
Preparation and bioactivity of SiO2 functional films on titanium by PACVD
CHENG Xiao-min(程晓敏)1, NIE Bang-min(聂邦民)1, S. KUMAR2
1. School of Materials Science and Engineering, Wuhan University of Technology, Wuhan 430070, China;
2. Ian Wark Research Institute, University of South Australia, North Terrace, Adelaide, Australia
Received 28 August 2007; accepted 3 December 2007
Abstract: SiO2 functional films were deposited on the surface of titanium by plasma assisted chemical vapour deposition(PACVD) and the composition of films was studied by XPS. Samples deposited with SiO2 films were immersed in different concentration simulated body fluid(SBF) for biomimetic deposition of hydroxyapatite(HA). The results show that SiO2 functional films deposited on titanium surface with PACVD have good bioactivity. Hydroxyapatite is formed while titanium coated with SiO2 is immersed in simulated body fluid for seven days.
Key words: titanium; SiO2 film; bioactivity; plasma assisted chemical vapour deposition; simulated body fluid
1 Introduction
Titanium alloy has high specific strength and good corrosion resistance. When hydroxyapatite is deposited on the surface of titanium alloy, it has both the advantages of metallic materials and bioactive ceramic materials, and can be used as a kind of perfect bone tissue implant materials[1-2]. Titanium alloy has no good bioactivity. When titanium alloy was planted into body directly, it is easy to form fibrous connective tissue and hard to form chemical bond combining with surrounding bone tissue[3]. SiO2 has good bioactivity. When it is deposited on titanium, SiO2 functional layer can improve the bioactivity of titanium markedly. At present, bioactivity and mechanism of SiO2 films have got great attention of lots of researchers. SiO2 films with the method of sol-gel[4] have so many influencing factors, so it is hard to control the quality. There also exist problems of preparation of CaO-P2O5-SiO2 series bioactive glass, such as complex process and long time treatment[5-8]. TiO2-SiO2 films[9] prepared with sol-gel method on the surface of NiTi alloy do not exhibit perfect bioactivity. The method of PACVD of SiO2 films is provided with advantages of low temperature, good combining interface and easy processing[10]. Bioactivity and preparation of SiO2 films by PACVD have seldom been reported recently. In this work, SiO2 films were deposited on titanium substrates by PACVD and the bioactivity of samples in SBF was studied.
2 Experimental
2.1 SiO2 films deposited by PACVD
PDC-32G plasma assisted reactor was used in experiment. Schematic map of PACVD processing is shown in Fig.1. Titanium was rinsed gently with NaOH, HF and HNO3. TEOS and air were used as precursors. Water bath temperature is 8 ℃ and reactor pressure is 5 Pa. First, dry air was introduced into the chamber at 10 mL/s and the plasma reactor was activated for 20 min. Hydrocarbons and oxides were removed[11]. Second, another air flow at 2 mL/s was used as a carrier gas for transporting the TEOS vapour to plasma chamber and the deposition process was 30 min. Then, SiO2 films with about 40 nm in thickness were obtained.

Fig.1 Schematic map of PACVD processing
2.2 Deposition of HA in simulated body fluid
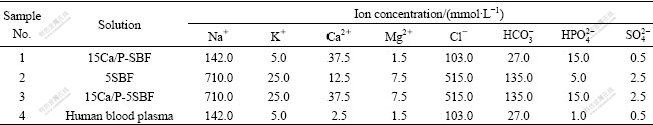
SBF with different concentrations was prepared by dissolving reagents of NaCl, NaHCO3, KCl, K2HPO4・3H2O, MgCl2・6H2O, CaCl2 and Na2SO4 into deionized water. The compositions of SBFs with different concentrations are listed in Table 1. Titanium pieces coated with SiO2 films were cut into the size of 10 mm×10 mm×1 mm, then put into different concentration SBFs. The SBF containers were put into electric-heated thermostatic water bath for 4 d and 7 d, respectively. SBF was changed every two days. Samples were removed from SBF and rinsed gently with deionized water and analyzed by scanning electron microscopy (JSM-5610LV) and multi-technique electron spectrometer (ESCALAB MK II).
