Prediction of structural and electronic properties of Cl2 adsorbed on TiO2(100) surface with C or CO in fluidized chlorination process:A first-principles study
来源期刊:中南大学学报(英文版)2021年第1期
论文作者:温良英 张生富 杨帆 彭琴 赵岩 徐健 扈玫珑 杨仲卿
文章页码:29 - 38
Key words:first-principles; co-adsorption model; titanium dioxide; bonding
Abstract: Based on the first-principles calculations of density functional theory, co-adsorption models of C or CO with Cl2 on rutile TiO2 (100) surface were established. The adsorption structures and electronic properties during chlorination process were predicted. Then, the adsorption energy, charge density, electron density difference and density of state of the adsorption structures were calculated and analyzed. The stabilities of the adsorption structures and the charge distributions between atoms were studied. It was found that both C and CO could promote the adsorption reactions of Cl2 on TiO2 (100) surface, and C was more favorable to the adsorption process. The results show that the adsorption process of Cl2 on TiO2(100) surface was physisorption, and the co-adsorption processes of C or CO with Cl2 on TiO2(100) surface were chemisorptions.
Cite this article as: YANG Fan, WEN Liang-ying, PENG Qin, ZHAO Yan, XU Jian, HU Mei-long, ZHANG Sheng-fu, YANG Zhong-qing. [J]. Journal of Central South University, 2021, 28(1): 29-38. DOI: https://doi.org/10.1007/ s11771-021-4583-3.

J. Cent. South Univ. (2021) 28: 29-38
DOI: https://doi.org/10.1007/s11771-021-4583-3
YANG Fan(杨帆)1, 2, WEN Liang-ying(温良英)1, 2, PENG Qin(彭琴)1, 2, ZHAO Yan(赵岩)1, 2,
XU Jian(徐健)1, 2, HU Mei-long(扈玫珑)1, 2, ZHANG Sheng-fu(张生富)1, 2, YANG Zhong-qing(杨仲卿)3
1. School of Materials Science and Engineering, Chongqing University, Chongqing 400044, China;
2. Chongqing Key Laboratory of Vanadium-Titanium Metallurgy and Advanced Materials,Chongqing University, Chongqing 400044, China;
3. School of Power Engineering, Chongqing University, Chongqing 400044, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Abstract: Based on the first-principles calculations of density functional theory, co-adsorption models of C or CO with Cl2 on rutile TiO2 (100) surface were established. The adsorption structures and electronic properties during chlorination process were predicted. Then, the adsorption energy, charge density, electron density difference and density of state of the adsorption structures were calculated and analyzed. The stabilities of the adsorption structures and the charge distributions between atoms were studied. It was found that both C and CO could promote the adsorption reactions of Cl2 on TiO2 (100) surface, and C was more favorable to the adsorption process. The results show that the adsorption process of Cl2 on TiO2(100) surface was physisorption, and the co-adsorption processes of C or CO with Cl2 on TiO2(100) surface were chemisorptions.
Key words: first-principles; co-adsorption model; titanium dioxide; bonding
Cite this article as: YANG Fan, WEN Liang-ying, PENG Qin, ZHAO Yan, XU Jian, HU Mei-long, ZHANG Sheng-fu, YANG Zhong-qing. [J]. Journal of Central South University, 2021, 28(1): 29-38. DOI: https://doi.org/10.1007/ s11771-021-4583-3.
1 Introduction
Titanium dioxide is widely used as colorant in coatings and plastics industry [1, 2]. The preparation of TiCl4 by chlorination of TiO2 with C is an important intermediate step in the production of titanium dioxide and titanium sponge [3-5]. In the process of preparing TiCl4 by chlorination of TiO2 with C, fluidized chlorination technology has attracted extensive attention due to its advantages such as optimized mass and heat transfer conditions at multiphase interfaces, short process flow, recyclability of Cl2 and continuous automation of operation [6, 7]. It has become the main technology of producing TiCl4 in the world.
The first-principles calculation of density functional theory (DFT) can explore the microscopic mechanism of chlorination from the atomic point of view [8, 9]. In recent years, DFT has made a significant contribution to the calculation of the interface reactions among metal oxides [10]. Many scholars have studied the adsorption mechanism of Cl on TiO2 surface by DFT and experiments. INDERWILDI et al [11] found that during the adsorption of TiCl4 on the surface of TiO2 (110), TiCl4 was dissociated, and chlorine was generated. The adsorption, diffusion and desorption of Cl atom on the surface of TiO2 (110) during the preparation of nano TiO2 particles by oxidation of TiCl4 were revealed. HEBENSTREIT et al [12] studied the adsorption process of chlorine on TiO2 (110) surface, and found that chlorine adsorbed on the bridge oxygen of TiO2 (110) surface at 200-300 °C. There would be bulges containing several Cl atoms with a width of 15  , and no long-range order was found in the adsorption layer. HIEHATA et al [13] studied the dissociation and adsorption of chlorine on the rutile TiO2 (110) surface. It was found that 94% of Cl adatoms would reach the top of Ti atoms on TiO2 surface, while the rest 6% of Cl adatoms could not. BATZILL et al [14] studied the charged impurities effect of chlorine adsorbed on TiO2 (110) surface, and found that when charged impurities existed, the adsorption amount of Cl on TiO2 (110) surface decreased, indicating that charged impurities had an inhibitory effect on the adsorption process. The fluidized chlorination process involves a complex interaction of many substances and is accompanied by the extensive use of corrosive gases. It is difficult to obtain the structures and electronic properties of the adsorbed intermediates by means of experiments. Rutile TiO2 consists of three low index surfaces, namely (110), (100) and (001) surface. Compared with the surface of TiO2(001), the surface of TiO2(110) and TiO2(100) has higher stability at high temperature [15]. However, in recent years, more studies have been reported on Cl2 adsorbed on TiO2(110) surface [16, 17], while few reports have been reported on TiO2(100) surface. In the fluidized chlorination process, coke is an important raw material for chlorination metallurgy, and CO gas inevitably exists. Therefore, it is of great significance to study the adsorption structures and electronic properties of C or CO with Cl2 adsorbed on TiO2(100) surface.
