Na2O��������ƻ�������γɻ����뾧��ģ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2016���3��
�������ߣ������� ������ �ں��� Ϳ�ӷ�
����ҳ�룺849 - 858
�ؼ��ʣ�����ƣ�Na2O���ӣ��γɻ���������ṹ���ս�����ģ��
Key words��calcium aluminate; Na2O-doping; formation mechanism; crystal structure; sintering; computer simulation
ժ Ҫ�����÷������Լ�CaCO3��Al2O3��SiO2��Na2CO3�ϳ�Na2O������������ϣ��о�Na2O���Ӷ�����ƻ������γɻ�����12CaO��7Al2O3(C12A7)����ṹ��Ӱ�졣�����������������Na2O��������4.26%(��������)ʱ����Na2O������ҪΪ2Na2O��3CaO��5Al2O3��Na2O��Al2O3������Na2O��Ҫ������12CaO��7Al2O3�ڣ�����ʹ��ᾧ�Ƚ��͡�2Na2O��3CaO��5Al2O3�Ľᾧ��Ҳ����������Na2O�������߶����͡�2Na2O��3CaO��5Al2O3��12CaO��7Al2O3������;�������ַ�ʽ��һ���ɳ�ʼ��Ӧ���ֱ�ӷ�Ӧ���ɣ������ɷ�Ӧ�м����CaO��Al2O3�Ľ�һ��ת����Ӧ���ɡ�����ṹģ������������Na2O���ӵ�12CaO��7Al2O3�����ڣ���O��Na���Ĺ���������O��Ca����O��Al���Ĺ����ԣ�ʹ12CaO��7Al2O3�������������ߣ���ѧ������ߡ���������Na2O������0������4.26%ʱ��Al2O3�Ľ�������34.81%������88.17%��
Abstract: Calcium aluminate clinkers doped with Na2O were synthesized using analytically pure reagents CaCO3, Al2O3, SiO2 and Na2CO3. The effects of Na2O-doping on the formation mechanism of calcium aluminate compounds and the crystal property of 12CaO��7Al2O3 (C12A7) cell were studied. The results show that the minerals containing Na2O mainly include 2Na2O��3CaO��5Al2O3 and Na2O��Al2O3, when the Na2O content in clinkers is less than 4.26% (mass fraction). The rest of Na2O is mainly doped in 12CaO��7Al2O3, which results in the decrease of the crystallinity of 12CaO��7Al2O3. The crystallinity of 2Na2O��3CaO��5Al2O3 is also inversely proportional to the Na2O content in clinkers. The formation processes of 2Na2O��3CaO��5Al2O3 and 12CaO��7Al2O3 can be divided into two ways, which are the direct reactions of raw materials and the transformation of CaO��Al2O3, respectively. The simulation shows that the covalency of O��Na bond in Na2O-doped 12CaO��7Al2O3 cell is weaker than those of O��Ca and O��Al bonds. The free energy of the unit cell increases because of Na2O doping, which results in the improvement of chemical activity of 12CaO��7Al2O3. The leaching efficiency of Al2O3 in clinker is improved from 34.81% to 88.17% when the Na2O content in clinkers increases from 0 to 4.26%.
Trans. Nonferrous Met. Soc. China 26(2016) 849-858
Yong-pan TIAN, Xiao-lin PAN, Hai-yan YU, Gan-feng TU
School of Metallurgy, Northeastern University, Shenyang 110819, China
Received 23 April 2015; accepted 10 November 2015
Abstract: Calcium aluminate clinkers doped with Na2O were synthesized using analytically pure reagents CaCO3, Al2O3, SiO2 and Na2CO3. The effects of Na2O-doping on the formation mechanism of calcium aluminate compounds and the crystal property of 12CaO��7Al2O3 (C12A7) cell were studied. The results show that the minerals containing Na2O mainly include 2Na2O��3CaO��5Al2O3 and Na2O��Al2O3, when the Na2O content in clinkers is less than 4.26% (mass fraction). The rest of Na2O is mainly doped in 12CaO��7Al2O3, which results in the decrease of the crystallinity of 12CaO��7Al2O3. The crystallinity of 2Na2O��3CaO��5Al2O3 is also inversely proportional to the Na2O content in clinkers. The formation processes of 2Na2O��3CaO��5Al2O3 and 12CaO��7Al2O3 can be divided into two ways, which are the direct reactions of raw materials and the transformation of CaO��Al2O3, respectively. The simulation shows that the covalency of O��Na bond in Na2O-doped 12CaO��7Al2O3 cell is weaker than those of O��Ca and O��Al bonds. The free energy of the unit cell increases because of Na2O doping, which results in the improvement of chemical activity of 12CaO��7Al2O3. The leaching efficiency of Al2O3 in clinker is improved from 34.81% to 88.17% when the Na2O content in clinkers increases from 0 to 4.26%.
Key words: calcium aluminate; Na2O-doping; formation mechanism; crystal structure; sintering; computer simulation
1 Introduction
The low-grade bauxites and non-bauxite sources such as nepheline, clay and fly ash are widely distributed in China. It is suitable to extract alumina from these sources by the lime sintering process since dry materials can be used during the sintering process, reducing the energy consumption of the alumina production industry. However, the application of the lime sintering process is restricted by the defects including the high consumption of calcium, large amount of slag and poor leaching property of clinkers. The clinker of the lime sintering process mainly consists of 12CaO��7Al2O3 (Ca12Al14O33, C12A7), 2CaO��SiO2 (C2S), CaO��Al2O3 (CaAl2O4, CA) and 2CaO��Al2O3��SiO2 (C2AS). The calcium aluminate compounds, which can decompose in the sodium carbonate solution, are mainly C12A7 and CA. The previous researches [1,2] showed that the doping of Na2O in the sintering process could improve the leaching performance of clinkers and reduce the consumption of CaO. Moreover, the results showed that the lattice constants of C12A7 increase with the increase of the molar ratio of Na2O to Al2O3 (N/A ratio) of clinker. However, the details of phase structures were not analyzed, and the mechanisms by which Na2O affects the calcium aluminate compounds were unknown.
