
Thermal stability of nanocrystalline in surface layer of
magnesium alloy AZ91D
ZHANG Jin(张 津)1, 2, OU Xin-bing(欧信兵)2, 3
1. Institute of Advanced Material, University of Science and Technology Beijing, Beijing 100083, China;
2. College of Materials Science and Engineering, Chongqing University of Technology, Chongqing 400050, China;
3. Nanjing Kerun Lubricants Co., Ltd., Nanjing 211100, China
Received 23 September 2009; accepted 30 January 2010
Abstract: Isothermal and isochronal annealing was conducted to study the thermal stability of the nanocrystalline in the surface layer of Mg alloy AZ91D induced by high-energy shot peening (HESP). Field emission scanning electron microscope (FESEM) and X-ray diffractometer were used to characterize the microstructure. Results showed that nanocrystalline produced by HESP on the surface layer of the magnesium alloy AZ91D was 60-70 nm on average. The nanocrystalline could remain stable at about 100 ?C, and grew up slowly between 100 ?C and 200 ?C. When the annealing temperature reached 300 ?C, the growth rate of the nanocrystalline increased significantly. The kinetic coefficient n of the nanocrystalline growth was calculated to be 2-3 and the grain growth activation energy Q=39.7 kJ/mol, far less than the self-diffusion activation energy of magnesium atoms in the coarse polycrystalline material.
Key words: high energy shot peening (HESP); magnesium alloy; AZ91D; nanocrystalline; thermal stability
1 Introduction
Service performance of materials was directly influenced by their surface state. Although magnesium alloys have lots of advantages, such as low density, good casting property and high damping capacity, their surface hardness is low and their corrosion resistance and abrasion resistance are poor[1]. Surface nano- crystallization (SNC) induced by severe plastic deformation (SPD) could improve surface hardness, tensile strength, friction and wear properties of metals or alloys[2]. It has been found that the SNC could play an active role in surface hardening of the steel and other alloys and metals, such as Al alloys[3], Mg alloys[4], pure Ti[5] and Co[6], which shows that the affecting objects of the SNC are increasingly widespread. However, the strengthening effects caused by the nanocrystalline necessitate its good thermal stability in engineering applications, and thermal stability could vary with different nanocrystalline structures produced in alloys[7-10].
Thermal stability is a key factor related to practical applications of nanostructured materials. The thermal stability of surface nanocrystalline (SNC) directly affects its application range and environment. Once the surface nanocrystalline grows up to the coarse grain, it will lose all of its outstanding performances. Therefore, the study on the thermal stability of surface nanocrystalline is of great significance for SNC technology in practical applications.
In this work, SNC is successfully implemented on the surface of magnesium alloy AZ91D by high-energy shot peening (HESP). In order to study the thermal stability of the nanocrystalline, isothermal and isochronal annealing is conducted under different conditions. This research could provide a basis for the surface grain refinement and surface properties improvement of magnesium alloy.
2 Experimental
The experimental material was die-cast magnesium alloy AZ91D, with the dimension of 140 mm×110 mm×20 mm and the chemical composition (mass fraction, %) of 8.5-9.5 Al, 0.9-0.95 Zn, 0.17-0.40 Mn, ≤0.05 Si, ≤0.025 Cu, ≤0.001 Ni, ≤0.004 Fe and balance Mg.
Before the treatment of HESP, samples were treated in advance for derusting, degreasing and edging. During the treatment, stainless balls with a diameter of 1.5 mm were accelerated by high pressure gas at room temperature and the distance between the spray-gun and the specimen was 200-250 mm. The entire surface of the sample was shot peened by the high velocity balls in 5 min. In order to make the deformation more uniform, the specimen was moved left and right during the treatment.
After HESP, the sample was cut into small pieces with the size of 10 mm×10 mm×20 mm. In order to study the thermal stability of the nanocrystalline, isothermal and isochronal annealing treatment was conducted at different temperatures (from 40 to 300 ?C) for different times (from 5 to 60 min). The sample was put into SX-2.5-12 electric resistance furnace after the required temperature was achieved, and then the sample was isothermally heated for the required time before being taken out and cooled in air to room temperature. Table 1 lists the temperature and time for isothermal heat treatment.
Table 1 Temperature and time for isothermal heat treatment

The surface microstructure of the sample after HESP was observed by using FEI Nova 400 field emission scanning electron microscope (FESEM). Before observation, the sample was mechanically polished firstly using silicon carbide paper to grade 1 000, then on a polishing cloth with a liquid suspension of 0.04 μm alumina, and finally etched in a mixed solution consisting of 1 mL nitric acid, 1 mL acetic acid, 1 mL ethanedioic acid and 150 mL distilled water.