, and no long-range order was found in the adsorption layer. HIEHATA et al [13] studied the dissociation and adsorption of chlorine on the rutile TiO2 (110) surface. It was found that 94% of Cl adatoms would reach the top of Ti atoms on TiO2 surface, while the rest 6% of Cl adatoms could not. BATZILL et al [14] studied the charged impurities effect of chlorine adsorbed on TiO2 (110) surface, and found that when charged impurities existed, the adsorption amount of Cl on TiO2 (110) surface decreased, indicating that charged impurities had an inhibitory effect on the adsorption process. The fluidized chlorination process involves a complex interaction of many substances and is accompanied by the extensive use of corrosive gases. It is difficult to obtain the structures and electronic properties of the adsorbed intermediates by means of experiments. Rutile TiO2 consists of three low index surfaces, namely (110), (100) and (001) surface. Compared with the surface of TiO2(001), the surface of TiO2(110) and TiO2(100) has higher stability at high temperature [15]. However, in recent years, more studies have been reported on Cl2 adsorbed on TiO2(110) surface [16, 17], while few reports have been reported on TiO2(100) surface. In the fluidized chlorination process, coke is an important raw material for chlorination metallurgy, and CO gas inevitably exists. Therefore, it is of great significance to study the adsorption structures and electronic properties of C or CO with Cl2 adsorbed on TiO2(100) surface.
In this work, we have investigated the co- adsorption processes of C and Cl2, CO and Cl2 on TiO2 (100) surface based on the first-principles calculations method of DFT, and the adsorption structures and electronic properties during chlorination process were predicted. The co-adsorption processes were studied through the analysis of the adsorption structure, adsorption energy, charge density, electron density difference and density of state, respectively. This work revealed the behavioral mechanism for promoting the chlorination adsorption reaction of C or CO with Cl2 on TiO2 (100) surface, and it is of great significance to improve the efficiency of the chlorination reaction, providing a theoretical support for further study on the fluidized chlorination mechanism of the titanium oxide.
2 Calculation method and parameter selection
In this work, the chlorination adsorption process of C or CO with Cl2 on the surface of TiO2(100) was calculated by using the CASTEP module of Material Studio software package [18-22]. In the first-principles calculations, the plane wave ultrasoft pseudopotential method can neglect the interactions between the inner electrons [23]. This method can be used to calculate the interactions between electrons and ions in the process of chlorination adsorption. The structure and energy characteristics of the titanium oxides are important parameters in the calculation of the chlorination adsorption. The selection of these parameters is directly related to the accuracy of the calculation results. At present, in the first-principles calculations process, the GGA-PBE functional theory is mainly used to describe it [24]. Therefore, the GGA-PBE [25-29] is selected as the exchange correlation term in the calculation of this work. PERRON et al [30] found that the surface energy of TiO2 (100) fluctuated with the number of layers, and when TiO2 (100) surface was composed of 8-10 layers of atoms, the surface energy of TiO2 (100) converged to 0.01 J/m2. Thus, we set the atomic layers of the surface model of TiO2 (100) to 9. During the calculation, all the atoms in the upper 5 layers are relaxed and the other 4 layers are fixed. We have tested the convergence of the energy cutoff. When the energy cutoffs are 260, 300, 340, 360, 380 and 400 eV, the system energies are -29742.41, -29764.81, -29770.53, -29771.25, -29771.06 and -29771.13 eV, respectively. When the energy cutoff is 360 eV, the system energy tends to be stable. In order to ensure the accuracy of the calculation results, we finally set the energy cutoff as 400 eV. The thickness of vacuum layer is set to 15  , and a grid of K-point in Brillouin zone is divided into 2×3×1. The convergence tolerance of energy, maximum force and maximum displacement are 2×10-5 eV/atom, 0.05 eV/
, and a grid of K-point in Brillouin zone is divided into 2×3×1. The convergence tolerance of energy, maximum force and maximum displacement are 2×10-5 eV/atom, 0.05 eV/ and 0.002
and 0.002  , respectively. The supercell structure used in the calculation is composed of multiple TiO2 unit cells, which are directly found in the standard structure database of Material Studio software and then optimized. Figure 1 shows the optimized structure of TiO2 (100) surface, and the surface contains Ti 5c (fivefold coordinated Ti atom), Ti 6c (sixfold coordinated Ti atom), O 2c (twofold coordinated O atom) and O 3c (threefold coordinated Ti atom), with lattice parameters a=9.19
, respectively. The supercell structure used in the calculation is composed of multiple TiO2 unit cells, which are directly found in the standard structure database of Material Studio software and then optimized. Figure 1 shows the optimized structure of TiO2 (100) surface, and the surface contains Ti 5c (fivefold coordinated Ti atom), Ti 6c (sixfold coordinated Ti atom), O 2c (twofold coordinated O atom) and O 3c (threefold coordinated Ti atom), with lattice parameters a=9.19  , b=5.92
, b=5.92  , c= 21.39
, c= 21.39  . In the calculation, the adsorption energy Ea of X (X=Cl2, C+Cl2, CO+Cl2) is defined as follows.
