Ni���������ܽ�-�����ͽ����������Ʊ�B-BiVO4������Ե�Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���9��
�������ߣ����� �� ������ лԪ�� ������ ���� ��ͮ
����ҳ�룺2022 - 2030
�ؼ��ʣ������ӣ������ӣ�BiVO4; ��������ܽ�-�����������շ�
Key words��co-doping; Ni doping; BiVO4; photocatalyst; sol-gel method; impregnation method
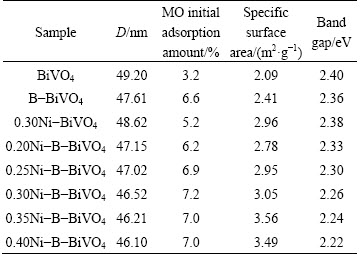
ժ Ҫ��Ϊ�˽�һ�����B-BiVO4�Ĺ�����ԣ�ͨ���ܽ�-�����ͽ����������Ʊ�Ni����B-BiVO4���(Ni-B-BiVO4)��ͨ��XPS��XRD��SEM��BET��EDS��UV-Vis���ֶζ���Ʒ���б�������������������Ӿ�����ı���Ʒ�ľ��ͺ���ò����������Ni����Ʒ�ľ���������С����δ���ӡ�B�Լ�Ni��������ȣ�Ni-B-BiVO4��Ʒ�Ĺ�������������ƫ�ƣ���N-B�����Ӻ���Ʒ��V4+ �ͱ����ǻ������ࡣ�����Ni������Ϊ0.30%(��������)ʱ�����Ʊ���0.3Ni-B-BiVO4��50 min�ڶԼ��ȵĽ����ʴ�95%���ң��Ҹ���Ʒ����Ч�����Ǽ���(MB)�����II��(AOII)������B(RhB)��Һ����������Ʒ���Ե������Ҫ��B��Ni��Эͬ�������¡�
Abstract: To enhance the photocatalytic activity of B-BiVO4, Ni-doped B-BiVO4 photocatalyst (Ni-B-BiVO4) was synthesized through sol-gel and impregnation method. The photocatalysts were characterized by XPS, XRD, SEM, EDS, BET and UV-Vis DRS techniques. The results showed that single or double doping did not change the crystalline structure and morphology, but the particle size decreased with Ni doping. The band gap energy absorption edge of Ni-B-BiVO4 shifted to a longer wavelength compared with undoped, B or Ni single doped BiVO4. More V4+ and surface hydroxyl oxygen were observed in BiVO4 after Ni-B co-doping. When the optimal mass fraction of Ni is 0.30%, the degradation rate of MO in 50 min is 95% for 0.3Ni-B-BiVO4 sample which also can effectively degrade methyl blue (MB), acid orange (AOII) II and rhodamine B (RhB). The enhanced photocatalytic activity is attributed to the synergistic effects of B and Ni doping.
Trans. Nonferrous Met. Soc. China 27(2017) 2022-2030
Min WANG, Guang-jun YANG, Mei-yan YOU, Yuan-hua XIE, You-zhao WANG, Jin HAN, Tong ZHU
School of Mechanical Engineering and Automation, Northeastern University, Shenyang 110165, China
Received 29 May 2016; accepted 26 September 2016
Abstract: To enhance the photocatalytic activity of B-BiVO4, Ni-doped B-BiVO4 photocatalyst (Ni-B-BiVO4) was synthesized through sol-gel and impregnation method. The photocatalysts were characterized by XPS, XRD, SEM, EDS, BET and UV-Vis DRS techniques. The results showed that single or double doping did not change the crystalline structure and morphology, but the particle size decreased with Ni doping. The band gap energy absorption edge of Ni-B-BiVO4 shifted to a longer wavelength compared with undoped, B or Ni single doped BiVO4. More V4+ and surface hydroxyl oxygen were observed in BiVO4 after Ni-B co-doping. When the optimal mass fraction of Ni is 0.30%, the degradation rate of MO in 50 min is 95% for 0.3Ni-B-BiVO4 sample which also can effectively degrade methyl blue (MB), acid orange (AOII) II and rhodamine B (RhB). The enhanced photocatalytic activity is attributed to the synergistic effects of B and Ni doping.
Key words: co-doping; Ni doping; BiVO4; photocatalyst; sol-gel method; impregnation method
1 Introduction
In past decades, bismuth vanadate (BiVO4) has attracted considerable attention for its visible light-driven photocatalytic activity in water splitting and organic pollutant degradation [1-3]. However, the poor photocatalytic efficiency of pure BiVO4, which is due to its low quanta yield ratio, is one of the restrictions for its widespread application in the wastewater treatment [4-6].
Therefore, the challenge to overcome the above drawbacks remains. Metal doping has proven to be an effective approach to enhance the photocatalytic activity of BiVO4 [7-15]. ZHANG et al [8] reported that Ag, Co and Ni doping of BiVO4 can effectively enhance the photocatalytic activity for the degradation of methyl blue and 2,4-dichlorophenol. CHEN et al [9] prepared the Ni-BiVO4 photocatalysts with a hydrothermal method. Compared with pure BiVO4, the Ni-BiVO4 catalyst shows a red shift in the absorption band in the visible region and a narrow band gap (2.35 eV). The Ni doping enhanced photocatalytic activities for the degradation of methylene blue (MB) under visible light irradiation. ZHANG et al [10] prepared a CuO-BiVO4 hetero- junction composite, which exhibited an enhanced photocatalytic activity for methylene blue (MB) degradation under visible light irradiation. It was found that the 5%CuO-loaded (mass fraction) composite can apparently enhance the photocatalytic activity for MB dye. Additionally, it was reported that F [16], S [17], N [18,19] and B [20-22] can also effectively enhance the photocatalytic activity of BiVO4. JIANG et al [16] prepared F-doped BiVO4 with a hydrothermal method using NH4F as the fluoride source. It has been shown that F can effectively enhance the photodegradation rate of phenol. ZHAO et al [17] reported the sulfur doping of BiVO4 prepared through a dodecylamine-assisted alcohol-hydrothermal route with Na2S as sulfur source. Our team prepared N- [18] and B- [20,21] doped BiVO4 by sol-gel method, a catalyst that can also effectively enhance the photocatalytic degradation of methyl orange under visible light irradiation.
