions of Zn-0.5Ni and Zn-10Ni (mass fraction, %). The composition and microstructure of the prepared Zn-0.5Ni and Zn-10Ni alloys were mentioned previously [8,9].
Figure 1 shows the molecular structures and abbreviation on use for the surfactants under investigation. These compounds were introduced to the corrosive medium at concentrations of 100-500 mg/L to test the inhibition on Zn and Zn-Ni alloys.

Fig. 1 Molecular structures of polyethylene glycol (PEG-400; n=9) (a) and cetyltrimethylammonium bromide (CTMAB) (b)
2.2 Electrochemical study
The electrochemical measurements were accomplished on the working disk electrode embedded in a Teflon holder. The metal and alloys surfaces were completely polished using emery papers with various roughness degrees (400, 600, 800, 1000 and 1200) and micro cloth until the surfaces become smooth like a bright mirror. Then, the electrodes were washed with pure ethanol and doubly distilled water and finally stored in a moisture-free desiccator. A saturated calomel electrode (Cl-(sat)/Hg2Cl2/Hg; SCE) and a Pt-sheet with a large surface area were used as reference (which all potentials are referred) and counter electrodes, respectively.
The cell description is given elsewhere [17]. To get rid of any oxide-layer on the Zn and Zn-Ni alloy surfaces, the electrodes were held at -1.5 V for 15 min before each experiment. A VersaSTAT4 potentiostat/galvanostatic instrument was utilized for the measurements.
2.2.1 Electrochemical impedance spectroscopy (EIS) measurement
EIS measurements were implemented using a VersaSTAT 4 potentiostat/galvanostatic instrument with a phase-sensitive detector (amplifier) (Model 5208) controlled by means of the frequency response analyzer (FRA) contained in a single unit. EIS measurements were achieved in 8 mol/L KOH saturated with ZnO at 30 ��C after 25 min immersion. The frequency ranged from 100 kHz to 0.5 Hz with amplitude of 10 mV during the experiment. All EIS experiments were accomplished at the potential of corrosion for the subjected electrode (��corr). The EIS data were fitted and analyzed using the Z-View software.
2.2.2 Potentiodynamic polarization technique
The PDP experiments were accomplished by using electrodes of surface area of 0.196 cm2. The anodic and cathodic polarization experiments were performed together at one experiment run. The potential was changed automatically from -0.25 up to 1.5 V vs (SCE) at a sweep rate of 1 mV/s.
2.2.3 Battery discharge performance
The studied electrodes of Zn alloys were assembled by using the discharge technique. The electrolyte was 8 mol/L KOH solutions saturated with ZnO in the presence of 500 mg/L PEG-400, and 500 mg/L CTMAB individually, and 250 mg/L PEG-400 + 250 mg/L CTMAB together at 25 ��C and constant current (10 mA).
2.3 Surface morphology
The micrographs of the inhibited and uninhibited Zn, Zn-0.5Ni and Zn-10Ni alloy surfaces were captured using scanning electron microscope (JEOL, model 5300). The energy of the accelerated beam was employed to be 10 kV and a working distance of 10 mm.
2.4 Computational methods
The quantum calculations based on the approximations of DFT approach were employed for the inhibitor structure. Energy calculations and optimization were implemented with B3LYP functional and 6-311G (d, p) basis set [18] as performed in Gaussian 03 program package [19].
3 Results and discussion
3.1 PDP measurements
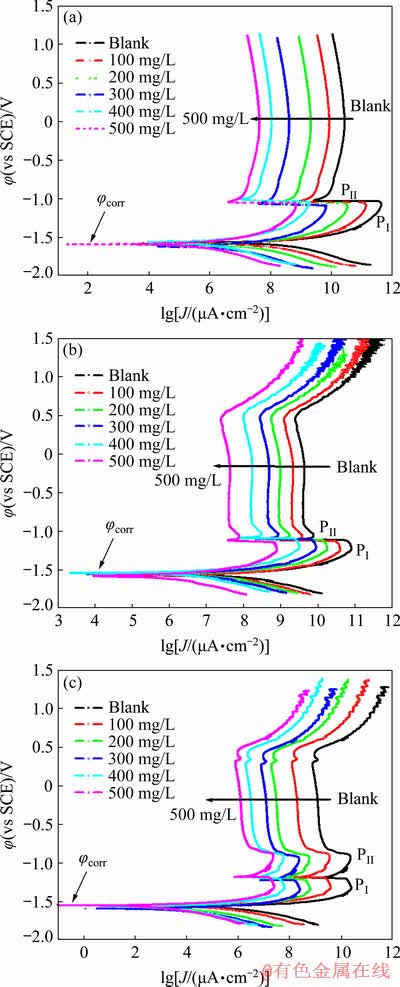
Fig. 2 Potentiodynamic polarization plots of Zn (a), Zn-0.5Ni (b) and Zn-10Ni (c) alloys in 8 mol/L KOH solution saturated with ZnO containing different concentrations of PEG-400+CTMAB (1:1, volume ratio) at 30 ��C and scan rate of 1 mV/s
Figures 2(a, b, c) display the potentiodynamic cathodic and anodic polarization plots of Zn, Zn-0.5Ni and Zn-10Ni alloys in ZnO-saturated 8 mol/L KOH solution containing various concentrations of PEG-400+CTMAB (volume ratio of 1:1) at 30 ��C. The polarization plots were swept from -0.25 mV vs the corrosion potential (��corr) to 1.5 mV (vs SCE). As seen from Fig. 2 the anodic polarization branches of Zn, Zn-0.5Ni and Zn-10Ni alloys exhibit an active/passive transmission. In the case of Zn, the dissolution current increases linearly with the utilized potential (in the active region), followed by the occurrence of two peaks (PI and PII). The first PI could be attributed to the Zn active dissolution to Zn2+ ions, while the second PII may be due to the dehydration of Zn(OH)2 to ZnO as previously reported [20,21].
In the same circumstances, the anodic polarization plots of Zn-0.5Ni alloy (Fig. 2(b)) showed two peaks, the first peak (PI) at -1.21 V (vs SCE) can be related to the electro-formation of ZnO layer. While the second peak (PII) at -1.055 V (vs SCE) is due to the Ni(OH)2 formation.
The potentiodynamic anodic ��-J curves of Zn-10Ni alloys (Fig. 2(c)) showed two active dissolution peaks (PI and PII), permanent passivation and eventually passive area breakdown. The two peaks (PI and PII) are well assigned at -1.33 and -0.929 V (vs SCE). These peaks are attributed to the formation of Zn(OH)2/ZnO and NiO/��-NiOOH, respectively.
