��ԭ���������Լ��ĸ���ЧӦ������Ƭ״���ϳ��е�����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���11��
�������ߣ��� �� �¿Ʊ� ���Ȼ �� � ������ �� ��
����ҳ�룺3732 - 3738
�ؼ��ʣ�����Ƭ״������ԭ����������Լ�����������
Key words��silver nanoplates; reducing agents; surfactants; capping effect
ժ Ҫ���о���ԭ���������Լ�����Ƭ״���γɵ�Ӱ�죬��������������ԭ�������������ơ���ʯ��أ��Լ�3�ֳ��ñ�����Լ�������ϩ������ͪ��ʮ����������ơ�����������Ӱ�졣�����������������ԭ���������Լ������п�����������(111)����ܽ�ϵĻ��Ի��ţ��Ӷ���������ЧӦ��ֹ�þ������������մﵽ�γ�Ƭ״����Ŀ�ġ��������ʾ��±�����ӵ��컯������Ƭ״������������Ҳ����һ����Ӱ�졣
Abstract: The effect of reducing agents and surfactants on controlling the silver nanoplates was studied. Two reducing agents, trisodium citrate dehydrate (TSC) and potassium tartrate, were used to reduce the AgNO3 solution. In this redox system, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS) and arabic gum were served as surfactants. The results showed that reducing agents and surfactants both act as the capping agent adhering to the certain facets of silver seeds to block this surface to grow. The relative intensity of reducing agents also takes an active part in influencing the growth rate and direction of silver seeds. It was also found that halides can accelerate the speed of Ostwald ripening by adding Cl-, Br- and I- into the aqueous and have some effects on the morphology of the nanoplates.
Trans. Nonferrous Met. Soc. China 24(2014) 3732-3738
Yang YAN, Ke-bin CHEN, Hao-ran LI, Wei HONG, Xiao-bin HU, Zhou XU
State Key Laboratory of Metal Matrix Composites, Shanghai Jiao Tong University, Shanghai 200240, China
Received 16 December 2013; accepted 12 September 2014
Abstract: The effect of reducing agents and surfactants on controlling the silver nanoplates was studied. Two reducing agents, trisodium citrate dehydrate (TSC) and potassium tartrate, were used to reduce the AgNO3 solution. In this redox system, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS) and arabic gum were served as surfactants. The results showed that reducing agents and surfactants both act as the capping agent adhering to the certain facets of silver seeds to block this surface to grow. The relative intensity of reducing agents also takes an active part in influencing the growth rate and direction of silver seeds. It was also found that halides can accelerate the speed of Ostwald ripening by adding Cl-, Br- and I-into the aqueous and have some effects on the morphology of the nanoplates.
Key words: silver nanoplates; reducing agents; surfactants; capping effect
1 Introduction
The shape-controlled synthesis of metallic nanoparticles has become a hot topic in the synthetic field. The morphology of noble metals has a great effect on their properties and chemical and physical performances [1,2]. Silver nanoparticles which are easy to prepare and with lots of good properties like catalysis and photic property catch researcher��s eyes [3]. Research shows that the type, size, morphology and dimension of silver nanoparticles in different degree affect their own optical, electrical, and magnetic properties. As a result, the synthesis of shape-controlled silver nanocrystal becomes attractive. That is, by changing the temperature, chemicals, density and other factors, we can change the shape, size and crystalline type of silver nanoparticles [4]. The experimental results have proved that silver nanoparticles have various shapes and each of them has unique characteristics. One-dimensional (1D) nano- structures (wires, rods, and tubes) are expected to play an important role in fabricating nanoscale devices [5]. Two-dimensional (2D) and three-dimensional (3D) nanostructures like nanoplates, nanodisks, nanocubes, nanospheres and octahedron carry authority in applications of silver nanocrystal [6]. GAO et al [7] and BENJAMIN et al [8] have achieved this work. Their teams have successfully synthesized nanocubes, nanowires, rods and other high-index nanoparticles by different methods. PIETROBON and KITAEV [9] have found the way to synthesize decahedral nanoparticles. Silver nanoplates, also referred to as nanoprisms or nanodisks, are 2D plasmonic nanostructures that have attracted intensive attention due to their strong shape- dependent optical properties and related applications. Many researchers try to find the exact answer of how to control the shape of nano-silver [10]. In this way we can synthesize the silver nanoparticles in proper shape for different applications.