Table 1 Ion concentration and composition of SBFs and human blood plasma
3 Results and discussion
3.1 SiO2 functional films deposited by PACVD
Results of XPS survey spectrum of SiO2 films deposited on titanium are shown in Fig.2. It can be seen that there exist three kinds of elements, C, O and Si. Quantitative calculation is carried out with the sensitive factor method. Quantitative data of C, O and Si are listed in Table 2.
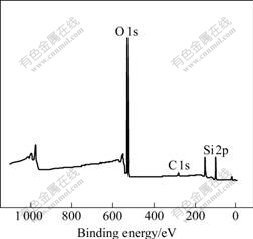
Fig.2 XPS survey spectrum of SiO2 films
Table 2 Quantitative analysis results of PACVD deposition
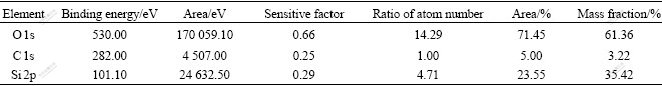
It is shown from Table 2 that a small quantity of C (3.22%) is found in surface films. The molar ratio of O to Si in coating is 3?1, a little larger than the stoichiometric value of 2.0 for SiO2. The increase of the x(O)/x(Si) ratio has also been observed by other researchers in silicon oxide coating. It has been explained on the basis of increased Si―O―H and Si―O―Cx―Oy types of bonds in place of Si―O―Si bonds[12].
The reaction may be written as follows:
SiO4(C2H5)4+12O2=SiO2+10H2O+8CO2 (1)
From the reaction, it is known that enough oxygen would reduce carbon content in SiO2 functional films and more pure SiO2 can be obtained[13].
3.2 HA deposition induced in SBF
3.2.1 Microstructure
SEM images of titanium coated with SiO2 films immersed in No.1 SBF for 12 h and 7 d are shown in Figs.3(a) and 3(b) respectively. Fig.3(c) shows the microstructure of the sample immersed in No.2 SBF for 7 d, and Fig.3(d) shows that in No.3 SBF for 7 d. It is observed that a lot of HA has been formed when SiO2 films are immersed in SBF for more than 12 h. It is obvious to observe that the quantity of HA increases markedly in Fig.3(b). More active sites[14-15] were supplied by noncontinuous surface, which is easy for Ca2+ and PO43- to be supersaturated in some region and favorable for HA to nucleate and grow[16-17]. Flocculent (Fig.3(c)) and granular structures (Fig.3(d)) are found all over the surface. However, granular HA, with a characteristic of denseness and uniformity, almost aggregates in succession. This means that more HA is formed on the sample immersed in No.3 SBF.

Fig.3 Microstructures of titanium coated with SiO2 films: (a) In No.1 SBF, 12 h; (b) In No.1 SBF, 7 d; (c) In No.2 SBF, 7 d; (d) In No.3 SBF, 7 d
3.2.2 Composition of HA deposition layer
XPS survey spectrum of the sample immersed in No.1 SBF for 7 d is shown in Fig.4. Ca, P, C and O are found in the surface layer. The results show that some HA has been deposited on the SiO2 film surface.
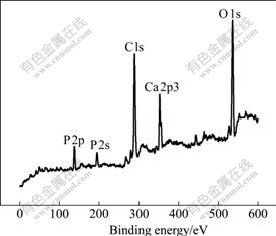
Fig.4 XPS survey spectrum of sample induced to deposit HA for 7 d
The amount of Ca, P and O elements in coating was scanned through high resolution spectra and calculated by atom sensitive factor method respectively[18-21]. The results are listed in Table 3.
Table 3 Quantitative analysis results of 7 d deposition in SBF
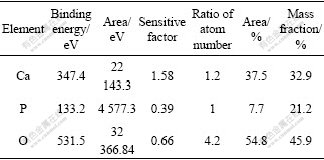
It is shown from Table 3 that the molar ratio of Ca to P is 1.2?1, which is lower than that in HA. The reason is that some CaHPO4 is formed in the surface layer.