In the Na2O-doped CaO-Al2O3 system, the amounts of C12A7 and 2Na2O��3CaO��5Al2O3 (Na4Ca3(AlO2)10) reach the maximum as the sodium content in CA increases to 10% (mass fraction) [3]. Additionally, the crystallinity of C12A7 decreased with the doping of Na2O while that of CA increased [4]. FUKUDA et al [5] reported that Al3+ ions were substituted by Si4+ ions and Ca2+ ions were substituted by Na+ ions, when 3CaO��Al2O6 (C3A) was doped with Na+ and Si4+ ions. Many reports [6-11] showed that the properties of crystal structures could be analyzed by the Material Studio. BRIK et al [12] showed that the structural and electronic properties of the two Cr3+-bearing systems (NaCrSi2O6 and LiCrSi2O6) could be studied by the CASTEP module of Materials Studio package with two ab initio DFT-based calculations. WU et al [13] observed that the coordination number of Al atom as well as its amphoteric property in the melt of xCaO��(1-x)Al2O3 (x represents mole fraction) and the microstructure properties of xCaO��(1-x)Al2O3 could be studied by the molecular dynamics simulation using Materials Studio. PAN et al [14] reported that the free energy of the C12A7 cell calculated by the Materials Studio increased because of the absence of some Ca atoms in C12A7 cells. In this work, the compounds containing Na2O, the doping mechanism of Na2O in compounds and the chemical activity of the C12A7 crystal structure with or without Na2O doping as well as leaching characteristics of clinkers were investigated. Meanwhile, the phase constitutions, the distribution characteristics of phases in micro-regions and the crystal structure of C12A7 doped with Na2O were also investigated through Material Studio.
2 Experimental
2.1 Materials
Analytically pure reagents (CaCO3, Al2O3, SiO2 and Na2CO3) were used in the current work. In Na2O-doped CaO-Al2O3-SiO2 system, the mass ratio of Al2O3 to SiO2 (A/S) was about 1.6 and the molar ratio of CaO to Al2O3 (C/A) was about 0.85. The C/A ratios of Na2O-doped CaO-Al2O3 system were 0.6 and 1.0. The amounts of Na2O in clinker were represented by the values of the molar ratios of Na2O to Al2O3 (N/A). The mixed materials were sintered at 1200 ��C or 1350 ��C in a MoSi2 resistance furnace (KSL-1700-A2). Then, the clinkers were ground to 100 ��m.
2.2 Methods
The X-ray diffraction (XRD) data were collected on a Philips PW3040/60 with Cu K�� (��=1.54056 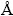 ) radiation operated at 40 kV and 40 mA. The K�� ray included two kinds of wavelength, which were K��1 and K��2, respectively. The data were analyzed by the MDI Jade with the database PDF2-2004. The cubic spline method was used to subtract the background and the peaks generated by the K��2 radiation were also subtracted as the ratio of K��1/K��2 was 2.0. The Pseudo-Voigt function was used during the fitting processes of spectra, and nearly 100 peaks were fitted for every spectrum. The microstructural analyses were carried out by scanning electron microscopy (SEM, SHIMADZU SSX-550) and energy-dispersive X-ray spectroscopy (EDS, DX-4). The samples were polished flat in water-free ethylene glycol using SiC papers, cleaned by ultra-sound in water-free ethylene glycol and coated with gold by sputtering.
) radiation operated at 40 kV and 40 mA. The K�� ray included two kinds of wavelength, which were K��1 and K��2, respectively. The data were analyzed by the MDI Jade with the database PDF2-2004. The cubic spline method was used to subtract the background and the peaks generated by the K��2 radiation were also subtracted as the ratio of K��1/K��2 was 2.0. The Pseudo-Voigt function was used during the fitting processes of spectra, and nearly 100 peaks were fitted for every spectrum. The microstructural analyses were carried out by scanning electron microscopy (SEM, SHIMADZU SSX-550) and energy-dispersive X-ray spectroscopy (EDS, DX-4). The samples were polished flat in water-free ethylene glycol using SiC papers, cleaned by ultra-sound in water-free ethylene glycol and coated with gold by sputtering.
The Visualizer module of Materials Studio was used to establish three-dimensional models of C12A7 cells with or without Na2O doping, which provided the foundation models for the subsequent calculation. The lattice parameter, the unit cell volume and free energy of the crystal structure, as well as the population and length of chemical bond were analyzed through the geometry optimization of the crystals. The CASTEP module of Materials Studio was used during the optimization process. The generalized gradient approximation (GGA) and the corresponding exchange-correlation potential (PBE) were used during the geometry optimization of the crystals through the BFGS algorithm while the Ultrasoft was selected as pseudopotential. The energy cut-off value was 340.0 eV. The SCF tolerance was 1.0��10-6 eV/atom. The results were obtained after large numbers of iterations. The original crystallographic data of the calculation were taken from the inorganic crystal structure database (ICSD).