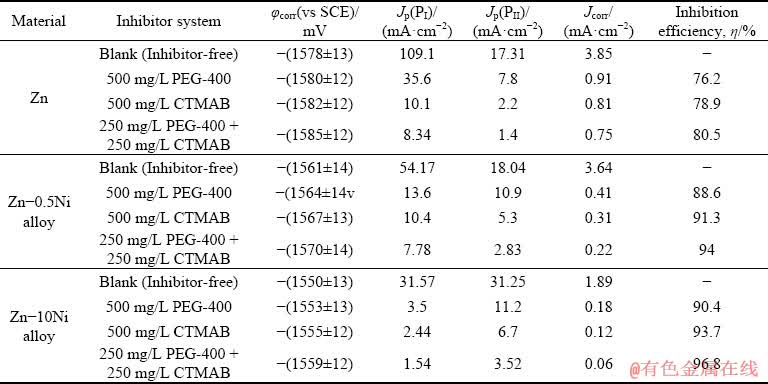
By the comparison between the anodic PDP plots of Zn and Zn-Ni alloys in ZnO-saturated 8 mol/L KOH solution (free inhibitors), it was found that, peak PI decreases while peak PII increases with increasing the Ni content in the alloy from 0.5% to 10% (Table 1). These outcomes indicate that, the content of Ni in the alloy composition plays a significant function in detraction of the dissolution rate of Zn in the studied medium. The increase in Ni content minimizes both the passivation current density (Jpass) and the anodic peak corrosion current density (Jpeak) of the alloys under the same conditions, subsequently progress their stability against corrosion. Accordingly, the obtained results displayed excellent resistance against corrosion of Zn-Ni alloy compared with Zn, and 10% Ni alloy showed superior corrosion resistance [22].
Table 1 Electrochemical parameters of Zn, Zn-0.5Ni and Zn-10Ni alloys derived from potentiodynamic polarization curves
The effect of various concentrations of PEG-400, CTMAB and PEG-400+CTMAB (1:1) on the cathodic/anodic potentiodynamic curves for Zn and studied Zn-Ni alloys in ZnO-saturated 8 mol/L KOH solution was precisely studied. The data showed that an increment in the inhibitor concentration leads to a decrease in the passivation current density (Jpass) as well as the peak height (Jp). This can be interpreted as the fact that the increase in the concentration of inhibitor facilitates the adsorption of the inhibitor molecule, and as a result, diminishes the peak and passivation current densities. The current density (Jcorr) and potential (��corr) of corrosion were detected at the point at which the extrapolations of the anodic and cathodic Tafel lines were intersected. The efficiency of corrosion inhibition (��) has been determined based on the following equation [23]:
 (1)
(1)
where Juninh and Jinh are corrosion current densities in the blank and inhibited solutions, respectively.
The data in Figs. 3(a) and (b) demonstrated that, Jcorr decreases, and �� increases as the inhibitor concentration (Cinh) increases. One can suggest from these results that a restriction in the electrodes processes takes place in both anodic and cathodic directions, because of the coverage of the metal and alloys surfaces by inhibitor species. However, the �� in the case of Zn-Ni alloys is higher than that of pure Zn at the same studied inhibitor concentrations. This manifests that the inhibitor molecule interacts more favorably with the alloy surface than that with the pure Zn [24]. It is also noticed that the inhibitor capacity promotes with increasing inhibitor concentration. This can be illustrated as that the adsorbed amount and the coverage area of surfactant molecule increase with increasing inhibitor concentration [25]. Furthermore, the ��corr partially shifts to more negative directions, and the shift increases with increasing the inhibitor concentrations of both pure Zn and its alloys (Fig. 3(c)). This implies that the changes in the hydrogen evolution overpotentials towards more negative values in the inhibited solutions have a positive impact on self-discharge and charge capacities [21]. This exhibits that the investigated inhibitors have capability to adsorb and prohibit the active dissolution centers on the surfaces of Zn and Zn-Ni alloys.
Figure 4 shows the dependence of the breakdown potential (��b) on the concentration of CTMAB for Zn-0.5Ni and Zn-10Ni alloys in 8 mol/L KOH solution saturated with ZnO at 30 ��C. It is also perceived that the ��b of the oxide layer formed on the alloy surfaces shifts to a less positive potential value with increasing the CTMAB concentration. These outcomes indicated that the inhibitor is preferentially adsorbed on the surface of alloys, therefore preventing oxide development. Therefore, the reduction process of the oxide film on surface of alloy takes place more easily in the presence of inhibitor than that in the absence of inhibitor. Subsequently, the addition of inhibitor to the corrosive media reinforces the electrochemical reactions of the alloys in the passive area, contributes to repression of H2 evolution and diminishes the corrosion rate. Moreover, the surface of alloy is reactivated in the passive area.
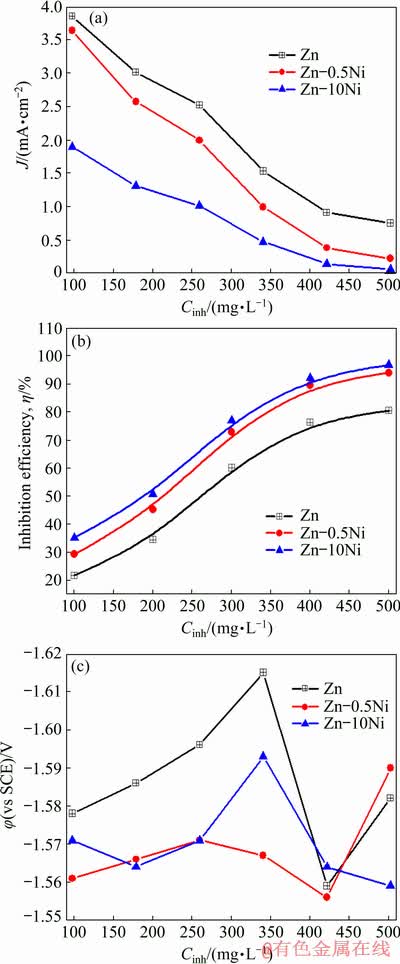
Fig. 3 Relationship between corrosion current density, Jcorr (a), inhibition efficiency, �� (b) and corrosion potential, ��corr (c) of Zn, Zn-0.5Ni and Zn-10Ni alloys in ZnO-saturated 8 mol/L KOH solution containing different concentrations of PEG-400+CTMAB (1:1, volume ratio) at 30 ��C
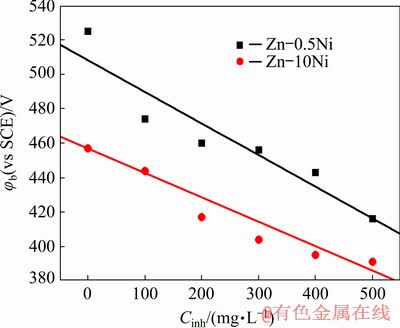
Fig. 4 Dependence of breakdown potential ��b on concentration of CTMAB for Zn-0.5Ni and Zn-10Ni alloys in ZnO-saturated 8 mol/L KOH solution at 30 ��C
Figure 5 shows the anodic/cathodic potentio- dynamic polarization curves of Zn and Zn-10Ni alloy in ZnO-saturated 8 mol/L KOH solution containing various inhibitors. Figure 5 indicates that the inhibitors reduce both cathodic and anodic current densities. This elucidates that the inhibitors prohibit both the cathodic and anodic reactions for the corrosion of Zn and its alloys.
