A hybrid zeolitic imidazolate framework Co-IM-mIM membrane for gas separation
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2017���8��
�������ߣ������� ������ LI Hai-long(���) ������ WANG S ������
����ҳ�룺1727 - 1735
Key words��Co-IM-mIM membrane; secondary growth; gas permeation; separation
Abstract: A zeolitic imidazolate hybrid membrane (Co-IM-mIM) containing two imidazolate ligands deposited on a macroporous ��-alumina support was prepared by pre-depositing and secondary growth technique. XRD, TGA and SEM characterizations demonstrate that a stable and thin, but dense and pure-phase Co-IM-mIM membrane can be obtained on the macroporous-alumina discs in Teflon-lined autoclave at 120 ��C after pre-depositing by dip-coating at room temperature. No visible cracks, pinholes or other defects were observed on the membrane layer. The gas separation studies of Co-IM-mIM membrane were carried out at 25 ��C and 1��105 Pa, showing ideal selectivity of 6.95, 5.25, 3.40 for H2/CO2, H2/N2 and H2/CH4, respectively, and a permeance of 17.37�� 10-6 mol/(m2��s��Pa) for H2. The influence of temperature and trans-membrane pressure on hydrogen separation and permeation was also carried out. The gas permeation and selectivity demonstrate that this membrane may have potential applications for efficient H2 separation.
Cite this article as: GAO Zheng-cai, LI Li-qing, LI Hai-long, CHEN Ruo-fei, WANG S, WANG Yang-gang. A hybrid zeolitic imidazolate framework Co-IM-mIM membrane for gas separation [J]. Journal of Central South University, 2017, 24(8): 1727-1735. DOI: https://doi.org/10.1007/s11771-017-3580-z.
J. Cent. South Univ. (2017) 24: 1727-1735
DOI: https://doi.org/10.1007/s11771-017-3580-z

GAO Zheng-cai(������)1, LI Li-qing(������)1, LI Hai-long(���)1,
CHEN Ruo-fei(������)1, WANG S2, WANG Yang-gang(������)3
1. School of Energy Science and Engineering, Central South University, Changsha 410083, China;
2. Department of Chemical Engineering, Curtin University, GPO Box U1987, Perth WA 6845, Australia;
3. School of Physical Science and Technology, Lanzhou University, Lanzhou 730000, China
 Central South University Press and Springer-Verlag GmbH Germany 2017
Central South University Press and Springer-Verlag GmbH Germany 2017
Abstract: A zeolitic imidazolate hybrid membrane (Co-IM-mIM) containing two imidazolate ligands deposited on a macroporous ��-alumina support was prepared by pre-depositing and secondary growth technique. XRD, TGA and SEM characterizations demonstrate that a stable and thin, but dense and pure-phase Co-IM-mIM membrane can be obtained on the macroporous-alumina discs in Teflon-lined autoclave at 120 ��C after pre-depositing by dip-coating at room temperature. No visible cracks, pinholes or other defects were observed on the membrane layer. The gas separation studies of Co-IM-mIM membrane were carried out at 25 ��C and 1��105 Pa, showing ideal selectivity of 6.95, 5.25, 3.40 for H2/CO2, H2/N2 and H2/CH4, respectively, and a permeance of 17.37��10-6 mol/(m2��s��Pa) for H2. The influence of temperature and trans-membrane pressure on hydrogen separation and permeation was also carried out. The gas permeation and selectivity demonstrate that this membrane may have potential applications for efficient H2 separation.
Key words: Co-IM-mIM membrane; secondary growth; gas permeation; separation
1 Introduction
In the past 10 years, metal organic frameworks (MOFs) have attracted significant attention due to their potential applications in gas storage and separation. Several hundreds of MOF materials have been synthesized, and the number continues to grow [1-4]. Further, the intrinsic nature of MOFs by metal clusters or cations connected with organic linker molecules makes the advantages such as highly diversified structures and pore size as well as specific adsorption affinities as compared to other materials, leading MOFs as fascinating candidates for fabrication of superior molecular sieve membranes [5-7]. Nowadays, various MOFs membranes have been well recognized both experimentally and computationally [8-10].
Zeolitic imidazolate frameworks (ZIFs), a MOF subfamily based on transition metals and imidazolate linkers, are of particular interest due to their versatile structures, permanent porosity and uniform pore size analogous to those of zeolites [11-13]. To date, researchers have put great efforts in practical application of this nanoporous material and several ZIF membranes with reasonable gas separation performances have been reported [5, 14-16]. ZIF-8 is one of the mostly studied ZIF compounds because of its potential functional applications in gas separation [17-19]. Although many MOF membranes have been prepared, only few membranes, containing two imidazolate ligands, were studied in permeation experiments. On the other hand, the controllable modification of their structure and functionality still remains a challenge. Up to now, it is possible to tune the properties of MOFs for specific applications via chemical or structural modification. The common approach is to use a linker with functional group or organic ligands to change the chemical or structural characteristics of the materials [20-22]. BURROWS et al [20] described the syntheses, structures and properties of mixed-ligand, mixed-metal and core�Cshell MOFs, and highlighted some of the potential benefits in functionality of the materials. Therefore, the strategy was adopted in this work to prepare a novel hybrid ZIF membrane for efficient H2 separation, which hopefully can approach the ideal membrane demands.