ZHANG et al [4] had a general explanation of the inner factors of reducing agent and surfactants in controlling the morphology of silver nanoparticles. There are two main theories that are well received. One is the face blocking theory. Capping agent like reducing agent and surfactant selectively adheres to a particular crystal facet of the growing nanocrystal and thus slows the growth rate of that facet relatively to the others [11,12]. The other factor controlling the morphology is the starting nuclei of the nanocrystal [13,14]. However, the mechanism of action of capping theory has not been totally made clear. How these capping drugs adhere to the certain facets still cannot be answered. And also There is still a question why the yield of silver triangular nanoplates is different between strong and weak reductants.
In this study, a series of experiments were conducted to discuss how the reducing agent, surfactant and halide ions control the crystal��s shape. In the experiment, sodium citrate acts as not only the reducing agent but also capping agent. This capping function was discussed in details by comparing with that of citrate and tartrate. The results also show that surfactants in this system have the same function as capping agent.
2 Experimental
2.1 Chemicals
Hydrogen peroxide (H2O2), sodium potassium tartrate, silver nitrate (AgNO3), sodium borohydride (NaBH4), sodium citrate tribasic dehydrate (TSC) were obtained from Sigma-Aldrich. Polyvinylpyrrolidone (PVP, relative molecular mass of about 29000), sodium dodecyl sulfate (SDS), arabic gum, sodium chloride (NaCl), sodium bromide (NaBr), and sodium iodide (NaI) were obtained from Sinopharm. All chemicals were analytical reagent used as-received without further treatment.
2.2 Synthesis of Ag nanoparticles
First, a standard reaction solution was prepared. 50 ��L silver nitrate (0.05 mol/L), 0.5 mL trisodium citrate (75 mmol/L) and 60 ��L H2O2 (30%) were put into 24.14 mL deionized water at room temperature with vigorous stirring. At the same time 250 ��L NaBH4 (100 mmol/L) was added into the aqueous solution. The solution turned into light yellow. But after about 3 min, the solution suddenly became red, aubergine, then purple and at last turned blue. In the next series of experiments, certain surfactants were added into the solution. And different surfactants were used for comparison. The solution turned blue in a longer time of 10-30 min or even kept the color of light yellow. At the last part of the experiments, different anions were mixed in the prepared solution. The anions were differed by nuclear charge number, configuration of extra-nuclear electron and the spatial structure of the anions. Most of the anions did not have obvious effect on the synthesized silver nanoplates. Only the halide ions could change the color of the solution from blue to yellow, which means the morphology changed.
2.3 Characterization
The morphology of Ag nanoparticles was characterized using a Tecnai T12 transmission electron microscope (TEM). The measurement of optical property was conducted using a Varian Cary 50 UV/vis spectrophotometer (190-1100 nm).
3 Results and discussion
Many experiments have achieved the synthesis of nanocrystalline metal like gold, platinum and silver which use sodium citrate tribasic (TSC) as reducing agents [15-17]. The silver nanocrystal was prepared by reducing the aqueous solution of AgNO3. When Ag+ was reduced by TSC, the resultant solution was blue while the solution was yellow when reduced by potassium tartrate. And when PVP was added, the reaction didn��t stop after 3 min. With the content of PVP growing, the response time became longer and longer, however, the color did not change much. In contrast, when tiny amount of halides was added into the system, the color of the reaction would change immediately. In this work, reducing agents and surfactants which acted as capping agent have been proved playing essential parts in making the silver nanoplates. And this effect will be discussed in details in the following parts.