In recent years, photocatalysts co-doped with metals and nonmetals have attracted more and more attention due to their enhanced photocatalytic activity [23-27].
For example, PATEL et al [24] prepared TiO2, N-doped TiO2, V-doped TiO2, and V-N-co-doped TiO2 thin films. It was observed that the co-doped TiO2 showed the best photocatalytic performance compared with the mono-doped and pure TiO2 because of higher visible light absorption and possible decrease in recombination of photo-generated charges. KIMA et al [25] synthesized Fe-N-co-doped TiO2 photocatalyst with a sonochemical method. The photocatalytic activity of Fe-N-co-doped TiO2 for the degradation of indigo carmine dye (ICD) under solar simulator was enhanced when compared with TiO2-P25 and N-TiO2. WANG et al [26,27] prepared B and rare earth ions (Eu and La) co-doped BiVO4via a one-step sol-gel method. It shows that the synergetic effects between two co-doping elements can effectively enhance the photocatalytic activity of BiVO4 under visible light irradiation.
Different preparation methods may result in different effects. Therefore, in the present work, we used sol-gel and impregnation two-step method to prepare B and Ni co-doped BiVO4 photocatalysts to further enhance the photocatalytic activity for the degradation of methyl orange (MO), methyl blue (MB), acid orange II (AOII), and rhodamine B (RhB). The serials xNi-B-BiVO4 samples prepared by this method were characterized by XPS, XRD, SEM, BET and DRS techniques. The effects of the doping nickel amount on the properties of B-doped BiVO4 were also investigated, and the synergetic effects between boron and nickel were studied.
2 Experimental
2.1 Photocatalyst preparation
In this experiment, we prepared the Ni-B co-doped BiVO4 in two steps as follows.
Step 1: The B-BiVO4 and pure BiVO4 precursors were prepared through sol-gel method according to Ref. [21].
Step 2: 3 g B-doped BiVO4 precursor was impregnated at 80 ��C with an aqueous solution of Ni(NO3)2��6H2O until the solution was dry. The resulting powder was collected and calcined in air in a muffle furnace at 500 ��C for 5 h, and then cooled to room temperature to obtain xNi-B-BiVO4 nanoparticles. The Ni doping content (x) was chosen as 0.20%, 0.25%, 0.30%, 0.35% and 0.40%, respectively, which is the mass fraction of Ni(NO3)2��6H2O to BiVO4. Ni-doped BiVO4 was prepared with the same impregnation method and its precursor is pure BiVO4. Pure BiVO4 and B-BiVO4 were prepared in the same way as references for direct comparison [21].
2.2 Characterization and measurements
The crystal phases of the samples were investigated by X-ray diffraction (XRD) with Cu K�� radiation (model D/max RA, Rigaku, Japan). The accelerating voltage and the applied current were 40 kV and 150 mA, respectively. The morphology of the samples was checked by a scanning electron microscope (SEM, S-3000N, Hitachi, Japan), coupled to an energy-dispersive X-ray spectrometer (EDX, Oxford Instrument). The binding energies of Bi, V, Ni, B and O were measured at room temperature using an X-ray photoelectron spectroscope (XPS, VGESCALAB MARK II) using Mg K�� radiation. The Brunauer-Emmett-Teller (BET) surface area of the sample was obtained from nitrogen adsorption- desorption isotherms determined at liquid nitrogen temperature on an automatic analyzer (Autosorb-iQ-MP, Quantachrome, USA). The diffuse reflectance spectra (DRS) were investigated by a UV-Vis spectro- photometer (TU-1901, Puxi, China) equipped with an integrating sphere assembly using BaSO4 as the reflectance standard. The spectra were recorded in the wavelength range of 230 to 800 nm at (25��1) ��C.
2.3 Photocatalytic activity tests
The photocatalytic activities of the as-synthesized samples were measured by the photodegradation of MO, AOII and RhB solutions. A 250 W halogen lamp equipped with a cutoff filter smaller than 420 nm was used as the visible light source and was placed at approximately 14 cm from the reactor. The photodegradation experiment was performed as follows: 0.01 g of catalyst was added into a 50 mL solution of 15 mg/L MO (the concentrations of MB, AOII and RhB are 10, 15 and 15 mg/L, respectively). Prior to irradiation, the suspension was stirred in the dark for 60 min until the adsorption/desorption equilibrium was established. At given time intervals, the collected samples were filtered through a 0.45 ��m millipore filter to remove the catalyst particles. The filtrate concentration was monitored by recording the absorbance at 464 nm using a UV-1800 UV-Vis spectrophotometer (Puxi, China).
3 Results and discussion
3.1 XPS analysis
The analysis of XPS spectra was performed to investigate the chemical states and chemical compositions of the samples. The Ni 2p high-resolution XPS scan spectrum is shown in Fig. 1(a). From Fig. 1(a), the Ni 2p spectrum of 0.3Ni-B-BiVO4 consists of two strong symmetrical peaks at Eb=856.4 and 861.2 eV, corresponding to the Ni 2p3/2 and Ni 2p1/2 signals, which are characteristic of the Ni2+ species [9].
