
Densities of molten Ni-(Cr, Co, W) superalloys
XIAO Feng(肖 锋)1, YANG Ren-hui(杨仁辉)1, FANG Liang(方 亮)2,
LIU Lan-xiao(刘兰霄)1, ZHAO Hong-kai(赵红凯)1
1. Materials Interfacial Physical-Chemistry Research Institute, Chongqing Institute of Technology,Chongqing 400050, China;
2. Department of Applied Physics, Chongqing University, Chongqing 400044, China
Received 16 April 2007; accepted 10 September 2007
Abstract: In order to obtain more accurate density for molten Ni-(Cr, Co, W) binary alloy, the densities of molten pure Ni and Ni-Cr, Ni-Co, Ni-W alloys were measured with a sessile drop method. It is found that the measured densities of molten pure Ni and Ni-Cr, Ni-Co, Ni-W alloys decrease with increasing temperature in the experimental temperature range. The density of alloys increases with increasing W and Co concentrations while it decreases with increasing Cr concentration in the alloy at 1 773-1 873 K. The molar volume of Ni-based alloys increases with increasing W concentration while it decreases with increasing Co concentration. The effect of Cr concentration on the molar volume of the alloy is little in the studied concentration range. The accommodation among atomic species was analyzed. The deviation of molar volume from ideal mixing shows an ideal mixing of Ni-(Cr, Co, W) binary alloys.
Key words: Ni-Cr; Ni-Co; Ni-W; superalloy; density; molar volume
1 Introduction
The density of molten metals and alloys is one of the most important thermal physical properties for studying their structure. We are particularly interested in the density of the molten Ni-based alloys because they are used in castings of critical components in gas turbine engines. Up to the present, the density of the molten Ni-Cr or Ni-Co alloy was only measured by EREMENKO et al[1], KATO et al[2], MUKAI et al[3], WATANABE et al[4] and DZHEMILEV et al[5] while the density of the molten Ni-W alloys has not been reported in the literatures. Moreover, there is a large scatter in the available data. As a result, it is difficult to use the available data for the mathematical simulation techniques for material phenomena. In order to obtain more accurate density data of the molten Ni-(Cr, Co, W) binary alloys, the densities of molten Ni-(Cr, Co, W) binary alloys were measured with a sessile drop method over the temperature range of 1 773-1 873 K in a high-purity Ar+3%H2 atmosphere in this work.
2 Experimental
A sessile drop method[6-8] was used to measure the densities of Ni-(Cr, Co, W) binary alloys. As shown in Fig.1, the experimental apparatus consists of a heating furnace with a Ta cylindrical heater, five concentric Mo reflectors and a thermocouple located close to the sample, two He-Ne laser generators, and two high resolution photographic systems including two digital cameras, two computers and two band-pass filters, which were set up between the camera and the sample to filtrate other light except He-Ne laser with a wavelength of 632 nm. The temperature at the place of the sample is calibrated using the melting point of pure Ni.
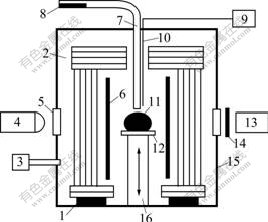
Fig.1 Schematic diagram of experimental apparatus: 1 Alumina support; 2 Mo reflector; 3 Evacuating system; 4 He-Ne laser generator; 5 Observation window; 6 Ta heater; 7 Alumina tube; 8 Sample before experiment; 9 Temperature controller; 10 Thermocouple; 11 Sample in experiment; 12 Substrate; 13 Digital camera; 14 Band-pass filter; 15 Chamber; 16 Sample elevator
The used alloy samples were non-doped materials with 99.99% (mass fraction) purity. Since a few impurities in substrates also can affect the result, high purity (99.99%, mass fraction) polycrystalline alumina substrates with dimensions of 20(diameter)×5(thickness) were used. The alloy sample was cleaned in acetone for three times and set in the sample container. When the temperature reached the experimental one, the solid sample in the container was moved to the end of the alumina tube connecting with the furnace chamber and molten down. By controlling the pressure difference in the chamber and the alumina tube, the molten sample dropped smoothly on the surface of the horizontal alumina substrate in the furnace. Then, the molten sample drop was photographed. The coordinates of approximately 99 points of the droplet profile were determined from the photographs and the density was automatically calculated using a kind of software. In the apparatus, two He-Ne laser generators and two high resolution photographic systems were used to record the shape of molten sample from two perpendicular directions at horizontal plane. As a result, it is easy to observe and find the distortion of molten sample if it exists. In this work, the density of the molten alloy at 1 773, 1 823 and 1 873 K in an Ar+3%H2 atmosphere was studied. The total maximum relative uncertainty for the method in this work was estimated as ±1.25%[9-13].
