Ni������Cu-0.4Be�Ͻ�ʱЧ��֯�����ܵ�Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2021���3��
�������ߣ������ ��ͬͬ ���� ��˫�� ���»� л����
����ҳ�룺679 - 691
Key words��Cu-Be alloy; alloying; phase transformation; microstructure; mechanical properties; electrical conductivity
ժ Ҫ���о�Ni����(0��2.10%����������)��Cu-0.4Be(����������%)�Ͻ�ʱЧ�����ࡢǿ�Ⱥ͵����ʵ�Ӱ�죬������Ӱ��������������������NiԪ�ؿɴٽ�ǿ������������������ʱЧǿ�����á���Ni������0����2.10%ʱ���Ͻ�ʱЧ���ǿ�ȳ���������С�ı仯���ƣ���Ni����Ϊ1.50%ʱ�ﵽ���1.50%Ni�Ͻ�ķ�ֵʱЧ����Ϊ400 ��C����60 min��ʱЧ��Ͻ�Ŀ���ǿ�Ⱥ�����ǿ�ȷֱ�Ϊ611 MPa��565 MPa���ֱ��Dz���Ni�Ͻ��2.8����6.1������Ni�Ͻ�ĵ���������ʱЧʱ����ӳ������ӣ�������Ni������������½�����400 ��C������ʱЧʱ����ӳ���Cu-0.4Be-1.5Ni�Ͻ����á�����á��������ת�䣻��ʱЧʱ��Ϊ60 minʱ���Ͻ��г��ִ�����ɢ�ֲ�����������á���ͦá��࣬����ǿ�������������ǺϽ���нϸ�ǿ�ȵ���Ҫԭ��
Abstract: The effects of Ni content (0-2.10 wt.%) on the precipitated phase, strength and electrical conductivity of Cu-0.4wt.%Be alloy were investigated, and the influencing mechanism was analyzed. The results showed that the addition of Ni promoted the precipitation of strengthening phase in the alloy and remarkably enhanced the strengthening effect. When the Ni content was increased from 0 to 2.10 wt.%, the strength of the aged alloy initially increased and then decreased, and approached the maximum when the Ni content was 1.50 wt.%. The peak-aging parameters of the alloy containing 1.50 wt.% Ni were the aging temperature of 400 ��C and the aging time of 60 min, where the tensile strength and yield strength of the aged alloy were 611 and 565 MPa, respectively, which were 2.8 times and 6.1 times those of the alloy without Ni. The electrical conductivity of the alloy with Ni increased with the aging time, and decreased with the increase of Ni content. With an increase of the aging time at 400 ��C, phase transition sequence of the Cu-0.4Be-1.5Ni alloy was �á� phase ���á� phase ���� phase. For the aging time of 60 min, a large number of dispersed nano-scale coherent �á� phase and �á� phase formed in the alloy with a remarkable strengthening effect, which was mainly responsible for the high strength of the alloy.
Trans. Nonferrous Met. Soc. China 31(2021) 679-691
Yan-bin JIANG1,2, Tong-tong ZHANG2, Yu LEI2, Shuang-jiang HE2, Xin-hua LIU2,3, Jian-xin XIE2,3
1. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
2. Key Laboratory for Advanced Materials Processing (Ministry of Education), University of Science and Technology Beijing, Beijing 100083, China;
3. Beijing Advanced Innovation Center for Materials Genome Engineering, University of Science and Technology Beijing, Beijing 100083, China
Received 30 April 2020; accepted 30 November 2020
Abstract: The effects of Ni content (0-2.10 wt.%) on the precipitated phase, strength and electrical conductivity of Cu-0.4wt.%Be alloy were investigated, and the influencing mechanism was analyzed. The results showed that the addition of Ni promoted the precipitation of strengthening phase in the alloy and remarkably enhanced the strengthening effect. When the Ni content was increased from 0 to 2.10 wt.%, the strength of the aged alloy initially increased and then decreased, and approached the maximum when the Ni content was 1.50 wt.%. The peak-aging parameters of the alloy containing 1.50 wt.% Ni were the aging temperature of 400 ��C and the aging time of 60 min, where the tensile strength and yield strength of the aged alloy were 611 and 565 MPa, respectively, which were 2.8 times and 6.1 times those of the alloy without Ni. The electrical conductivity of the alloy with Ni increased with the aging time, and decreased with the increase of Ni content. With an increase of the aging time at 400 ��C, phase transition sequence of the Cu-0.4Be-1.5Ni alloy was �á� phase ���á� phase ���� phase. For the aging time of 60 min, a large number of dispersed nano-scale coherent �á� phase and �á� phase formed in the alloy with a remarkable strengthening effect, which was mainly responsible for the high strength of the alloy.