For the preparation of MOF membranes, various fabrication techniques such as in-situ growth, secondary growth, layer-by-layer growth and electrospinning technology were discussed in Refs. [23, 24]. The synthesis of continuous and thin MOFs membranes still remains a challenge. SHAH et al [25] discussed the common challenges facing polycrystalline MOF membranes such as poor membrane�Csubstrate bonding, poor membrane stability and macroscopic crack formation during membrane formation and activation.
Here we report a new hybrid membrane consisting of cobalt coordinated with imidazole (IM) and 2-methylimidazole (mIM). The hybrid membrane was prepared by pre-depositing at room temperature and secondary growth at 120 ��C. Macroporous ��-alumina (��-Al2O3) disks were selected as the supports of membranes. Therefore, the Co-IM-mIM nanocrystals can infiltrat into the macroporous support when pre-depositing, and the support becomes smooth without concave. ZIF membrane started growing from the nanocrystals embedded in the precursor layer. It is shown that Co-IM-mIM powders have shown a good stability upon heat and water solvents. In gas permeation tests, the Co-IM-mIM membrane showed a high selectivity and permeance with a H2/CO2 ideal selectivity of 6.95 and H2 permeances of about 17.37��10-6 mol/(m2��s��Pa). Thus, the Co-IM-mIM membrane would be ideal in practical applications because it could be selective to H2 over other gases with high selectivity and permeance.
2 Experimental
2.1 Materials
Cobalt nitrate hexahydrate (Co(NO3)2��6H2O, ��99%, AR), imidazole (C3H4N2, 99%), 2-methylimidazole (C4H6N2, 99%) and methanol (CH4O, ��99.5%, AR) were purchased from Aladdin Chemical Co. Ltd. Gases for permeation experiments, such as nitrogen (N2), hydrogen (H2), methane (CH4) and carbon dioxide (CO2), with purities >99.50% were obtained from Changsha High-tech Gas Co, Ltd.
2.2 Synthesis of Co-IM-mIM nanocrystals
Co-IM-mIM nanocrystals were prepared according to the following method. Firstly, a solid mixture of Co(NO3)2��6H2O (2.91 g, 10.0 mmol), imidazole (2.72 g, 40.0 mmol) and 2-methylimidazole (3.28 g, 40.0 mmol) were dissolved in 300 mL methanol. Secondly, the mixture was dispersed with ultrasonication at 25 ��C for 10 min. Subsequently, the mixture was decanted into an open conical flask and stirred at room temperature for 6 h. The product was collected by centrifuging, and then washed with methanol 3 times. Finally, the product was dried at 80 ��C overnight in an oven.
In order to compare the influence of temperature on synthesis, another Co-IM-mIM nanocrystal was prepared by a modified solvothermal protocol. Firstly, a solid mixture of Co(NO3)2��6H2O (2.91 g, 10.0 mmol), imidazole (2.72 g, 40.0 mmol) and 2-methylimidazole (3.28 g, 40.0 mmol) was dissolved in 300 mL methanol. Secondly, the mixture was dispersed with ultrasonic at 25 ��C for 10 min. Subsequently, the mixture was loaded into a Teflon-lined autoclave. The autoclaves were sealed and heated to 120 ��C for 12 h. Finally, the product was collected by centrifuging, and then washed with methanol 3 times. The product was dried at 80 ��C overnight in an oven.
2.3 Membrane synthesis
Co-IM-mIM membranes were prepared according to the following method. Macroporous-alumina discs (3 mm in thickness and 40 mm in diameter��Angtai company, China) were cleaned with deionized (DI) water and dried at 120 �� for 24 h, and then were cooled down to room temperature. One side of the discs was first surfacely deposited with the Co-IM-mIM nanocrystals 3 times by dip-coating with a methyl alcohol pre-depositing sol which contained ca. 4% nanocrystals. The discs were immediately removed after dip-coating and air-dried in an oven at 80 ��C for 4 h and cooled down to room temperature. For subsequent solvothermal growth, a solution was used, which was prepared by dissolving 0.58 g (2.0 mmol) cobalt nitrate hexahydrate, 0.65 g (8.0 mmol) 2-methylimidazole, and 0.50 g imidazole (8.0 mmol) in 80 mL methanol. The deposited alumina discs were placed vertically in a 100 mL Teflon-lined autoclave, after which the reaction solution was loaded into the Teflon-lined autoclave. Then the autoclave was sealed and heated to 120 ��C for 12 h and was cooled to room temperature. The membranes were taken out and intensively washed three times with fresh methanol, and then immersed in 30 mL fresh methanol for another 12 h to exchange the solvent that is occluded inside the channels of Co-IM-mIM. Finally, the disc was dried in vacuum at 80 ��C for 6 h.
Another Co-IM-mIM hybrid membrane was prepared by using an in-situ growth method as described above using the alumina discs without pre-depositing.
2.4 Characterization
The membranes and powder samples were determined by X-ray diffraction (XRD, X�� PertPro MPD, PANalytical B.V., NED) with a PANalytical powder diffractometer operated at 40 mA and 40 kV using Cu/K�� as the radiation source. The scanning range was from 5�� to 40�� with a step size of 4(��)/s. Thermogravimetric analysis (TGA) was performed on a NETZSCH STA 409 apparatus in the range of 25 to 1000 ��C. The microporosity of the Co-IM-mIM crystal was determined by N2 adsorption at -196 ��C using an SA3100 specific surface area analyzer��Beckman-Coulter Instrument Co., USA). The membrane morphologies were observed via scanning electron micrographs (SEM) (Helios Nanolab 600i), where gold-coated specimens were used to increase the conductivity and the measurements were operated under 10-20 kV acceleration voltage. The membrane was first simply broken and cleaned in air flow to remove dust.
