
Hemolysis effect and calcium-phosphate precipitation of
heat-organic-film treated magnesium
GAO Jia-cheng(高家诚), QIAO Li-ying(乔丽英), LI Long-chuan(李龙川), WANG Yong(王 勇)
College of Materials Science and Engineering, Chongqing University, Chongqing 400030, China
Received 8 August 2005; accepted 22 December 2005
Abstract: A heat-organic-films process was employed to induce calcium-phosphate apatites formation on magnesium, consequently the corrosion resistance and hemolysis properties of magnesium were improved for biomedical applications. Firstly, magnesium samples were heat-treated at 773 K for 10 h; secondly, stearic acid films were coated on the surface of the heat-treated magnesium. Then the surface modified magnesium was soaked in simulated body fluid (SBF) to test its corrosion resistance. The results show that the heat treatment process allows magnesium to form a dense oxide layer with a thickness of around 20 μm, thereby the surface modified magnesium has higher corrosion resistance. After 24 h in SBF island apatite was deposited on magnesium. The unevenly precipitates were characterized by XRD and FTIR as the mixture of hydroxyapatite(HA) and octacalcium phosphate(OCP). The preliminary hemolysis experiment indicates that untreated magnesium has hemolytic effect (about 60%); whereas the heat-organic film treated samples has no hemolytic effect. The mechanism of fast nucleation and growth of calcium-phosphate apatites on surface modified magnesium in SBF was also discussed.
Key words: magnesium; biomaterials; heat-organic films; corrosion resistance; hemolysis
1 Introduction
Various kinds of metal-based biomaterials have been widely used in clinic. However the application of NiTi and 316L is limited by the potential health hazard owing to the release of Ni and Cr ions[1]. Titanium alloy, which has extensively served as substitute for human hard tissues, was reported to cause metallic allergy[2] and stress shielding effects[3]. Thus there is an interest to develop a new kind of metal-based biomaterial whose mechanical properties are more similar to human hard organization. In fact, magnesium and its alloys exhibit many attractive properties like no toxicity in digestion system, the elastic modulus and the density similar to those of human bone, magnesium is also a needful element for body, and its content is the second in positive ion of human cell. These make them possible to be a new kind of metallic implants. However, the corrosion resistance of magnesium is poor, especially in some aggressive environments like physiological conditions. That greatly limits the possibility of using magnesium in biomaterials fields.
Recently, there is an attractive strategy to deposit inorganic phases on the metal with surfactant monolayers. This biomimetic approach is a complex process that involves the controlled nucleation and growth of inorganic phases through the functional sites on the films. Studies of biomimetic synthesizing calcium-phosphate coatings on titanium implants have been reported[4, 5]. Studies of biomineralization on magnesium, however, are lacking. On the other hand, to investigate the possibility of using magnesium as implants, biocompa- tibility tests on magnesium, such as hemocompatibility tests, are necessary. In this communication, the surface modification of pure magnesium through heat treatment and organic films is described first, then the preliminary hemolysis tests on treated and untreated magnesium samples are examined. In the end, the mechanism of precipitating apatite coatings on the treated samples is hypothesized.
2 Experimental
2.1 Heat organic film treatment
Magnesium with the purity of 99.99% was cut into rectangular specimens with the size of 20 mm×10 mm×2 mm. Then the specimens were abraded by waterproof abrasive papers, and then they were ultrasonically cleaned for 10 min in acetone and 10 min in distilled water respectively.
The cleaned and polished samples were heat-treated at 773 K for 10 h, then the heat-treated samples were soaked into beakers that contained 100 mL 0.1 mmol/L stearic acid (CH3(CH2)16COOH)/chloroform mixture for 1h, the samples were then taken out of the beakers through careful operation to make the chloroform completely evaporate. All reagents used in the experiments were analytical grade and the experiments were carried out in triplicate.
2.2 Soaking experiments in SBF solution
The specimens processed by the methods above were soaked in SBF solution at 37±0.5 ℃. The compositions of SBF solution are listed in Table 1. All reagents were analytical grade. As soon as the soaking experiments were finished, the specimens were taken out for morphological and structural examination.
Table 1 Ion concentrations of simulated body fluid(SBF) (mmol/L)

2.3 Preliminary study of hemolysis tests
The samples were divided into three groups, namely untreated, heat-treated and heat-organic films-treated magnesium samples, for hemolysis test. All the samples were sterilized for 5 min by ultraviolet germicidal lamp. The extraction preparation was performed according to ISO 10993―12[6], i.e. 3 cm2 of material/mL of extrac- tion liquid and incubating at 37 ℃ for 72 h. The extraction liquid was phosphate buffered saline(PBS).