The anodic dissolution processes of Zn and its alloys are also polarized by the inhibitor addition but the anodic dissolution reaction is not affected so dramatically as that of H2 evolution processes.
It is noticed that there is no distinction in the polarization of the anodic dissolution process between the two inhibitors and their composites. This indicates that the enhancement of corrosion inhibition of Zn and its alloys by the PEG-400 and CTMAB inhibitors fundamentally refers to their effect on H2 evolution reaction.
It can be observed from Table 1 that PEG-400 and/or CTMAB can protect Zn and its alloys with Ni from corrosion to different ranges. The �� values of the studies inhibitor systems in the same conditions are permanently in the sequence of (from high to low) PEG-400+CTMAB > CTMAB > PEG-400, indicating that there is a synergism between PEG-400 and CTMAB on the surfaces of Zn and Zn-Ni alloys. It is recognized that the effect of synergistic inhibition due to the joint behavior of compounds in total impact is greater than the sum of the singular effects.
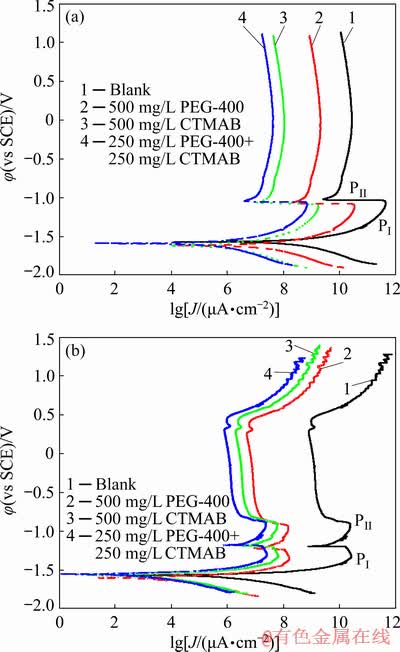
Fig. 5 Potentiodynamic polarization curves of Zn (a) and Zn-10Ni alloy (b) in ZnO-saturated 8 mol/L KOH solution at 30 ��C
Among the compounds investigated in this study, the interaction through the hydrogen bonding between oxygen atoms in polyethylene groups of PEG-400 molecules and surface metal hydroxyl groups on Zn and its alloys surfaces is predominant. Moreover, the hydrocarbon tails of the CTMAB surfactant would contribute to the expected enhancement of the adsorption density and consequently, the improvement of the inhibition efficiency of PEG/CTMAB system.
3.2 EIS spectra
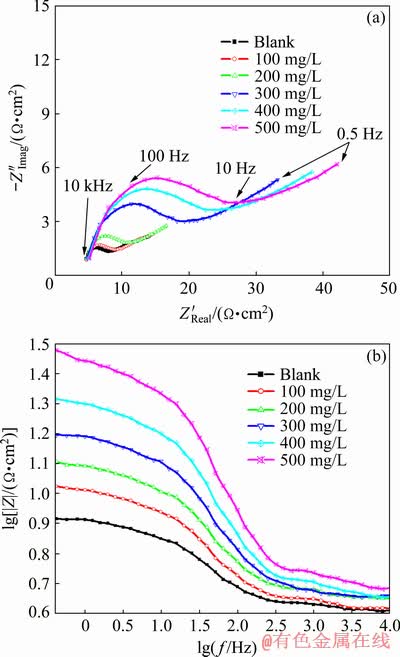
Fig. 6 Nyquist plot (a) and Bode plot (b) for Zn in ZnO-saturated 8 mol/L KOH solution containing various concentrations of PEG-400+CTMAB (volume ratio 1:1) measured at ��corr and 30 ��C
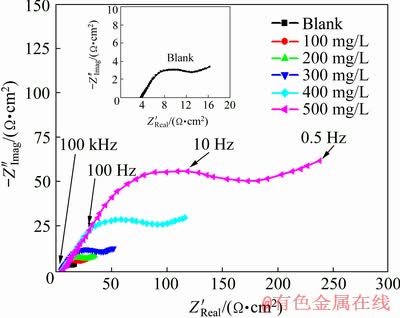
Fig. 7 Nyquist plot of Zn-0.5Ni alloy in ZnO-saturated 8 mol/L KOH solution containing various concentrations of PEG-400+CTMAB (volume ratio 1:1) measured at ��corr and 30 ��C
It is well known that the EIS is a helpful method for investigating the mechanism of corrosion processes and adsorption phenomena. In order to back the data obtained from the PDP measurements, EIS of Zn and its alloys in ZnO-saturated 8 mol/L KOH solutions containing different concentrations of PEG-400, CTMAB and PEG-400+CTMAB (1:1) was examined. Figures 6(a) and 7 show the Nyquist plots for Zn and Zn-0.5Ni alloy in ZnO-saturated 8 mol/L KOH solutions in the absence and presence of various concentrations of PEG-400+CTMAB (1:1), measured at ��corr and 30 ��C. The obtained data of the impedance spectra in the inhibited and uninhibited solutions were analyzed and interpreted. The data of the charge transfer resistance, Rct, and constant phase element (CPE) describing the interface double-layer capacity (Qcdl) were calculated via both Nyquist and Bode curves of the impedance diagrams. However, the Warburg impedance (Zw) is estimated from the following equations [26]:
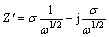 (2)
(2)
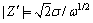 (3)
(3)
where �� is angular frequency, �� is the Warburg parameter, ��, can be accurately detected from the slope of the Warburg plot (the slope of Z�� (real part) vs 1/��1/2; ��=2��f, f is frequency, or by fitting to an equivalent circuit model which includes a Warburg impedance. Nevertheless, most programs of equivalent circuit modeling return ��Zw�� instead of ��, Zw is the Warburg impedance (Zw =W) which can be determined by the formula given as follows:
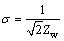 (4)
(4)
The recorded plot displays one capacitive loop at the higher values (HF) frequency followed by the Warburg tail at lower frequency (LF) values (Fig. 6(a)). Figures 6(a) and 7 exhibit that Zw is the Warburg impedance related to the anodic diffusion of soluble species from the metal and alloys surfaces to the solution bulk [27,28]. The capacitive loop diameter Rct, and Zw increase while the double- layer capacity (Qcdl) decreases with increasing inhibitor concentration. These results may give clear proof of the inhibition of the corrosion process.
Electrochemical impedance parameters for Zn, Zn-0.5Ni and Zn-10Ni alloys in 8 mol/L KOH solutions saturated with ZnO at ��corr and 30 ��C are shown in Table 2.