2.5 Permeation experiments
The membrane properties were studied with permeation tests. The measurements were carried out using home-made gas permeation equipment (Fig. 1). The supported Co-IM-mIM membranes were tightly sealed in the permeation cell by using silicone and lock nut with the effective remaining membrane area of 1.53��10-4 m2. Permeances in mol/(m2��s��Pa) were calculated on the basis of the measured flow rates (in mL/min) at room temperature (25 ��C) and ambient pressure (101.3 kPa) and from the applied partial pressure at the feed side of the membrane (60 kPa).
3 Results and discussions
3.1 Characterization of Co-IM-mIM crystals
Figures 2(a) and (b) show X-ray diffraction (XRD) patterns of single crystal Co-IM-mIM. The XRD pattern of the Co-IM-mIM membrane after secondary growth at 120 ��C for 24 h (Fig. 2(a)) is similar to the XRD pattern of the as-synthesized Co-IM-mIM crystal by stirring the synthesis solution at room temperature (Fig. 2(b)). Through the comparison of XRD patterns between samples a and b, it could prove that the structures of Co-IM-mIM materials on the substrate were the same to the pure powder ones. That is to say, the membrane is purely composed of Co-IM-mIM.
Membrane stability is crucial in its applications. The thermal stability of Co-IM-mIM is shown from TGA profile at 25 ��C to 1000 ��C�� (Fig. 3). Two mass losses were observed: at first, a slight degradation was between 25 ��C and 495 ��C, due to the loss of the methanol and other residual organic solvents; and the second one was occurring between 495 ��C and 1000 ��C, attributing to the structural decomposition of Co-IM-mIM. Therefore, Co-IM-mIM is thermally stable up to 495 ��C.
Figures 4(a) and (b) show the microporosity of the Co-IM-mIM crystal, It can be seen that the Co-IM-mIM samples as-synthesized at 298 K and 393 K both show the presence of micropores. The Co-IM-mIM sample as-synthesized at 298 K has a small pore volume with pores size about 1 nm; however, the Co-IM-mIM sample as-synthesized at 393 K shows a higher pore volume and pore size about 0.95 nm. The Co-IM-mIM membrane has larger micropores compared to others synthetic membranes (Table 2). This will influence the permeability and permselectivity performance of the membranes [26].
The morphology of pure Co-IM-mIM single crystal was characterized by scanning electron microscopy (SEM). SEM image shows that the crystal structure of synthesized sample is tetragonal rectangular solid with about 5 ��m in length (Fig. 5(a)). As shown in Figs. 5(a) and (b), it can be seen that two samples have a little difference, the Co-IM-mIM single crystal collected on the autoclave after secondary growth at 120 ��C for 12 h was bigger than the Co-IM-mIM single crystal collected on the beaker after stirring the synthesis solution at room temperature, which indicates that higher temperature is beneficial to the agglomerate of the Co-IM-mIM single crystal.
Although the Co-IM-mIM crystals synthesized at room temperature grow smaller, the yield is too low, only 15%. The yield of Co-IM-mIM crystal prepared by a modified solvothermal protocol is about four times as much as room-temperature synthesis. Considering synthesized Co-IM-mIM sample synthesized at 393 K shows a higher pore volume (Fig. 4). In this work, the Co-IM-mIM membrane was prepared by solvothermal secondary growth technique after pre-depositing.
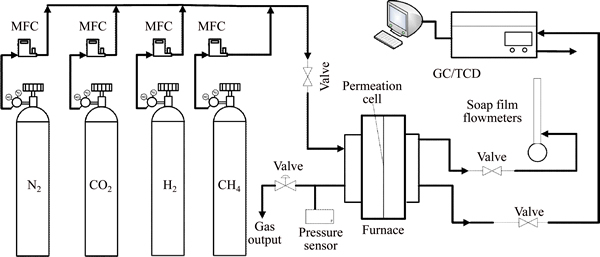
Fig. 1 Schematic diagram of separation experimental system
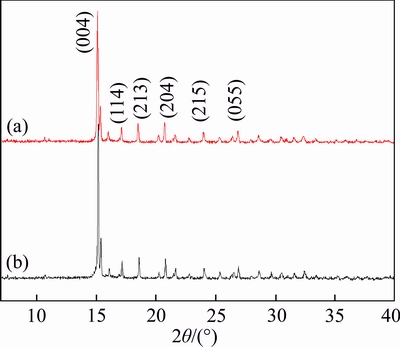
Fig. 2 XRD patterns of Co-IM-mIM crystal collected on Co-IM-mIM membrane (a) and as-synthesized Co-IM-mIM crystal at room temperature (b)
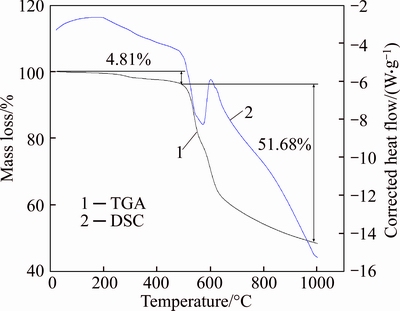
Fig. 3 TGA and DSC of Co-IM-mIM crystal
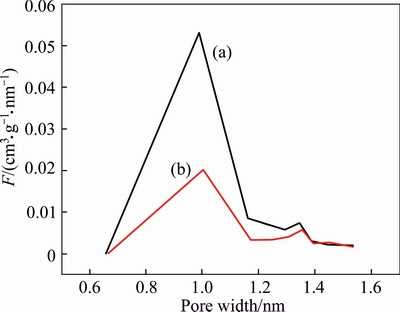
Fig. 4 Micropore size distribution of Co-IM-mIM as- synthesized at 393 K (a) and as-synthesized at 298 K (b)
3.2 Characterization of Co-IM-mIM membranes
We used two ways to synthesize membranes in order to investigate the influence of pre-depositing on membrane layer growth. Figure 6(a) shows the reference membrane synthesized by in-situ growth method presents in poorly intergrown film and some uncovered places.