2.3 Leaching conditions
The leaching experiments were carried out in a thermostatic water bath. The sintered clinkers were leached by the sodium carbonate and sodium hydroxide solution at 80 ��C for 30 min. The liquid-to-solid ratio for leaching was 10. The sodium carbonate concentration of the solution was 80 g/L (in the form of Na2O) and the sodium hydroxide concentration of the solution was 15 g/L (in the form of Na2O). The leaching residues were washed to neutrality with hot water and dried under 90 ��C. The chemical compositions of clinkers and residues were analyzed by X-ray fluorescence (XRF-1800). The alumina leaching efficiencies (��AO) of Na2O-doped CaO-Al2O3-SiO2 system clinker and Na2O-doped CaO-Al2O3 system clinker were calculated according to Eq. (1):
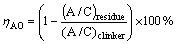 (1)
(1)
where (A/C)residue and (A/C)clinker are the mass ratios of Al2O3 to CaO of leached residues and sintered clinkers, respectively.
3 Results and discussion
3.1 Phase species and formation mechanism
As shown in Fig. 1(a), pure Na4Ca3(AlO2)10 was synthesized after sintering at 1200 ��C for 30 min. There is no Na4Ca3(AlO2)10 card in the PDF2-2004. But the XRD pattern of Na4Ca3(AlO2)10 is similar to that in Ref. [3]. CA was synthesized after sintering at 1600 ��C for 60 min. CA and Na2CO3 were sintered at 1350 ��C for 60 min when the N/A ratio is 0.33. The XRD patterns of clinkers are shown in Fig. 1(b). Most of CA transforms to C12A7 and Na4Ca3(AlO2)10. Some Na2O��Al2O3 (NA) also exists in clinkers.
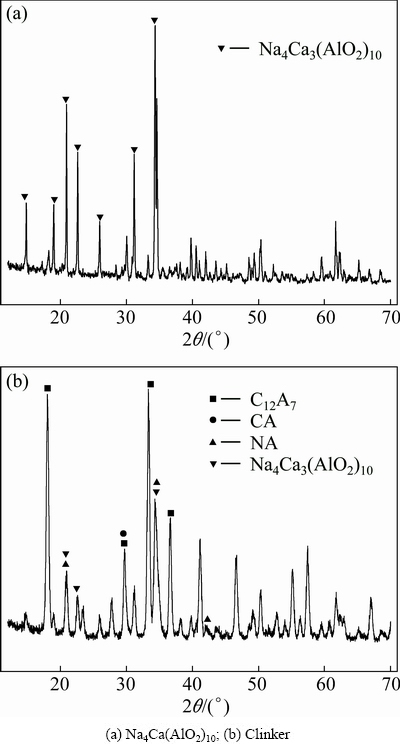
Fig. 1 XRD patterns of Na2O-CaO-Al2O3 system clinkers
Table 1 Chemical compositions of Na2O-doped clinkers (mass fraction, %)


Fig. 2 XRD patterns of clinkers with different N/A ratios
The compositions of Na2O-doped CaO-Al2O3-SiO2 system clinkers are shown in Table 1. The phases of clinkers with different N/A ratios are shown in Fig. 2. The phases containing Al2O3 consist of C12A7, CA, NA, CaO��2Al2O3 (CA2), Na4Ca3(AlO2)10 and C2AS. The phases containing SiO2 consist of C2AS, ��-C2S and ��-C2S. The phases containing Na2O consist of Na4Ca3(AlO2)10 and NA. In addition, trace amounts of Ca8.5NaAl6O18 and Ca8.25Na1.5Al6O18 are also found. When the N/A ratio is less than 0.04, small amount of CA2 and some un-reacted CaO and Al2O3 exist in clinkers. As the N/A ratio increases to 0.17, CaO and Al2O3 react completely. The amount of CA2 is also inversely proportional to the amount of Na2O in clinkers. No CA2 exists in clinkers as the N/A ratio increases to 0.17, which indicates that Na2CO3 accelerates the transformation reactions of CA2. The melting temperature of Na2CO3 is about 850 ��C and the decomposition temperature is about 1750 ��C, so Na2CO3 melts at the clinkering temperature of 1350 ��C. The diffusion speeds of ions are accelerated by the melting of Na2CO3, which results in higher reaction velocities of CaO, Al2O3 and CA2. The sintering temperature is higher than the formation temperature of Na4Ca3(AlO2)10. So, some Na4Ca3(AlO2)10 form before the sintering temperature reaches 1350 ��C in the Na2O-doped CaO-Al2O3-SiO2 system clinkers. The formation processes of Na4Ca3(AlO2)10 and C12A7 can be divided into two ways, which are the transformation of CA (Eq. (2) [3]) and the direct reaction among Na2CO3, CaO and Al2O3, respectively.
