
Influence of SiO2 nano-particles on microstructures and properties of Ni-W-P/CeO2-SiO2 composites prepared by pulse electrodeposition
WANG Jun-li(王军丽)1 , XU Rui-dong(徐瑞东)2, ZHANG Yu-zhi(章俞之)3
1. Research Center for Analysis and Measurement, Kunming University of Science and Technology,
Kunming 650093, China;
2. Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology,
Kunming 650093, China;
3. Key Laboratory of Inorganic Coating Materials, Chinese Academy of Sciences, Shanghai 200050, China
Received 6 July 2009; accepted 10 January 2010
Abstract: Ni-W-P base composites containing CeO2 and SiO2 nano-particles were prepared on common carbon steel surface by pulse co-deposition of Ni, W, P, CeO2 and SiO2 nano-particles. The influence of SiO2 concentrations in bath on microstructures and properties of Ni-W-P/CeO2-SiO2 composites was studied, and the characteristics were assessed by chemical compositions, element distribution, surface morphologies, deposition rate and microhardness. The results indicate that when SiO2 concentration in bath is controlled at 20 g/L, the composites possess the fastest deposition rate, the highest microhardness, compact microstructures, smaller crystallite sizes and uniform distribution of W, P, Ce and Si within Ni-W-P matrix metal. Increasing SiO2 concentration in bath from 10 to 20 g/L leads to the refinement in grain size and the inhomogeneity of microstructures. While when SiO2 concentration is increased to 30 g/L, the crystallite sizes increase again and some bosses with nodulation shape appear on the surface of composites.
Key words: pulse electrodeposition; nano-particle; composite; microstructure and property
1 Introduction
The need for composites with improved resistance to highly aggressive environments is high as a result of a growing demand for extended safe service life of industrial objects. Composite electrodeposition is a valuable new surface intensification technology to obtain particle-reinforced metal matrix composites allowing to co-deposit inorganic and organic solid particles with matrix metal[1-4]. The composites possess comprehensive performance of matrix metal and second phase particle. At present, there are some reports about Ni-W-P base multi-element composites prepared by direct current electrodeposition[5-9], while studies on components design, preparation technology, micro- structures and properties of the multi-element composites prepared by pulse electrodeposition are not sufficient owing to the existing problems of complexity of the bath system and plating device.
The grain sizes, chemical compositions, microstructures and properties of the composites not only depend on bath compositions, but also process conditions [10-14]. W is a hard element and high mass fraction in the composites is helpful for improving microhardness [15]. CeO2 and SiO2 nano-particles deposited into Ni, Ni-P or Ni-W-P can lead to refinement in grain size and increase microhardness, wear and oxidation resistance [16-19]. Compared with the direct current electro- deposition, pulse electrodeposition can improve cathode activation polarization and decrease concentration polarization, avoiding the shortages of single direction and persistence of direct current by means of utilizing the relaxation of pulse current. Therefore, it is more favorable for preparing the composites and has already attracted much attention. In this work, Ni-W-P base
composites containing CeO2 and SiO2 nano-particles were prepared under different SiO2 concentrations in bath.
2 Experimental
The basic compositions of electroplating bath used for electrodepositing Ni-W-P/CeO2-SiO2 composites were as follows: 75 g/L NiSO4・6H2O, 120 g/L H3C6H5O7・H2O, 100 g/L Na2WO4・2H2O, 6 g/L NaH2PO2・H2O, 20 g/L SiO2 (average size: 30 nm) 10 g/L CeO2, (average size: 30 nm), and 6 mg/L cationic surfactant. Bath temperature was maintained at 60 ℃, pH value was about 5.5 and plating time was 2 h. Double-pulse parameters were as follows. Forward and reverse pulse duty cycles were controlled at 10%, forward and reverse pulse average current densities were controlled at 6 A/dm2 and 0.6 A/dm2, and forward and reverse pulse working times were controlled at 100 ms and 10 ms, respectively.