3.1 Reducing agent
ZHANG et al [4] conducted a series of work that not all the reducing agent can make the Ag+ into nanoplates. Their result showed that reducing agent with specially appointed structure can act as the capping agent. The capping structure can adhere to the (111) facet of the silver seeds to prevent this facet growing. In this work, TSC and potassium tartrate were taken as examples. Figures 1 and 2 respectively show the TEM images and absorption spectra of the silver triangular nanoplates that were reduced by TSC and potassium tartrate. Both of these two groups resulted in nanoplates. This results testified ZHANG et al��s conclusion that the structure with dicarboxylate groups is just like a lid that caps on the silver (111) facet blocking the growth rate of it, and as a result this facet retains [18]. Figure 3 shows the molecular structure of TSC. We can see that there are two carbon atoms between the nearest two carboxylate groups. Potassium tartrate has the same character. This structure is the key to the synthesis of silver nanoplates for it tends to adhere to (111) facet of silver seed. Meanwhile, the molecular structures tell us that there are two dicarboxylate groups in TSC while there is only one in potassium tartrate. So in this experiment we can see from Fig. 1 that 90% of the resultant reduced by TSC is triangular nanoplates while silver nanoplates reduced by potassium tartrate are almost disks and hexagonal nanoplates [19,20] whose yield of triangular plates is rarely about 50%. This phenomenon proved ZHANG et al��s conclusion further. The dicarboxylate groups act as capping agent in this kind of reactions.

Fig. 1 TEM images showing morphology of products reduced by TSC (a) and potassium tartrate (b)

Fig. 2 UV/vis spectra of silver nanoparticles reduced by different reducing agents by controlling concentration of H2O2 (b)
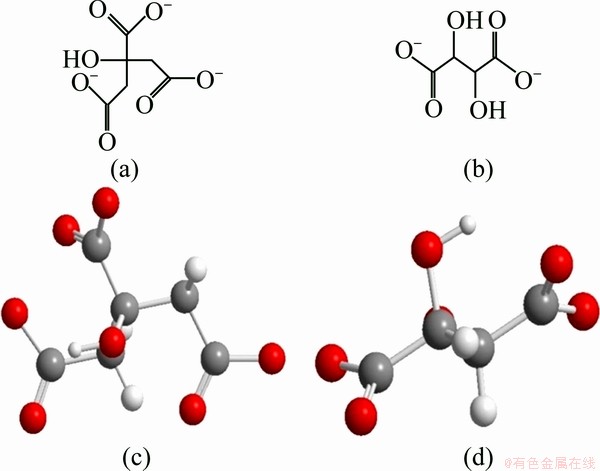
Fig. 3 Molecular structures of sodium citrate (TSC) (a) and potassium tartrate (b); space structures of TSC (c) and potassium tartrate (d)
If we consider this dicarboxylate groups can lead to the structure of nanoplates, the reducibility of reducing agents may also affect the growth of the (100) and (110) facets. Research showed that twinning defects and surface stacking dislocations play roles in controlling the morphology of silver nanocrystal, and silver seeds have much more defects when they are reduced by weak reducing agent [21-23]. Figure 4 shows that the fine structures of the triangular nanoplates and nanodisks consist of (111), (110) and (100) facets [24,25]. Generally silver is face-centered cubic. The surface energy successively increases in the order of (111), (100) and (110). And facet which grows fast swallows that grows slow [26]. Figure 4 shows that when the growth rate of (100) and (110) is similar, the triangular plates will turn into disks. As the results show, TSC and potassium tartrate have different effects on the (110) facet for they have different yield of triangular nanoplates. It is inferred that weak reducing agent potassium tartrate brings more defects to the silver seeds than TSC does, which will change and balance the growth rate of (100) and (110) facets. In order to test this surmise we conducted experiments that proper H2O2 was added into the system. It can etch the seeds in order to increase the twinning defects and surface stacking [27-29]. Nanoplates in Fig. 5(b) are 100 nm and those in Fig. 5(a) are 20 nm. It is clear that the yield of triangular plates reduced in the resultant for they had the same initial mass of Ag+ and acute angel also reduced. This experiment supported the result above. Therefore, a conclusion can be drawn that the reducibility of reducing agents can control the morphology of silver nanocrystal by changing the scale of defects.