Fig. 5 SEM images of Co-IM-mIM crystals as-synthesized at 298 K (a) and as-synthesized at 393 K (b)
Figure 6(b) shows the membrane synthesized by pre-depositing and then secondary growth. It can be seen that randomly oriented Co-IM-mIM crystals were compact and uniform on the surface of samples, and no visible cracks, pinholes or other defects were observed on the membrane layer. This indicates that the pre-depositing is essential for the formation of a continuous and defect free Co-IM-mIM membrane. We can more clearly see the influence of pre-depositing on membrane layer growth from Fig. 6(c). On the same ��-alumina support, the lower left corner part of Fig. 6(c) was synthesized by in-situ growth method, and the top right corner of Fig. 6(c) was synthesized by pre-depositing and then secondary growth.
3.3 Gas separation performance of Co-IM-mIM membrane
3.3.1 Single gas separation performance of Co-IM-mIM membrane
The gas permeability and ideal selectivity of Co-IM-mIM membrane were studied at the volumetric flow rates of the single gas (H2, CH4, N2, and CO2) at 25 ��C and 1��105 Pa using home-made gas permeation equipment. The permeation results for single gas are given in Figs. 7 and 8. The membrane exhibits remarkably higher hydrogen permeance compared with other gases of CO2, CH4 and N2. The permeancesdecrease in an order of H2 > CH4 > N2 > CO2 , which does not agree with increasing kinetic diameter of the permeating molecules (Table 1). It has a linear relationship with the inverse of the square root of their molecular mass (MW), as shown in Fig. 9. This illustrates that the permeation behaviors of H2, CH4, CO2 and N2 mainly follow Knudsen diffusion, similar to the reports for other MOF membranes [27, 28]. The permeation of hydrogen was 17.37��10-6 mol/(m2��s��Pa), which is similar to that of a ZIF membrane prepared on a ��-alumina support [25]. But the permeation of hydrogen is higher than most MOFs membranes prepared by other researchers (Table 2). This may be due to higher micropore (0.95 nm) of the Co-IM-mIM membrane. It is also noted that the ideal selectivity (Fig. 8) is much higher than the Knudsen constant and the values of Spermeance of H2 to other three gases are all larger than 1.0, also larger than those in most other MOFs membranes reported up to now [38-41]. As previously noted, it was indicated that the Co-IM-mIM hybrid membrane may be a promising candidate for efficient H2 separation in terms of permeation flux and selectivity.

Fig. 6 SEM images of Co-IM-mIM hybrid membrane synthesized using in-situ growth method (a), and images of Co-IM-mIM synthesized by pre-depositing and then secondary growth (b) and comparison of Co-IM-mIM membrane synthesized by in-situ growth method and pre-depositing and then secondary growth (c)
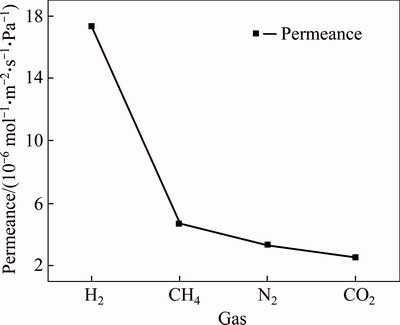
Fig. 7 Single gas permeances result of Co-IM-mIM membrane at 25 ��C under a trans-membrane pressure drop of 1��105 Pa
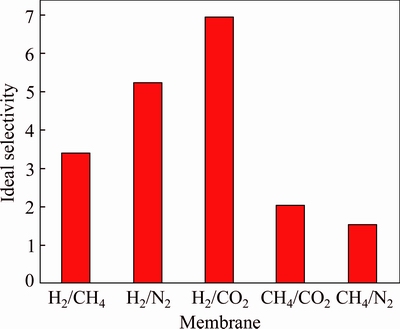
Fig. 8 Ideal selectivity results of Co-IM-mIM membrane at 25 ��C under a trans-membrane pressure drop of 1��105 Pa
Table 1 Single gas permeances (mol/(m2��s��Pa)) and ideal selectivity for ��-Al2O3 supported Co-IM-mIM membrane at 25 ��C and a trans-membrane pressure drop of 1��105 Pa
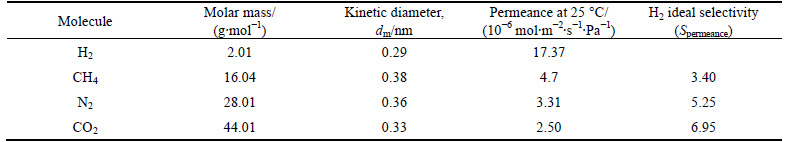
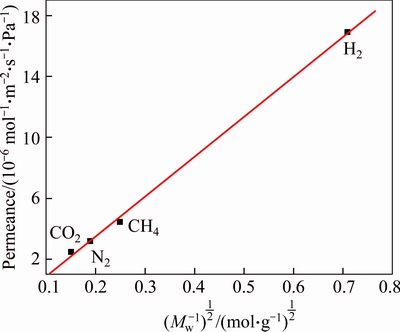
Fig. 9 Permeation behaviors of Co-IM-mIM membrane at 25 ��C under a trans-membrane pressure drop of 1��105 Pa
3.3.2 Thermal effect on hydrogen separation
Previous works have suggested that temperature may have an important effect on the gas separation performance of MOF membranes [42]. Therefore, single gas permeations for H2, CH4, N2 and CO2 at different temperatures (from 25 to 70 ��C) were evaluated.Figure 10 represents the permeance and ideal selectivity of the membrane at different temperature. It is found that the permeance values of H2, CH4, N2 gases show a slight decrease with the increasing temperature, while CO2 shows an increase with the increase of temperature. This different behaviour of CO2 was reported by ZHAO et al [43]. The permeance of gas depends on the relative values of activation energy for diffusion and exothermic heat of adsorption.