39(CaO��Al2O3)+10Na2CO3=2(12CaO��7Al2O3)+5(2Na2O��3CaO��5Al2O3)+10CO2�� (2)
The intensities of characteristic peaks of different phases are shown in Table 2. With the doping of Na2O and the absolute reaction of CaO and Al2O3, the intensity of the characteristic peak of C2AS at 2�� value of 31.47�� increases firstly and then decreases as the N/A ratio increases from 0.04 to 0.24. The increase of the amount of C2AS may be due to the higher reaction velocities of CaO, Al2O3 and SiO2 accelerated by the melting of Na2CO3, and the decrease may be due to the lower forming velocity of C2AS compared with other compounds. C2S exists as the form of ��-C2S when the temperature is above 800 ��C and then transforms to ��-C2S with the decrease of temperature. The Na2O-doped clinkers contain large amounts of ��-C2S, which indicates that Na2O restrains the transformation of ��-C2S. Moreover, the decrease of the intensity of the characteristic peak of ��-C2S also suggests the reducing of ��-C2S content. The intensities of the characteristic peaks at 2�� values of 18.23�� and 21.10�� increase as the N/A ratio increases from 0 to 0.24. The results suggest the increase of the C12A7 and Na4Ca3(AlO2)10 contents. On the contrary, the content of CA decreases with the increase of Na2O content in clinkers. CA disappears as the N/A ratio increases to 0.17. Moreover, the results are in accordance with the phase diagram, which suggests that the zone of C2AS gradually decreases while the zone of Na4Ca3(AlO2)10 increases with the N/A ratio increasing from 0.17 (Fig. 3(a)) to 0.33 (Fig. 3(b) [15]). No CA exists in the system when the N/A ratio is equal to 0.33.
Table 2 Intensities of peaks of phases in Na2O-doped CaO-Al2O3-SiO2 system clinkers
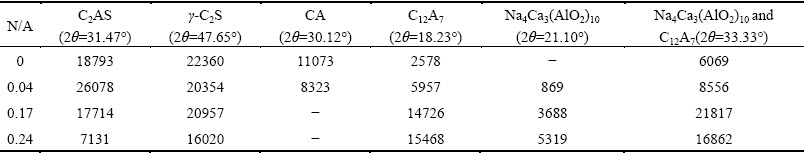
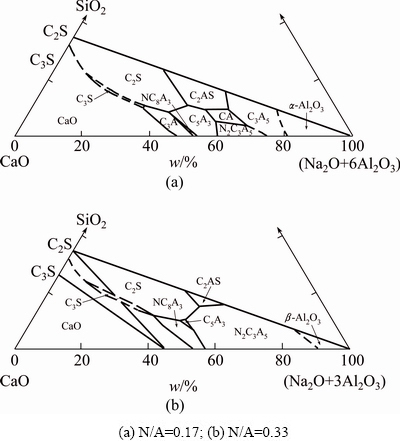
Fig. 3 Na2O-CaO-Al2O3-SiO2 quarternary system phase diagram [15]
The characteristic peak at 2�� value of 33.33�� is the peak of C12A7 when the N/A ratio is 0 and then becomes a mixed peak of C12A7, Na4Ca3(AlO2)10 and NA when the clinkers are doped with Na2O. Small amount of NA exists in the clinkers, so the contribution of CA to the intensity of the mixed peak can be ignored. The crystal faces of C12A7 represented by the characteristic peaks at 2�� values of 18.23�� and 33.33�� are (211) and (420), respectively. The intensity of the characteristic peak of C12A7 at 2�� value of 33.33�� is stronger than that of the characteristic peak of C12A7 at 2�� value of 18.23�� when the N/A ratio is 0. The intensity of the characteristic peak at 2�� value of 33.33�� decreases from 21817 to 16862, while the intensity of the characteristic peak at 2�� value of 18.23�� increases from 14726 to 15468 when the N/A ratio increases from 0.17 to 0.24. The results suggest that the C12A7 formed by the transformation of CA prefers to growing in the crystal face (211).
3.2 Microstructures of clinkers
As shown in Fig. 4, the representative microstructure of the clinker, when the N/A ratio is 0.04, displays three gray scales: the dark gray, the medium gray, and the light gray. The compositions of the micro-regions of Points A, B and C are listed in Table 3. The molar ratio of CaO to SiO2 in the micro-region of Point A is close to 2, indicating that the light gray regions stand for C2S. The molar ratio of CaO to Al2O3 at Point B is close to 1.71. So, the dark area is the distribution area of C12A7. The Na2O content in C12A7 is 4.33% (mass fraction). The chemical formula can be written as 0.93Na2O��10CaO��7Al2O3. At Point C where CaO is divided into two parts, the molar ratios of CaO to Al2O3 and CaO to SiO2 are close to 1 and 2, respectively. Therefore, the region of Point C consists of CA and C2S and the medium gray region stands for CA. Na2O is mainly doped in C12A7 since the Na2O content in the region of C12A7 is the highest. The analysis results of Ca and O elements in different areas do not show significant differences as the Na, Al and Si elements do.

Fig. 4 BSE images of sintered clinkers at N/A ratio of 0.04 and C/A ratio of 0.88
Table 3 Mole fractions of elements in clinkers determined by EDS (%)

Map scanning results are shown in Fig. 5. The high light areas in Figs. 5(b)-(f) are the main distribution areas of Na, Al, Si, Ca and O elements, respectively. The high light area of the picture is the main distribution area of the element. As shown in Fig. 5(b), it is obvious that Na2O mainly exists in the dark gray area, which is the distribution area of C12A7. Figure 5(d) shows the scopes of C2S and C12A7 clearly. According to Figs. 5(e) and (f), the distributions of O and Ca do not show significant differences, which is consistent with the analysis results of EDS.