The B 1s XPS spectra of the B-doped and Ni-B- co-doped samples are shown in Figs. 1(b) and (c). Each XPS spectrum of B 1s exhibits an asymmetric broad peak from 184 to 191 eV, which is a typical characteristic peak for B 1s species. The asymmetric peak is decomposed into two components at Eb=184.2 and 190.8 eV for B-BiVO4, and Eb=186.9 and 188.8 eV for 0.3Ni-B-BiVO4. The peaks at Eb=184.2 and 186.9 eV are assigned to B4C [20] which shows no photocatalytic activity. The standard binding energies for B 1s in B2O3, H3BO3 (B��O bond), and VB2 (V��B bond) are 193.6 [28], 193.0 [29] and 188.3 eV [21], respectively. There is no doubt that the B atoms are not bonded by means of B��V��B bond or B��O bond. It may be concluded that some of the B atoms might be doped in the BiVO4 lattice and replaced the O atoms to form a B��Bi��O bond [20].
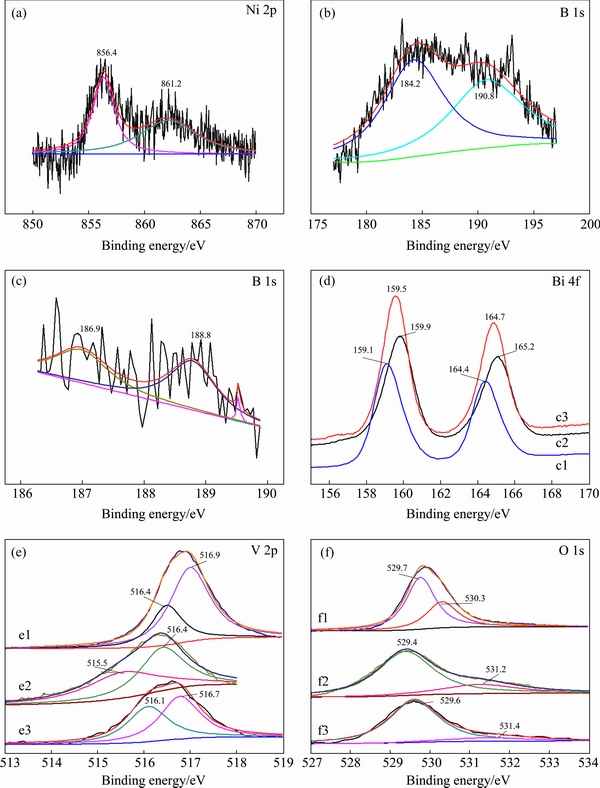
Fig. 1 Ni 2p (a), B 1s (b, c), Bi 4f (d), V 2p (e) and O 1s (f) XPS spectra of BiVO4 (1), B-BiVO4 (2) and 0.3Ni-B-BiVO4 (3)
After further doping with Ni, the binding energy of B 1s changes to 188.8 eV, which is comparable to the standard binding energy for B 1s in NiB (188.9 eV), probably because the doping Ni2+ ions replace some Bi3+ ions to form B��Ni��O bonds [9]. However, the binding energy of Ni in the 0.3Ni-B-BiVO4 sample is 856.4 eV, which is higher than that of Ni in NiB (852.4-854.2 eV). We can therefore deduce that no NiB phase appears in BiVO4 (as shown in Fig. 1(a)). Moreover, the electron density of B increases due to the lower electronegativity of Ni compared with that of Bi, while the binding energy of B 1s decreased. The XPS results show that both boron and nickel are successfully doped into the BiVO4 lattice.
Figure 1(d) shows the Bi 4f high-resolution XPS scan spectra of pure BiVO4, B-BiVO4, and 0.3Ni-B-BiVO4. The binding energies of Bi 4f appear at 159.1-159.5 eV and 164.4-165.2 eV for Bi 4f7/2 and Bi 4f5/2, respectively, which are characteristics of the Bi3+ species [12,17]. However, the binding energies are different: Eb=159.1 and 164.4 eV for BiVO4, Eb=159.9 and 165.2 eV for B-BiVO4, Eb=159.5 and 164.7 eV for 0.3Ni-B-BiVO4. Compared with undoped BiVO4 (159.1 and 164.4 eV for Bi 4f7/2 and Bi 4f5/2, respectively), the Bi 4f peaks of B-doped (159.9 and 165.2 eV), and Ni-B co-doped samples (159.5 and 164.7) slightly shift by 0.8 and 0.4 eV, respectively, towards higher binding energy. The results indicate that B or/and Ni have been incorporated in the BiVO4 crystal lattice, and the electron density of Bi consequently decreases because of the lower electronegativity of B with respect to O and the lower electronegativity of Ni with respect to Bi.
The V 2p XPS spectra of pure BiVO4, B-BiVO4 and 0.3Ni-B-BiVO4 are shown in Fig. 1(e). The asymmetric V 2p3/2 signals are decomposed into two peaks at 1) Eb=516.1 and 516.7 eV for BiVO4, 2) Eb= 515.5 and 516.4 eV for B-BiVO4, and 3) Eb=515.7 and 516.3 eV for 0.3Ni-B-BiVO4 and are attributed to the surface V4+ and V5+ species, respectively [20]. The surface V4+/V5+ molar ratio is 0.85 for BiVO4, 0.98 for B-BiVO4 and 1.24 for 0.3Ni-B-BiVO4. The molar ratio increases with B and Ni doping into the crystal lattice of BiVO4. According to the electro-neutrality principle, one can deduce that the BiVO4 samples are oxygen-deficient, and the amount of nonstoichiometric oxygen is dependent on the surface V4+/V5+ molar ratios.