This similar trend was also observed for other membranes [44]��[����[��Zhao, 2011 #31;Kanezashi, 2009 #65��]����]��. In the temperature range, due to the different permeance values of single gas, it is found that the H2/CO2 ideal selectivity decreases from 6.84 at 25 ��C to 5.58 at 70 ��C. While the H2/CH4 and H2/N2 ideal selectivities increase from 3.80 at 25 ��C to 3.92 at 70 ��C and 5.37 at 25 ��C to 5.43 at 70 ��C, respectively. Nevertheless, such permeance value of single gas is useful for the optimal operation conditions for use of the membrane.
3.3.3 Pressure drop effect on hydrogen separation
In order to evaluate the effect of trans-membrane pressure on the gas separation performance of Co-IM-mIM membranes, gas separation tests were carried out by increasing the trans-membrane pressure drop from 0.6��105 to 1.5��105 Pa (Fig. 11). As expected, it is found that the permeance values of single gas show a slight increase with the increase of trans-membrane pressure. This similar trend was also observed from other membranes [45]. Nevertheless, the variation in the ideal selectivity is observed with different trans-membrane pressure. An increase of the ideal selectivity for H2/CO2 can be clearly seen before 1.0��105 Pa and an decrease after 1.0��105 Pa, while a decrease of the ideal selectivity for H2/CH4 and H2/N2 can also be seen before 1.0��105 Pa and an increase after 1.0��105 Pa. The adsorption ability of Co-IM-mIM membrane will change under different pressure conditions because at high pressures, high concentration of a gas such as CO2 may swell the polymer and increase the mobility of the polymer chains, which leads to a change in membrane��s selectivity [46]. Therefore, this discrepancy of ideal selectivity can be explained by the variations in ratios of adsorption ability of Co-IM-mIM membrane towards single gas in the trans-membrane pressure range [47]. Moreover, the ideal selectivities of H2/N2, H2/CO2, and H2/CH4 still maintain high values, which shows the Co-IM-mIM membranes are stable under different trans-membrane pressures. The permeance value of single gas in this temperature and pressure range is useful for the optimal operation conditions of the membrane.
Table 2 Comparison of H2 permeance and selectivity of as-synthesized Co-IM-mIM membrane with some reported MOFs membranes
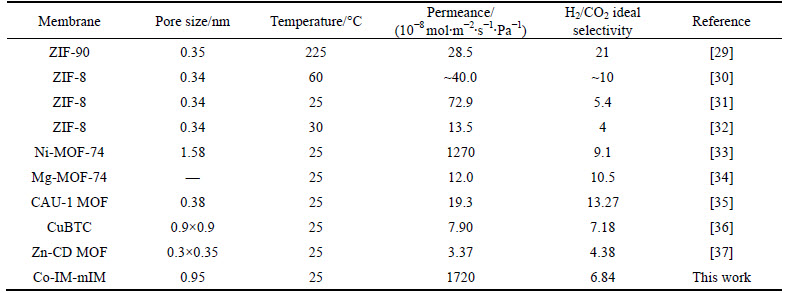
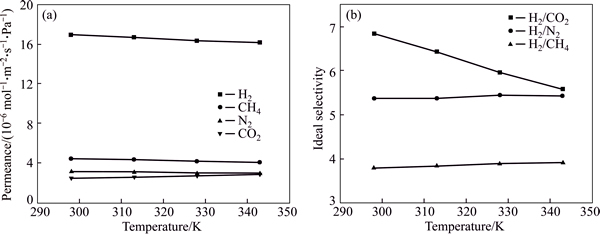
Fig. 10 Gas permeances (a) and ideal selectivities (b) for H2/CO2, H2/N2 and H2/CH4 of Co-IM-mIM membrane as a function of temperature with a trans-membrane pressure drop of 0.6��105 Pa
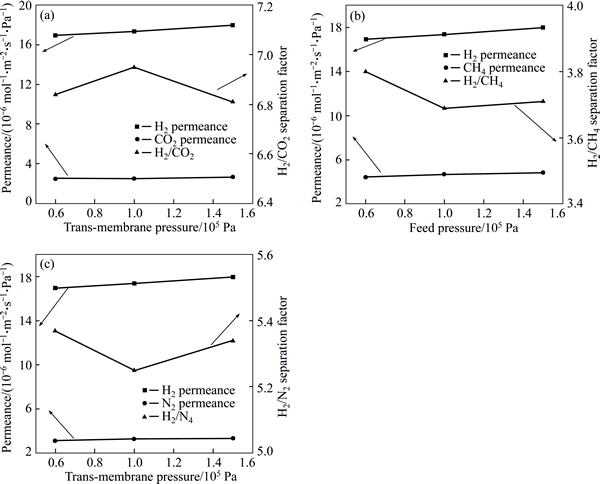
Fig. 11 Gas permeances and ideal selectivities for H2/CO2 (a), H2/CH4 (b) and H2/N2 (c) of Co-IM-mIM membrane as a function of trans-membrane pressure drop at 25 ��C
4 Conclusions