To guarantee better dispersion of CeO2 and SiO2 nano-particles within Ni-W-P matrix metal, before pulse electrodeposition, the electroplating bath was dispersed by ultrasonic for 30 min. Then, mechanical stirring was used during the experiments and the stirring speed was controlled at 750 r/min. 316L stainless steel was used as anode material, common carbon steel with dimensions of 30 mm×60 mm×2 mm was used as substrate materials. Each experiment was carried out with a fresh solution.
The chemical compositions, element distribution, deposition rate, microhardness and microstructures were evaluated as the characteristics of the composites. The microhardness was tested by 401MVA microhardness tester. The thickness was measured by metallurgical microscope and deposition rate was expressed by the thickness deposited in unit time. The microstructures, element distribution and chemical compositions were examined by scanning electron microscope (XL30 ESEM) and energy disperse spectroscopy (PHOENIX type).
3 Results and discussion
3.1 Influence of SiO2 concentrations in bath on chemical compositions of composites
Fig.1 shows the influence of SiO2 concentrations in bath on chemical compositions of Ni-W-P/CeO2-SiO2 composites. As shown in Fig.1, mass fraction of SiO2 in the composites increases with increasing SiO2 concentrations in bath. When the concentration is controlled at 20 g/L, SiO2 mass fraction reaches the highest value of 3.98%; but over 20 g/L, it begins to decrease again. While mass fraction of CeO2 decreases with the increase of SiO2 concentrations in bath. The mass fraction of P is the highest when SiO2 concentration is controlled at 15 g/L and the change of W mass fraction is little. The main reason may be that when SiO2 concentration is lower than 20 g/L, with increasing its concentration, the amounts of SiO2 nano-particles in bath increase and the probability imbedded into Ni-W-P matrix metal increases on the cathodic surface, leading to the increase of SiO2 deposition amounts and mass fraction in the composites. When SiO2 concentration is over 20 g/L, too high particle concentrations in bath result in the increase of electrolyte viscidity and serious coacervation of nano-particles absorbed on the cathodic surface. Therefore, the area offered electric chemical reaction is deduced and true density current is improved, leading to hydrogen evolution increasing on the cathodic surface and the decrease of deposition amounts. In addition, increasing SiO2 concentration in bath leads to the increase of SiO2 particle deposition amounts on the cathodic surface, making the probability of CeO2 nano-particles imbedded in the composites decrease, so the deposition amounts in the composites are also reduced.
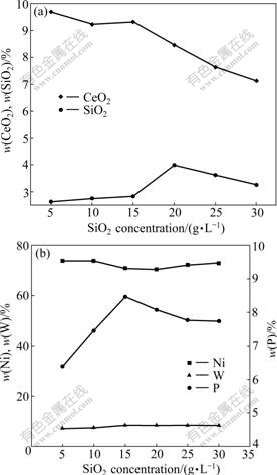
Fig.1 Influence of SiO2 concentrations in bath on chemical compositions of composites: (a) SiO2 and CeO2; (b) Ni, W and P
3.2 Influence of SiO2 concentrations in bath on element distribution of composites
When SiO2 concentration in bath is controlled at 20 g/L, the energy spectrogram analysis results of line scanning and area scanning are shown in Fig.2. As shown in Fig.2(a), on the level measurement points of line scanning, the mass fraction of W is 1.53%-16.85% (Average 7.75%), that of P is 1.23%-14.46% (Average 8.77%), that of Ce is 3.24%-15.74% (Average 6.48%) and that of Si is 0-4.84% (Average 2.17%). It can be seen form Fig.2(b) that the result of area scanning is close with that of line scanning, displaying that the distribution of every element in the composites is very uniform.