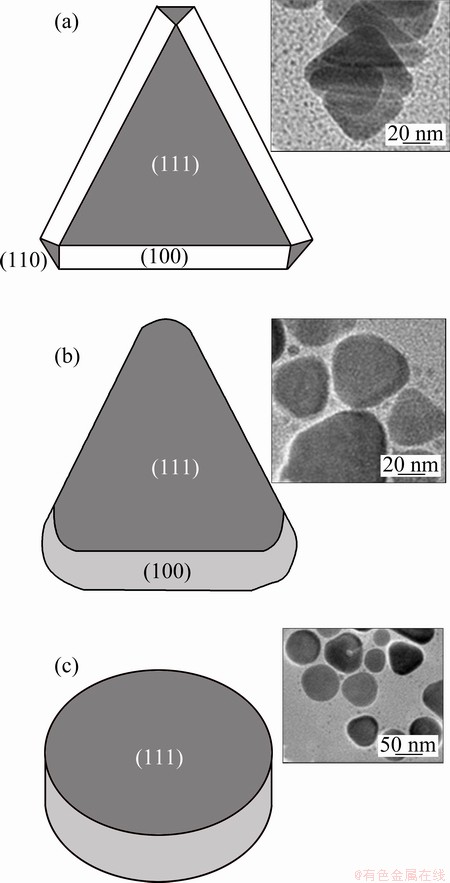
Fig. 4 Structural models showing facet composition of triangular nanoplates (a), alteration triangular nanoplates (b) and silver nanodisks (c) (The insets in (a), (b) and (c) are corresponding TEM images)
Generally, the morphology of silver nanocrystal can be controlled by changing the activity of reducing agent, and we can find reducing agents that can block different specific facets so that we can easily control the morphology of nanocrystal.
3.2 Surfactant
HUANG et al found that TSC controlled the morphology by changing the proportion of AgNO3 and PVP [30]. They have successfully prepared silver nanowires, nanoplates and nanobelts. So surfactants surely can control the morphology of the silver nanocrystal [31,32]. Experiments were conducted with different kinds and contents of surfactants, and the TEM images of the resultants are shown in Fig. 6. It is obvious that these results didn��t match the result of HUANG et al. This difference perhaps was caused by the concentration of surfactants. The concentration was much higher in our study than that in HUANG et al��s work. As a result surfactants began to play a particular part in controlling the shape compared with the previous results. Resultants still keep the shape of nanoplates like triangular, disks and hexagon. Table 1 shows that more surfacants cannot change the morphology of nanoplates but can slow the speed of this reaction. ROCKENBERGER et al [33] demonstrated that the PVP tends to bind tightly with the silver atoms on the (111) facet. The surfactants are usually high polymer with many functional groups. These functional groups will coat the silver in case of the particle reunion. In this way the growth rate of silver seeds reduces. As a result the silver nanocrystal can grow larger. Also (100) and (110) facets will grow in an equivalent speed. This phenomenon is in favour of silver atoms transfer and changes a small amount of triangular nanoplates into nanodisks. The color of the resultants didn��t have obvious change as shown in Fig. 7 which shows the absorption spectra of resultants with 0, 100 and 200 ��L PVP solution. In other words, surfactants like PVP with polar groups act as the capping agent in these kinds of experiments and they won��t have a great effect on the morphology as HUANG��s work.