Hemolysis test was performed according to the standard of ISO 10993―4[7]. Human venous blood donated by non-smokers was anticoagulated with potassium oxalate (2% w/v), diluted 1∶5 with PBS and centrifuged for 5 min at 700g at room temperature. The supernatant was discarded and the erythrocytes were diluted 1∶9 with PBS, then 5 mL of suspension was incubated with 4 mL of the extracts or with the blank (PBS) at 37 ℃ for 4 h without any agitation. The positive control was 5 mL of distilled water with 4 mL PBS. The negative control was 5 mL of suspension with 4 mL of the extracts of Ti6Al4V. After the incubation, the suspensions were centrifuged at 700g for 5 min. Then a 1 mL supernatant sample from each kind of extractions was withdrawn and the absorbance at 540 nm was determined using a spectrophotometer. The hemolysis ratio(HR) can be obtained through the following equation:
HR=(Dt-Dnc)/(Dpc-Dnc)
where Dt is the absorbance of the samples, Dpc and Dnc are the absorbance of the positive and negative reference respectively.
2.4 Surface characterization
A scanning electron microscope (Amray-X2000, 20 kV, XL30-TMP, 20 kV) equipped with energy dispersive spectroscope(EDS) was used to observe the micro- structure and morphology of the surface of treated samples at various time intervals. The phases of the samples which had been soaked in SBF were examined with X-ray diffraction analyzer. The samples at various processing steps were analyzed by Fourier Transform Infrared (FTIR, Perkin-Elmer, Spectrum GX) spectrum with auto image FTIR microscope. Photoelectrical scale and pH meter were employed to monitor the mass change of the untreated and treated samples and pH variation of SBF solution. The hemolysis result was conducted by the analysis of variance (ANOVA). P<0.003 was considered statistically significant.
3 Results and discussion
3.1 Heat treatment
It has been reported that heat treatment technique on magnesium can effectively improve the corrosion resistance in saline[8]. The effectiveness of the heat treatment may be attributed to the formation of corrosion resistant MgO layer on the surface of the samples. Figs.1(a) and (b) show the cross sectional morphologies of samples after they were heat-treated at 773 K for 10 h. It can be observed that an oxide layer with the thickness of 20 μm was formed after heat treatment. No obvious boundary crack between the surface oxide layer and the substrate is found. EDS analysis shows that the O atomic concentration in the surface layer is around two times as high as that in the substrate, which coincides with our hypothesis. That may explain the phenomena that in our experiments, the treated samples did not suffer from any mass loss while untreated magnesium samples almost completely dissolved when these two groups of samples were soaked in SBF for 14 d(shown in Fig.6).
3.2 Heat-organic films treatment
Fig.2 shows the morphological features of magnesium samples after they were heat-organic films-treated. The island-like precipitates are formed on the treated samples after they were immersed in SBF solution for 24 h. At the high resolution, cracks can be observed on the precipitates, which probably result from the release of hydrogen bubbles as magnesium reacted with the aqueous solution. The XRD pattern (Fig.3) identifies that the apatite is the mixture of OCP and HA. MgO phase is also detected on the samples by EDS analysis (Fig.1), which indicates that MgO may play a key role in the corrosion resistance.
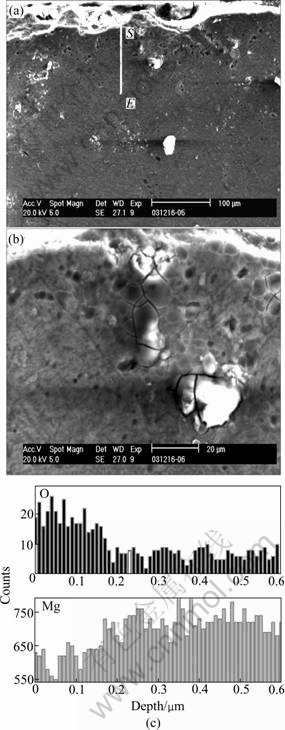
Fig.1 SEM morphologies of heat-treated samples at 773 K for 10 h ((a) and (b)) and EDS analysis of cross section of heat- treated samples(c)
Fig.4 shows the FTIR spectra of samples subject to various steps of surface modification. The absorption bands at 2 360 cm-1 can be attributed to CO2. For curve b in Fig.4, the presence of broad absorption band at characteristic stretching bands of COO- at 1 520 cm-1 and 794 cm-1 indicates a successful introduction of COO- functional groups on magnesium samples. The curve c in Fig.4 shows the spectrum of treated samples after immersed in SBF for 24 h. The absorption peak at 1 110 cm-1 arises from the asymmetric stretch of the phosphate group[9,10]. The O―H stretch at 3 696 cm-1 is also observed. In addition, a broad absorption peak attributed to the hydrogen bond appears around 3 380 cm-1. The existence of the characteristic band of  (862 cm-1) signals the presence of a calcium- deficient apatite. The presence of
(862 cm-1) signals the presence of a calcium- deficient apatite. The presence of  group band at 1 470 cm-1 indicates that the apatite formed contains carbonate-containing apatite, similar to minerals formed
group band at 1 470 cm-1 indicates that the apatite formed contains carbonate-containing apatite, similar to minerals formed
in bone[11]. The spectra of two random spots on A and B (Fig.5) further reveal that calcium and phosphate apatite do not evenly deposit on the surface of samples.