The X-ray diffraction analysis was carried out on a DX-2500 X-ray diffractometer with Cu target radiation
(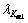 =1.540 56?) in the continuous scanning mode. The experimental parameters were: tube potential 35 kV, tube current 25 mA, scanning range 30?-90?, scanning velocity 0.06 (?)/s. The average grain size and microstrain were calculated in terms of the diffraction line broadening after removing the influence of instrument broadening, which was obtained on a standard AZ91D magnesium alloy specimen without any strain and grain refinement.
=1.540 56?) in the continuous scanning mode. The experimental parameters were: tube potential 35 kV, tube current 25 mA, scanning range 30?-90?, scanning velocity 0.06 (?)/s. The average grain size and microstrain were calculated in terms of the diffraction line broadening after removing the influence of instrument broadening, which was obtained on a standard AZ91D magnesium alloy specimen without any strain and grain refinement.
3 Results and discussion
3.1 Microstructure characterization of surface layer after HESP
The XRD patterns of AZ91D magnesium alloy before and after HESP are shown in Fig.1. As shown in Fig.1(a), two samples both consisted of α-Mg and β-Mg17Al12 phase, which meant there were not any new phases formed after HESP.
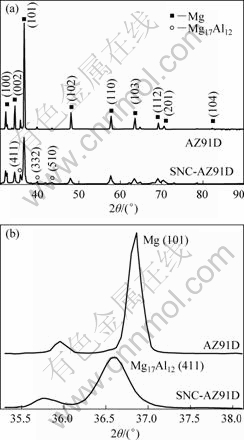
Fig.1 XRD patterns for magnesium alloys AZ91D before and after HESP: (a) Full pattern; (b) Mg (101) and Mg17Al12 (411) enlarged pattern
From Fig.1(b), it could be found that after HESP, the diffraction intensity decreased greatly, the diffraction line broadened evidently and the diffraction peaks shifted left to different extents, the detailed XRD data of which are listed in Table 2.
According to the principle of XRD, the peak position shift is mainly due to the macro-stress in samples and the peak broadening may be attributed to grain refinement, micro-strain and instrument broadening. The change of diffraction intensity corresponds to the lattice distortion. So, it could be concluded that after HESP there were three different kinds of stress coexisting in the Mg alloy AZ91D. However, only microstrain needs to be considered when the average grain size and micro-stain of surface layer of as-treated sample were calculated in terms of values of full width at half maximum (FWHM).
The calculation was done according to the following equation[11]:
Table 2 XRD data for magnesium AZ91D before and after HESP treatment
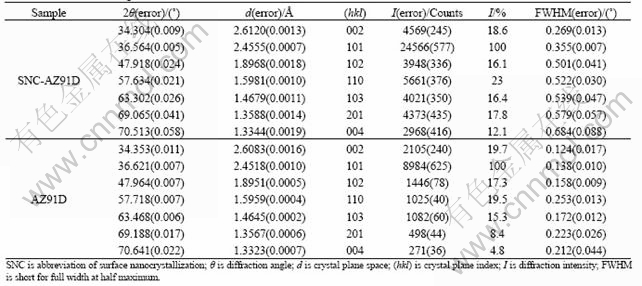
WS?cos θ=K?λ/D+4?ε?sin θ (1)
where WS is the FWHM value of the sample, θ the diffraction angle, K a constant usually taken as value of 1, λ the X-ray wavelength, D and ε represent the average grain size and average microstrain, respectively. In calculation, at least two WS values were measured. sin θ/λ and WS?cos θ/λ were used as abscissa and ordinate, respectively. Thus, a straight line should be resulted according to the least square method. The slope and the intercept of the line represent 2ε and 1/D, respectively.
Fig.2 shows the calculation results. It could be found that after HESP, the average grain size and microstrain in the surface layer of Mg alloy AZ91D were (66.8±18.3) nm and (0.367 8±0.040 45)%, respectively. Fig.3 shows FESEM image of the surface layer morphology, from which it could be observed that the average grain size was 60-70 nm, very close to the XRD calculation results and similar to the results of Ref.[12]. So, it could be concluded that surface nanocrystallization of magnesium alloy AZ91D was realized by HESP.