3.2.3 Mechanism of HA deposition
The reaction of HA deposition is written as follows:
10Ca2++2OH-+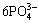 =Ca10(PO4)6(OH)2 (2)
=Ca10(PO4)6(OH)2 (2)
Hydrolyzation reaction of SiO2 films can be written as follows:
(Si―O―Si)+H2O=Si―OH+HO―Si (3)
When Si―O―Si bonds break out, lots of Si―OH groups[22-23] are formed, which generates polysilicon acid, then chain and netlike structure[24] with lots of negative charge are produced.
Ca2+ will be adsorbed on the surface[25-27] because of much negative charge existed in Si―OH structure and the static gravitation effect. Then Ca2+ can adsorb  . HA crystal nucleus forms with the increase of Ca2+,
. HA crystal nucleus forms with the increase of Ca2+, 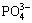 and OH-. Once the crystal nucleus forms, it grows up and HA coating forms at last[4,28].
and OH-. Once the crystal nucleus forms, it grows up and HA coating forms at last[4,28].
4 Conclusions
1) SiO2 functional films, which contains small quantity of carbon, are obtained by taking the following parameters: 5 Pa chamber pressure, 8 ℃ water bath temperature, 10 mL/s air flow and 30 min plasma deposition.
2) 3.22% carbon is found in SiO2 functional films deposited by PACVD.
3) The atomic ratio of Ca to P in the samples immersed in SBF for 7 d is 1.2?1.
4) Better HA coating can be obtained when the titanium coated with SiO2 films is immersed in 60 ℃ 15 Ca/P-5SBF for 7 d.
Acknowledgements
Special thanks to KIM, University of South Australia and Andrews, Flinders University, for his help in some experiments.
References
[1] SHANG Da-shan, LI Mu-qin. Recent progress and prospect in bioactive composite coatings on metal substrate [J]. J Jiamusi Univ (Nat Sci Edn), 2002, 20(3): 271-275.
[2] CHEN Cai-feng. Research progress of preparing metal-base coat of nanometer scale [J]. Journal of Ceramics, 2002, 23(4): 241-245.
[3] ZHUANG Y, HU R, CHEN F. Surface biochemical modification for the titanium implants [J]. Journal of Biomedical Engineering, 2005, 22(3): 618-621. (in Chinese)
[4] GAO Yan-jiao, LIU Rong-fang, XIAO Xiu-feng. Preparation and bioactivity of silica films coated on titanium[J]. Applied Chemistry, 2006(3): 313-318. (in Chinese)
[5] CHENG Xiao-feng, WANG Ying-jun, ZHAO Na-ru. SEM and FTIR analysis on microstructure and bioactivity of the porous materials from the sol-gel derived glass[J]. Journal of Chinese Electron Microscopy Society, 2003, 22(4): 304-310. (in Chinese)
[6] CHENG Xiao-feng, LI yu-li, ZHAO Na-ru. Nano-structure analysis of the sol-gel derived bioactive glass [J]. Bulletin of the Chinese Ceramic Society, 2007, 26(4): 247-251. (in Chinese)
[7] ZHAO Na-ru, WANG Ying-jun, CHEN Xiao-feng. In vitro study of a sol-gel bioactivity glass degradability and ineralization deposit[J]. Journal of Materials Science & Engineering, 2004, 22(5): 723-725. (in Chinese)
[8] ZHANG Xiao-kai, LIU Wei, CHEN Xiao-feng. Mophology character of the sol-gel derived bioactive glass in SBF solution[J]. Chinese Journal of Chemical Physics, 2004, 17(4): 495-499. (in Chinese)
[9] LIU Jing-xiao, YANG Da-zhi, CAI Ying-ji. Surface characteristics and bioactivity of TiO2 and TiO2-SiO2 films[J]. Journal of the Chinese Ceramic Society, 2001, 29(4): 350-354. (in Chinese)
[10] WANG Yong-zheng, GONG Guo-quan, CUI Jin-zhong. Preparation and application of SiO2 thin films[J]. Vacuum & Cryogenics, 2003, 3(4): 228-232. (in Chinese)
[11] WILSON D H. Cleaning and finishing of reactive and refractive alloys[M]// WOOD W G. Metals Handbook. Ohio, USA: American Society for Metals, 1982: 650-658.