Key words: Cu-Be alloy; alloying; phase transformation; microstructure; mechanical properties; electrical conductivity
1 Introduction
Cu-Be alloys are widely used in marine engineering, aerospace, new energy and other high-tech fields due to their excellent properties such as high strength, high electrical conductivity, high elasticity, fatigue resistance, non-magnetism and non-sparking when subjected to impact [1-6]. With the development of miniaturization, light- weight, high density and multifunction of electronic components, higher performances of Cu-Be alloy are required.
Cu-Be alloy is a typical precipitation- strengthening alloy. CuBe phase precipitated in Cu matrix can improve the mechanical properties of the alloy [7-9]. However, the rapid growth of Cu matrix grain and rapid precipitation of the CuBe phase in the Cu-Be alloy happen during heat treatment, and especially, the lamellar discontinuous phase is easy to form at grain boundary [10], which has an adverse effect on the mechanical properties of the alloy. Adding alloying elements (Ni, Co, Mg, etc) can improve the comprehensive properties of the alloy [11-16]. For example, Ni element is added with Be element in order to form BeNi compound, which can improve the precipitation-strengthening effect of Cu-Be alloy. PENG et al [17] studied the effect of precipitation on the strength and electrical conductivity of Cu-Be-Ni alloy aged at 525 ��C. It was found that a great number of dispersed �á� phase formed in the alloy during peak-aging, which strengthened the alloy remarkably and improved the electrical conductivity of the alloy. XIE et al [9] studied the precipitation behavior and properties of Cu-2.0wt.%Be-0.34wt.%Ni alloy during aging at 320 ��C, and the tensile strength and electrical conductivity of the alloy after peak-aging were 1230 MPa and 25% (IACS), respectively. On one hand, however, adding excessive Ni may induce coarsening and inhomogeneous distribution of the precipitate phase, which decreased the mechanical properties of the alloy. On the other hand, the increase of Ni content in the Cu matrix can enhance electron scattering extent, which results in the reduction of the electrical conductivity of the alloy. Therefore, reasonably controlling Ni content is an effective method to improve the mechanical properties and electrical conductivity of Cu-Be alloy.
Compared with high beryllium content copper alloy (Be: 1.6-2.1 wt.%), low beryllium content copper alloy (Be: 0.2-0.7 wt.%) has lower strength but higher electrical conductivity, and works as connector material, which is widely used in electronic components. In this work, the effects of Ni content on the microstructure, strength and electrical conductivity of Cu-0.4Be-xNi alloy and its influence mechanism were studied, which can provide a basis for controlling precipitation and improving the performance of Cu-0.4Be alloy by reasonably controlling Ni content.
2 Experimental
2.1 Material and heat treatment
The raw materials were electrolytically pure Cu (99.95 wt.%), electrolytically pure Ni (99.9 wt.%) and Cu-3.5wt.%Be master alloy. Ingots with different chemical compositions and diameter of 100 mm were prepared by vacuum medium-frequency induction melting with a melting temperature of 1250 ��C and pouring temperature of 1200 ��C. The composition of the alloy was tested by chemical analysis, as given in Table 1. Samples of 20 mm in thickness and 60 mm in width were cut from the alloy ingots and hot rolled at 850 ��C to 2 mm (hereinafter called as hot-rolled sample), and then cold rolled to 1 mm (hereinafter called as cold-rolled sample).
Table 1 Chemical compositions of Cu-0.4Be-xNi alloy (wt.%) [18]