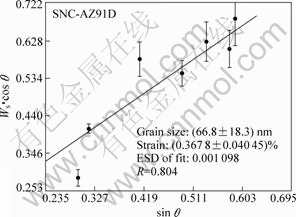
Fig.2 Average micro-strain and grain size of AZ91D after HESP treatment

Fig.3 FESEM image of surface of AZ91D alloy treated by HESP
3.2 Thermal stability of nanostructured surface layer
Fig.4 shows the nanocrystalline growth with the increase of isochronal annealing temperature in the range of 100-200 ?C for 60 min. It could be seen from Fig.4 that, when the temperature was low, 100-120 ?C, the grain almost kept stable. With the annealing temperature increasing, the grain size became larger and the grain growth rate gradually increased.
During isothermal annealing at 200 ?C and 300 ?C, the nanocrystalline growth with the increase of heat treatment time is shown in Fig.5. From Fig.5, it could be found that at 200 ℃ the grain size increased very slowly, up to 100 nm for 60 min. However, the grain growth rate increased dramatically when the heat treatment temperature was up to 300 ℃. The grain size reached 100 nm in 6 min, which meant that the grain growth rate at 300 ?C was 10 times that at 200 ?C.
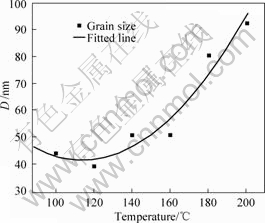
Fig.4 Effect of annealing temperature on grain size in HESP surface layer of magnesium AZ91D after heating for 60 min
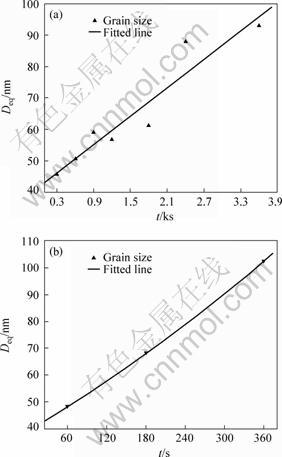
Fig.5 Variation of grain size versus heat treatment time in surface layer of AZ91D after HESP treatment: (a) 200 ?C; (b) 300 ?C
According to the above analysis, it was concluded that the nanostructured surface layer could be stable at about 100 ?C, and grew slowly below 200 ?C, however grew dramatically at 300 ?C.
For the traditional polycrystalline materials, the variation of grain size d with time t can be described by the following relationship:
d n-d0n=Kt (2)
where d0 is the initial grain diameter at t=0; K and n are the time-independent constants[13].
K has a temperature dependence described by the following equation[14]:
K=K0exp[-Q/(RT)] (3)
where K0 is the proportion coefficient, Q is the grain growth activation energy, R is the Boltzmann constant, and T is the absolute temperature.
Taking the natural logarithm of Eq.(2) yields
ln t=nln(d/d0)-ln K (4)
Thus, if ln t and ln(d/d0) were used as the ordinate and abscissa, respectively, a straight line should be obtained, and the slope and intercept of the line represent the n and -ln K, respectively.
Fig.6 shows the plots of ln t vs ln(d/d0) at 200 ?C and 300 ?C. The initial grain size d0 was taken as 24.9 nm, which was the average result calculated by XRD. The n and K values can be obtained from Fig.6, and K0 and Q values could be obtained according to Eq.(3) (Table 3).
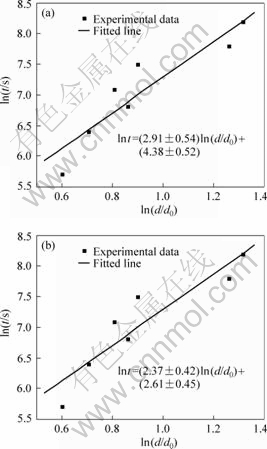
Fig.6 Plot of ln t vs ln(d/d0) at different temperatures: (a) 200 ?C; (b) 300 ?C
Table 3 Calculation results of kinetic parameters
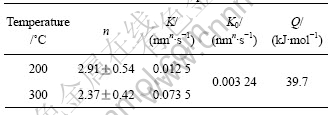
The value of n varies with the difference of grain growth rate. Generally speaking, the value of n is equal to 2 for normal grain growth in a pure, single phase system; 3 for grain growth in the presence of solutes, and 4 for the presence of pores[15]. From Table 3, we can see that the calculated values of n were closed to 3. The result indicated that it was reasonable to use Eq.(2) to describe the growth law of the nanocrystalline in the surface layer of magnesium alloy AZ91D, and it also showed that alloying elements in the Mg alloy had great influence on the grain growth.
Grain growth is due to the migration of grain boundaries, which is the diffusion of atoms in essence. Therefore, the activation energy of grain growth could also be used to characterize the driving force and difficulty degree of atoms diffusion. The activation energy calculated in the experiment was extremely low, only 39.7 kJ/mol, much less than the self-diffusion activation energy for the coarse-grained pure magnesium metal in Ref.[16], about 200.3 kJ/mol and 279.7 kJ/mol along the directions parallel and vertical to c axis. This result meant that the driving force of atoms diffusion in nanostructured materials was much higher, thus atoms diffusion was much easier in the nanocrystalline materials than in the conventional coarse-grained material.