Fig. 5 SEM image (a) and scanning maps (b-f) of clinkers at N/A ratio of 0.04 and C/A ratio of 0.88

Fig. 6 BSE images of sintered clinkers at N/A ratio of 0.24 and C/A ratio of 0.87 in different zones
Table 4 Mole fractions of elements in clinkers determined by EDS

BSE images of clinkers at N/A ratio of 0.24 are shown in Fig. 6. The analysis results of EDS are given in Table 4. It can be clearly seen that the phases distribute in different regions by mole fraction of elements. The lightest area is the distribution area of C2S. It can be calculated that the Na2O content doped in C2S is about 1.97%. The regions represented by Points E and F are the distribution areas of C12A7 and the mass fractions of Na2O doped in C12A7 at Points E and F are about 4.40% and 4.45%, respectively. So, the chemical formulae can be written as 0.92Na2O��9.37CaO��7Al2O3 and 0.94Na2O��9.61CaO��7Al2O3, respectively. The Na2O content doped in C12A7 increases slightly from 4.33% to 4.45% when the N/A ratio is increased from 0.04 to 0.24. According to analysis results of Point G, the darker area at the edge of C2S represents the region of CA. Trace amount of CA is formed when the N/A ratio of clinker is 0.24 and the content of Na2O in the CA is about 1.43%. The results of element analysis of Points H and I confirm the existence of NA and Na4Ca3(AlO2)10. Moreover, NA and Na4Ca3(AlO2)10 are distributed in the same micro-regions.
The crystal system of CA is monoclinic and the space group is P21/n. The crystal system of C12A7 is cubic and the space group is I-43d. MIZUKAMI et al [16], SUSHKO et al [17] and HOSONO et al [18] reported that the C12A7 cell was constituted by sub-nanometer size cages, which was approximately 0.4 nm in diameter. Each C12A7 cell consisted of two molecules with twelve cages. Two free oxygen ions existed in the twelve cages. The free oxygen ion can be substituted by the hydroxide ion, chloride ion and fluoride ion without changing the crystal structure [19-23]. The radii of Ca2+ and Na+ ions are 0.130 and 0.102 nm, respectively. The radii of OH-, Cl- and F- ions are 0.137, 181 and 133 nm, respectively. They are all larger than the radius of Na+ ion. Therefore, Na+ ion may replace Ca2+ ion or the free oxygen ion of C12A7 without changing the crystal system and space group. The Ca2+ and O2- ions of C12A7 cells are not distributed symmetrically, while those of CA cells are distributed symmetrically. Therefore, it is easier for polar molecules such as H2O and Na2O to penetrate the cells or react with the cells.
The crystallinities of phases are calculated through fitting the spectra using MDI Jade, which are shown in Fig. 7. Due to the doping of Na2O in the crystal structures, the crystallinities of C12A7 and ��-C2S decrease with the rise of Na2O content in clinkers. The crystallinity of Na4Ca3(AlO2)10 decreases with the increase of Na2O content in clinkers. The reason may be the coexistence of NA and Na4Ca3(AlO2)10 and the unbalanced distribution of Na2O between NA and Na4Ca3(AlO2)10. On the contrary, the crystallinity of CA increases slightly from 96.87% to 97.72% when the N/A ratio increases from 0 to 0.04. This is due to the fact that the ions in the defects of CA crystal structure have higher activity. Thus, the ions are easier to react with Na2CO3, which reduces the defects of CA grains. The increase of the crystallinity of CA also shows that it is difficult for Na2O to insert the cells of CA. With the doping of Na2O in clinkers, the crystallinity of ��-C2S slightly decreases. Yet, the crystallinity of ��-C2S rises slightly with the increase of Na2O content in clinkers. However, the crystallinity of ��-C2S in different clinkers is around 96.5%.
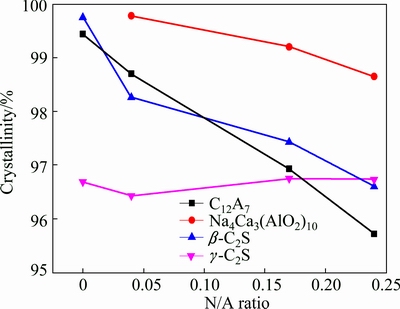
Fig. 7 Crystallinities of phases in clinkers at C/A ratio of about 0.85
3.3 Simulation of Na2O-doped C12A7 crystal
The analysis results of EDS show that Na2O mainly coexists with C12A7 compared with other compounds. Material Studio was used to analyze the effect of Na2O addition on the C12A7 cell. The setting values of the parameters during the optimization process of the crystal structures of C12A7 cells with or without Na2O doping are the same to ensure the comparability of the results. As shown in Table 5, the convergence standards during the calculation process are also the same with each other. The crystal structure data illustrated in Table 6 are derived from the ICSD.
Table 5 Parameter settings for optimization of crystal structure of C12A7 cells during calculation

The primitive cell of C12A7, simulated by MS using the crystal structure data in Table 6, is shown in Fig. 8. The crystal structure of C12A7 has a high symmetry. Any Ca atom can be chosen to be replaced by Na atom. The results will be the same, since only one kind of Ca site exists in the structure. The cell, one Ca atom of which is substituted by one Na atom, is shown in Fig. 9.