In summary, we have prepared a Co-IM-mIM membrane on a macroporous ��-alumina support by pre-depositing and secondary growth technique. The pre-depositing is essential for the formation of a defect free Co-IM-mIM membrane. The single-gas permeation experiments indicated that the Co-IM-mIM membrane demonstrates good selectivities with high permeance. The ideal selectivities of H2/CO2, H2/CH4 and H2/N2 through Co-IM-mIM membrane were 6.95, 3.40 and 5.25 with a high permeance of 17.37��10-6 mol/(m2��s��Pa) for H2 at room temperature. Such characteristics of the Co-IM-mIM membrane offer good potential toward applications for efficient H2 separation. Although some results have been obtained through the experiments, further researches based on Co-IM-mIM membrane such as the influences of mixed gas on membrane separation performance, and effects of mixed membrane and membrane modification on gas separation, etc, would be taken into consideration in the future by authors.
References
[1] LI Jian-rong, KUPPLER R J, ZHOU Hong-cai. Selective gas adsorption and separation in metal�Corganic frameworks [J]. Chemical Society Reviews, 2009, 38: 1477-1504.
[2] YAGHI O M, O��KEEFFE M, OCKWIG N W, CHAE H K, EDDAOUDI M, KIM J. Reticular synthesis and the design of new materials [J]. Nature, 2003, 423: 705-714.
[3] JIANG Lan-lan, WANG Xian-you, WU Hao, WU Chun, ZHAO Qing-lan, SONG Yun-feng. Effect of carbonization temperature on structure and electrochemical performance of porous carbon from metal framework [J]. Journal of Central South University: Science and Technology, 2013, 44(10): 4012-4018. (in Chinese)
[4] YUAN Chang-fu, LIU Jin, HAN Peng-fei, XU Jun-yi, LI Jie. Li-transfer promotion and interface stabilization of MOF-5 on PEO-based polymer electrolyte [J]. Journal of Central South University: Science and Technology, 2015, 46(4): 1189-1196. (in Chinese)
[5] HUANG Ai-sheng, DOU Wei, CARO J. Steam-stable zeolitic imidazolate framework ZIF-90 membrane with hydrogen selectivity through covalent functionalization [J]. Journal of the American Chemical Society, 2010, 132: 15562-15564.
[6] LTY A, YEUNG K L. An investigation of the relationship between microstructure and permeation properties of ZSM-5 membranes [J]. Journal of Membrane Science, 2001, 194: 33-55.
[7] LIN Li-gang, ZHANG Long-hui, ZHANG Chao, DONG Mei-mei, LIU Chun-yu, WANG An-dong, CHU Yi-jian, ZHANG Yu-zhong, CAO Zhan-ping. Membrane adsorber with metal organic frameworks for sulphur removal [J]. Rsc Advances, 2013, 3: 9889-9896.
[8] NUNES S, PEINEMANN K. 1.06 advanced polymeric and organic�Cinorganic membranes for pressure-driven processes [J]. Comprehensive Membrane Science and Engineering, 2010, 2017: 113-129.
[9] YOO Y, LAI Z, JEONG H K. Fabrication of MOF-5 membranes using microwave-induced rapid seeding and solvothermal secondary growth [J]. Microporous & Mesoporous Materials, 2009, 123: 100-106.
[10] ZHAO Zhen-xia, MA Xiao-li, KASIK A, LI Zhong, LIN Y S. Gas separation properties of metal organic framework (MOF-5) membranes [J]. Industrial & Engineering Chemistry Research, 2012, 52: 1102-1108.
[11] PHAN A, DOONAN C J, URIBE-ROMO F J, KNOBLER C B, O��KEEFFE M, YAGHI O M. Synthesis, structure, and carbon dioxide capture properties of zeolitic imidazolate frameworks [J]. Acc Chem Res, 2010, 43: 58-67.
[12] BANERJEE R, PHAN A, WANG B, KNOBLER C, FURUKAWA H, O'KEEFFE M, YAGHI O M. High-throughput synthesis of zeolitic imidazolate frameworks and application to CO2 capture [J]. Science, 2008, 319: 939-943.
[13] PARK K S, NI Z,  A P, CHOI J Y, HUANG R, URIBE-ROMO F J, CHAE H K, O��KEEFFE M, YAGHI O M. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks [J]. Proceedings of the National Academy of Sciences, 2006, 103: 10186-10191.