. In the calculation, the adsorption energy Ea of X (X=Cl2, C+Cl2, CO+Cl2) is defined as follows.
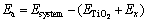
where Esystem, ETiO2 and Ex represent the energy of TiO2 and X (X=Cl2, C+Cl2, CO+Cl2), the energy of TiO2 and the energy of X (X=Cl2, C+Cl2, CO+Cl2), respectively.
3 Results and discussion
3.1 Adsorption structures of C or CO with Cl2 on TiO2(100) surface
When C atom adsorbed on TiO2 (100) surface alone, C bonds with O atom on the surface. When CO molecule adsorbed on TiO2 (100) surface, there is no obvious interaction between CO molecule and TiO2 (100) surface. Figure 2(a) shows the structure of Cl2 molecule adsorbed on the TiO2 (100) surface. After the adsorption, the chemical bond of Cl2 molecule is not broken, and the bond length is 1.99  , indicating that Cl2 molecule does not interact with TiO2(100) surface significantly.
, indicating that Cl2 molecule does not interact with TiO2(100) surface significantly.
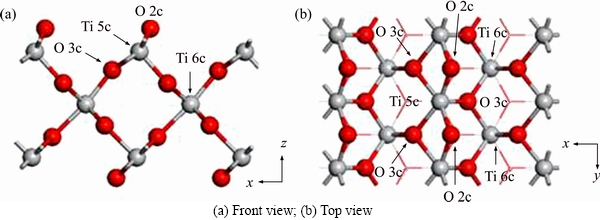
Figure 1 Optimized structure of TiO2 (100) surface:
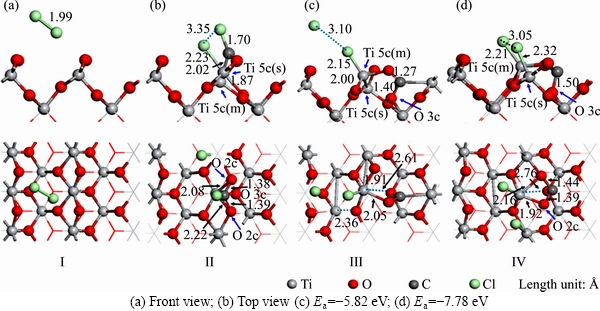
Figure 2 Co-adsorption structures of C and Cl2 adsorbed on TiO2 (100) surface:(The top is front view, and bottom is top view)
Figures 2(b)-(d) show the structures of C and Cl2 adsorbed on TiO2 (100) surface. Cl2 molecules will be dissociated into two Cl atoms, which will bond with the surface Ti 5c and C atom or enter the vacuum layer. In Figure 2(b), the Cl atoms bond with C and surface Ti 5c(s), and the bond lengths are 1.70 and 2.23  , respectively. The C atom bonds with surface Ti 5c(m) and O 2c on both sides, and the bond lengths of them are 2.02, 1.38 and 1.39
, respectively. The C atom bonds with surface Ti 5c(m) and O 2c on both sides, and the bond lengths of them are 2.02, 1.38 and 1.39  , respectively. The bond lengths of the Ti 5c―O 2c on both sides extend to 2.08 and 2.22
, respectively. The bond lengths of the Ti 5c―O 2c on both sides extend to 2.08 and 2.22  , respectively. The bond length of the Ti5c(m)―O3c extends to 1.87
, respectively. The bond length of the Ti5c(m)―O3c extends to 1.87  , and O 3c is obviously depressed from the surface. In Figure 2(c), after the dissociation of Cl2 molecule, one of the Cl atoms bonds with surface Ti 5c(m), and the bond length is 2.15
, and O 3c is obviously depressed from the surface. In Figure 2(c), after the dissociation of Cl2 molecule, one of the Cl atoms bonds with surface Ti 5c(m), and the bond length is 2.15  . The other Cl atom is free in the vacuum layer. Ti 5c(m) and O 3c are obviously raised from the surface, and the chemical bond between Ti 5c(m) and O3c breaks. The distance between Ti 5c(m) and O 3c extends to 2.61
. The other Cl atom is free in the vacuum layer. Ti 5c(m) and O 3c are obviously raised from the surface, and the chemical bond between Ti 5c(m) and O3c breaks. The distance between Ti 5c(m) and O 3c extends to 2.61  . C atom bonds with O 2c and O 3c, and the lengths of them are 1.27 and 1.40
. C atom bonds with O 2c and O 3c, and the lengths of them are 1.27 and 1.40  , respectively. In Figure 2(d), two Cl atoms bond with Ti 5c on both sides, and the bond lengths of them are 2.21 and 2.32
, respectively. In Figure 2(d), two Cl atoms bond with Ti 5c on both sides, and the bond lengths of them are 2.21 and 2.32  , respectively. The bond lengths of Ti 5c(m)―O 2c on both sides extend to 1.92 and 2.16
, respectively. The bond lengths of Ti 5c(m)―O 2c on both sides extend to 1.92 and 2.16  , respectively. Both Ti 5c(m) and O 3c are raised from the surface, and the Ti 5c(m)―O 3c bond on the surface breaks. C atom bonds with O 2c on both sides, and the bond lengths are 1.39 and 1.44
, respectively. Both Ti 5c(m) and O 3c are raised from the surface, and the Ti 5c(m)―O 3c bond on the surface breaks. C atom bonds with O 2c on both sides, and the bond lengths are 1.39 and 1.44  , respectively. In Figure 2, the adsorption process of structure (a) is physisorption, and the adsorption energy is -0.07 eV, indicating that Cl2 could not interact significantly with the adsorption surface. The adsorption processes of (b), (c) and (d) are all chemisorptions, and the adsorption energies of them are -7.62, -5.82 and -7.78 eV, respectively, indicating that the stability of structure (d) is higher than that of other structures.