Table 6 Crystal structure data and atomic coordinates of C12A7
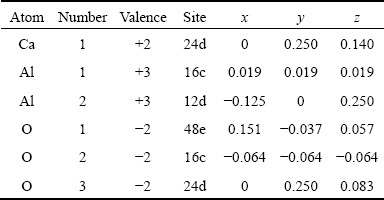
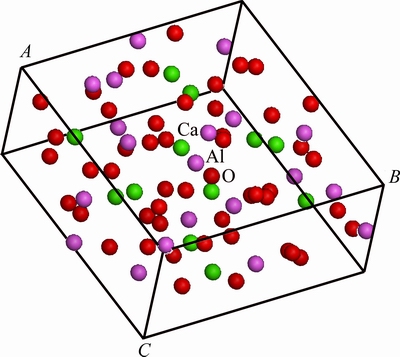
Fig. 8 Model of C12A7 unit cell

Fig. 9 Model of C12A7 unit cell when one Ca atom is replaced by one Na atom
The crystal structure of C12A7 mainly contains O��Al, O��Ca and O��O bonds. The bond length of O��Al is about 0.17 nm with the 3s and 3p orbital electrons of Al atom and the 2s, 2p orbital electrons of O atom forming the hybrid orbit. The bond length of O��Ca is about 0.24 nm with the 3s, 3p and 4s orbital electrons of Ca atom and the 2s, 2p orbital electrons of O atom forming the hybrid orbit. The bond population and length of C12A7 are presented in Table 7. The bond population is useful for evaluating the bonding character. A high value of the population of one chemical bond suggests that the bond is a covalent bond, whereas a low value suggests that the bond is an ionic bond [24,25]. As shown in Table 7, the population of the O��Al bond is much higher than that of the O��Ca bond, suggesting that the covalent character of the O��Al bond is stronger than that of the O��Ca bond. The O��O bonds are separated into two kinds, which show the covalent character and the ionic character, respectively. The bond of O��O with larger population is composed of the two free oxygen atoms in the cage-like structures. The computer cannot simulate the free oxygen atoms, so the free oxygen atoms may not form chemical bonds in the real materials.
Table 7 Bond population and length of C12A7

Table 8 Bond population and length of C12A7 doped with one Na atom
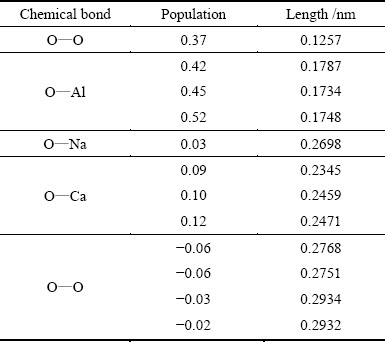
The bond population and length of C12A7 doped by one Na atom are shown in Table 8. The positions of the atoms in the crystals are changed in order to minimize the free energy of the cells and improve the stability of the cells during the simulation. The data in Table 8 suggest that the doping of Na atom does not significantly change the atomic distances and populations of O��Al and O��O bonds. Due to the replacement of Na atom, the species of O��Ca bond decrease from the original 4 to 3. As shown in Table 8, the population of O��Na bond is 0.03, which is lower than that of O��Ca bond. In addition, the O��Na bond has a larger atomic distance than O��Ca bond. Consequently, the covalency of the O��Na bond is weaker than that of the O��Ca bond.
The lattice parameter, the unit cell volume and the free energy of C12A7 unit cell and Na2O-doped C12A7 cell are illustrated in Table 9. The doping of the Na atom has no significant effect on the lattice parameters and cell volumes. The free energy of the unit cell is increased because of the Na2O addition. The free energy is the internal energy converted into other kind of energy during reactions. The simulation processes have been done under the hypothesis that the external environment conditions such as temperature and pressure are stable. Therefore, the free energy is the characteristic of the stability of the crystal structure. According to the principle of minimum energy, a lower free energy value corresponds to a higher activity. The increase of the free energy shows that the doping of Na atom can increase the chemical activity of C12A7, which benefits the leaching process of C12A7 in the sodium carbonate solution.
Table 9 Structural parameters of C12A7 and Na2O-doped C12A7

3.4 Leaching properties of sintered clinkers
CA, C12A7 and Na4Ca3(AlO2)10 can decompose in sodium carbonate solution. The processes are shown in Eqs. (3)-(5). Sodium hydroxide is also needed during the leaching process. The function of sodium hydroxide is to keep the stability of sodium aluminate in solution.
CaAl2O4+Na2CO3+4H2O=CaCO3��+2NaAl(OH)4 (3)
Ca12Al14O33+12Na2CO3+33H2O=12CaCO3��+14NaAl(OH)4+10NaOH (4)
Na4Ca3(AlO2)10+3Na2CO3+20H2O=3CaCO3��+10NaAl(OH)4 (5)
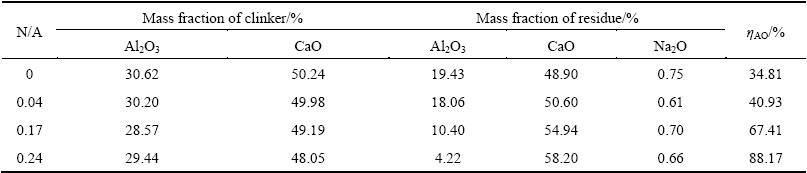
The leaching results of clinkers are listed in Tables 10 and 11. As given in Table 10, the leaching efficiency of CA clinkers containing small quantity of C12A7 is 86.28%. The leaching efficiency increases to 96.79% when the N/A ratio increases from 0 to 0.33. The leaching efficiency of pure Na4Ca3(AlO2)10 is 97.15%, which suggests that Na4Ca3(AlO2)10 has a better leaching performance than CA. Na4Ca3(AlO2)10 is easier to react with carbon sodium solution. Thus, the molecular polarity of Na4Ca3(AlO2)10 is higher than that of CA. As given in Table 11, the leaching efficiency of Al2O3 of the clinkers increases from 34.81% to 88.17% as the N/A ratio increases from 0 to 0.24. The C/A ratio of raw materials can be decreased to 0.85 when mixed with Na2O and the clinkers can also have proper leaching performance.