The Bode modules of the lg |Z| against lg f of Zn electrode in the absence and presence of various concentrations of PEG-400+CTMAB (1:1) are presented Fig. 6(b). The LF straight line confirms that the corrosion of Zn in ZnO-saturated 8 mol/L KOH solutions is diffusion-controlled. It is recognized that the Zn anodic dissolution and the cathodic O2 reduction processes occur simulta- neously on the electrode surface. The process of diffusion could be ascribed to either the migration of aggressive ions and soluble products of corrosion at interface of electrode/environment or the dissolved oxygen diffusion to the surface of Zn.
Table 2 Electrochemical impedance parameters for Zn, Zn-0.5Ni and Zn-10Ni alloys in 8 mol/L KOH solutions saturated with ZnO at ��corr and 30 ��C
The EIS plot of Zn-10Ni alloy in 8 mol/L KOH solutions saturated with ZnO containing various concentrations of PEG-400+CTMAB (1:1) is presented in Fig. 8. This diagram has a similar behavior throughout all investigated conditions. This proves that there is nearly no variation in the mechanism of corrosion after the addition of the inhibitor [29]. It can be observed that the impedance spectra comprise a large capacitive loop at HF values and an inductive one at LF values. Furthermore, the capacitive loop at HF is ascribed to the electron charge transfer and the double-layer. Whilst, the inductive loop at LF is due to the by-adsorption of inhibitor molecules on the surface of the alloy [30]. Further examination reveals that the diameter of the capacitive loop is increased with increasing inhibitor concentration which manifests the adsorption of inhibitors on the alloy surface. Consequently, the inhibitor adsorption leads to a clear increase in polarization resistance and then the corrosion rate of Zn-10Ni alloy is reduced.
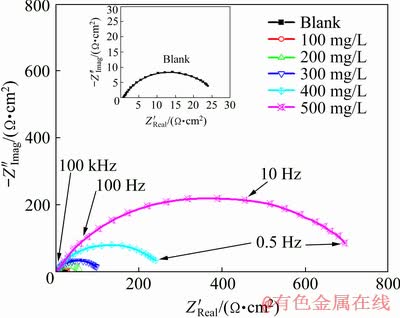
Fig. 8 Nyquist plot of Zn-10Ni alloy in ZnO-saturated 8 mol/L KOH solution containing various concentrations of PEG-400+CTMAB (volume ratio 1:1) measured at ��corr and 30 ��C

Fig. 9 Equivalent circuit models used for EIS data of semicircle with Warburg tail (Zn and Zn-0.5Ni alloy) (a) and semicircle without Warburg tail (Zn-10Ni alloy) (b)
The equivalent circuit models (ECM) used to fit the empirical data in Figs. 6(a), 7 and 8 are presented in Figs. 9(a) and (b), respectively. The impedance diagrams including Warburg impedance are analyzed utilizing the ECM shown in Fig. 9(a) and without Warburg tail in Fig. 9(b). It can be noticed that the obtained Nyquist diagram is analogous to that stimulated by the ECM. From Rct, Zw and Qcdl are estimated by the analysis of the complex-plane impedance plot and the ECM. From careful inspection of data obtained in Table 2, it was found that, the values of both Rct and Zw increase while Qcdl decreases with an increment in the inhibitor concentration, and these outcomes are convenient with those obtained from the PDP measurements. Meanwhile, the corrosion inhibition performance was reinforced with an increase of inhibitor concentration. It may be explained that the adsorption amount and the inhibitor coverage on the metal and alloy surfaces increase with increasing dose. Moreover, the addition of the inhibitor does not mutate the feature of the impedance diagram. This indicates that comparable corrosion mechanisms for Zn and Zn-0.5Ni alloy in ZnO- saturated 8 mol/L KOH in the presence and absence of the inhibitors can occur.
From Fig. 10, it is clear that after the inhibitors PEG-400, CTMAB and PEG-400+CTMAB are added to the solution, Rct increases, but Qcdl decreases dramatically in all cases compared with the blank solution. However, a maximum increase of Rct is noticed when the solutions consist of PEG-400+CTMAB. This attitude is related to the increase of the surface coverage via the inhibitor molecule, which increases the efficiency of inhibition. The values of inhibition capacity (��) are accurately determined from the values of Rct as claimed by the following equation [31,32]:
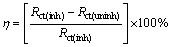 (5)
(5)
where Rct(inh) and Rct(uninh) are the resistances of charge transfer in the inhibited and uninhibited solutions, respectively. The addition of PEG-400, CTMAB and PEG-400+CTMAB results in lower Cdl values. Based on the model of Helmholtz [33],
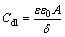 (6)
(6)
where ��0 can be defined as the permittivity of the vacuum (constant value), �� is the dielectric constant of the medium (tabulated value), �� is the thickness of the protective film of the inhibitor and A is the metal and alloy surface area. Thus, the diminution of the value of double-layer capacitance (Cdl) is due to the decrease of the local dielectric constant and/or the increase of the electrical double-layer thickness.
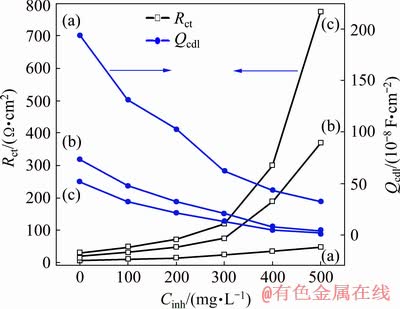
Fig. 10 Comparison between charge-transfer resistance, Rct, and constant phase element, Qcdl of Zn (a), Zn-0.5Ni (b) and Zn-10Ni (c) in ZnO-saturated 8 mol/L KOH solutions containing different concentrations of PEG-400+CTMAB (volume ratio 1:1) at 30 ��C
This suggests that the molecules of inhibitors are aggregated and adsorbed at the electrode/KOH interface as an outcome of the replacement of H2O molecules by the inhibitor species [34]. It is also observed that �� values increase with the concentration of inhibitors and follows the order (from low to high): PEG-400 < CTMAB < PEG-400+CTMAB. The efficiencies of inhibition estimated from EIS measurements showed the same trend as those obtained from PDP measurements.
By comparison between the Nyquist diagrams for Zn and its alloys (Zn-0.5Ni and Zn-10Ni) in ZnO-saturated 8 mol/L KOH at ��corr and 30 ��C (Fig. 11). It is observed that the semicircle diameter of the Nyquist diagram is enlarged by increasing the Ni content in the studied alloy. The greater diameter of semicircle is received in the case of Zn-10Ni alloy with hiding Warburg tail. This attitude displays very substantial outcomes that Ni content in the alloy plays a significant function in minimizing the corrosion rate in alkaline media. Accordingly, the corrosion resistance is enhanced with the increase of the Ni content in the alloy composition.
To compare the Nyquist curves of Zn-10Ni alloy in the solutions containing CTMAB, PEG-400, and CTMAB+PEG-400, the impendence results of the Zn-10Ni alloy are presented in a single plot (Fig. 12).