, respectively. In Figure 2, the adsorption process of structure (a) is physisorption, and the adsorption energy is -0.07 eV, indicating that Cl2 could not interact significantly with the adsorption surface. The adsorption processes of (b), (c) and (d) are all chemisorptions, and the adsorption energies of them are -7.62, -5.82 and -7.78 eV, respectively, indicating that the stability of structure (d) is higher than that of other structures.
Figures 3(a)-(c) show the adsorption structures of CO and Cl2 adsorbed on TiO2 (100) surface. Cl2 molecules will be dissociated into two Cl atoms, which will bond with the surface Ti 5c or C atom. C atoms of the CO molecule in the structures (a) and (b) can form C-O2c bond, and in structure (c), it can form CO2 molecule by being bonded with O 3c.
It can be seen in Figure 3(a) that the C atom of CO molecule adsorbed on TiO2 (100) surface can form C―O 2c bond with a bond length of 1.34  . At this time, the Ti 5c(s)―O 2c bond breaks. The bond lengths of Ti 5c(m)―O 2c and Ti 5c(m)―O 3c increased to 1.87 and 2.24
. At this time, the Ti 5c(s)―O 2c bond breaks. The bond lengths of Ti 5c(m)―O 2c and Ti 5c(m)―O 3c increased to 1.87 and 2.24  , respectively. One Cl atom bonds with C atom, and the bond length is 1.77
, respectively. One Cl atom bonds with C atom, and the bond length is 1.77  . The other Cl atom bonds with Ti 5c(m) and Ti 5c(s), and the bond lengths of them are 2.44 and 2.42
. The other Cl atom bonds with Ti 5c(m) and Ti 5c(s), and the bond lengths of them are 2.44 and 2.42  , respectively. In Figure 3(b), it can be seen that after the CO molecule adsorbed on TiO2 (100) surface, the O 2c significantly shifts downward, and the bond length of Ti 5c(m)―O 2c extends to 2.04
, respectively. In Figure 3(b), it can be seen that after the CO molecule adsorbed on TiO2 (100) surface, the O 2c significantly shifts downward, and the bond length of Ti 5c(m)―O 2c extends to 2.04  . The C atom and O 2c forms a stable C―O 2c double bond, and the bond length is 1.31
. The C atom and O 2c forms a stable C―O 2c double bond, and the bond length is 1.31  . The chemical bond between Ti 5c(m) and O3c is broken, and O 3c moves up obviously. The Cl atoms bond with the Ti 5c(m) and Ti 5c(s), and the bond lengths of them are 2.21 and 2.34
. The chemical bond between Ti 5c(m) and O3c is broken, and O 3c moves up obviously. The Cl atoms bond with the Ti 5c(m) and Ti 5c(s), and the bond lengths of them are 2.21 and 2.34  , respectively. In Figure 3(c), it can be seen that the C atom bonds with O 3c, and the bond length is 1.20
, respectively. In Figure 3(c), it can be seen that the C atom bonds with O 3c, and the bond length is 1.20  . The bonds of Ti 6c―O 3c and Ti 5c(m)―O 3c are all broken, and Ti 5c(m) is obviously raised up. The coordination numbers of Ti 6c on both sides and Ti 5c(m) decrease, and the activities of them increase. After the dissociation of Cl2 molecule, the two Cl atoms bond with Ti 5c on both sides, and the bond lengths are 2.23
. The bonds of Ti 6c―O 3c and Ti 5c(m)―O 3c are all broken, and Ti 5c(m) is obviously raised up. The coordination numbers of Ti 6c on both sides and Ti 5c(m) decrease, and the activities of them increase. After the dissociation of Cl2 molecule, the two Cl atoms bond with Ti 5c on both sides, and the bond lengths are 2.23  . The adsorption processes of structures V-VII are all chemisorptions, and the adsorption energies of them are -2.21, -1.56 and -0.44 eV, respectively, indicating that the stability of structure V is higher than that of other structures. The adsorption energies of structures V-VII are higher than those of structures II-IV in Figure 2. That is to say, the stabilities of the adsorption structures formed by C and Cl2 on TiO2 (100) surface are higher than that formed by CO and Cl2, and C atom is more conductive to promoting the adsorption reaction of Cl2 on TiO2 (100) surface than CO molecule.
. The adsorption processes of structures V-VII are all chemisorptions, and the adsorption energies of them are -2.21, -1.56 and -0.44 eV, respectively, indicating that the stability of structure V is higher than that of other structures. The adsorption energies of structures V-VII are higher than those of structures II-IV in Figure 2. That is to say, the stabilities of the adsorption structures formed by C and Cl2 on TiO2 (100) surface are higher than that formed by CO and Cl2, and C atom is more conductive to promoting the adsorption reaction of Cl2 on TiO2 (100) surface than CO molecule.