The rolled samples were conducted by solid- solution treatment at 950 ��C followed by water- quenching, hereinafter called as solid-solution treatment. The above samples were then aged at 250, 300, 350, 350, 400, 425, 450, 475, 500 and 550 ��C for 60 min, respectively. The hardness of the aged samples was tested to determine the aging temperature TS corresponding to the peak hardness. The samples with different chemical compositions were aged at TS-25 ��C, TS and TS+25 ��C for 30, 60, 120, 180, 240, 480 and 960 min, respectively. The microstructure and property evolutions as well as reasonable aging parameters of the alloys were obtained by measuring the microstructure, strength and electrical conductivity of the samples.
2.2 Microstructure observation and property test
Tensile samples with different compositions and heat treatments were prepared according to GB/T 228��2010 standard. MTS material test machine was used to perform the tensile tests at room temperature under a strain rate of 0.05 mm/s, using samples with a gauge length of 50 mm. Three samples of each condition were tested and the average value of the properties was taken as the test result. The hardness of the sample with a size of 20 mm �� 20 mm after removal of surface oxidation was measured by HXD-1000T hardness tester with a load of 50 g and loading time of 10 s. Five points were measured for each sample and the average value was used as the test result.
Samples of 50 mm in length, 3 mm in width and 1 mm in thickness were cut from the aged alloys. The electrical resistance was measured by Amber Applent AT-510 precision DC resistance tester. Three samples were tested for each group and the electrical conductivity was calculated by the average value of three measurements.
Slices of 0.5 mm in thickness were cut from the aged alloys and then ground to 30-50 ��m. The samples for transmission electron microscopy (TEM) observation were prepared by using MTP-1A magnetically driven double-jet electrolytic thinner with the electrolyte containing 70 vol.% methanol and 30 vol.% nitric acid at -30 ��C and electric current of 45 mA. The amount, morphology and distribution of the precipitated phase in the samples were observed by FEI Tecnai F20 transmission electron microscope. The size of precipitated phase was obtained from five TEM images taken from different areas under the same magnification.
3 Results and discussion
3.1 Effects of Ni content on hardness and strength of Cu-0.4Be alloy
The Cu-Be alloys with different Ni contents after solid-solution treatment were aged at 250, 300, 350, 375, 400, 425, 450, 475, 500 and 550 ��C for 60 min, respectively. The hardness of the aged alloys was tested to determine the appropriate aging temperature range. Figure 1 shows the hardness curves of the aged samples with the aging temperature. With increasing the aging temperature, the hardness of alloy increased initially and then declined. The hardness of the alloy without Ni after solid-solution treatment was HV 86. When the aging temperature was raised to 300 ��C, the hardness was increased to HV 115. With further raising the aging temperature, the hardness reduced slightly. With an increase of Ni content, the hardness of the aged alloy increased initially and then fell. When the Ni content was 1.50 wt.%, the hardness of the alloy was the maximum. The aging temperature corresponding to the peak hardness of the alloy with the Ni content of 0.45, 0.98, 1.50 and 2.10 wt.% was 425, 425, 400 and 425 ��C, respectively, and the peak hardness was HV 239, 345, 383 and 344, respectively, which was much higher than that of the alloy without Ni. It is indicated that the addition of Ni element had a remarkable strengthening effect in the Cu-Be alloy and the maximum strengthening effect was achieved for the Ni content of 1.50 wt.%.
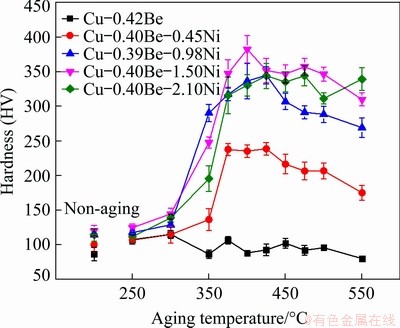
Fig. 1 Hardness curves of Cu-0.4Be alloy with different Ni contents changing with aging temperature (aging time of 1 h)
For purpose of further obtaining the optimal aging parameters of the alloy, heat treatment was carried out at 375, 400 and 425 ��C for different aging time, respectively. Figure 2 shows the hardness curves of the alloys with the aging time. The hardness of the alloy without Ni changed slightly with the aging time at 375, 400 and 425 ��C, and was HV 80-110, indicating a low aged strengthening effect. With prolonging the aging time, the hardness of the alloy containing Ni initially increased to a maximum value and then slightly decreased. Taking the alloy with 1.50 wt.% Ni aged at 400 ��C for an example, when the aging time was 30 min, the hardness of the alloy increased from HV 120 of the solid-solution state to HV 315. With prolonging the aging time, the increment of the hardness decreased, and the peak value (HV 383) of the hardness was achieved for the aging time of 1 h. When the aging time was 8 h, the hardness of the alloy was HV 300. In addition, for the alloy with the same Ni content, the aging time required to reach the peak hardness decreased with an increase of the aging temperature.
The hardness of the alloys with different Ni contents reached the peak value after aging at 400 ��C for 60 min. Therefore, the tensile strength and yield strength of the alloy with different Ni contents after aging at 400 ��C for different aging time were tested at room temperature, as shown in Fig. 3.
From Fig. 3, for the alloy with Ni addition, the tensile strength and yield strength changed slightly with an increase of the aging time. When the aging time was 10 min, the tensile strength and yield strength of the alloy were 220 and 92 MPa, respectively, and were decreased to 212 and 70 MPa when the aging time was increased to 480 min.