As shown in Fig. 1(f), XPS spectrum of each O 1s shows a broad asymmetric peak, which can be fitted into two peaks. One stronger peak at ~530.07 eV corresponds to the bulk oxygen bonded to V or Bi [16]. The other peak at 530-531eV is attributed to the surface hydroxyl oxygen [16]. The molar ratios of Oads/Olatt in the undoped, B-doped, and B-Ni co-doped samples evaluated by the XPS analysis are 0.14, 0.85 and 1.59, respectively. Obviously, the B and Ni co-doping further increases the surface hydroxyl.
3.2 XRD and SEM analysis
To investigate the crystalline forms and crystallinities of the pure and doped samples, the XRD patterns of the as-prepared samples are presented in Fig. 2. These peaks are attributed to monoclinic BiVO4 (JCPDS cards No. 75-1866), as indexed in Fig. 2. No other phases can be found. However, the diffraction peaks observed for the doped samples are sharper than those of pure BiVO4, which indicates an increase of the crystallinity. Through using the Debye-Scherre equation, the crystallite sizes of all the samples, calculated for the (121) peak, are shown in Table 1. It shows that the crystallite sizes decrease when the amount of Ni doping increases. Because the ionic radius of Ni2+ (0.0690 nm) is smaller than that of Bi3+ (0.1110 nm), the Ni2+ ion might replace Bi3+ ion in B-BiVO4, as verified by the XPS results.
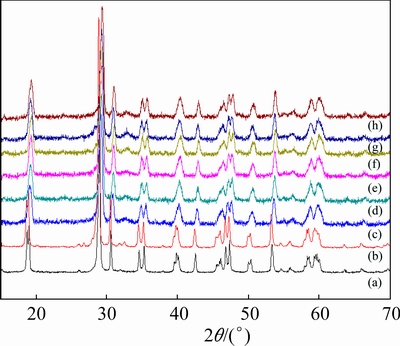
Fig. 2 XRD patterns of BiVO4 (a), B-BiVO4 (b), 0.30Ni-BiVO4 (c), 0.20Ni-B-BiVO4 (d), 0.25Ni-B-BiVO4 (e), 0.30Ni-B-BiVO4 (f), 0.35Ni-B-BiVO4 (g) and 0.40Ni-B- BiVO4 (h)
Table 1 Some selected properties of samples
The surface morphologies and particle sizes of pure BiVO4, B-BiVO4, and Ni-B-BiVO4 were observed by SEM (Fig. 3). The BiVO4 samples display a sphere-like morphology, and the particles are uniform in size. There is little difference in the morphologies between pure BiVO4 and B-BiVO4. But, the size of some particles decreases after Ni doping, indicating that Ni-doping can inhibit particle growth. The composition of the 0.30Ni-B-BiVO4 sample was determined by energy dispersive X-ray spectroscopy (EDX). As shown in Fig. 3(d), the signals for Bi, V, O and Ni are quite obvious.
The BET specific surface areas of the as-prepared samples were measured using a nitrogen adsorption BET method (shown in Table 1). Ni-B co-doping can slightly enhance the specific areas of samples.
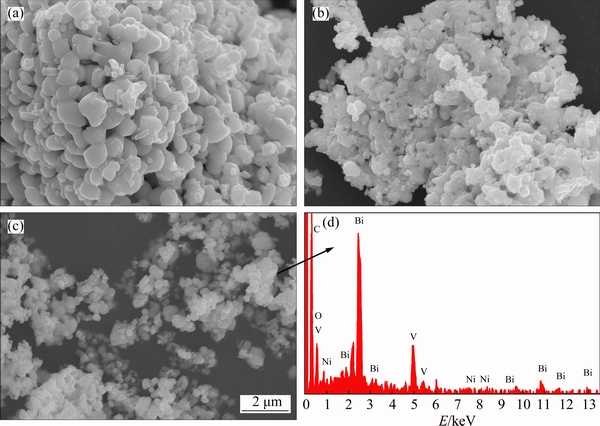
Fig. 3 SEM images of pure BiVO4 (a), B-BiVO4 (b), 0.30Ni-B-BiVO4 (c) and EDX spectrum of 0.30Ni-B-BiVO4 (d)
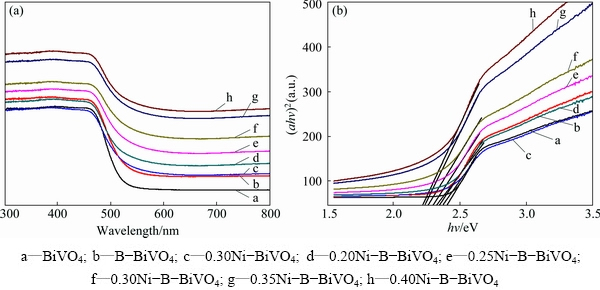
Fig. 4 UV-Vis absorption spectra (a) and (ahv)2-hv curves (b) of different samples
3.3 UV-Vis diffuse reflectance spectra
Figure 4 shows the UV-Vis diffuse reflectance spectra (DRS) of the Ni-B co-doped BiVO4 samples compared with pure, B-doped and Ni-doped BiVO4. Due to the introduction of B dopants, a red shift of the absorption spectrum occurred. WANG et al [21] proposed that the visible light response was due to the narrowing of the band gap induced by mixing B 1s and O 2p states. The d-electrons of the doping Ni ions transfer to the conducting band of BiVO4 [8], leading to stronger absorption ability in the visible region, more red shifting, and a narrower band gap. Furthermore, with the increase of the doping nickel amount, the absorptions of all samples are strengthened in the visible region. It is generally accepted that the photocatalytic performance is determined by the light absorption ability, charge separation efficiency, and transfer rate to the surface of the photocatalysts. Consequently, the absorption ability of visible light for BiVO4 is strengthened because nickel and boron co-doping can improve the photocatalytic activity. The Kubelka-Munk function, i.e., ahv=c(h�CEg)n, was used to estimate the band gap energy of the as-prepared samples, where a is the absorption coefficient, hv is the photon energy, c is a constant (c=1), Eg is the band gap energy, and n is a constant and is dependent on the type of semiconductor. In the case of BiVO4, n is equal to 0.5 and 2 for direct and indirect band gaps, respectively [17]. The results indicate that the band gap energies of BiVO4, B-BiVO4, 0.3Ni-BiVO4 and xNi-B-BiVO4 (x=0.20, 0.25, 0.30, 0.35, 0.40) are 2.40, 2.36, 2.38, 2.33, 2.30, 2.26, 2.24 and 2.22, respectively.