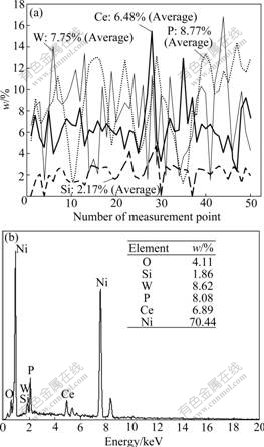
Fig.2 Energy spectrogram analysis results of line scanning and area scanning (SiO2 concentration: 20 g/L): (a) Line scanning; (b) Area scanning
When SiO2 concentration in bath is increased to 30 g/L, the energy spectrogram analysis results of line scanning and area scanning are shown in Fig.3. As shown in Fig.3(a), on the level measurement points of line scanning, the mass fraction of W is 0.03%-15.35% (Average 6.38%), that of P is 1.14%-14.58% (Average 8.64%), that of Ce is 1.74%-14.24% (Average 5.00%) and that of Si is 0-7.85% (Average 3.93%). It can be seen form Fig.3(b) that there is the large deviation from the results of line scanning. In particular, the average content of Si obtained by area scanning is obvious lower than that by line scanning. It can be concluded that the distribution of SiO2 in the composites is very uneven and may have a serious coacervation under too high SiO2 concentration in bath.
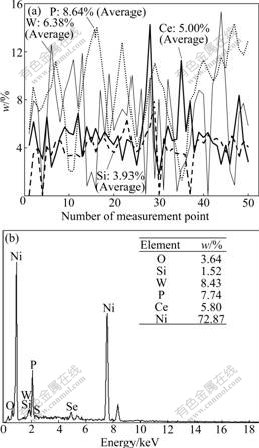
Fig.3 Energy spectrogram analysis results of line scanning and area scanning (SiO2 concentration: 30 g/L): (a) Line scanning; (b) Area scanning
3.3 Influence of SiO2 concentrations in bath on microstructures of composites
Fig.4 shows the influence of SiO2 concentrations in bath on surface morphologies of the composites. It can be seen form Fig.4 that increasing SiO2 concentrations in bath from 10 g/L to 20 g/L leads to the refinement in grain size and the inhomogeneity of microstructures. While when SiO2 concentration is improved to 30 g/L, the crystallite sizes begin to increase again and surface smooth decreases. The main reason may be that when SiO2 concentration is lower than 20 g/L, increasing its concentration leads to the increase of deposition rate (as shown in Fig.5). So, the nucleation rate of crystallites is faster than the growing up rate. In the meantime, the increase of SiO2 deposition amounts in the composites provides a large amount of nucleation points for matrix metal, hindering the continuous growth of Ni-W-P

Fig.4 Influence of SiO2 concentrations in bath on surface morphologies of composites: (a) 10 g/L; (b) 15 g/L; (c) and (e) 20 g/L; (d) and (f) 30 g/L
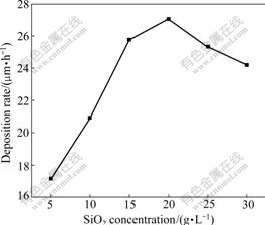
Fig.5 Influence of SiO2 concentrations in bath on deposition rate of composites
matrix metal, and resulting in the refinement in grain size. When SiO2 concentration is improved to 30 g/L, too high particle concentrations in bath result in the serious coacervation of nano-particles, which is unfavorable to refinement in grain size, so, the crystallite sizes increase again. As shown in Figs.4(e) and 4(f), it is obvious that when SiO2 concentration is controlled at 20 g/L, the outline of Ni-W-P matrix metal crystallites with spherical type is very clear, and the distribution of CeO2 and SiO2 nano-particles within matrix metal is uniform. When SiO2 concentration is improved to 30 g/L, the coacervation of nano-particles is serious and the distribution is very uneven. In the meantime, there appear a lot of small boss with nodulation shape on the composites surface.