Fig. 2 Hardness curves of Cu-0.4Be alloy with different Ni contents changing with aging time

Fig. 3 Strength of Cu-0.4Be alloy with different Ni contents changing with aging time at 400 ��C
With an increase of the aging time, the tensile strength and yield strength of the alloys containing Ni increased initially and then decreased, which were similar to the change of the hardness with the aging time, and the yield strength decreased slightly after reaching the peak value. In addition, the tensile strength and yield strength of the alloy with the Ni content of 1.50 wt.% were the maximum after aging at 400 ��C for different aging time. For the alloy containing 1.50 wt.% Ni, when the aging time was increased from 10 to 60 min, the tensile strength and yield strength of the alloy increased rapidly from 403 and 297 MPa to the maximum values of 611 and 565 MPa, respectively. With further increasing the aging time, the tensile strength and yield strength of the alloy fell slowly. When the aging time was 480 min, the tensile strength and yield strength were reduced to 534 and 506 MPa, respectively.
The aging time for the peak strength of the alloys with Ni content of 0.45, 0.98, 1.50 and 2.10 wt.% after aging at 400 ��C was respectively 120, 60, 60 and 60 min, and the tensile strength was respectively 515, 575, 611 and 595 MPa, and the yield strength was respectively 384, 476, 565 and 560 MPa. Figure 4 shows the influence of the Ni content on the strength of the aged alloys. It can be seen that the tensile strength and yield strength of the alloy initially increased to the maximum and then decreased with the increase of the Ni content. The alloy with the Ni content of 1.50 wt.% had the maximum tensile strength and yield strength.

Fig. 4 Strength of Cu-0.4Be alloy changing with Ni content
3.2 Effect of Ni content on electrical conductivity of Cu-0.4Be alloy
Figure 5 displays the electrical conductivity of Cu-Be alloys changing with the aging time at 400 ��C. With an increase of the aging time, the electrical conductivity of the alloys with different Ni contents increased gradually. For the aging time of 10-60 min, the electrical conductivity rapidly increased with an increase of the aging time. With further prolonging the aging time, the increment of the electrical conductivity reduced. Taking the alloy with Ni content of 1.50 wt.% as an example, when the aging time was 60 min, the electrical conductivity increased rapidly from 30.5% (IACS) of the solid-solution state to 40.3% (IACS). The electrical conductivity increased to 47.4% (IACS) when the aging time was 480 min.

Fig. 5 Electrical conductivity of Cu-0.4Be alloy with different Ni contents changing with aging time at 400 ��C
Ni content had a significant effect on the electrical conductivity of Cu-Be alloy. Prior to aging, the electrical conductivity of the alloy with the Ni content of 0, 0.45, 0.98, 1.50 and 2.10 wt.% after solid-solution treatment was 41.0%, 37.1%, 33.5%, 30.5% and 29.8% (IACS), respectively. When the aging time exceeded 10 min, the electrical conductivity of the alloy decreased with the increase of the Ni content. Taking aging at 400 ��C for 60 min as an example, the electrical conductivity of the alloy with the Ni content of 0, 0.45, 0.98, 1.50 and 2.10 wt.% was 42.5%, 47.6%, 44.7%, 40.3% and 39.6% (IACS), respectively.
3.3 Effect of Ni content on microstructure of Cu-0.4Be alloy
The mechanical and electrical properties of Cu-Be alloy were closely related to the phase precipitation behavior during aging. From the experimental results in Section 3.1 and Section 3.2, it was indicated that both the strength and electrical conductivity of the aged alloys containing Ni changed similarly with the aging time, and the alloy with 1.50 wt.% Ni after aging at 400 ��C had the maximum strength. In order to analyze the morphology, the number and distribution of precipitated phases in the alloys during aging, taking the alloy with 1.50 wt.% Ni (hereinafter called as Cu-0.4Be-1.50Ni) as an example, the microstructures of the alloy treated by under-aging, peak-aging and over-aging were observed by TEM, as shown in Fig. 6, Fig. 7 and Fig. 8, respectively.
After aging at 400 ��C for 10 min, a large number of ��" phase and ��' phase diffusively formed in the alloy, and the ��' phase was disk-shaped with a diameter of about 3 nm and a thickness (0.1-0.3 nm) of 1-2 atomic layers, as shown in Fig. 6. The bright field image of ��' phase showed no-contrast strain lines parallel to  plane in the matrix, indicating the coherent relationship between the precipitated phase and the matrix [19].
plane in the matrix, indicating the coherent relationship between the precipitated phase and the matrix [19].