3 Results and discussion
3.1 Densities of molten Ni-(Cr, Co, W) alloys
The densities of the molten Ni-(Cr, Co, W) alloys measured in this work are shown in Fig.2. The densities of molten binary alloys decrease with increasing temperature. STEINBERG[14], FANG and XIAO[15] proposed that the density data of molten metals and alloys could be represented by the following equation:
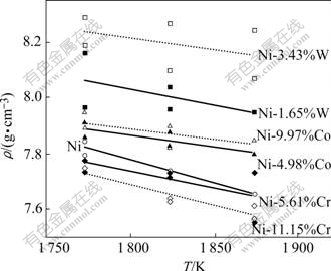
Fig.2 Temperature dependence of densities of Ni-(Cr, Co, W) alloy
r=rL+k(T-TL) (1)
where rL, in g/cm3, stands for the density at the liquidus temperature TL and k, in g/(cm3?K), is the temperature coefficient of density at constant pressure that is expressed as
 (2)
(2)
where T is the temperature, K.
If we follow Steinberg formula, the densities of molten Ni, Ni-Cr, Ni-Co and Ni-W (molar fraction) alloys can be described as
r(Ni)=7.90-1.70×10-3(T-1 728) (3)
r(Ni-1.65%W)=8.10-1.17×10-3(T-1 743) (4)
r(Ni-3.43%W)=8.29-1.61×10-3(T-1 753) (5)
r(Ni-4.98%Co)=7.91-6.00×10-4(T-1 730) (6)
r(Ni-9.97%Co)=7.97-1.20×10-3(T-1 732) (7)
r(Ni-5.61%Cr)=7.87-1.83×10-3(T-1 725) (8)
r(Ni-11.15%Cr)=7.84-1.80×10-3(T-1 717) (9)
The density of molten Ni at the melting point and its temperature coefficient obtained are in good agreement with the values reported by MUKAI and XIAO[3] using the following modified sessile drop method:
r=7.91-1.43×10-3(T-1 728) (1 728-1 923 K) (10)
The alloy concentration dependence of the densities is shown in Fig.3. It is found that the density of alloys trends to increase with increasing W and Co concentration, while it trends to decrease with increasing Cr concentration at 1 773-1 873 K.

Fig.3 Effects of alloy concentration on density of molten alloys
3.2 Molar volume for molten Ni-(Cr, Co, W) alloys
The molar volume of molten Ni-(Cr, Co, W) alloys can be calculated from the molar mass and the density of alloys by
Vmol=malloy/ρalloy (11)
where Vmol, malloy and ralloy are the molar volume, the molar mass and the density of alloys, respectively. Inserting the values of malloy and ralloy for every alloy into Eqn.(11), the molar volume of alloys can be expressed as
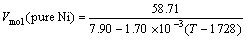 (12)
(12)
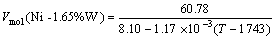 (13)
(13)
 (14)
(14)
 (15)
(15)
 (16)
(16)
 (17)
(17)
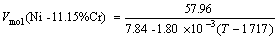 (18)
(18)
The molar volume of the Ni-(Cr, Co, W) binary alloys is plotted as isothermal molar volume at different temperatures in Fig.4. The molar volume of Ni-W alloys trends to increase with increasing W concentration although the density increases with it. The reason is that the increasing rate of molar mass of the alloys with W concentration is larger than that of the density with W concentration. As a result, both the density and the molar volume of the alloys increase with increasing W concentration in the alloys.

Fig.4 Relationship between molar volume of Ni-(Cr, Co, W) binary alloys and alloy concentration
The molar volume of Ni-Co alloys trends to decrease with increasing Co concentration. The effect of Cr concentration on the molar volume of the alloys is little in the studied concentration range.
The study of the accommodation among atomic species is an important task, which is expressed as a deviation, DVmix, of the liquid solution from ideal volumetric mixing:
DVmix=Vmol-Videal (19)
where Vmol is the molar volume calculated from the density and Videal is the molar volume in ideal volumetric mixing.
For the Ni-based binary alloys:
(20)

where x(Ni) and x(i) are the molar fractions of Ni and other elements; m(Ni) and m(i) the molar masses of Ni and other elements, r(Ni-i), r(Ni) and r(i) are the densities of Ni-based alloys, pure Ni and pure alloy element, respectively.
In the following calculation, the density of Ni measured in present work and that of Co from Ref.[6] are used:
r(Co)=7.75-1.65×10-3(T-1 768) (T>1 768 K) (21)
Because W and Cr do not exist in the liquid state at the temperature lower than their melting points (3 695 K and 2 180 K), it is difficult to strictly verify the molar volume in Eqn.(20) of the molten Ni-W and Ni-Cr system. However, if one assumes that the relationship proposed by LEVIN and AYUSHINA[16] can be used to calculate the density of molten W and Cr in metastable state at the temperature lower than melting point, the density of metastable W and Cr can be calculated as[17]:
r(W)=17.6 g/cm3 (at melting point 3 695 K) (22)
r(Cr) =6.30-1.10×10-3(T-2 180) (T>2 180 K) (23)
Then the deviation of molar volume from ideal mixing was calculated and the results are shown in Fig.5. The deviation of molar volume of Ni-(Cr, Co, W) binary alloys from ideal mixing trends to be zero. This shows an ideal mixing of the alloys.