3.4 Influence of SiO2 concentrations in bath on microhardness of composites
Fig.6 shows the influence of SiO2 concentrations in bath on microhardness of the composites. Obviously, the microhardness increases with the increase of SiO2 concentrations in bath and reaches the highest value with HV666 at 20 g/L, but it will decrease gradually if SiO2 concentrations increase continuously.
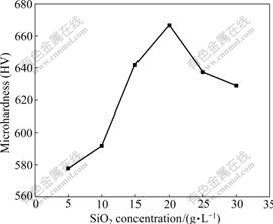
Fig.6 Influence of SiO2 concentrations in bath on microhardness of composites
In general, the microhardness of the composites is relative not only with microstructures but with chemical compositions. CeO2 particles can lead to refinement in grain size and increase the microhardness of the composites. Some mechanical properties such as microhardness and abrasion resistance can also be improved with increasing mass fraction of SiO2 particle in the composites. In addition, the high W content in Ni, Ni-P or Ni-W-P base composites is also helpful for improving the microhardness. In this work, when SiO2 concentration in bath is controlled at 20 g/L, mass fraction of SiO2, 3.98%, and mass fraction of W, 8.62%, are the highest, CeO2 mass fraction of 8.46% is also higher. In the meantime, the grain size is small and the grain structure is compact (see Fig.4), so the microhardness is higher. When SiO2 concentration in bath is improved to 30 g/L, the mass fractions of nano-particles decrease obviously, and their distribution in Ni-W-P matrix metal is also very uneven owing to the coacervation, leading to the decrease in the microhardness.
4 Conclusions
1) Ni-W-P-CeO2-SiO2 composites were prepared on common carbon steel surface by pulse electrodeposition of Ni, W, P, CeO2 and SiO2 nano-particles in bath.
2) When SiO2 concentration in bath is controlled at 20 g/L, the deposition rate is the fastest and microhardness is the highest. The average contents of W, P, Si and Ce are very close by means of line scanning and area scanning, displaying that the distribution of every element within the composites is very even.
3) Increasing SiO2 concentration in bath from 10 g/L to 20 g/L leads to the refinement in grain size and the inhomogeneity of microstructures. The crystallite sizes increase again when the SiO2 concentration is improved to 30 g/L. In the meantime, there appear a lot of small boss with nodulation shape on the composite surface.
References
[1] EL-SHERIK A M, ERB U. Adhesion and corrosion performance of nanocrystalline Ni coatings [J]. Plating and Surface Finishing, 1995, 82(9): 85-89.
[2] ZHITOMIRSKY I. Electrochemical processing and characterization of nickel hydroxide-polyelectrolyte coatings [J]. Materials Letters, 2004, 58(3/4): 420-424.
[3] WANG Z, SHEMILT J, XIAO P. Novel fabrication technique for the production of ceramic/ceramic and metal/ceramic composite coatings [J]. Scripta Materialia, 2000, 42(7): 653-659.
[4] MARCO M. Electrodeposition of composites: An expanding subject in electrochemical materials science [J]. Electrochimica Acta, 2000, 45(20): 3397-3402.
[5] XU Rui-dong, GUO Zhong-cheng, PAN Jun-yi. Corrosion resistance of electrodeposited RE-Ni-W-P-SiC-PTFE composite coating in phosphoric and ferric chloride [J]. Trans Nonferrous Met Soc China, 2006, 16(3): 666-670.
[6] GUO Zhong-cheng, XU Rui-dong, ZHU Xiao-yun. Studies on the wear resistance and structure of electrodeposited RE-Ni-W-P-SiC-PTFE composite materials [J]. Surface and Coatings Technology, 2004, 187(2/3): 141-145.
[7] XU Rui-dong, WANG Jun-li, HE Li-fang, GUO Zhong-cheng. Study on the characteristics of Ni-W-P composite coatings containing nano-SiO2 and nano-CeO2 particles [J]. Surface and Coatings Technology, 2008, 202(8): 1574-1579.