3.4 Photocatalytic activity
The photocatalytic activities of BiVO4, B-BiVO4, Ni-BiVO4, and xNi-B-BiVO4 were evaluated by examining the degradation of MO. The change in MO concentration was determined by measuring the change of its characteristic absorption band at 452 nm. Figure 5(a) shows the degradation rate of MO over pure, B-doped, Ni-doped, and Ni-B co-doped BiVO4 samples under visible light irradiation for 50 min. Figure 5(b) presents the kinetic curves for the photocatalytic degradation of MO, and the apparent rate constants of the samples were calculated according to the following formula: ln(c0/ct)=kt, where c0 and ct are the initial and measured concentrations after irradiation for t min, respectively [21].
From Fig. 5(b), the reaction did follow a first-order mechanism, and the apparent rate constants obtained for different catalysts with different Ni contents are listed in Table 1. Furthermore, the results for the blank test demonstrate that the degradation of MO is slow without a photocatalyst. Compared with pure BiVO4, the MO photocatalytic degradation rate of the B-doped sample is higher. With Ni doping, the MO photocatalytic degradation rate of Ni-B co-doped BiVO4 increases with Ni doping content up to 0.30%, but decreases when the amount of Ni is higher than 0.30%. The degradation rate of MO can reach 95% in 50 min, which shows that the optimal mass fraction of Ni doping is 0.30%.
The 0.3Ni-B-BiVO4 was also used to photodegrade methyl blue (MB), acid orange II (AOII), rhodamine B (RhB) solutions. The changes in the absorption spectra of MB, AOII, RhB and MO solutions during the photodegradation process by 0.3Ni-B-BiVO4 at different irradiation time are shown in Fig. 6. It was observed that the maximum absorption peaks of these four dye solutions decrease with increasing irradiation time. This indicated that the concentration of the dye solutions decreased in the presence of 0.3Ni-B-BiVO4 and visible light illumination. So, we can conclude that the Ni-B co-doped BiVO4 has a good photocatalytic activity for organic pollutants.
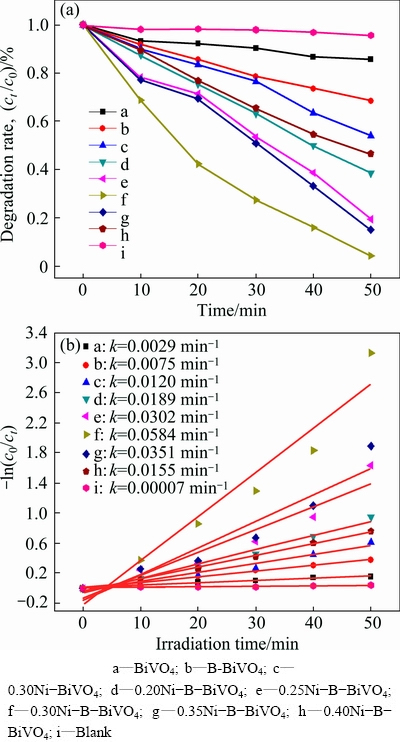
Fig. 5 MO degradation rate under visible light illumination for 50 min in the presence of B-BiVO4 with various nickel doping, pure BiVO4 and without photocatalyst (a), and ln(c0/ct) vs time curves of different samples (b)
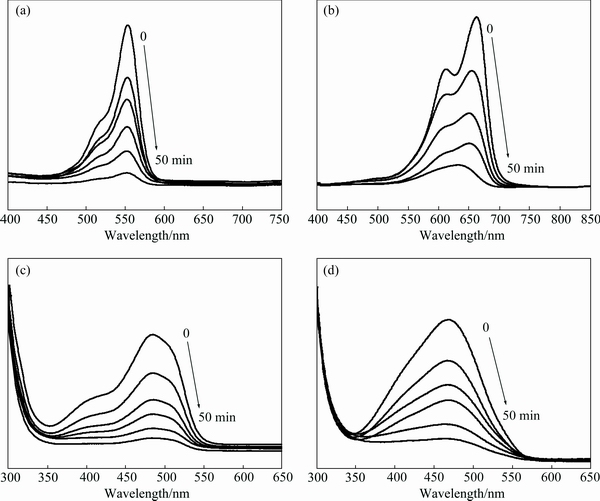
Fig. 6 UV-Vis absorption spectra of photocatalytic degradation of 10 mg/L RhB (a), 15 mg/L MB (b), 15 mg/L AOII (c) and 15 mg/L MO (d) by 0.3Ni-B-BiVO4 samples at different irradiation time
3.5 Mechanism for photocatalytic activity of Ni-B co-doped BiVO4
In contrast with the undoped, B or Ni single doped BiVO4 samples, the enhanced degradation of organic pollutants with the Ni-B co-doped sample under visible light irradiation can be explained as follows. Firstly, it is well known that the photoactivity of a catalyst is determined by the light absorption ability. The co-doped sample exhibits an enhanced visible light absorption property due to the B and Ni present in the substitutional positions in the BiVO4 lattice. The dopants introduce more energy levels in the BiVO4 band gap, thus leading to a narrower band gap in Ni-B-BiVO4 catalysts. They improve the quantity of the photo-induced electrons and holes, and promote the photocatalytic rate. Secondly, the separation efficiency of photogenerated electrons and holes is an important factor determining the light quantum yield. The lower the recombination rate of the sample is, the higher the photoactivity is. The Ni-doping in B-BiVO4 can effectively inhibit the recombination of photogenerated electrons and holes, thereby improving the quantum efficiency. Additionally, more V4+ ions, formed by Ni doping in B-BiVO4, can also capture photo-induced electrons to form V5+, which is favorable for photocatalytic efficiency. Furthermore, the Ni-B co-doping further increases the concentration of surface hydroxyl oxygen (Oads) in BiVO4 (as shown in Section 3.1), which is beneficial to photocatalytic process. The hydroxyl groups may accept photogenerated holes to produce strong oxidative hydroxyl free radicals (��OH) and H+ species. On the other hand, the surface hydroxyl groups can also act as adsorption centers for O2 and organic molecules [30]. This reaction would occur via the following mechanism:
BiVO4+hv��e+h+ (1)
Ni2++hv��Ni3++e (2)
V4++hv��V5++e (3)
Ni3++e��Ni2+ (4)
V5++e��V4+ (5)
H2O+h+����OH+H+ (6)
O2+e��O2�C (7)
O2�C+H+����OH (8)
��OH+organic pollutants��oxidation products of organic pollutants (9)
Photo-induced e/h+ pairs are generated on the surface of the photocatalyst during irradiation (Eq. (1)). The photo-induced electron (e) can be easily trapped by Ni3+ and V5+ ions (Eqs. (4) and (5)), thus effectively reducing the recombination of e/h+ pairs. The photo-induced h+ oxidizes H2O molecules adsorbed on the photocatalyst surface into ��OH and H+ species (Eq. (6)), while the photo-induced e can be captured by O2 molecules adsorbed on the catalyst surface to form O2�C species (Eq. (7)), and H+ reduces the active O2�C species into ��OH species (Eq. (8)). It is known that ��OH species are the main active species in the photocatalytic reaction [29]. The organic molecules are oxidized by the ��OH species into inorganic molecules (Eq. (9)). Therefore, we think that active V5+, Ni3+, and Oads species contribute to the direct or indirect generation of ��OH species on the surface of the photocatalyst under visible light illumination, which promote the photocatalytic degradation of MO. In other words, doping Ni2+ into the B-BiVO4 lattice improves photocatalytic activity by increasing the concentrations of V4+ and Oads species.
In addition, it is generally accepted that photocatalytic performance is also influenced by the crystal structure, particle morphology and surface area [3,31]. In the present work, the samples had some similar physical properties such as monoclinic scheelite crystal structure and sphere-like particle morphology. The crystal structure and particle morphology are therefore not the main reasons for the higher photocatalytic activity of Ni-B co-doped BiVO4. However, as seen from the BET results, the surface areas of Ni-B co-doped BiVO4 samples are larger than those of the undoped, B or Ni single doped BiVO4 samples. We can therefore conclude that the increase in surface area by Ni-B co-doping may be another cause for the higher photocatalytic activity because photocatalytic reactions mainly take place on the photocatalyst��s surface. The adsorption of target pollutants on the photocatalyst is also a key step in the photocatalytic reaction. Consequently, the stronger adsorption of organic pollutants and the improved microstructural properties both benefit to the enhanced photoactivity of the co-doped sample. In conclusion, all of the above factors resulting from Ni-B co-doping cause a synergetic effect to enhance the visible light activity.
However, Ni2+ ions can also act as recombination centers for the electrons and holes. Therefore, an adequate amount of doping Ni in the B-BiVO4 catalysts is important so that the recombination of photogenerated electrons and holes can be suppressed effectively. As a consequence, the efficiency of the photocatalytic reaction is improved.
4 Conclusions
1) Ni-doped BiVO4 samples with different Ni2+ doping concentrations were successfully prepared through sol-gel and impregnation two-step method. Compared with the undoped, B-, Ni-single doped BiVO4, the Ni-B co-doped BiVO4 exhibits higher photoactivity under visible light, which shows higher photocatalytic performance for the MO degradation, and the highest degradation rate with 0.3Ni-B-BiVO4 can reach approximately 96% under visible light irradiation for 50 min. The 0.3Ni-B-BiVO4 sample can also effectively photodegrade RhB, MB and AOII.
2) All of the samples had single-phase monoclinic crystal structure and sphere-like particle morphology. Ni-B co-doped BiVO4 leads to the narrower band gaps, the appearance of Ni2+ species and increased amounts of V4+ and Oads species. Moreover, Ni-B co-doping increases the concentration of surface hydroxyls and the adsorption ability to organic substances.
3) The enhanced visible light activity for Ni-B co-doped BiVO4 is ascribed to the synergetic effect of the increased visible light absorption ability, charge separation efficiency, surface hydroxyl and adsorption ability to organic pollutants.
References
[1] HE Rong-an, CAO Shao-wen, ZHOU Peng, YU Jia-guo. Recent advances in visible light Bi-based photocatalysts [J]. Chinese Journal of Catalysis, 2014, 35(7): 989-1007.