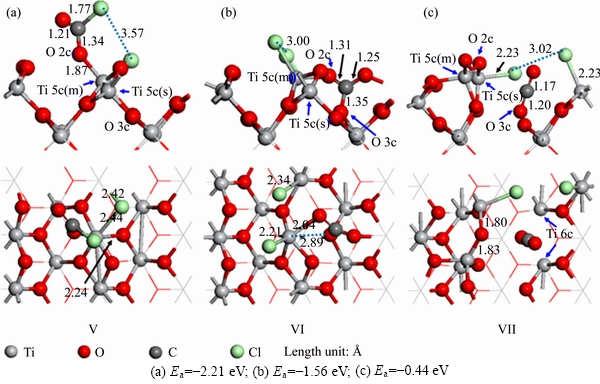
Figure 3 Co-adsorption structures of CO and Cl2 adsorbed on TiO2 (100) surface:(The top is the front view, and the bottom is the top view)
3.2 Charge transfer
Table 1 shows the charge transfer quantities of valence electrons of TiO2 (100) clean surface and C, O, Ti and Cl atoms in the adsorption structures of II-IV. Among the three adsorption structures, O 2c and Cl2 are electron acceptors, and Ti 5c(m) and C are electron providers. In the three adsorption structures of II-IV, C atom loses 0.09, 0.23 and 0.21 e, respectively. Among them, C atom loses more electrons in the structure of Figure 2(c) than those in other structures. Cl2 obtained 0.16, 0.43 and 0.44 e in the structures of II-IV, respectively, and the obtained electrons amounts of Cl2 in the structure of Figure 2(d) are more than those in other structures.
Table 2 shows the charge transfer quantities of valence electrons of TiO2 (100) clean surface and CO, O, Ti and Cl2 in the adsorption structures of V, VI and VII. Among them, Ti5c refers to the Ti atoms of the Ti 5c―Cl bonds. In the three adsorption structures, Ti 5c and CO molecules are all electron providers, and O 2c and Cl2 are electron acceptors. In the three adsorption structures of V, VI and VII, CO loses 0.09, 0.17 and 0.42 e, respectively. CO loses more electrons in the structure of VII than those in other structures. Cl2 obtained 0.31, 0.41 and 0.55 e in the structures of V, VI and VII, respectively, and the obtained electrons amounts of Cl2 in the structure of VII are more than those in other structures.
3.3 Electron density difference
Figure 4 shows the electron density differences for the adsorption of C and Cl2 on TiO2(100) surface in Figures 4(a)-(c) are corresponding to in Figures 2(b)-(d), respectively. C―Cl, Ti―Cl and C―O bonds are formed when C and Cl2 are co-adsorbed on TiO2(100) surface. In Figure 4(a), Cl atoms bond with C and Ti atoms, respectively. The surrounding region of C―Cl bond is yellow, indicating that it is an electron depleted region and the stability of the C―Cl bond is low. In Figure 4(b), the Cl2 molecule is dissociated into two Cl atoms, and one of them bonds with the Ti atom. The surrounding region of Ti―Cl bond is blue, indicating that it is an electron accumulated region and there is a strong interaction between the Ti and Cl atoms. When C atom forms bond with O, it is surrounded by yellow color, indicating that it is the electron depleted region and C atom outputs electrons during bonding process. The color of the region between C and Ti atoms is blue, indicating that it is an electron accumulated region and there is a strong interaction between C and Ti. In Figure 4(c), Cl atoms bond with Ti, and C atom bonds with O. The color of the region between Cl and Ti atoms is blue, indicating that there are electron accumulated region and strong interaction between Ti and Cl atoms.
Table1 Mulliken charge analysis of adsorption of C and Cl2 adsorbed on TiO2 (100) surface (Unit: e)
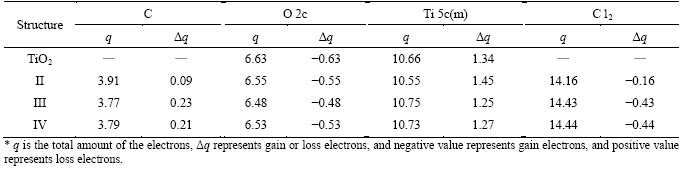
Table 2 Mulliken charge analysis of adsorption of CO and Cl2 on TiO2 (100) surface (Unit: e)
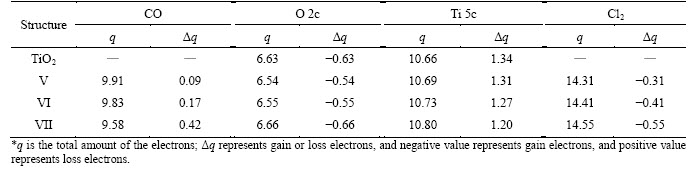
Figure 5 shows the electron density differences for the adsorption of CO and Cl2 on TiO2(100) surface in Figures 5(a)-(c) are corresponding to Figures 3(a)-(c), respectively. When CO and Cl2 co-adsorbed on TiO2(100) surface, C―Cl, Ti―Cl and C―O bonds are formed. In Figure 5(a), Cl atoms bond with C and Ti atoms, respectively. There is blue color around the Ti―Cl bond, indicating that it is an electron accumulated region and there is a strong interaction between Ti and Cl atoms. In Figure 5(b), Cl2 molecule is dissociated into two Cl atoms, which are all bonded with Ti atoms. The blue color around the Ti―Cl bonds indicates that there are electron accumulated regions and a strong interaction between the Ti and Cl atoms. When C atom bonds with O atom, the surrounding region of the bond is yellow, indicating that it is an electron depleted region, and the C atom outputs electrons during bonding process. In Figure 5(c), C atom is bonded with O atom, and the surrounding region of C atom is yellow, indicating that the surrounding region of C atom is an electron depleted region and CO outputs electrons during the adsorption process, and it is consistent with the charge analysis results of Mulliken.
3.4 Density of state
Figure 6 shows the partial density of states of TiO2 (100) clean surface and C atom, Cl atom and CO adsorbed on TiO2 (100) surface. Figures 6(b), (c) and (d) are corresponding to the structures of II, III and IV in Figure 2, and Figures 6(e) and (f) are corresponding to the structures of V and VI in Figure 3, respectively. C-Cl, Ti 5c―Cl and Ti 5c― O 2c bonds are formed or influenced when C or CO co-adsorbed with Cl2 on TiO2 (100) surface. Therefore, studying the density of state variations of C―Cl, Ti 5c―Cl and Ti 5c―O 2c bonds are of great significance to understand the bonding process of the adsorption structures.