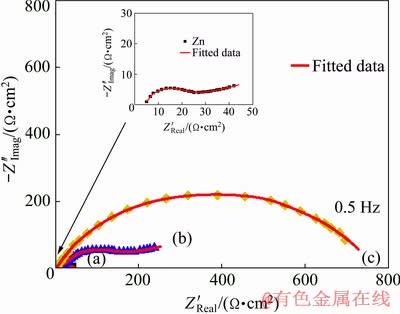
Fig. 11 Comparison between Nyquist plots for Zn (a), Zn-0.5Ni (b) and Zn-10Ni (c) in ZnO-saturated 8 mol/L KOH solutions containing 250 mg/L PEG-400 + 250 mg/L CTMAB at ��corr and 30 ��C
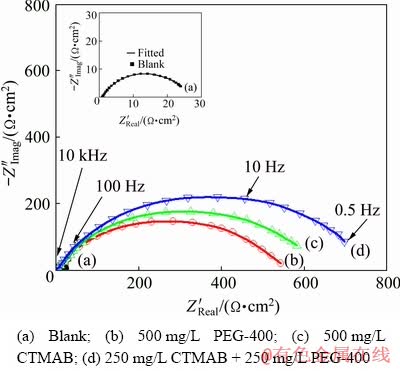
Fig. 12 Nyquist plots of Zn-10Ni alloy in 8 mol/L KOH solution saturated with ZnO measured at ��corr and 30 ��C
It is observed from Fig. 12 that Nyquist plots in the presence of PEG-400 and CTMAB alone are identical to those obtained when PEG-400+ CTMAB is added to ZnO-saturated 8 mol/L KOH solution. However, the higher resistance shows in the presence of PEG-400+CTMAB. The impedance response of Zn-10Ni increases significantly in the solution containing PEG-400+ CTMAB in comparison with that noted in the presence of PEG-400 or CTMAB, which individually implies the synergistic inhibition performance between PEG-400 and CTMAB.
3.3 Adsorption isotherm
Adsorption isotherms can supply essential data on the interaction between the electrode surface and inhibitor. The surface coverage (��) desired for efficient structure of an adsorption isotherm can be obtained for various concentrations of CTMAB, PEG-400, and CTMAB+PEG-400 from the measurements of potentiodynamic polarization by utilizing the equation: ��=��/100 [35,36].
Many adsorption isotherm models such as Temkin, Langmuir, Freundlich, Frumkin, and Flory�CHuggins were assessed and the Langmuir adsorption model was found to supply the best fit of the adsorption behavior of CTMAB, PEG-400 and CTMAB mixed with PEG-400 onto Zn and its alloys surfaces. The Langmuir model can be identified by the famous equation as follows [37]:
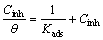 (7)
(7)
where �� is the surface coverage, and Kads is the constant of adsorption equilibrium. The relation between Cinh/�� and Cinh at 30 ��C is illustrated as in Fig. 13 for PEG-400 (a), CTMAB (b), and CTMAB+PEG-400 (c) on Zn-10Ni alloy in ZnO- saturated 8 mol/L KOH solution. Plotting of Cinh/�� and Cinh gives straight lines with regression coefficients (r2) of 0.988, 0.995 and 0.993 for inhibitors PEG-400, CTMAB, and PEG-400+ CTMAB, respectively. The intercept and slope were detected. The slope was found to be near unity. This confirms that the adsorption of investigated inhibitors on electrode surfaces follow the Langmuir adsorption model, which indicates that the inhibitor species can be adsorbed on the electrode surface forming a barrier. This barrier isolates the interaction between the surface of metal and the corrosive solution.
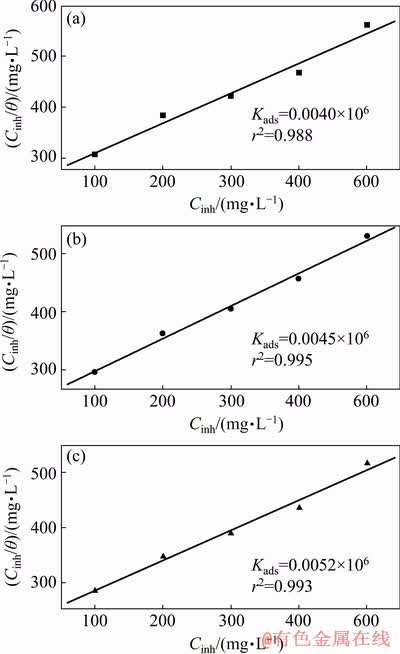
Fig. 13 Langmuir adsorption isotherms of PEG-400 (a), CTMAB (b), and PEG-400+CTMAB (1:1) (c) on Zn- 10Ni alloy in ZnO-saturated 8 mol/L KOH solution
It is found from Fig. 13 that the Kads value of CTMAB is greater than that of PEG-400, indicating that CTMAB is adsorbed on alloy surface preferably. The Kads value of CTMAB+PEG-400 is larger than that of CTMAB or PEG-400 individually, confirming that more molecules are adsorbed on the alloy surface in the solution containing the mixed inhibitor.
3.4 Surface examinations by SEM
Figure 14 shows SEM micrographs of the corrosion products formed on the surfaces of Zn, Zn-0.5Ni and Zn-10Ni alloys, after 48 h of immersion in ZnO-saturated 8 mol/L KOH solution in the absence (Figs. 14(a), (c) and (e)) and presence of 250��10-6 PEG-400 + 250��10-6 CTMAB (Figs. 14(b), (d) and (f)) at 30 ��C. The results of SEM observed in the uninhibited medium exhibit intensive porous film of the corrosion product (ZnO/ZnO2 mixture) which wraps most surface of the electrode. Nevertheless, the corrosion product formed on the surface of Zn-0.5Ni and Zn-10Ni alloys (Figs. 14(c) and (e)) offers two corrosion layers (upper and inner layer).