[2] CHEN Yuan, ZHOU Ke-chao, HUANG Su-ping, LI Zhi-you, LIU Guo-cong. Hydrothermal synthesis and photocatalytic property of BiVO4 nanosheets [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(7): 1570-1579. (in Chinese)
[3] XU Lei, WEI Yong-ge, GUO Wan, GUO Yi-hang, GUO Ying-na. One-pot solvothermal preparation and enhanced photocatalytic activity of metallic silver and graphene co-doped BiVO4 ternary systems [J]. Applied Surface Science, 2015, 332: 682-693.
[4] ZHANG X F, DU L L, WANG H, DONG X L, ZHANG X X, MA C, MA H C. Highly ordered mesoporous BiVO4: Controllable ordering degree and super photocatalytic ability under visible light [J]. Microporous and Mesoporous Materials, 2013, 173: 175-180.
[5] ZHOU Ying, LI Wei, WAN Wen-chao, ZHANG Rui-yang, LIN Yuan-hua. W/Mo co-doped BiVO4 for photocatalytic treatment of polymer-containing wastewater in oilfield [J]. Superlattices and Microstructures, 2015, 82: 67-74.
[6] SARKAR S, CHATTOPADHAYY K K. Visible light photocatalysis and electron emission from porous hollow spherical BiVO4 nanostructures synthesized by a novel route [J]. Physica E: Low-dimensional Systems and Nanostructures, 2014, 58: 52-58.
[7]  G. Heterostructured Er3+ doped BiVO4 with exceptional photocatalytic performance by cooperative electronic and luminescence sensitization mechanism [J]. Applied Catalysis B: Environmental, 2014, 158-159: 242-249.
G. Heterostructured Er3+ doped BiVO4 with exceptional photocatalytic performance by cooperative electronic and luminescence sensitization mechanism [J]. Applied Catalysis B: Environmental, 2014, 158-159: 242-249.
[8] ZHANG Bin, ZHAO Xu, LIU Hui-jiuan, QU Jiu-hui, HUANG C P. Synthesis of visible-light sensitive M-BiVO4 (M=Ag, Co, and Ni) for the photocatalytic degradation of organic pollutants [J]. Separation and Purification Technology, 2011, 77: 275-282.
[9] CHEN Yuan, YANG Jia-tian, PANG Qi, ZHOU Ke-chao, HUANG Jian-ying. Hydrothermal synthesis of visible light driven Ni-BiVO4 photocatalysts and its photocatalytic performance [J]. Journal of Synthetic Crystals, 2013, 42: 1230-1236. (in Chinese)
[10] ZHANG Jin, CUI Hao, WANG Bing, LI Chuang, ZHAI Jian-ping, LI Qin. Preparation and characterization of fly ash cenospheres supported CuO-BiVO4 heterojunction composite [J]. Applied Surface Science, 2014, 300: 51-57.
[11] GAO Xiao-ming, FU Feng, ZHANG Li-ping, LI Wen-hong. The preparation of Ag-BiVO4metal composite oxides and its application in efficient photocatalytic oxidative thiophene [J]. Physica B: Condensed Matter, 2013, 419: 80-85.
[12] CHALA S, WETCHAKUN K, PHANICHPHANT S, INCEESUNGVORN B, WETCHANUM N. Enhanced visible- light-response photocatalytic degradation of methylene blue on Fe-loaded BiVO4 photocatalyst [J]. Journal of Alloys and Compounds, 2014, 597: 129-135.
[13] LUO Yang-yang, TAN Guo-qiang, DONG Guo-hua, ZHANG Li-li, HUANG Jing, YANG Wei, ZHAO Cheng-cheng, REN Hui-jun. Structural transformation of Sm3+ doped BiVO4 with high photocatalytic activity under simulated sun-light [J]. Applied Surface Science, 2015, 324: 505-511.
[14] ZHANG Yao-hong, YI Zhi-guo, WU Guo-hua, SHEN Qing. Novel Y doped BiVO4 thin film electrodes for enhanced photoelectric and photocatalytic performance [J]. Journal of Photochemistry and Photobiology A: Chemistry, 2016, 327: 25-32.
[15] LUO Yang-yang, TAN Guo-qiang, DONG Guo-hua, REN Hui-jun, XIA Ao. A comprehensive investigation of tetragonal Gd-doped BiVO4 with enhanced photocatalytic performance under sun-light [J]. Applied Surface Science, 2016, 364: 156-165.
[16] JIANG Hai-yan, DAI Hong-xin, DENG Ji-guan, LIU Yu-xi, ZHANG Lei, JI Ke-meng. Porous F-doped BiVO4: Synthesis and enhanced photocatalytic performance for the degradation of phenol under visible-light illumination [J]. Solid State Sciences, 2013,17: 21-27.
[17] ZHAO Zhen-xuan, DAI Hong-xing, DENG Ji-guang, LIU Yu-xi, AU C K. Effect of sulfur doping on the photocatalytic performance of BiVO4 under visible light illumination [J]. Chinese Journal of Catalysis, 2013, 34(8): 1617-1626.
[18] WANG Min, LIU Qiong, CHE Yin-sheng, ZHANG Li-fang, ZHANG Dong. Characterization and photocatalytic properties of N-doped BiVO4synthesized via a sol-gel method [J]. Journal of Alloys and Compounds, 2013, 548: 70-76.
[19] TAN Guo-qiang, ZHANG Li-li, REN Hui-jun, HUANG Jing, YANG Wei, XIA Ao. Microwave hydrothermal synthesis of N-doped BiVO4nanoplates with exposed (040) facets and enhanced visible-light photocatalytic properties[J]. Ceramics International A, 2014, 40: 9541-9547.