Fig. 5 TEM images showing morphology and scale of products

Fig. 6 TEM images of silver nanoplates with different surfactants
Table 1 Response time and color of resultants with different surfactants added into reaction system
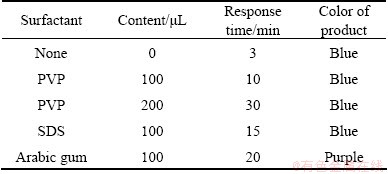

Fig. 7 Absorption spectra of reaction product solution with different additions of PVP
3.3 Halide
In this experiment, NaCl, NaBr and NaI were added into the reaction solution. The solution immediately turned into yellow after halide was added. From Fig. 8 we can see that there is only one obvious absorption peak in the solution without halides while three peaks with halides. The number and value of adsorption peaks all indicate that halides have some effect on the morphology of nano-silver. Figure 9 shows the TEM images of the blue and yellow resultants. The reaction product changes from triangular nanoplates into nanodisks. We also tried to use other anions, but there is no marked change. YANG et al [34] and CHEN et al [35] have achieved that they can change silver nanospheres into silver triangular nanodisks by adding NaCl in the reaction. Halides play an essential role in these two experiments and they have the same feature to change the relative unstable silver nanocrystals into stable state. The reasons are that halides are anion ligands and these anion ligands easily combine with oxygen. Halides and oxygen form a powerful etching agent. In fact silver triangular nanoplates are not the most stable state. We put the reaction product without surfactant in shading environment at room temperature for 7 d. The solution would turn into yellow or even colorless. With time going on, silver atoms will transfer from one facet to another and ions in the solution can also produce oxidation etching to form the most stable nanodisks. This phenomenon is called Ostwald ripening [36]. In this experiment, halide with oxygen which is a powerful etching agent can etch the unstable twin crystal and silver triangular nanoplates grow into nanodisks. Halides accelerate the Ostwald ripening phenomenon. They make the silver nanocrystal achieve the most stable state in a short time. Some researchers have tried to use this phenomenon to control the morphology of silver nanocrystal and have made certain progress.

Fig. 8 Absorption spectra of silver nanoparticles solution without and with halides

Fig. 9 TEM images of silver nanoplates with Cl- (a) and without halides (b) (The insets show the color of products)
4 Conclusions
The form and mechanism of reducing agent, surfactants and halides to control the morphology of silver nanocrystal were studied. Reducing agents with dicarboxylate groups like TSC and potassium tartrate and surfactants with various function groups are very important in making triangular nanoplates. Also, they act as capping agent during the synthesis and adhere to (111) facet blocking the growth of it. This is the key reason why silver nanoplates exist in this reaction system. In addition, halides serve as the etching agent which accelerates the Ostwald ripening to make the silver keep the most stable state.
References
[1] TIAN N, ZHOU Z Y, SUN S G, DING Y, WANG Z L. Synthesis of tetrahexahedral platinum nanocrystals with high-index facets and high electro-oxidation activity [J]. Science, 2007, 316(5825): 732-735.
[2] HAES A J, van DUYNE R P. A nanoscale optical biosensor: Sensitivity and selectivity of an approach based on the localized surface plasmon resonance spectroscopy of triangular silver nanoparticles [J]. Journal of the American Chemical Society, 2002, 124(35): 10596-10604.
[3] WILEY B, SUN Y, MAYERS B, XIA Y. Shape-controlled synthesis of metal nanostructures: The case of silver [J]. Chemistry-A European Journal, 2005, 11(2): 454-463.
[4] ZHANG Q, LI N, GOEBL J, LU Z, YIN Y. A systematic study of the synthesis of silver nanoplates: Is citrate a ��magic�� reagent? [J]. Journal of the American Chemical Society, 2011, 133(46): 18931-18939.
[5] NIKHIL R, LATHA G, CATHERINE G. Wet chemical synthesis of silver nanorods and nanowires of controllable aspect ratio [J]. Chem Commm, 2001(7): 617-618.
[6] JIN R, CAO Y, MIRKIN C, KELLY K, SCHATZ G, ZHENG J. Photoinduced conversion of silver nanospheres to nanoprisms [J]. Science, 2001, 294: 1901-1903.
[7] GAO Y, JIANG P, SONG L, XIE S. Studies on silver nanodeca hedrons synthesized by PVP-assisted N,N-dimethylformamide(DMF) reduction [J]. Journal of Crystal Growth, 2006, 289: 376-380.
[8] BENJAMIN W, THURSTON H, SUN YG, XIA Y N. Polyol synthesis of silver nanoparticles: Use of chloride and oxygen to promote the formation of single-crystal, truncated cubes and tetrahedrons [J]. Nano Lett, 2004, 4(9): 1733-1739.