Fig.5 Deviation of molar volume from ideal mixing in Ni-based alloys
4 Conclusions
1) Densities of molten Ni-(Cr, Co, W) binary alloys were measured at 1 773-1 873 K with a sessile drop method. The densities of molten binary alloys decrease with increasing temperature. The densities of alloys increase with increasing W and Co concentration, while decrease with increasing Cr concentration in Ni-Cr alloys at 1 773-1 873 K.
2) The molar volumes of Ni-(Cr, Co, W) binary alloys increase with increasing W concentration while decrease with increasing Co concentration. The effect of Cr concentration on the molar volume of the alloys is little in the studied concentration range. And the deviation of molar volume from ideal mixing shows an ideal mixing of the alloys.
References
[1] EREMENKO V N, NAIDICH Y V. LAVRINENKO I A. The density and free surface energy of liquid alloys of the system nickel- chromium [R]. New York and London: Consultants Bureau, 1970.
[2] KATO M, IKUTA S, HISADA K, HASEGAWA J. The density of liquid Ni-based alloys [R]. Nagoya: Government Industrial Research Institute, 1987, 36: 152-166.
[3] MUKAI K, XIAO F. Density of Ni-Cr alloy in liquid and solid-liquid coexistence states [J]. Materials Transactions, 2002, 43: 1153-1160.
[4] WATANABE S, AMATATU M, SAITO T. Densities of Fe-Ni, Co-Ni, Co-Mo and Co-W alloys in liquid state [J]. Transaction of Japan Institute of Metal, 1971, 12: 337-3342. (in Japanese)
[5] DZHEMILEV N K, POPEL S I, TSAREVSKII B V. Density and surface properties of iron-cobalt-nickel melts at 1 550? [J]. Russian Journal of Physical Chemistry, 1967, 41: 24-27. (in Russian)
[6] NOGI K, OISHI K, OGINO K. Wettability of solid oxides by liquid pure metals [J]. Materials Transactions JIM, 1989: 30: 137-145.
[7] FUJII H, NAKAE H. Equilibrium contact angle in the magnesium oxide/aluminium system [J]. Acta Materialia, 1996, 44: 3567-3573.
[8] XIAO Feng, FANG Liang. Solidification shrinkage of Ni-Cr alloys [J]. Materials Science and Engineering B, 2006, 132: 193-196.
[9] XIAO Feng, FANG Liang. Density and structure analysis of molten Ni-W alloys [J]. Journal of Materials Science & Technology, 2004, 20: 410-413.
[10] XIAO Feng. Density of liquid steels over the temperature range of 1 803-1 873 K [J]. Journal of Iron Steel Research International, 2004, 11: 37-40.
[11] FANG Liang, XIAO Feng. Density of Ni-Al alloys in liquid and solid-liquid coexistence state measured by a modified pycnometric method [J]. Journal of Materials Science & Technology, 2004, 20: 405-410.
[12] XIAO Feng. Evaluation of modified pycnometric method for accurately measuring the density of molten nickel [J]. Materials Review, 2004, 18: 250-251.
[13] FANG Liang, XIAO Feng. Measurement of the density of molten Ni-Ta alloys by a modified pycnometric method [J]. Rare Metal Materials & Engineering, 2005, 34: 521-525.
[14] STEINBERG D J. A simple relationship between the temperature dependence of the density of liquid metals and their boiling temperature [J]. Metallurgy Transaction, 1974, 5A: 1341-1343.
[15] FANG Liang, XIAO Feng. Density of Ni-Co-Al ternary alloys in liquid and mushy states measured by a modified pycnometric method [C]// International Conference on Aerospace Materials, Beijing: 2006.
[16] LEVIN E S, AYUSHINA G D. Study of some physico-chemical properties of Ni-Al melts [J]. Izv Akad Nauk SSSR, Met, 1971(1): 227-229.
[17] DAVID R L. Handbook of chemistry and physics [M]. London: CRC Press, 4-124.
Foundation item: Project(2000-2005) supported by the New Energy and Industrial Technology Development Organization in Japan; Project(2004527) supported by Scientific Research Foundation for the Returned Overseas Chinese Scholar; Project(200594) supported by Chongqing Bureau of Personnel, China; Project(CSTC2005BA4016-1) supported by the National Scientific Foundation of Chongqing Municipality, China; Project(2003ZD31) supported by Chongqing Institute of Technology, China
Corresponding author: XIAO Feng; Tel: +86-23-66966286; E-mail: xiaofeng@cqit.edu.cn
(Edited by LI Xiang-qun)