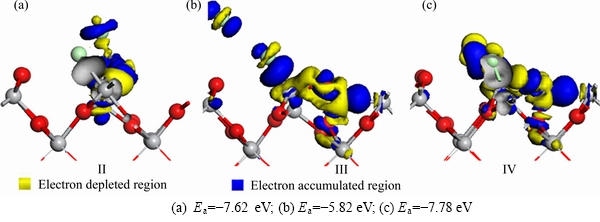
Figure 4 Electron density difference maps for adsorption of C and Cl2 on TiO2(100) surface:(The top is front view, and bottom is top view)
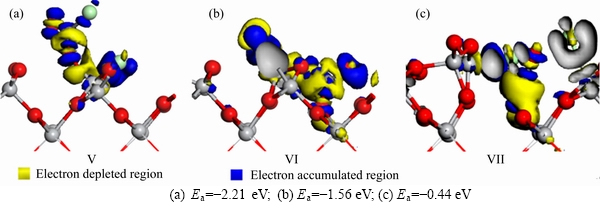
Figure 5 Electron density difference maps for adsorption of CO and Cl2 on TiO2(100) surface:(The top is the front view, and the bottom is the top view)

Figure 6 Partial density of states (PDOS) of O 2c, Ti 5c, C atoms and CO:(EF represents the Fermi energy level)
In the density of state diagram of TiO2 (100) clean surface, the Ti 3d and O 2p orbitals have overlapping peaks at -4.03, -2.95, -2.44 eV on the left side of Fermi energy level, indicating that there is a bonding interaction between Ti 5c and O 2c. The Cl atom in the density of state diagram in Figure 6(b) refers to the Cl bonded with C, and the overlapping peaks of C 2p and Cl 3p orbitals are at -7.64, -0.05 and 0.84 eV, respectively, indicating that there is a bonding interaction between C and Cl atom. The σ bond is formed at -10.13 eV for C 2s and O 2p orbitals, and π bonds are formed at -7.64, -6.88 and -0.05 eV for C 2p and O 2p orbitals, respectively. The high overlapping peak values indicate that there is a bonding interaction between C and O 2c. In Figure 6(c), σ bond is formed at -10.23 eV for C 2s and O 2p orbitals and π bonds are formed at -8.23, -7.52 and -1.78 eV for C 2p and O 2p orbitals. The high overlapping peaks on the left side of Fermi energy level indicate that there is a bonding interaction between C and O 2c. The overlapping peak of Ti 3d and Cl 3p at -4.02 eV on the left side of Fermi energy level indicates that there is a bonding interaction between the two atoms. In Figure 6(d), the σ bond is formed at -9.34 eV for C 2s and O 2p orbitals, and π bonds are formed at -6.43 and -0.05 eV for C 2p and O 2p orbitals, respectively. The high overlapping peaks indicate that there is a bonding interaction between C and O 2c. The overlapping peaks of Ti 5c and Cl atom at -3.07, -2.54, -1.38 and -0.02 eV on the left side of Fermi energy level indicate that there is a bonding interaction between Ti 5c and Cl atom. In Figure 6(e), there are obvious overlapping peaks of CO and O 2c at -7.94, -5.91 and -0.02 eV on the left side of Fermi energy level, respectively, indicating that there are bonding interactions between CO and O 2c. The overlapping peaks of CO molecule and Cl atom exist at -7.94, -5.91, -4.74, -0.92 and -0.02 eV on the left side of Fermi energy level, indicating that there is bonding interaction between CO molecule and Cl atom. In Figure 6(f), the overlapping peaks of CO and O 2c exist at -9.16, -7.78, -7.15 and -4.42 eV on the left side of Fermi energy level, indicating that there are bonding interactions between CO and O 2c. The overlapping peaks between Ti 5c and Cl atoms exist at -13.23 and -2.59 eV on the left side of Fermi energy level, indicating that there is a bonding interaction between Ti 5c and Cl atom.
4 Conclusions
Based on the first-principles calculations method of the density functional theory, the chlorination adsorption structures and electronic properties of TiO2(100) surface with C or CO are predicted, and the following results are obtained.
1) When CO and Cl2 adsorbed on TiO2 (100) surface at the same time, the adsorption energies of the three structures are higher than that of C and Cl2. That is to say, the stabilities of the adsorption structures of C and Cl2 on TiO2 (100) surface are higher than that of CO and Cl2 on TiO2 (100) surface. C atom is more favorable to promote the adsorption reaction of Cl2 on TiO2 (100) surface than CO molecule.
2) In the adsorption process of C and Cl2 on TiO2 (100) surface, O 2c and Cl2 are electron acceptors, and Ti 5c (m) and C are electron providers. In the adsorption process of CO and Cl2 on TiO2 (100) surface, O 2c and Cl2 are electron acceptors, and Ti 5c and CO molecules are electron providers.
3) In the structure systems formed by the adsorption of C and Cl2 on TiO2 (100) surface, there are bonding interactions between C atom and O 2c, and there are also bonding interactions between Cl and C or Ti 5c. In the structure systems formed by the adsorption of CO and Cl2 on TiO2 (100) surface, there are bonding interactions between CO and O 2c, and there are also bonding interactions between Cl and Ti 5c.