[9] PIETROBON B, KITAEV V. Photochemical synthesis of monodisperse size-controlled silver decahedral nanoparticles and their remarkable optical properties [J]. Chemistry of Materials, 2008, 20(16): 5186-5190.
[10] SUN Y, MAYERS B, XIA Y. Transformation of silver nanospheres into nanobelts and triangular nanoplates through a thermal process [J]. Nano Lett, 2003, 3(5): 675-679.
[11] PASTORIZA-SANTOS I,  L M. Colloidal silver nanoplates: State of the art and future challenges [J]. J Mater Chem, 2008, 18(15): 1724-1737.
L M. Colloidal silver nanoplates: State of the art and future challenges [J]. J Mater Chem, 2008, 18(15): 1724-1737.
[12] MILLSTONE J E, HURST S J,  G S, CUTLER J I, MIRKIN C A. Colloidal gold and silver triangular nanoprisms [J]. Small, 2009, 5(6): 646-664.
G S, CUTLER J I, MIRKIN C A. Colloidal gold and silver triangular nanoprisms [J]. Small, 2009, 5(6): 646-664.
[13] BERRIMAN R, HERZ R. Twinning and the tabular growth of silver bromide crystals [J]. Nature, 1957, 180: 293-294.
[14] LEE H Y, KIM J S, KIM D Y. Fabrication of BaTiO3 single crystals using secondary abnormal grain growth [J]. Journal of the European Ceramic Society, 2000, 20(10): 1595-1597.
[15] MA Y, KUANG Q, JIANG Z, XIE Z, HUANG R, ZHENG L. Synthesis of trisoctahedral gold nanocrystals with exposed high-index facets by a facile chemical method [J]. Angewandte Chemie International Edition, 2008, 47(46): 8901-8904.
[16] MING T, FENG W, TANG Q, WANG F, SUN L, WANG J, YAN C. Growth of tetrahexahedral gold nanocrystals with high-index facets [J]. Journal of the American Chemical Society, 2009, 131(45): 16350-16351.
[17] TIAN N, ZHOU Z Y, SUN S G. Platinum metal catalysts of high-index surfaces: From single-crystal planes to electrochemically shape-controlled nanoparticles [J]. The Journal of Physical Chemistry C, 2008, 112(50): 19801-19817.
[18] LI X, WANG L, YAN G. Review: Recent research progress on preparation of silver nanowires by soft solution method and their applications [J]. Crystal Research and Technology, 2011, 46(5): 427-438.
[19]  G S, MIRKIN C A. Rapid thermal synthesis of silver nanoprisms with chemically tailorable thickness [J]. Advanced Materials, 2005, 17(4): 412-415.
G S, MIRKIN C A. Rapid thermal synthesis of silver nanoprisms with chemically tailorable thickness [J]. Advanced Materials, 2005, 17(4): 412-415.
[20] ZHANG Q, HU Y, GUO S, GOEBL J, YIN Y. Seeded growth of uniform Ag nanoplates with high aspect ratio and widely tunable surface plasmon bands [J]. Nano Lett, 2010, 10(12): 5037-5042.
[21] PENN R L, BANFIELD J F. Imperfect oriented attachment: dislocation generation in defect-free nanocrystals [J]. Science, 1998, 281(5379): 969-971.
[22] DJALALI R, CHEN YF, MATSUI H. Au nanocrystal growth on nanotubes controlled by conformations and charges of sequenced peptide templates [J]. Journal of the American Chemical Society, 2003, 125(19): 5873-5979.
[23] CASULA M F, JUN Y W, ZAZISKI D J, CHAN E M, CORRIAS A, ALIVISATOS A P. The concept of delayed nucleation in nanocrystal growth demonstrated for the case of iron oxide nanodisks [J]. Journal of the American Chemical Society, 2006, 128(5): 1675-1682.
[24] JIANG X, ZENG Q, YU A. Thiol-frozen shape evolution of triangular silver nanoplates [J]. Langmuir, 2007, 23(4): 2218-2223.