The leaching residues are almost washed to neutrality. The contents of Na2O in residues, as shown in Tables 10 and 11, are almost the same. Moreover, the Na2O content in residues of clinkers without Na2O is higher than that of clinkers doped with Na2O. Thus, most of Na2O in residues derives from the sodium carbonate or sodium hydroxide. Na2O in compounds almost all dissolves in solution. It can be concluded that the improvement of leaching property of clinkers is attributed to the increase of Na4Ca3(AlO2)10 and C12A7 contents in clinkers and the improvement of the chemical activity of Na2O-doped C12A7.
Table 10 Leaching results of clinkers of Na2O-doped CaO-Al2O3 system

Table 11 Leaching results of clinkers of Na2O-doped CaO-Al2O3-SiO2 system
4 Conclusions
1) The phases containing Na2O in clinkers of Na2O-doped CaO-Al2O3-SiO2 system mainly include Na4Ca3(AlO2)10 and Na2O��Al2O3. The rest of Na2O is mainly doped in C12A7. It is easier for Na2O to insert into the C12A7 cell than to insert into CA cell. The crystallinities of Na4Ca3(AlO2)10 and C12A7 decrease gradually with the increase of Na2O content in clinkers while that of CA increases slightly.
2) The doping of Na2O promotes the formation of C12A7 and Na4Ca3(AlO2)10 and prohibits the formation of C2AS. The formation processes of Na4Ca3(AlO2)10 and C12A7 can be divided into two ways, which are the direct reactions among raw materials and the transformation of CA, respectively.
3) The covalency of O��Al is the strongest in the C12A7 cells. The covalency of the O��Na bond is weaker than that of the O��Ca bond, which results in the increase of the free energy of C12A7 crystal structure and the decrease of the structural stability of C12A7.
4) C12A7 and Na4Ca3(AlO2)10 have better leaching properties than CA. The leachability of clinker is increased because of the formation of Na4Ca3(AlO2)10, the increasing amount of C12A7 and the doping of Na2O in C12A7 cells.
References
[1] YU Hai-yan, PAN Xiao-lin, WANG Bo, ZHANG Wu, SUN Hui-lan, BI Shi-wen. Effect of Na2O on formation of calcium aluminates in CaO-Al2O3-SiO2 system [J]. Transaction of Nonferrous Metals Society of China, 2012, 22(12): 3108-3112.
[2] WANG Bo, SUN Hui-lan, GUO Dong, ZHANG Xue-zheng. Effect of Na2O on alumina leaching property and phase transformation of MgO-containing calcium aluminate slags [J]. Transaction of Nonferrous Metals Society of China, 2011, 21(12): 2752-2757.
[3] OSTROWSKI C, ZELAZNY J. Solid solutions of calcium aluminates C3A, C12A7 and CA with sodium oxide [J]. Journal of Thermal Analysis and Calorimetry, 2004, 75(3): 867-885.
[4] TIAN Yong-pan, TU Gan-feng, PAN Xiao-lin, YU Hai-yan. Phase transformation and leaching performance of Na2O-doping calcium aluminate compounds [J]. Journal of the Chemical Industry and Engineering Society of China, 2015, 66(4): 1151-1156. (in Chinese)
[5] FUKUDA K, INOUE S, YOSHIDA H. Substitution of sodium and silicon in tricalcium aluminate [J]. Journal of the American Ceramic Society, 2003, 86(1): 112-114.
[6] KANG E T, LEE S J, HANNON A C. Molecular dynamics simulations of calcium aluminate glasses [J]. Journal of Non-Crystalline Solid, 2006, 352(8): 725-736.
[7] EUGENIE V U, MICHAEL C W, NORBERT J K, AYDIN A G. Glass-forming ability in calcium aluminate-based systems [J]. Journal of the American Ceramic Society, 1993, 76(2): 449-453.
[8] SHAHRIAR I, JEKABS G, GUNNAR S, JESPER L, TOBIAS J, GIANLUIGI A B, CARMEN M A, HAKAN E. Phase formation of CaAl2O4 from CaCO3-Al2O3 powder mixtures [J]. Journal of the European Ceramic Society, 2008, 28(4): 747-756.
[9] CHEN Guo-hua. Mechanical activation of calcium aluminate formation from CaCO3-Al2O3 mixtures [J]. Journal of Alloys and Compounds, 2006, 416(1): 279-283.
[10] YI H C,  J Y, MOORE J J, SCHOWENGERDT F D, ROBINSON L A, MANERBINO A R. Preparation of calcium aluminate matrix composites by combustion synthesis [J]. Journal of Materials Science, 2002, 37(21): 4537-4543.
J Y, MOORE J J, SCHOWENGERDT F D, ROBINSON L A, MANERBINO A R. Preparation of calcium aluminate matrix composites by combustion synthesis [J]. Journal of Materials Science, 2002, 37(21): 4537-4543.
[11] MARTIN R M. Electronic structure: Basic theory and practical methods [M]. London: Cambridge University Press, 2004: 566-569.
[12] BRIK M G, AVRAM N M, GRUIA A S. Calculation of the spectral, structural, and electronic properties of NaCrSi2O6 and LiCrSi2O6 crystals [J]. Optical Materials, 2013, 35(10): 1772-1775.
[13] WU Y Q, YOU J L, JIANG G C. Molecular dynamics study of the structure of calcium aluminate melts [J]. Journal of Inorganic Materials, 2003, 18(3): 619-626.