Fig.2 Surface morphological features of heat-organic films- treated samples under low(a) and high(b) resolution

Fig.3 XRD spectrum of heat-organic films-treated samples soaked in SBF for 24 h

Fig.4 FTIR spectra of samples: (a) Heat-treated; (b) Heat- organic films-treated; (c) First heat-organic films-treated and then soaked in SBF for 24 h
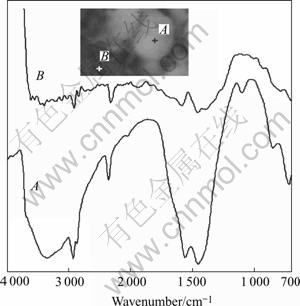
Fig.5 FTIR spectra with microscope of samples after soaked in SBF for 24 h
3.3 Hemolysis test results
The in vitro hemocompatibility tests are indispensable to evaluate the biomaterials contacting blood. Magnesium ion has been proved one of the most important bivalent ions associated with the formation of biological apatites. It also plays an important role in the changes in bone matrix that determines bone fragility[12]. The study of in vitro cell toxicity of magnesium on marrow cells has been carried in our lab and the results have been released elsewhere. However, literatures about hemocompatibility on magnesium have not been seen. In our preliminary hemolysis study, we have chosen the indirect method, which consists of the incubation of erythrocyte suspension with the extract of the test materials. The use of extract, permitting assay condition more easily standardizable, can evaluate the hemolysis induced by the chemicals that are released by the test material into the extraction medium. In our tests, the untreated magnesium extract has obvious hemolytic effect on the human red cell suspension as compared with blank (PBS) and negative controls (Table 2). While the heat-treated samples’ extract shows slight hemolytic effect, the extraction of heat-organic films-treated magnesium causes no hemolysis. The very different hemolysis may be explained by the different corrosion resistance of the samples. In our extraction preparation experiment, the untreated samples are seriously corroded after soaked in PBS for 72 h and the extraction becomes gel-like solution, whereas the heat-organic films-treated samples have high corrosion resistance and there is no hemolytic effect. Therefore it seems that one reason for hemolysis is the dissolution of high concentration magnesium ions in PBS extraction.
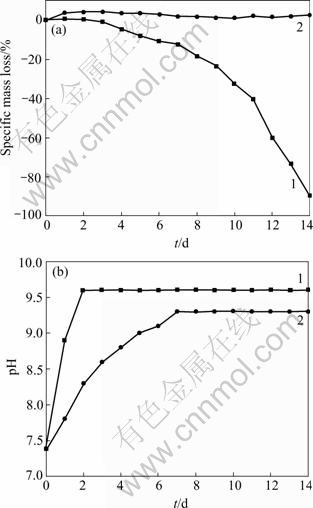
Fig.6 Specific mass loss and pH value of untreated samples(1) and heat-organic films-treated samples(2) after soaked in SBF for 14 d
Table 2 Results of hemolysis test

3.4 Mechanism of precipitating Ca-P based apatite on magnesium substrate in SBF
The inorganic-organic interfaces are postulated as lattice matching, hydrogen bonding, stereochemistry and electrostatics. In different model systems each parameter plays a potential role in controlling inorganic-organic interface nucleation and growth. LETELLIER et al[13] concluded that non-specific electrostatic interactions appeared to be the dominant factor. In our experiments, when the heat-organic films-treated samples are immersed in the SBF solution, the negatively charged COO- groups on stearic acid organic layers can function as“recognized sites”which bind with positively charged Ca2+ ions from the SBF solution, forming higher degree of supersaturation near the substrate and thus initiating Ca-P apatites nucleation on the substrate. We also found that apatites can only be detected on the heat-organic films-treated samples, whereas no Ca-P based precipitates are observed on untreated and heat-treated only samples during the immersion test in SBF. The result indicates that stearic acid layers with COO- functional groups play an important role in inducing calcium and phosphate ions precipitation. In our studies, the mixture of HA and OCP but not pure HA was obtained on the treated samples. Actually, it is difficult to obtain pure HA in aqueous solution under room temperature even though HA is thermodynamically preferred when the pH of the solution is higher than 4.2[14]. The formation of HA usually proceeds via a complex transformation from the precursor phase of amorphous calcium phosphate or tri-calcium phosphate, often through an intermediate OCP phase, in which  and
and  groups present.
groups present.