Contributors
The overarching research goals were developed by YANG Fan and WEN Liang-ying. PENG Qin and ZHAO Yan sorted out relevant data in the early stage. The initial draft of the manuscript was written by YANG Fan. WEN Liang-ying, XU Jian, HUMei-long, ZHANG Sheng-fu, and YANG Zhong-qing supervised the process. YANG Fan and WEN Liang-ying replied to reviewers’ comments and revised the final version.
Conflict of interest
All authors declare that they have no conflict of interest.
References
[1] DAI Wen-wu, ZHAO Zong-yan. Structural and electronic properties of low-index stoichiometric BiOI surfaces [J]. Materials Chemistry and Physics, 2017, 193: 164-176. DOI: 10.1016/j.matchemphys.2017.02.017.
[2] ULRIKE D. The surface science of titanium dioxide [J]. Surface Science Reports, 2003, 48: 53-229. DOI: 10.1016/S0167-5729(02)00100-0.
[3] WANG Yu-ming, LIU Rui-feng, ZHOU Rong-hui, SHAO Bao-shun, WEI Qing-song, YUAN Zhang-fu, XU Chong. Thermodynamics on the reaction of carbochlorination of titania for getting titanium tetrachloride [J]. Computers and Applied Chemistry, 2006, 23: 263-266. DOI: 10.1016/ S1003-6326(06)60040-X.
[4] ADIPURI A, ZHANG Guang-qing, OSTROVSKI O. Chlorination of titanium oxycarbide produced by carbothermal reduction of rutile [J]. Metallurgical and Materials Transactions B, 2008, 39: 23-34. DOI: 10.1007/ s11663-007-9117-3.
[5] SHON H, VIGNESWARAN S, KIM I, CHO J, KIM J. Preparation of titanium dioxide (TiO2) from sludge produced by titanium tetrachloride (TiCl4) flocculation of wastewater [J]. Environmental Science and Technology, 2007, 41: 1372-1377. DOI: 10.1021/es062062g.
[6] EL-SADEK M H, FOUAD O A, MORSI M B, EL-BARAWY K. Controlling conditions of fluidized bed chlorination of upgraded titania slag [J]. Transactions of the Indian Institute of Metals, 2019, 72: 423-427, 2018. DOI: 10.1007/ s12666-018-1493-7.
[7] XIONG Shao-feng, YUAN Zhang-fu, XU Cong, XI Liang. Composition of off-gas produced by combined fluidized bed chlorination for preparation of TiCl4 [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(1): 128-134. DOI: 10.1016/S1003-6326(09)60109-6.
[8] ZHONG Hong, ER De-quan, WEN Liang-ying. Theoretical study on influence of CaO and MgO on the reduction of FeO by CO [J]. Appllied Surface Science, 2017, 399: 630-637. DOI: 10.1016/j.apsusc.2016.12.123.
[9] ZHONG Hong, WEN Liang-ying, LI Jian-long, XU Jian, HU Mei-long, YANG Zhong-qing. The adsorption behaviors of CO and H2 on FeO surface: A density functional theory study [J]. Powder Technology, 2016, 303: 100-108. DOI: 10.1016/ j.powtec.2016.09.017.
[10] ZHONG Hong, WEN Liang-ying, ZOU Cong, ZHANG Sheng-fu, BAI Chen-guang. Density functional theory study on the carbon-adhering reaction on Fe3O4(111) surface [J]. Metallurgical and Materials Transactions B, 2015, 46: 2288-2295. DOI: 10.1007/s11663-015-0379-x.
[11] INDERWILDI O, KRAFT M. Adsorption, diffusion and desorption of chlorine on and from rutile TiO2(110): A theoretical investigation [J]. Chemphyschem, 2007, 8: 444-451. DOI: 10.1002/cphc.200600653.
[12] HEBENSTREIT E, HEBENSTREIT W, GEISLER H, VENTRICE C, HITE D, SPRUNGER P, DIEBOLD U. The adsorption of chlorine on TiO2(110) studied with scanning tunneling microscopy and photoemission spectroscopy [J]. Surface Science, 2002, 505: 336-348. DOI: 10.1016/ S0039-6028(02)01385-7.
[13] HIEHATA K, SASAHARA A, ONISHI H. Kelvin probe force microscope observation of chlorine-adsorbed TiO2(110) surfaces [J]. Japanese Journal of Applied Physics, 2008, 47: 6149-6152. DOI: 10.1143/JJAP.47.6149.
[14] BATZILL M, HEBENSTREIT E, HEBENSTREIT W, DIEBOLD U. Influence of subsurface, charged impurities on the adsorption of chlorine at TiO2(110) [J]. Chemical Physics Letters, 2003, 367: 319-323. DOI: 10.1016/S0009-2614 (02)01635-4.
[15] RAMAMOORTHY M, VANDERBILT D. First-principles calculations of the energetics of stoichiometric TiO2 surfaces [J]. Physical Review B, 1994, 23(49): 722-727. DOI: 10.1103/PhysRevB.49.16721.
[16] INDERWILDI O, KRAFT M. Adsorption, diffusion and desorption of chlorine on and from rutile TiO2{110}: A theoretical investigation [J]. ChemPhysChem, 2007, 8: 444-451. DOI: 10.1002/cphc.200600653.
[17] HEINE A, ISABELA C, STUDT F, ABILD F, BLIGARD T, ROSSMESIL J. Electrochemical chlorine evolution at rutile oxide (110) surfaces [J]. Physical Chemistry Chemical Physics, 2010, 12: 283-290. DOI: 10.1039/b917459a.
[18] SEZGIN A, MEHMET. Stability and superconductivity properties of metal substituted aluminum diborides (M0.5Al0.5B2) [J]. Computational Materials Science, 2018, 154: 234-242. DOI: 10.1016/j.commatsci.2018.08.005.