A P, CHOI J Y, HUANG R, URIBE-ROMO F J, CHAE H K, O��KEEFFE M, YAGHI O M. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks [J]. Proceedings of the National Academy of Sciences, 2006, 103: 10186-10191.
[14] LI Yan-shuo, LIANG Fang-yi, BUX H, YANG Wei-shen, CARO J. Zeolitic imidazolate framework ZIF-7 based molecular sieve membrane for hydrogen separation [J]. Journal of Membrane Science, 2010, 354: 48-54.
[15] LIU Yun-yang, ZENG Gao-feng, PAN Yi-chang, LAI Zhi-ping. Synthesis of highly c-oriented ZIF-69 membranes by secondary growth and their gas permeation properties [J]. Journal of Membrane Science, 2011, 379: 46-51.
[16] LI S L. CO2 and CH4 permeation through zeolitic imidazolate framework (ZIF)-8 membrane synthesized via in situ layer-by-layer growth: An experimental and modeling study [J]. Rsc Advances, 2015, 5: 79098-79106.
[17] HERT G L, BUX H, CARO J, CHMELIK C, REMSUNGNEN T, KNAUTH M, FRITZSCHE S. Diffusion of CH4 and H2 in ZIF-8 [J]. Journal of Membrane Science, 2011, 377: 36-41.
[18] LI Lun-xi, YAO Jian-feng, CHEN Ri-zhi, HE Li, WANG Kun, WANG Huan-ting. Infiltration of precursors into a porous alumina support for ZIF-8 membrane synthesis [J]. Microporous and Mesoporous Materials, 2013, 168: 15-18.
[19] PAN Yi-chang, WANG Bo, LAI Zhi-ping. Synthesis of ceramic hollow fiber supported zeolitic imidazolate framework-8 (ZIF-8) membranes with high hydrogen permeability [J]. Journal of Membrane Science, 2012, 421: 292-298.
[20] BURROWS A D. Mixed-component metal�Corganic frameworks (MC-MOFs): Enhancing functionality through solid solution formation and surface modifications [J]. Cryst Eng Comm, 2011, 13: 3623-3642.
[21] THOMPSON J A, BLAD C R, BRUNELLI N A, LYDON M E, LIVELY R P, JONES C W, NAIR S. Hybrid zeolitic imidazolate frameworks: Controlling framework porosity and functionality by mixed-linker synthesis [J]. Chemistry of Materials, 2012, 24: 1930-1936.
[22] KAHR J, MOWAT J P, SLAWIN A M, MORRIS R E, FAIREN- JIMENEZ D, WRIGHT P A. Synthetic control of framework zinc purinate crystallisation and properties of a large pore, decorated, mixed-linker RHO-type ZIF [J]. Chemical Communications, 2012, 48: 6690-6692.
[23] SHAH M, MCCARTHY M C, SACHDEVA S, LEE A K, JEONG H K. Current status of metal�Corganic framework membranes for gas separations: Promises and challenges [J]. Industrial & Engineering Chemistry Research, 2012, 51: 2179-2199.
[24] QIU Shi-lun, XUE Ming, ZHU Guang-shan. Metal�Corganic framework membranes: from synthesis to separation application [J]. Chemical Society Reviews, 2014, 43: 6116-6140.
[25] SHAH M, MCCARTHY M C, SACHDEVA S, LEE A K, JEONG H K. Current status of metal�Corganic framework membranes for gas separations: Promises and challenges [J]. Industrial & Engineering Chemistry Research, 2012, 51: 2179-2199.
[26] TANCO M A L, TANAKA D A P, RODRIGUES S C, TEXEIRA M, MENDES A. Composite-alumina-carbon molecular sieve membranes prepared from novolac resin and boehmite. Part I: Preparation, characterization and gas permeation studies [J]. International Journal of Hydrogen Energy, 2015, 40: 5653-5663.
[27] CAO Fa, ZHANG Chun-juan, XIAO Yuan-long, HUANG Hong-liang, ZHANG Wen-juan, LIU Da-huan, ZHONG Chong-li, YANG Qing-yuan, YANG Zhu-hong, LU Xiao-hua. Helium recovery by a Cu-BTC metal�Corganic-framework membrane [J]. Industrial & Engineering Chemistry Research, 2012, 51: 11274-11278.
[28] LIU Yun-yang, HU En-ping, KHAN E A, LAI Zhi-ping. Synthesis and characterization of ZIF-69 membranes and separation for CO2/CO mixture [J]. Journal of Membrane Science, 2010, 353: 36-40.
[29] HUANG Ai-sheng, LIU Qian, WANG Nan-yi, CARO J. Organosilica functionalized zeolitic imidazolate framework ZIF-90 membrane for CO2/CH4 separation [J]. Microporous & Mesoporous Materials, 2014, 192: 18-22.
[30] GE Lei, ZHOU Wei, DU Ai-jun, ZHU Zhong-hua. Porous polyethersulfone-supported zeolitic imidazolate framework membranes for hydrogen separation [J]. Journal of Physical Chemistry C: Nanomaterials Interfaces & Hard Matter, 2012, 116: 13264-13270.