4 Conclusions
1) The nanocrystalline in the surface layer of magnesium alloy AZ91D produced by HESP, in size of 60-70 nm, could remain stable up to about 100 ?C; it grew slowly below 200 ?C; and increased significantly when the temperature reached 300 ?C.
2) The nanocrystalline growth could be analyzed according to the traditional laws of polycrystalline grain growth. The kinetic coefficient n of the nanocrystalline growth was calculated to be 2-3 and the grain growth activation energy Q=39.7 kJ/mol, far less than the self-diffusion activation energy of magnesium atoms in the coarse polycrystalline material.
Acknowledgement
Thanks are given to Professor ZHANG Xi-yan, Chongqing University, for his kind help on the preparation of SNC of Mg alloy, as well as his critical advice in the discussion.
References
[1] ZHANG Jin, ZHANG Zong-he. Magnesium alloys and application [M]. Beijing: Chemical Industry Press, 2004: 1-3. (in Chinese)
[2] LU Ke, LU Jian. Nanostructured surface layer on metallic materials induced by surface mechanical attrition treatment [J]. Materials Science and Engineering A, 2004, 375/376/377: 38-45.
[3] WU X, TAO N, HONG Y. Microstructure and evolution of mechanically-induced ultrafine grain in surface layer of Al-alloy subjected to USSP [J]. Acta Materialia, 2002, 50: 2075-2084.
[4] WEI Ying-hui, LIU Bao-sheng, HOU Li-feng. Characterization and properties of nanocrystalline surface layer in Mg alloy induced by surface mechanical attrition treatment [J]. Journal of Alloys and Compounds, 2008, 452(2): 336-342.
[5] ZHU K Y, VASSEL A, BRISSET F. Nanostructure formation mechanism of α-titanium using SMAT [J]. Acta Materialia, 2004, 52: 4101-4110.
[6] WU X, TAO N, HONG Y. Strain-induced grain refinement of cobalt during surface mechanical attrition treatment [J]. Acta Materialia, 2005, 53: 681-691.
[7] BA De-ma, MA Shi-ning, LI Chang-qing. Thermal stability of nanocrystallite in surface layer of Cr-Si alloy steel [J]. Material Engineering, 2008(8): 22-25, 30. (in Chinese)
[8] HU Lan-qing, MA Jin-fang, XU Bing-she. Surface nanocrystallization of Al-Zn-Mg alloy and its thermal stability [J]. Transactions of Materials and Heat Treatment, 2007, 28(8): 343-347. (in Chinese)
[9] KOCH C C. Structural nanocrystalline materials: An overview [J]. J Mater Sci, 2007, 42: 1403-1414.
[10] KOCH C C, SCATTERGOOD R O, DARLING K A, SEMONES J E. Stabilization of nanocrystalline grain sizes by solute additions [J]. J Mater Sci, 2008, 43: 7264-7272.
[11] KLUG H P, ALEXANDER L E. X-ray diffraction procedures for polycrystalline and amorphous materials[M]. 2nd ed. New York and London: John Wiley, 1974: 356-370.
[12] ZHANG M X, SHI Y N, SUN H Q, KELLY P M. Surface alloying of Mg alloys after surface nanocrystallization [J]. Journal of Nanoscience and Nanotechnology, 2007, 8: 1-5.
[13] WILLIAM D, Jr CALLISTER. Fundamentals of materials science and engineering[M]. 5th ed. New York and London: John Wiley, 2000: 219.
[14] ISONISHI K Z, OKAZAKI K J. Grain growth in nanocrystalline NbAl3 prepared by mechanical alloying [J]. Journal of Materials Science, 1993, 28: 3829-3834.
[15] CHEN Z W, SHEK C H, LAI J K L. An analysis of the grain growth kinetics in Mn2O3 nanocrystals [J]. Appl Phys A, 2005, 80: 703-707.
[16] ZHANG Lian-meng, HUANG Xue-hui, SONG Xiao-lan. Fundamentals of materials science[M]. Wuhan: Wuhan University of Technology Press, 2004: 409. (in Chinese)
(Edited by YANG Bing)
Corresponding author: ZHANG Jin; Tel: +86-10-82377393, +86-13683032221; E-mail: zhangjin@ustb.edu.cn
DOI: 10.1016/S1003-6326(09)60301-0