[8] SONG Yue-hai, WEI Gang, XIONG Rong-chun. Properties and structure of RE-Ni-W-P-SiC composite coating prepared by impulse electrodeposition [J]. Trans Nonferrous Met Soc China, 2007, 17(2): 363-367.
[9] XU Rui-dong, WANG Jun-li, GUO Zhong-cheng, WANG Hua. Study on particle-reinforced metal matrix nano-composites prepared by pulse electrodeposition [J]. Journal of Aeronautical Materials, 2008, 28(6): 46-50. (in Chinese)
[10] RICHOUX V, DILIBERTO S, BOULANGER C. Pulsed electrodeposition of bismuth telluride coatings: Influence of pulse parameters over nucleation and morphology [J]. Electrochimica Acta, 2007, 52(9): 3053-3060.
[11] DONG J K, YU M R, MOO H S. Effects of the peak current density and duty cycle on material properties of pulse-plated Ni-P-Fe electrodeposits [J]. Surface and Coatings Technology, 2005, 192(1): 88-93.
[12] ZIMMERMAN A F, CLARK D G, AUST K T. Pulse electrodeposition of Ni-SiC nanocomposite [J]. Materials Letters, 2002, 52(1/2): 85-90.
[13] MISHRA R, BALASUBRAMANIAM R. Effect of nanocrystalline grain size on the electrochemical and corrosion behavior of nickel [J]. Corrosion Science,2004, 46(12): 3019-3029.
[14] XU Rui-dong, WANG Jun-li, GUO Zhong-cheng, WANG Hua. Influence of process parameters on microstructure and properties of Ni-W-P-CeO2-SiO2 nano-composite films [J]. Journal of Materials Engineering, 2008(8): 17-21. (in Chinese)
[15] HUANG Ling, DONG Jun-xiu. Study on structure and microhardness of electrodeposited Ni-W alloy coatings [J]. Materials Protection, 1999, 32(10): 18-19. (in Chinese)
[16] WANG Hong-mei, JIANG Bin, XU Bin-shi. Microstructure and fretting wear behavior of Ni based composite coatings reinforced by SiO2 nanoparticles [J]. Tribology, 2005, 25(4): 289-293. (in Chinese)
[17] ZHU Cheng-yi, GUO Zhong-cheng. Effects of addition of RE on properties and structure of Ni-W-B-SiC composite coatings [J]. Chemical Metallurgy, 1999, 20(3): 225-228. (in Chinese)
[18] KARTHIKEYAN S, SRINIVASAN K N, VASUDEVAN T. Study on electroless Ni-P-Cr2O3 and Ni-P-SiO2 composite coatings [J]. Electroplating and Finishing, 2007, 26(1): 1-6.
[19] CAO Tie-hua, CHEGN Dan-hong, SANG Fu-ming. A study of the technology of (Ni-P)-nanometer SiO2 composite pulse electroplating [J]. Plating and Finishing, 2004, 26(6): 27-30. (in Chinese)
(Edited by LI Xiang-qun)
Foundation item: Project(20806035) supported by the National Natural Science Foundation of China; Project(2009CI026) supported by Back-up Personnel Foundation of Academic and Technology Leaders of Yunnan Province, China; Project(KKZ6200927001) supported by Opening Fund of Key Laboratory of Inorganic Coating Materials, Chinese Academy of Sciences; Project(2007E187M) supported by Applied Basic Research Plans of Yunnan Province, China; Project(08C0025) supported by Scientific Research Fund of Yunnan Provincial Education Department, China; Project(KKZ3200927029) supported by Training Foundation for Talents of Kunming University of Science and Technology, China; Project(2008-003) supported by Analysis and Measurement Research Fund of Kunming University of Science and Technology, China
Corresponding author: XU Rui-dong; Tel: +86-871-5157903; E-mail: rdxupaper@yahoo.com.cn
DOI: 10.1016/S1003-6326(09)60223-5