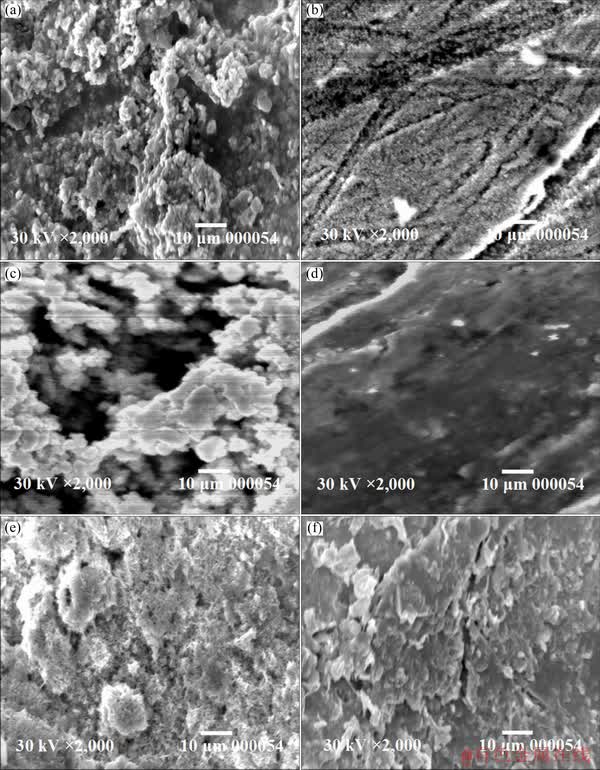
Fig. 14 SEM micrographs of Zn (a, b), Zn-0.5Ni alloy (c, d) and Zn-10Ni alloy (e, f) after immersion in ZnO-saturated 8 mol/L KOH solution for 48 h in the absence (a, c, e) and presence (b, d, f) of 250 mg/L PEG-400 + 250 mg/L CTMAB at 30 ��C
It is also seen that the inner layer is denser and coats most of the electrode surface. But, the upper layer particles are much greater in size, and the vacancies between the particles become wider. For this reason, the inner layer manifests readily through them. Therefore, the alloys surfaces seem to be fully covered by ZnO/ZnO2 and/or NiO. However, in the solution containing 250 mg/L PEG-400 + 250 mg/L CTMAB (Figs. 14(b), (d) and (f)), the corrosion product is reduced. From Figs. 14(b), (d) and (f) it is observed that the electrode surfaces are smoothly enhanced in the presence of the mixed inhibitors. Furthermore, less damage is observed in comparison with the electrode surface in the blank solutions. This manifests that the inhibitor molecule is strongly adsorbed on the surface and hinders the corrosion process.
3.5 Quantum chemical calculations
Table 3 Calculated dipole moment (DM), EHOMO, ELUMO, energy gap (DE), chemical hardness (��1) and electronic chemical potential (��)
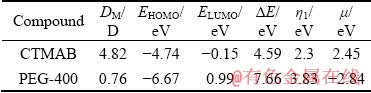
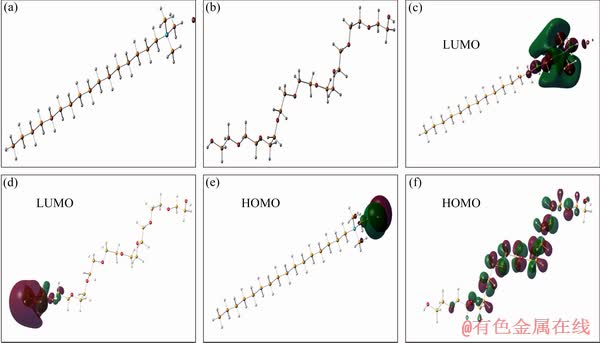
Fig. 15 3D optimized structures with HOMO and LUMO orbital occupation for investigated inhibitor models of CTMAB (a, c, e) and PEG-400 (b, d, f)
From the calculated theoretical indices, the inhibition performance of CTMAB and PEG-400 compounds can be accurately detected. The parameters including energies of the lowest unoccupied molecular orbital (ELUMO), the energies of the highest occupied molecular orbital (EHOMO), the energy gap (��E=ELUMO-EHOMO), chemical hardness (��1), dipole moment (DM) and electronic chemical potential (��) are given in Table 3. HOMO and LUMO distributions and 3D optimized structures of the investigated CTMAB and PEG-400 compounds are presented in Fig. 15. The structures of CTMAB and PEG-400 tend to have a zigzag form in a single dimension, indicating that they could act as inhibitors via the terminal ends. The HOMO-LUMO orbitals for CTMAB are localized in the ammonia-bromide terminal, which is normal due to the ionic characteristics of these groups. On the other hand, PEG-400 structures have delocalized HOMO-LUMO orbital occupation with the extension of (��CH2��CH2��O)n unit. It is reported in Ref. [38] that the higher the EHOMO energy level, the less the ionization potential and the easier the electrons donation by the inhibitor to the unoccupied d-orbital of the metal. According to the current calculations the order of HOMO energies from high to low is CTMAB>PEG-400. On the other hand, the lower the LUMO energy, the more the probability of compound to accept electrons from metal surfaces, as the ��E decreases and the inhibitor efficiency is improved, because the energy needed to eliminate an electron from the last occupied-orbital will be depressed [39]. One of the important parameters in the adsorption process is the separation energy ��E, where ��E=ELUMO- EHOMO is a function of the adsorption reactivity of the inhibitor species on the surface of metal. When ��E diminishes, the inhibitor molecule reactivity increases, causing an increase in the inhibition capacity of the inhibitor.
The calculations in Table 3 show that CTMAB has higher reactivity than PEG-400 and accordingly higher inhibition capacity which matches well with the empirical results. For the stability of the molecules which can be estimated from the chemical hardness value (��1), the higher the ��1 value of the inhibitor, the more stable the structure of it. The results indicate that CTMAB is less stable compared with PEG-400. It can be noticed that the molecules with higher ��E are the hard molecules, whilst the soft molecules have a smaller gap of ��E [40]. So, the CTMAB compound which has the lower ��E and hardness has higher inhibition efficiency.
The synergistic effect could be attributed to the various polarities of CTMAB and PEG-400. The polarity of CTMAB and PEG-400 can be indicated in terms of their dipole moments (DM). According to the theoretical parameters, the DM is 4.82 D for CTMAB and 0.76 D for PEG-400. The polarity of CTMAB is stronger than that of PEG-400 and thus CTMAB is adsorbed on metal and alloy surfaces preferably. The electrode surface cannot be covered completely by CTMAB due to the branched structure of the compound. The linear PEG-400 can be adsorbed on the remaining active sites. Therefore, the composite is better than the individual.
3.6 Battery discharge performance
Figure 16 shows the constant current discharge curves of Zn and Zn-Ni alloys using various inhibitors after stored at 30 ��C and constant current of 10 mA for 10 h.
Figure 16(a) shows that the stored discharge performance of the studied Zn electrode is affected by the inhibitors type. It is noticed that the inhibitors inhibit the self-discharge of anodic zinc, resulting in the improvement of the stored discharge capacity of the system. It is also noticed that the system using the mixed inhibitor of equal concentration of 250 mg/L for each has the largest discharge capacity (16 mA��h), confirming the synergistic effect between PEG-400 and CTMAB. Figure 16(b) shows that the Zn-10Ni alloy has the largest discharge capacity (20 mA��h) compared with Zn and other alloys in the synergistic conditions, matching with the quantum calculations and the above investigations.