The lattice matching may also explain the fast precipitation of Ca-P apatite on samples in SBF. The ED pattern of stearic acid monolayers can be roughly defined a rectangular unit cell with lattice parameters of a= 49 nm, b=74 nm and an area per molecule of 1 760 nm2 or a bit larger[15]. From the schematic model shown in Fig.7, it can be seen that on the (001) face of HA, positively charged Ca2+ ions match well with the negatively charged COO- sites well. This structural frame may bind the calcium ions on the structural sites of stearic acid films, and accelerate the nucleation and growth of apatite in SBF.

Fig.7 Schematic model of interaction between stearic acid films and (001) face of HA
4 Conclusions
1) The corrosion resistance and biocompatibility of the heat-organic films-modified magnesium have a great improvement. There is little mass loss in SBF for 14 d.
2) An oxide layer with the thickness of 20 μm is formed after the samples are heat-treated at 773 K for 10 h. The oxide layer contains two times as much O element as the inner substrate.
3) HA and OCP on the samples are observed after the heat-stearic acid films modified magnesium is subsequently soaked in SBF over 24 h. The calcium- phosphate, however, is not evenly like island morpholo- gical features.
4) The hemolysis rate of magnesium is about 60%, whereas the hemolysis rate of heat-organic film treated magnesium is 0. The corrosion resistance of the samples may be one of the key reason for the hemolysis of magnesium.
5) Non-specific electrostatic interactions, together with beneficial lattice matching between the (001) face of HA and the sites on stearic acid films seem to be the main reason of precipitation of Ca-P apatites on heat-organic films-treated samples.
References
[1] Gurrappa I. Corrosion and its importance in selection of materials for biomedical applications [J]. Corrosion Prevention & Control, 2001, 1: 23-27.
[2] Al-Abdullat y, tsutsumil s, Nalajima n. Surface modification of magnesium by NaHCO3 and corrosion behavior in Hank’s solution for new biomaterial applications [J]. Materials Transaction, 2001, 42(8): 1777-1780.
[3] Rashmir-raven a m, Richardson d c, Aberman h m. The response of cancellous and cortical canine bone to hydroxylapatite-coated and uncoated titanium rods [J]. Journal of Applied Biomaterials, 1995, 6(4): 237-242.
[4] MAO C, LI H, CUI F. Oriented growth of hydroxyapatite on (001) textured titanium with functionalized self-assembled silane monolayer as template [J]. Journal of Materials Chemistry, 1998, 8: 2795-2801.
[5] Adriana b, Elisa b, Barbara b. Nanocrystalline hydroxyapatite coatings on titanium: a new fast biomimetic method [J]. Biomaterials, 2005, 26(19): 4085-4089.
[6] ISO 10993―12. Biological Evaluation of Medical Devices-Part 12: Sample Preparation and Reference Materials, 1996.
[7] ISO 10993―4. Biological Evaluation of Medical Devices-Part 4: Selection of Tests for Interactions with Blood, Annex D, 1993.
[8] Kuwahara h, Al-Abdullat y, Mazaki n. Precipitation of magnesium apatite on pure magnesium surface during immersing in Hank’s solution [J]. Materials Transactions, 2001(7): 1317-1321.
[9] Dasarathy h, Riley c, Coble h d. Analysis of apatite deposits on substrates [J]. Journal of Biomedical Materials Research, 1993, 27(4): 477-482.
[10] Shirkhanzdeh m. Direct formation of nanophase hydroxyapatite on catholically polarized electrodes [J]. Journal of Materials Science: Materials in Medicine, 1998, 9(2): 67-72.
[11] Sho j, Ding h, Wu y. Biomimetic apatite layers on plasma-sprayed titanium coatings after surface modification [J]. Surface and Coatings Technology, 2001, 137(1): 97-103.
[12] Koma s r, Leeb j h, Kimb y t. Synthesis of Si, Mg substituted hydroxyapatites and their sintering behaviors [J]. Biomaterials, 2003, 24(8): 1389-1398.
[13] Letellier r s, Lochhead m j, Campbell a a. Oriented growth of calcium oxalate monohydrate crystals beneath phospholipid monolayers [J]. Biochemical & Biophysical Act, 1998, 1380(1): 31-45.
[14] Klein c p, BLIEK-HOGERVOST J M, Wolke j g c. Studies of the solubility of different calcium phosphate ceramic particles in vitro [J]. Biomaterials, 1990, 11(7): 509-512.
[15] Lu h b, Ma c l, Cui h. Controlled crystallization of calcium phosphate under stearic acid monolayers [J]. Journal of Crystal Growth, 1995, 155(1-2): 120-125.
Foundation item: Prject(2004AA4003) supported by Chongqing Science and Technology Committee
Corresponding author: GAO Jia-cheng; Tel: +86-23-65127307; E-mail: gaojch@cqu.edu.cn
(Edited by YUAN Sai-qian)