[20] WANG Min, ZHENG Hao-yan, LIU Qiong, NIU Chao, CHE Yin-sheng, DANG Ming-yan. High performance B-doped BiVO4photocatalyst with visible light response by citric acid complex method [J]. Spectrochimica Acta A: Molecular and Biomolecular Spectroscopy, 2013, 114: 74-79.
[21] WANG Min, NIU Chao, LIU Jun, WANG Qian-wu, YANG Chang-xiu, ZHENG Hao-yan. Enhanced visible-light-driven photocatalytic activity of B-doped BiVO4 synthesized using a corn stem template [J]. Materials Science in Semiconductor Processing, 2015, 30: 307-313.
[22] LI Yan-qing, JING Tao, LIU Yuan-yuan, HUANG Bai-biao, DAI Ying-dai, ZHANG Xiao-yang, QIN Xiao-yan, WHANGBO M H. Enhancing the efficiency of water oxidation by boron-doped BiVO4 under visible light: Hole trapping by BO4 tetrahedra [J]. Chem Plus Chem, 2015, 80: 1113-1118.
[23] ZHOU Yi, HUANG Ke-long, ZHU Zhi-ping, XIA Chang-bin. Eu2+/Gd3+-codoped nanocrystalline titania catalyst and its photocatalytic activity under natural light [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(S3): s1112-s1116.
[24] PATEL N, JAISWAL R, WARANG T, SCARDUELLI G, DASHORA A, AHUJA B L, KOTHARI D C, MIOTELLO A. Efficient photocatalytic degradation of organic water pollutants using V-N co-doped TiO2 thin films [J]. Applied Catalysis B: Environmental, 2014, 150-151: 74-81.
[25] KIMA T H,  V, GYAWALI G, CHO S H, SEKINO T, LEE S W. Synthesis of solar light responsive Fe, N co-doped TiO2 photocatalyst by sonochemical method [J]. Catalysis Today, 2013, 212: 75-80.
V, GYAWALI G, CHO S H, SEKINO T, LEE S W. Synthesis of solar light responsive Fe, N co-doped TiO2 photocatalyst by sonochemical method [J]. Catalysis Today, 2013, 212: 75-80.
[26] WANG Min, CHE Yin-sheng, NIU Chao, DANG Min-yang, ZHANG Dong. Effective visible light-active boron and europium co-doped BiVO4 synthesized by sol-gel method for photodegradion of methyl orange [J]. Journal of Hazardous Materials, 2013, 262: 447-455.
[27] WANG Min, CHE Yin-sheng, NIU Chao, DANG Min-yang, ZHANG Dong. Lanthanum and boron co-doped BiVO4with enhanced visible light photocatalytic activity for degradation of methyl orange [J]. Journal of Rare Earths, 2013, 31: 878-884.
[28] LONG Hui-jin, MENG Qing-ju, YUAN Jin, YANG Wen-sheng, CAO Ya-an. Study on photocatalytic activity of boron doped TiO2 catalyst [J]. Acta Chimica Sinica , 2008, 66: 657-661. (in Chinese)
[29] WEI Feng-yu, NI Liang-suoi. Photocatalytic performance and doping mechanism of B-S co-doped TiO2 [J]. Chinese Journal of Catalysis, 2007, 28: 905-909.
[30] UMAPATHY V, MANIKANDAN A, ARUL ANTONY S, RAMU P, NEERAJA P. Structure, morphology and opto-magnetic properties of Bi2MoO6 nano-photocatalyst synthesized by sol-gel method [J]. Transactions of Nonferrous Metal of China, 2015, 25: 3271-3278.
[31] JIANG Hai-yan, DAI Hong-xin, MENG Xue, ZHANG Lei, DENG Ji-guang, JI Ke-meng. Morphology dependent photocatalytic performance of monoclinic BiVO4 for methyl orange degradation under visible-light irradiation [J]. Chinese Journal of Catalysis, 2011, 32: 939-949.
�� �������������㣬лԪ���������ѣ��� ������ ͮ
������ѧ ��е�������Զ���ѧԺ������ 110165
ժ Ҫ��Ϊ�˽�һ�����B-BiVO4�Ĺ�����ԣ�ͨ���ܽ�-�����ͽ����������Ʊ�Ni����B-BiVO4���(Ni-B-BiVO4)��ͨ��XPS��XRD��SEM��BET��EDS��UV-Vis���ֶζ���Ʒ���б�������������������Ӿ�����ı���Ʒ�ľ��ͺ���ò����������Ni����Ʒ�ľ���������С����δ���ӡ�B�Լ�Ni��������ȣ�Ni-B-BiVO4��Ʒ�Ĺ�������������ƫ�ƣ���N-B�����Ӻ���Ʒ��V4+ �ͱ����ǻ������ࡣ�����Ni������Ϊ0.30%(��������)ʱ�����Ʊ���0.3Ni-B-BiVO4��50 min�ڶԼ��ȵĽ����ʴ�95%���ң��Ҹ���Ʒ����Ч�����Ǽ���(MB)�����II��(AOII)������B(RhB)��Һ����������Ʒ���Ե������Ҫ��B��Ni��Эͬ�������¡�
�ؼ��ʣ������ӣ������ӣ�BiVO4; ��������ܽ�-�����������շ�
(Edited by Wei-ping CHEN)
Foundation item: Projects (21207093, 51004072) supported by the National Natural Science Foundation of China for Youth; Project (LJQ2014023) supported by the Liaoning Excellent Talents in University, China; Project (L20150178) supported by the General Scientific Research Projects Foundation of Liaoning Educational Committee, China; Project (N140303002) supported by the Fundamental Research Funds for the Central Universities, China
Corresponding author: Tong ZHU; Tel: +86-13940281581; E-mail: tongzhu@mail.neu.edu.cn
DOI: 10.1016/S1003-6326(17)60227-9