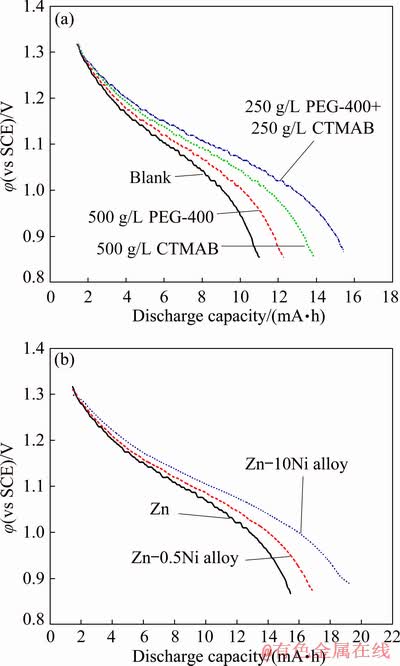
Fig. 16 Variation of potential with discharge capacity for Zn anode at different concentrations of inhibitors as labeled (a) and Zn, Zn-0.5Ni and Zn-10Ni anodes in the presence of 250 mg/L PEG-400 + 250 mg/L CTMAB (b) in 8 mol/L KOH solutions saturated with ZnO at 30 ��C and constant current of 10 mA for 10 h
4 Conclusions
(1) Solutions of PEG-400+CTMAB systems display more convenient inhibition characteristics compared to the individual inhibitor solutions, because of the strong adsorption on the surface of the electrode and formation of a protective layer.
(2) The EIS and PDP plots show that the inhibition capacity of the investigated inhibitor systems increases with increase in inhibitor concentrations.
(3) The inhibition capacity (��) by CTMAB is higher than that of PEG-400, while the �� of PEG-400+CTMAB is higher than that of CTMAB, indicating the synergistic effect between PEG-400 and CTMAB.
(4) It is potential to adding PEG-400, CTMAB or PEG-400+CTMAB to the ZnO-saturated 8 mol/L KOH electrolyte in the case of using Zn or Zn-Ni alloy as a good anode in alkaline batteries.
(5) The SEM images reveal that the surface is smooth and is almost free from damage, indicating that a good protective layer formed on the electrode surface has been adsorbed, also the highest inhibition capacity of PEG-400+CTMAB mixture is obtained.
(6) A good agreement between computational and experimental data implies the possibility of quantum chemical calculations to examine the corrosion inhibition of Zn and Zn-Ni alloy surfaces and their suitability for alkaline batteries applications.
Acknowledgments
The authors would like to thank Chemistry Department, Faculty of Science, Sohag University, and the deanship of scientific research, King Faisal University (Project No. 1811020) for financing of this work.
References
[1] ABDALLAH M, AHMED S A, ALTASS H M, ZAAFARANY I A, SALEM M, ALY A I, HUSSEIN E M. Competent inhibitor for the corrosion of zinc in hydrochloric acid based on 2,6-bis-[1-(2- phenylhydrazono) ethyl]pyridine [J]. Chem Eng Commun, 2019, 206(2): 137-148.
[2] EL-SAYED A, MOHRAN H S, ABD EL-LATEEF H M. Inhibitive action of ferricyanide complex anion on both corrosion and passivation of zinc and zinc�Cnickel alloy in the alkaline solution [J]. J Power Sources, 2011, 196: 6573-6582.
[3] CARAMIA V, BOZZINI B. Materials science aspects of zinc�Cair batteries: A review [J]. Mater Renewable Sustainable Energy, 2014, 3: 1-12.
[4] NAKATA A, ARAI H, YAMANE T, HIRAI T, OGUMI Z. Preserving zinc electrode morphology in aqueous alkaline electrolytes mixed with highly concentrated organic solvent [J]. J Electrochem Soc, 2016, 163: A50-A56.
[5] YIN Qi, WANG Zhen-yao, PAN Chen. Initial corrosion behavior of pure zinc in simulated tropical marine atmosphere [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(12): 2582-2591.
[6] LIANG H X, WANG Z L. Effect of indium addition on the electrochemical behavior of zinc electrodes in concentrated alkaline solutions [J]. Adv Mater Res, 2013, 721: 95-104.
[7] SHIMAA M A, HAMEDH A A L. Control of zinc corrosion in acidic media: Green fenugreek inhibitor [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(11): 3034-3045.
[8] ABD EL-LATEEF H M, EL-SAYED A, MOHRAN H S. Role of Ni content in improvement of corrosion resistance of Zn-Ni alloy in 3.5% NaCl solution. Part I: Polarization and impedance studies [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(8): 2807-2816.
[9] ABD EL-LATEEF H M, EL-SAYED A, MOHRAN H S. Role of nickel alloying on anodic dissolution behavior of zinc in 3.5% NaCl solution. Part II: Potentiodynamic, potentiostatic and galvanostatic studies [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(9): 3152-3164.
[10] SALEH M M, MAHMOUD M G, ABD EL-LATEEF H M. Comparative study of synergistic inhibition of mild steel and pure iron by 1-hexadecylpyridinium chloride and bromide ions [J]. Corrosion Science, 2019, 154: 70-79.
[11] RUSLING J F. Molecular aspects of electron transfer at electrodes in micellar solutions [J]. Coll Surf, 1997, 123: 81-88.
[12] EIN-ELI Y, AUINAT M, STAROSVETSKY D. Electro- chemical and surface studies of zinc in alkaline solutions containing organic corrosion inhibitors [J]. J Power Sources, 2003, 114: 330-337.
[13] COHEN-HYAMS T, ZIENGERMAN Y, EIN-ELI Y. In-situ STM studies of zinc in aqueous solutions containing PEG diacid inhibitor: Correlation with electrochemical performances of zinc-air fuel cells [J]. J Power Sources, 2006, 157: 584-591.
[14] DOBRYSZYCKI J, BIALLOZOR S. On some organic inhibitors of zinc corrosion in alkaline media [J]. Corrosion Science, 2001, 43: 1309-1319.
[15] EL-SAYED A, MOHRAN H S, ABD EL-LATEEF H M. Corrosion study of zinc, nickel, and zinc-nickel alloys in alkaline solutions by Tafel plot and impedance techniques [J]. Metall Mater Trans A, 2012, 43: 619-632.
[16] OKAMOTO H. Ni-Zn (nickel-zinc) [J]. J Phase Equilibria, 2003, 24: 280-281.
[17] ABD EL-REHIM S S, HASSAN H H, MOHAMED N F. Anodic behavior of tin in maleic acid solution and the effect of some inorganic inhibitors [J]. Corrosion Science, 2004, 46: 1071-1082.
[18] FRISCH M J, TRUCKS G W, SCHLEGEL H B, et al.Gaussian 03 (Revision C.02) [M].Wallingford CT: Gaussian Inc, 2004.
[19] DITCHFIELD R, HEHRE W J, POPLE J A. Self-consistent molecular-orbital methods. IX: An extended gaussian-type basis for molecular-orbital studies of organic molecules [J]. J Chem Phys, 1971, 54: 724-728.
[20] AMIN M A. Passivity and passivity breakdown of a zinc electrode in aerated neutral sodium nitrate solutions [J]. Electrochim Acta, 2005, 55: 1265-1274.