[12] VALLE C, GOULLET A, GRANIER A. In situ ellipsometry and infrared analysis of PECVD SiO2 films deposited in an O2/TEOS helicon reactor[J]. J Non-Cryst Solids, 1997, 216: 48-54.
[13] CHEN Zhao-feng, CHENG Lai-fei, XU Yong-dong. Fabrication of silica matrix composite by chemical vapor infiltration [J]. Acta Aeronautica et Astronautica Sinica, 2002, 23(1): 25-28. (in Chinese)
[14] DONG Jing, WANG Feng, KOU Fu-ming. Preparation of nanograde hydroxyl apatite by simulated body fluid [J]. Guangzhou Chemistry, 2006, 31(4): 12-16. (in Chinese)
[15] WANG De-ping, YU Rui. Surface change of calcium phosphate glass ceramics in simulated body fluid [J]. Journal of the Chinese Ceramic Society, 2004, 32(11): 1441-1444. (in Chinese)
[16] FU Tao, HUANG Ping, HAN Yong. Apatite formation induced by alkaline treatment on titanium alloy substrate [J]. Rare Metal Materials and Engineering, 2000(6): 15-16. (in Chinese)
[17] LIN Lan-yun, LIU Rong-fang, XIAO Xiu-feng. Advance in biomimetic deposition hydroxyapatite coating [J]. Bulletin of the Chinese Ceramic Society, 2005(1): 70-75. (in Chinese)
[18] SHEN Jian-feng, CHANG Cheng-kang, MAO Da-li. Fabrication and properties of HAP-bioglass-Ti6A14V composite [J]. Journal of Inorganic Materials, 2001, 16(5): 993-998. (in Chinese)
[19] LIU Shi-hong, WANG Dang-han. X-ray photoelectron spectroscopy [M]. Beijing: Science Press, 1988. (in Chinese)
[20] ATANASSOVA E, TYULIEV G. XPS study of N2 annealing effect on thermal Ta2O5 layers on Si[J]. Applied Surface Science, 2004, 25: 86-99.
[21] YU Jia-guo, ZHAO Xiu-jian, ZHAO Qing-nan. XPS study on TiO2 photocatalytic thin film prepared by the sol-gel method[J]. Chinese Journal of Materials Research, 2000, 14(2): 203-209. (in Chinese)
[22] UCHIDA M, KIM H M, KOKUBO T. Bonelike apatite formation induced on zirconia gel in a simulated body fluid and its modified solutions[J]. J Am Ceram Soc, 2001, 84: 2041-2044.
[23] ZHONG Ji-ping, DAVID C G. Processing and properties of sol-gel bioactive glasses[J]. J Biomed Mater Res Appl Biomater, 2000, 53: 694-701.
[24] ZHU Ren. Inorgnic chemistry [M]. Beijing: Higher Education Press, 2000: 409-412. (in Chinese)
[25] LIANG Fang-hui, WANG Ke-guang, ZHOU Lian. Enhancement of the bioactivity of alkali-heat treated porous titanium by pre-calcification [J]. Rare Metal Materials and Engineering, 2004, 33(10): 1013-1017. (in Chinese)
[26] CORBRIDGE D E C. Structural chemistry of phophorus [M]. Amsterdam-London-New York: Elsevier, 1974: 247-261.
[27] CUI Zhong-bo, CHEN Min-fang. Progress of bioimmetic process on surface of biomedical metal materials[J]. Journal of Tianjin University of Technology, 2006, 22(2): 18-23. (in Chinese)
[28] GAO Yan-jiao, LIU Rong-fang, XIAO Xiu-feng. Biomimetic deposition of carbonated hydroxyapatite coating by calcium enriched silica films based on titanium[J]. Journal of Fujian Normal University (Natural Science Edition), 2006, 22(2): 49-55. (in Chinese)
Corresponding author: CHENG Xiao-min; Tel: +86-13507117513; Fax: +86-27-87651779; E-mail: chengxm@whut.edu.cn
(Edited by YANG Bing)