[25] XIA Y, XIONG Y, LIM B, SKRABALAK S E. Shape-controlled synthesis of metal nanocrystals: Simple chemistry meets complex physics? [J]. Angewandte Chemie International Edition, 2009, 48(1): 60-103.
[26] CAO Y L, DING X L, LI H C, YI Z G, WANG X F, ZHU J J, KAN C. Morphology-controllable noble metal nanoparticles: Synthesis, optical property and growth mechanism [J]. Acta Physico-Chimica Sinica, 2011, 27(6): 1273-1286.
[27] GERMAIN V, LI J, INGERT D, WANG Z, PILENI M. Stacking faults in formation of silver nanodisks [J]. The Journal of Physical Chemistry B, 2003, 107(34): 8717-8720.
[28] SALZEMANN C, URBAN J, LISIECKI I, PILENI M P. Characterization and growth process of copper nanodisks [J]. Advanced Functional Materials, 2005, 15(8): 1277-1284.
[29] WILEY B, SUN Y, XIA Y. Synthesis of silver nanostructures with controlled shapes and properties [J]. Accounts of Chemical Research, 2007, 40(10): 1067-1076.
[30] HUANG S M, HWANG J J, LIU H J, LIN L H. Crystallization behavior of poly (L-lacticacid)/montmorillonite nanocomposites [J]. Journal of Applied Polymer Science, 2010, 117(1): 434-442.
[31] CALLEGARI A, TONTI D, CHERGUI M. Photochemically grown silver nanoparticles with wavelength-controlled size and shape [J]. Nano Letters, 2003, 3(11): 1565-1568.
[32] JUN Y W, CASULA M F, SIM J H, KIM S Y, CHEON J, ALIVISATOS A P. Surfactant-assisted elimination of a high energy facet as a means of controlling the shapes of TiO2 nanocrystals [J]. Journal of the American Chemical Society, 2003, 125(51): 15981-15985.
[33] ROCKENBERGER J, SCHER E C, ALIVISATOS A P. A new nonhydrolytic single-precursor approach to surfactant-capped nanocrystals of transition metal oxides [J]. Journal of the American Chemical Society, 1999, 121(49): 11595-11596.
[34] YANG J, ZHANG Q, LEE J Y, TOO H P. Dissolution�C recrystallization mechanism for the conversion of silver nanospheres to triangular nanoplates [J]. Journal of Colloid and Interface Science, 2007, 308(1): 157-161.
[35] CHEN B, JIAO X, CHEN D. Size-controlled and size-designed synthesis of nano/submicrometer Ag particles [J]. Crystal Growth & Design, 2010, 10(8): 3378-3386.
[36] MADRAS G, MCCOY B J. Ostwald ripening with size-dependent rates: Similarity and power-law solutions [J]. The Journal of Chemical Physics, 2002, 117(17): 8042-8049.
�� �����¿Ʊ����Ȼ���� 쿣��������� ��
�Ϻ���ͨ��ѧ ���������ϲ��Ϲ����ص�ʵ���ң��Ϻ� 200240
ժ Ҫ���о���ԭ���������Լ�����Ƭ״���γɵ�Ӱ�죬��������������ԭ�������������ơ���ʯ��أ��Լ�3�ֳ��ñ�����Լ�������ϩ������ͪ��ʮ����������ơ�����������Ӱ�졣�����������������ԭ���������Լ������п�����������(111)����ܽ�ϵĻ��Ի��ţ��Ӷ���������ЧӦ��ֹ�þ������������մﵽ�γ�Ƭ״����Ŀ�ġ��������ʾ��±�����ӵ��컯������Ƭ״������������Ҳ����һ����Ӱ�졣
�ؼ��ʣ�����Ƭ״������ԭ����������Լ�����������
(Edited by Sai-qian YUAN)
Foundation item: Project (51373097) supported by The National Natural Science Foundation of China
Corresponding author: Xiao-bin HU; Tel: +86-21-34202843; Fax: +86-21-34202843; E-mail: hxb@sjtu.deu.cn
DOI: 10.1016/S1003-6326(14)63522-6