[21] EL-SAYED A, MOHRAN H S, ABD EL-LATEEF H M. Effect of minor nickel alloying with zinc on the electrochemical and corrosion behavior of zinc in alkaline solution [J]. J Power Sources, 2010, 195: 6924-6936.
[22] SOHI M H, JALALI M. Study of the corrosion properties of zinc�Cnickel alloy electrodeposits before and after chromating [J]. J Mater Process Technol, 2003, 138: 63-66.
[23] EL-SAYED A, SHAKER A M, ABD EL-LATEEF H M. Corrosion inhibition of tin, indium and tin�Cindium alloys by adenine or adenosine in hydrochloric acid solution [J]. Corrosion Science, 2010, 52: 72-81.
[24] ABD EL-REHIM S S, HAMDY H H, AMIN M A. Corrosion inhibition study of pure Al and some of its alloys in 1.0 M HCl solution by impedance technique [J]. Corrosion Science, 2004, 46: 5-25.
[25] ABD EL-LATEEF H M. Experimental and computational investigation on the corrosion inhibition characteristics of mild steel by some novel synthesized imines in hydrochloric acid solutions [J]. Corrosion Science, 2015, 92: 104-117.
[26] MACDONALD J R. Impedance spectroscopy [M]. New York: John Wiley & Sons, 1987.
[27] BARCIA E, MATTOS O R, PEBERE N, TRIBOLLET B. Mass-transport study for the electro dissolution of copper in 1 mol/L hydrochloric acid solution by impedance [J]. J Electrochem Soc, 1993, 140: 2825-2832.
[28] DESLOUIS C, TRIBOLLET B, PAGURA C, MUSIANI M M. Electrochemical behaviour of copper in neutral aerated chloride solution. I: Steady-state investigation [J]. J Appl Electrochim, 1988, 18: 374-383.
[29] LIANG C, LIU Z, LIANG Q, HAN G C, HAN J, ZHANG S, FENG X Z. Synthesis of 2-aminofluorene bis-Schiff base and corrosion inhibition performance for carbon steel in HCl [J]. J Mol Liq, 2019, 277: 330-340.
[30] TAN K W, KASSIM M J, OO C W. Possible improvement of catechin as corrosion inhibitor in acidic medium [J]. Corrosion Science, 2012, 65: 152-162.
[31] BENEDETTI A V, SUMODJO P T A, NOBE K, CABOT P L, PROUD W G. Electrochemical studies of copper, copper-aluminium and copper-aluminium-silver alloys: Impedance results in 0.5 M NaCl [J]. Electrochim Acta, 1995, 40: 2657-2668.
[32] ABD EL-LATEEF H M, SHAKER S M A, KHALAF M M. Synthesis of polar unique 3d metal-imine complexes of salicylidene anthranilate sodium salt. Homogeneous catalytic and corrosion inhibition performance [J]. J Taiwan Inst Chem E, 2018, 88: 286-304.
[33] OGUZIE E E, LI Y, WANG F H. Effect of 2-amino-3- mercaptopropanoic acid (cysteine) on the corrosion behaviour of low carbon steel in sulphuric acid [J]. Electrochim Acta, 2007, 53: 909-914.
[34] OGUNDELE G I, WHITE W E. Some observations on corrosion of carbon steel in aqueous environments containing carbon dioxide [J]. Corrosion, 1986, 42: 71-78.
[35] OGUZIE E E. Inhibiting effect of crystal violet dye on aluminum corrosion in acidic and alkaline media [J]. Chem Eng Commun, 2009, 196: 591-601.
[36] TAO Z, ZHANG S, LI W, HOU B. Corrosion inhibition of mild steel in acidic solution by some oxo-triazole derivatives [J]. Corrosion Science, 2009, 51: 2588-2595.
[37] Y��CE A O, BASAK, KARDAS G, YAZICI B. Electrochemical and quantum chemical studies of 2-amino- 4-methyl-thiazole as corrosion inhibitor for mild steel in HCl solution [J]. Corrosion Science, 2014, 83: 310-316.
[38] SHAKER M S A, ABD EL-LATEEF H M, SOLIMAN K A. Anionic oxide vanadium Schiff base amino acid complexes as potent inhibitors and as effective catalysts for sulfides oxidation: Experimental studies complemented with quantum chemical calculations [J]. J Mol Liq, 2018, 250: 307-322.
[39] ABD EL-LATEEF H M, ABBASOV V M, ALIYEVA L I, QASIMOV E E, ISMAYILOV I T. Inhibition of carbon steel corrosion in CO2-saturated brine using some newly surfactants based on palm oil: Experimental and theoretical investigations [J]. Mater Chem Phys, 2013, 142: 502-512.
[40] ABD EL-LATEEF H M, SOLIMAN K A, TANTAWY A H. Novel synthesized Schiff base-based cationic gemini surfactants: Electrochemical investigation, theoretical modeling and applicability as biodegradable inhibitors for mild steel against acidic corrosion [J]. J Mol Liq, 2017, 232: 478-498.
Hany M. ABD EL-LATEEF1,2, Mahmoud ELROUBY1
1. Department of Chemistry, Faculty of Science, Sohag University, Sohag 82524, Egypt;
2. Department of Chemistry, College of Science, King Faisal University, P. O. Box 380 Al Hofuf 31982 Al-Hassa, Saudi Arabia
ժ Ҫ��ͨ������λ����/��������(PDP)�͵绯ѧ�迹��(EIS)�����۲���Ҷ���-400(PEG-400)��ʮ����������廯�(CTMAB)��п��Zn-Ni�Ͻ���ZnO���͵�8 mol/L KOH��Һ�и�ʴ��Эͬ�������á��绯ѧ�о�֤ʵ��PEG-400��CTMAB��������п����Ͻ�ĸ�ʴ����ЭͬЧӦ���о����֣�250 mg/L CTMAB + 250 mg/L PEG-400��ϻ�ʴ���Ļ�ʴЧ��Զ���ڵ�һ��ʴ��500 mg/L PEG-400��500 mg/L CTMAB�Ļ�ʴЧ������ʴʵ��ǰ��ֱ����Ʒ����ɨ��羵(SEM)�۲죬֤����ϻ�ʴ������ЭͬЧӦ����ˣ��ڼ�����Һ�м������о��Ļ�ʴ��������Ե�������ķŵ�Ч�ʺ����������õ绯ѧ������������������õ�����ԡ�
�ؼ��ʣ�ЭͬЧӦ����ʴ��п-���Ͻ𣻼��Ե��
(Edited by Wei-ping CHEN)
Corresponding author: Hany M. ABD EL-LATEEF, E-mail: hmahmed@kfu.edu.sa, hany_shubra@yhaoo.co.uk;
Mahmoud ELROUBY, E-mail: dr_mahmoudelrouby@hotmail.com
DOI: 10.1016/S1003-6326(19)65197-6