[14] PAN Xiao-lin, YU Hai-yan, TIAN Yong-pan, SU Hang, TU Gan-feng, BI Shi-wen. Computer simulation of crystal stability of 12CaO��7Al2O3 with calcium vacancy [J]. The Chinese Journal of Nonferrous Metals, 2014, 24(11): 2914-2920. (in Chinese)
[15] GREENE K T, BOGUE R H. Phase equilibrium relations in a portion of the system Na2O-CaO-Al2O3-SiO2 [J]. Journal of Research of the National Bureau of Standards (U.S.), 1946, 36(2) 185-207.
[16] MIZUKAMI F, CHATTERJEE A, NISHIOKA M. A periodic first principle study to design microporous crystal 12MO��7A12O3 for selective and active O-radicals encaging [J]. Chemical Physics Letters 2004, 390(4): 335-339.
[17] SUSHKO P V, SHLUGER A L, KATSURO H, MASAHIRO H, HIDEO H. Localisation assisted by the lattice relaxation and the optical absorption of extra-framework electrons in 12CaO��7A12O3 [J]. Materials Science and Engineering C, 2005, 25(5): 722-726.
[18] HOSONO H, KIM S W, MIYAKAWA M, MATSUISHI S, KAMIYA T. Thin film and bulk fabrication of room-temperature-stable electride C12A7:e utilizing reduced amorphous 12CaO��7A12O3(C12A7) [J]. Journal of Non-Crystalline Solids, 2008, 354(19): 2772-2776.
[19] PALACIOS L,  G, TORRE D L, BRUQUE S, JOSE L, SANTIAGO G G, SHEPTYAKOV D, MIGUEL A G. Crystal structures and in-situ formation study of mayenite electrides [J]. Inorganic Chemistry, 2007, 46(10): 4167-4176.
G, TORRE D L, BRUQUE S, JOSE L, SANTIAGO G G, SHEPTYAKOV D, MIGUEL A G. Crystal structures and in-situ formation study of mayenite electrides [J]. Inorganic Chemistry, 2007, 46(10): 4167-4176.
[20] NISHIOA Y, NOMURAB K, YANAGIA H, KAMIYA T, HIRANO M, HOSONO H. Short-channel nanowire transistor using a nanoporous crystal semiconductor 12CaO��7Al2O3 [J]. Materials Science and Engineering B, 2010, 173(1): 37-40.
[21] MIYAKAWA M, TODA Y, HAYASHI K, HIRANO M, KAMIYA T, MATSUNAMI N, HOSONO H. Formation of inorganic electride thin films via site-selective extrusion by energetic inert gas ions [J]. Journal of Applied Physics, 2005, 97(2): 023510.
[22] KIM S W, SHIMOYAMA T, HOSONO H. Solvated electrons in high-temperature melts and glasses of the room-temperature stable electride [Ca24Al28O64]4+��4e- applied [J]. Science, 2011, 333: 71-74.
[23] HAYASHI K, MATSUISHI S, KAMIYA T, HIRANO M, HOSONO H. Light-induced conversion of an insulating refractory oxide into a persistent electronic conductor [J]. Nature, 2002, 419: 462-465.
[24] MULLIKEN R S. Electronic population analysis on LCAO�CMO molecular wave functions: I [J]. Journal of Chemical Physics, 1955, 23: 1833-1840.
[25] KHADRAOUI Z, BOUZIDI C, HORCHANI-NAIFER K, FERID M. Crystal structure, energy band and optical properties of dysprosium monophosphate DyPO4 [J]. Journal of Alloys and Compounds, 2014, 617: 281-286.
�����ʣ������֣��ں��࣬Ϳ�ӷ�
������ѧ ұ��ѧԺ������ 110819
ժ Ҫ�����÷������Լ�CaCO3��Al2O3��SiO2��Na2CO3�ϳ�Na2O������������ϣ��о�Na2O���Ӷ�����ƻ������γɻ�����12CaO��7Al2O3(C12A7)����ṹ��Ӱ�졣�����������������Na2O��������4.26%(��������)ʱ����Na2O������ҪΪ2Na2O��3CaO��5Al2O3��Na2O��Al2O3������Na2O��Ҫ������12CaO��7Al2O3�ڣ�����ʹ��ᾧ�Ƚ��͡�2Na2O��3CaO��5Al2O3�Ľᾧ��Ҳ����������Na2O�������߶����͡�2Na2O��3CaO��5Al2O3��12CaO��7Al2O3������;�������ַ�ʽ��һ���ɳ�ʼ��Ӧ���ֱ�ӷ�Ӧ���ɣ������ɷ�Ӧ�м����CaO��Al2O3�Ľ�һ��ת����Ӧ���ɡ�����ṹģ������������Na2O���ӵ�12CaO��7Al2O3�����ڣ���O��Na���Ĺ���������O��Ca����O��Al���Ĺ����ԣ�ʹ12CaO��7Al2O3�������������ߣ���ѧ������ߡ���������Na2O������0������4.26%ʱ��Al2O3�Ľ�������34.81%������88.17%��
�ؼ��ʣ�����ƣ�Na2O���ӣ��γɻ���������ṹ���ս�����ģ��
(Edited by Wei-ping CHEN)
Foundation item: Projects (51174054, 51104041, 51374065) supported by the National Natural Science Foundation of China; Project (N130402010) supported by the Fundamental Research Funds for the Central Universities of China
Corresponding author: Xiao-lin PAN; Tel: +86-24-83686460; E-mail: panxl@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(16)64176-6

