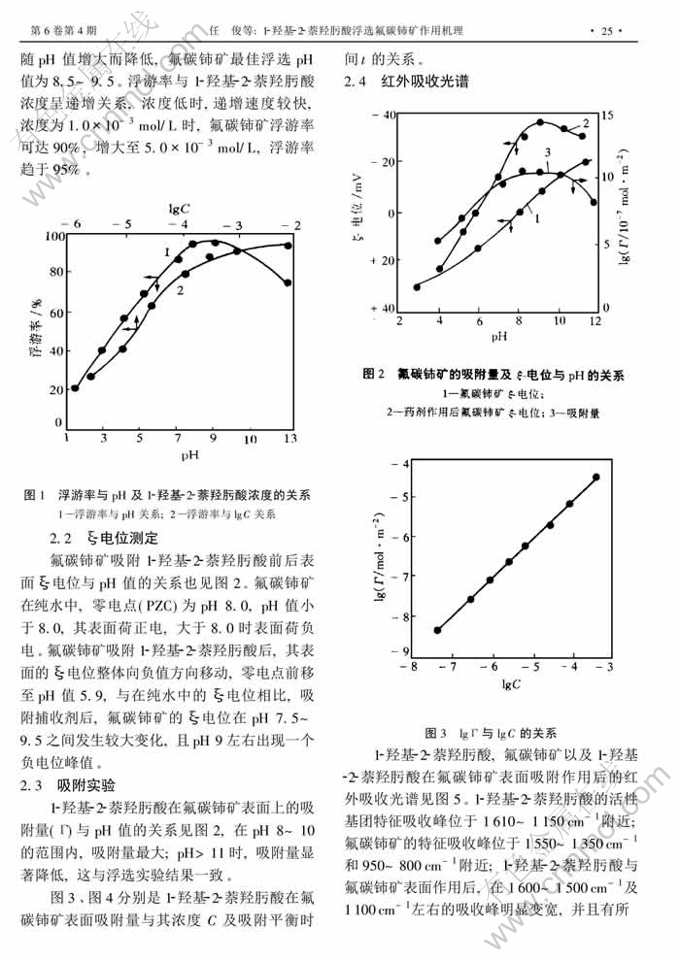
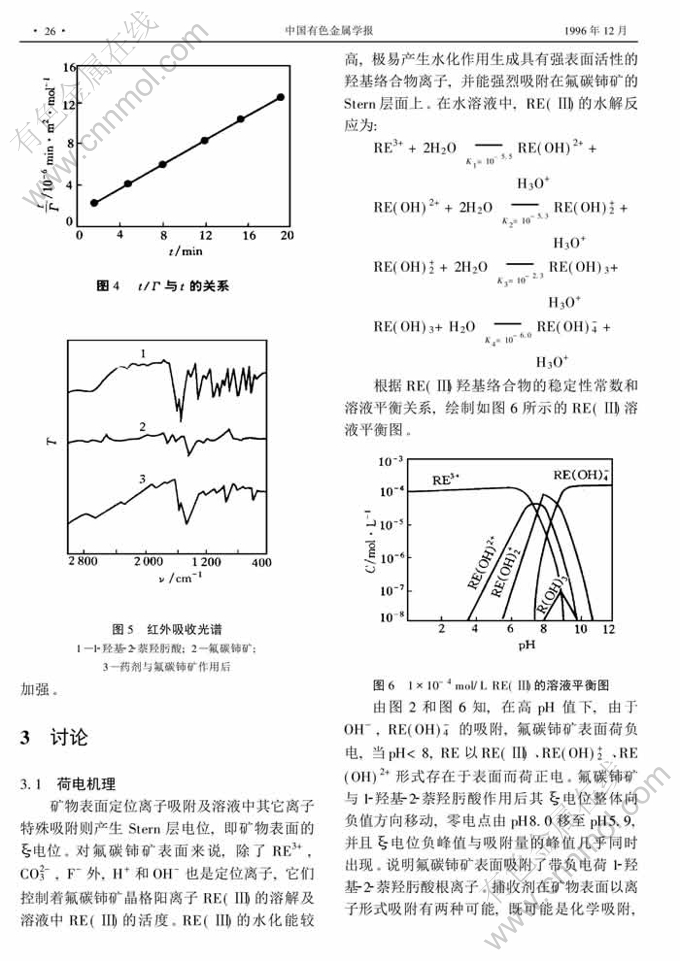
1-羟基-2-萘羟肟酸浮选氟碳铈矿作用机理①
来源期刊:中国有色金属学报1996年第4期
论文作者:任俊 卢寿慈 池汝安
文章页码:27 - 31
关键词:1-羟基-2-萘羟肟酸; 氟碳铈矿; 浮选; 电负性; 螯合特性
Key words:1-hydroxy-2-naphthyl hydroxamic acid; flotation; bastnaesite
摘 要:通过ξ-电位、吸附作用规律、药剂基团电负性理论及红外吸收光谱法论述了1-羟基-2-萘羟肟酸对氟碳铈矿的捕收性能及作用机理。1-羟基-2-萘羟肟酸主要是以羟肟基上的两个氧原子与氟碳铈矿表面的Re(Ⅱ)形成了O-C=N-O-Re(Ⅲ)-O五元环的螯合物而化学吸附,同时兼有多层的、不均匀的物理吸附,吸附方程Γ= 6.76×10-2・C1/1.02,吸附速率常数K = 2.64 × 105m2・mol-1・min-1。
Abstract: The collecting behaviour of bastnaesite with 1-hydroxy-2-naphthyl hydroxamic acid was investigated and its mechanism was discussed byξ-potential measurement, law of adsorption, means of theoretical calculation on the group electronegativity and method of infrared absorption spectrum. The result showed that two oxygen atoms from 1-hydroxy-2-naphthyl hydroxamic acid chelate RE(Ⅲ) on the surface of bastnaesite forms five-chain O―C=N―O―RE(Ⅲ)―O chelate which is chemically adsorbed on the surface of bastnaesite, together with the polylaminate. The nonhomogeneous physical adsorption equation is Г=6.76×10-2・C1/1.02. The constant of adsorption velocity is K=2.64×105m2・mol-1・min-1.