[19] TUTUIANU M, INDERWILDI O R, BESSLER W G, WARNATZ J. Competitive adsorption of NO, NO2, CO2 and H2O on BaO(100): A quantum chemical study [J]. Journal of Physical Chemistry B, 2006, 110: 17484-17492. DOI: 10.1021/jp055268x.
[20] MA C G, KRASNENKO V, BRIK M G. First-principles calculations of different (001) surface terminations of three cubic perovskites CsCaBr3, CsGeBr3, and CsSnBr3 [J]. Journal of Physics and Chemistry of Solids, 2018, 115: 289-299. DOI: 10.1016/j.jpcs.2017.12.052.
[21] CHEN Hao, LI Xue-chao, WAN Run-dong, KAO WS, LEI Ying. A DFT study of the electronic structures and optical properties of (Cr, C) co-doped rutile TiO2 [J]. Chemical Physics, 2018, 501: 60-67. DOI: 10.1016/j.chemphys.2017. 11.021.
[22] JIANG Zhao, GUO Shu-yi, FANG Tao. Theoretical investigation on the dehydrogenation mechanism of CH3OH on Cu(100) surface [J]. Journal of Alloys and Compounds, 2017, 698: 617-625. DOI: 10.1016/j.jallcom.2016.12.220.
[23] PAYNE M C, TETER M P, ALLAN D C, ARIAS T A, JOANNOPOULOS J D. Iterative minimization techniques for ab initio total-energy calculations: Molecular dynamics and conjugate gradients [J]. Reviews of Modern Physics, 1992, 64: 1045-1097. DOI: 10.1103/RevModPhys.64.1045.
[24] BERGERMAYER W, SCHWEIGER H, WIMMER E. Ab initio thermodynamics of oxide surfaces: O2 on Fe2O3(0001) [J]. Physical Review B, 2004, 69: 1324-1332. DOI: 10.1103/ PhysRevB.69.195409.
[25] DAI Yue-hua, GONG Shan-shan, ZHONG Zhi-sheng, GAO Feng-yu, WANG Fei-fei, DING Chen, YANG Jin, YANG Fei. Effect of graphene/TiO2 (001) interface on threshold voltage and nonlinearity [J]. Nano, 2018, 13: 23069-4. DOI: 10.1142/S1793292018300049.
[26] CHEN Hao, LI Xue-chao, WAN Run-dong. Theoretical studies on the electronic structure and optical absorption property of (Ni, C) co-doped anatase TiO2 [J]. Computational Condensed Matter, 2017, 13: 16-28. DOI: 10.1016/j.cocom. 2017.08.005.
[27] FAN Ya-ming, WENG Qi-yu, ZHUO Yu-qun, DONG Song-tao, HU Peng-bo, LI Duan-le. Theoretical study of As2O3 adsorption mechanisms on CaO surface [J]. Materials, 2019, 12: 677-690. DOI:10.3390/ma12040677.
[28] LIU Shuai, TANG Cheng-huang, ZHAN Yong-zhong. Theoretical prediction of transition metal alloying effects on the lightweight TiAl intermetallic [J]. Metallurgical and Materials Transactions A, 2016, 33: 1451-1460. DOI:10. 1007/s11661-015-3321-6.
[29] JIN Shang-xiao, LIU Na, ZHANG Shuai, LI De-jun. The simulation of interface structure, energy and electronic properties of TaN/ReB2 multilayers using first-principles [J]. Surface and Coatings Technology, 2017, 326: 417-423. DOI: 10.1016/j.surfcoat.2016.11.068.
[30] PERRON H, DOMAIN C, ROQUES J, DROT R, SIMONI E, CATALETTE H. Optimisation of accurate rutile TiO2 (110), (100), (101) and (001) surface models from periodic DFT calculations [J]. Theoretical Chemistry Accounts: Theory, Computation, and Modeling, 2007, 117: 565-574. DOI: 10.1007/s00214-006-0189-y.
(Edited by FANG Jing-hua)
中文导读
沸腾氯化过程中C和CO分别与Cl2在TiO2(100)表面的吸附结构及电荷属性分析:、第一性原理研究
摘要:基于密度泛函理论的第一性原理计算方法,构建C和CO分别与Cl2在金红石TiO2 (100)面共吸附模型,预测了氯化过程中的吸附结构及其电荷属性。之后,对吸附结构的吸附能、电荷密度、差分电荷密度和态密度等进行计算分析,研究了吸附结构的稳定性及原子间的电荷分布。发现C和CO均能促使Cl2在TiO2 (100)表面发生吸附反应,且C更有利于促进吸附反应的发生。结果表明:Cl2分子单独在TiO2(100)表面吸附的过程为物理吸附,C和CO分别与Cl2同时在TiO2(100)表面吸附的过程均为化学吸附。
关键词:第一性原理;共吸附模型;二氧化钛;成键
Foundation item: Projects(51674052, 51974046) supported by the National Natural Science Foundation of China; Project(cstc2018jcyjAX0003) supported by the Chongqing Research Program of Basic Research and Frontier Technology, China
Received date: 2020-03-22; Accepted date: 2020-07-25
Corresponding author: WEN Liang-ying, PhD, Professor; Tel: +86-23-65127306; E-mail: cquwen@cqu.edu.cn; ORCID: https://orcid. org/0000-0002-7897-8952; ZHANG Sheng-fu, PhD, Professor; E-mail: zhangsf@cqu.edu.cn; ORCID: https://orcid.org/0000-0002-0858- 7286