[31] TAO Kai, CAO Lu-jie, LIN Yi-chao, KONG Chun-long, CHEN Liang. A hollow ceramic fiber supported ZIF-8 membrane with enhanced gas separation performance prepared by hot dip-coating seeding [J]. Journal of Materials Chemistry, 2013, 268: 13046-13049.
[32] LIU Yi, WANG Nan-yi, LISA D, FRANK S,  C J. MOF membrane synthesis in the Confined space of a vertically aligned LDH network [J]. Chemical communications, 2014, 50: 4225-4227.
C J. MOF membrane synthesis in the Confined space of a vertically aligned LDH network [J]. Chemical communications, 2014, 50: 4225-4227.
[33] LEE D J, LI Qi-ming, KIM H, LEE K. Preparation of Ni-MOF-74 membrane for CO2 separation by layer-by-layer seeding technique [J]. Microporous & Mesoporous Materials, 2012, 163: 169-177.
[34] WANG Nan-yi, MUNDSTOCK A, LIU Yi, HUANG Ai-sheng, CARO J, Amine-modified Mg-MOF-74/CPO-27-Mg membrane with enhanced H2/CO2 separation [J]. Chemical Engineering Science, 2015, 124: 27-36.
[35] ZHOU Shu-yuan, ZOU Xiao-qin, SUN Fu-xing, REN Hao, LIU Jia, ZHANG Feng, ZHAO Nian, ZHU Guang-shan. Development of hydrogen-selective CAU-1 MOF membranes for hydrogen purification by ��dual-metal-source�� approach [J]. International Journal of Hydrogen Energy, 2013, 38: 5338-5347.
[36] NAGARAJU D, BHAGAT D G., BANERJEE R, KHARUL U K. In situ growth of metal-organic frameworks on a porous ultrafiltration membrane for gas separation [J]. J Mater Chem A, 2013, 1: 8828-8835.
[37] HUANG Kang, LIU Sai-nan, LI Qian-qian, JIN Wan-qin. Preparation of novel metal-carboxylate system MOF membrane for gas separation [J]. Separation & Purification Technology, 2013, 119: 94-101.
[38] BUX H, CHMELIK C, van BATEN J M, KRISHNA R, CARO J. Novel MOF-membrane for molecular sieving predicted by IR-diffusion studies and molecular modeling [J]. Advanced Materials, 2010, 22: 4741-4743.
[39] HU Yao-xin, DONG Xue-liang, NAN Jiang-pu, JIN Wan-qin, REN Xiao-ming, XU Nan-ping, LEE Young-moo. Metal�Corganic framework membranes fabricated via reactive seeding [J]. Chemical Communications, 2011, 47: 737-739.
[40] NAN Jiang-pu, DONG Xue-liang, WANG Wen-jin, JIN Wan-qin, XU Nan-ping. Step-by-step seeding procedure for preparing HKUST-1 membrane on porous ��-alumina support [J]. Langmuir, 2011, 27: 4309-4312.
[41] LI Yan-Shuo, BUX H, FELDHOFF A, LI Guo-Ling, YANG Wei-shen, CARO J. Controllable synthesis of metal�Corganic frameworks: From MOF nanorods to oriented MOF membranes [J]. Advanced Materials, 2010, 22: 3322-3326.
[42] ZOU Xiao-qin, ZHU Guang-shan, ZHANG Feng, GUO Ming-yi, QIU Shi-lun. Facile fabrication of metal�Corganic framework films promoted by colloidal seeds on various substrates [J]. Cryst Eng Comm, 2010, 12: 352-354.
[43] ZHAO Zhen-xia, MA Xiao-li, LI Zhong, LIN Y S. Synthesis, characterization and gas transport properties of MOF-5 membranes [J]. Journal of Membrane Science, 2011, 382: 82-90.
[44] KANEZASHI M, LIN Y S. Gas permeation and diffusion characteristics of MFI-type zeolite membranes at high temperatures [J]. The Journal of Physical Chemistry C, 2009, 113: 3767-3774.
[45] ALGIERI C, BERNARDO P, GOLEMME G, BARBIERI G, DRIOLI E. Permeation properties of a thin silicalite-1 (MFI) membrane [J]. Journal of Membrane Science, 2003, 222: 181-190.
[46] ADATOZ E, AVCI AK, KESKIN S. Opportunities and challenges of MOF-based membranes in gas separations [J]. Separation and Purification Technology, 2015, 152: 207-237.
[47] GUO Hai-ling, ZHU Guang-shan, HEWITT I J, QIU Shi-lun. ��Twin copper source�� growth of metal-organic framework membrane: Cu3(BTC)2 with high permeability and selectivity for recycling H2 [J]. Journal of the American Chemical Society, 2009, 131: 1646- 1647.
(Edited by YANG Hua)
Cite this article as: GAO Zheng-cai, LI Li-qing, LI Hai-long, CHEN Ruo-fei, WANG S, WANG Yang-gang. A hybrid zeolitic imidazolate framework Co-IM-mIM membrane for gas separation [J]. Journal of Central South University, 2017, 24(8): 1727-1735. DOI: https://doi.org/10.1007/s11771-017-3580-z.
Foundation item: Project(21376274) supported by the National Natural Science Foundation China; Project(2015BAL04B02) supported by the National Key Technology R&D Program of China
Received date: 2016-02-29; Accepted date: 2016-10-06
Corresponding author: LI Li-qing, PhD, Professor; Tel: +86-731-88879863; E-mail: liqingli@hotmail.com