Fig. 6 TEM images and diffraction pattern of Cu-0.4Be-1.50Ni alloy aged at 400 ��C for 10 min

Fig. 7 TEM images (a, b, c) and diffraction pattern (d) of Cu-0.4Be-1.50Ni alloy aged at 400 ��C for 60 min
When the aging time was 60 min, the numbers of the dispersed ��" phase and ��' phase increased remarkably, a part of ��" phase transformed into disk-like ��' phase, and the ��' phase grew up slightly, as shown in Fig. 7(a). From the HRTEM image in Fig. 7(b), the diameter of the disk-like ��' phase was about 3.5 nm and the thickness was 0.4-0.6 nm (2-3 atomic layers). Figure 7(d) shows the selected area electron diffraction pattern (SADP) of Fig. 7(a) along [011]�� zone axis. It was found that the diffraction bands along  were perpendicular to the non-contrast strain line of the ��' phase, and these diffraction bands were formed by the ordered arrangement of Be and Ni atoms in the ��' phase on (001)�� plane of the Cu matrix, which was in agreement with the report by RYOICHI et al [20]. Meanwhile, the light-dark fringes parallel to
were perpendicular to the non-contrast strain line of the ��' phase, and these diffraction bands were formed by the ordered arrangement of Be and Ni atoms in the ��' phase on (001)�� plane of the Cu matrix, which was in agreement with the report by RYOICHI et al [20]. Meanwhile, the light-dark fringes parallel to  trace appeared around the ��' phase (seen in Fig. 7(c)). These fringes can be considered as the elastic shear strain region of the coherent ��' phase [21], indicating that large lattice distortion occurred around the ��' phase, which had a great strengthening effect.
trace appeared around the ��' phase (seen in Fig. 7(c)). These fringes can be considered as the elastic shear strain region of the coherent ��' phase [21], indicating that large lattice distortion occurred around the ��' phase, which had a great strengthening effect.

Fig. 8 TEM images (a, b, c) and diffraction pattern (d) of Cu-0.4Be-1.50Ni alloy aged at 400 ��C for 480 min
When the aging time was increased to 480 min, the morphology, size and distribution of precipitated phase in the alloy changed greatly, as shown in Fig. 8. The alloy mainly consisted of numerous coherent ��' phase and incoherent �� phase, and most of the ��" phase transformed into disk-like ��' phase with a diameter of about 6 nm and a thickness of 0.8-1 nm, which were parallel- distributed in the Cu matrix, i.e., the length direction of the ��' phase was parallel to the  trace. Furthermore, a part of the ��' phase transformed into the �� phase, and the size of the �� phase was increased to 4-6 nm during the long- term aging.
trace. Furthermore, a part of the ��' phase transformed into the �� phase, and the size of the �� phase was increased to 4-6 nm during the long- term aging.
According to the above experimental results, the tensile strength of the alloy with the Ni content of 0, 0.45, 0.98, 1.50 and 2.10 wt.% (hereinafter called as Cu-0.4Be, Cu-0.4Be-0.45Ni, Cu-0.4Be-0.98Ni, Cu-0.4Be-1.50Ni and Cu-0.4Be-2.10Ni) achieved the maximum at 400 ��C for 60, 120, 60, 60 and 60 min (hereinafter referred to as the ��peak-aging��), respectively. In order to analyze the influence of Ni content on the microstructure of the alloy, TEM was used to observe the microstructure of the alloys with different Ni contents after peak-aging, as shown in Figs. 9-11.

Fig. 9 TEM image (a) and diffraction pattern (b) of Cu-0.4Be alloy aged at 400 ��C for 60 min

Fig. 10 TEM images (a, b) and diffraction pattern (c, d) of Cu-0.4Be-0.49Ni alloy aged at 400 ��C for 120 min
No precipitated phase was observed in the alloy without Ni after aging, as shown in Fig. 9. With an increase of the Ni content, the morphology, number and distribution of the precipitated phase in the aged alloy changed greatly. When the Ni content was 0.49 wt.%, numerous ��" and ��' phases were observed in the TEM bright-field image in [001]�� zone axis. The ��' phase was disk-shaped with a diameter of about 3 nm and a thickness of 0.3-0.4 nm, and its non-contrast strain line was parallel to [110]�� and  respectively, as shown in Fig. 10. When the Ni content was increased to 1.50 wt.%, the number of Ni atom in the Cu matrix increased, and the number of the ��' phase and ��" phase which were formed by Ni and Be atoms increased obviously. The size of the ��' phase changed slightly, with the diameter of about 3.5 nm and the thickness of 0.4-0.6 nm. The bright and dark stripes were parallel to the
respectively, as shown in Fig. 10. When the Ni content was increased to 1.50 wt.%, the number of Ni atom in the Cu matrix increased, and the number of the ��' phase and ��" phase which were formed by Ni and Be atoms increased obviously. The size of the ��' phase changed slightly, with the diameter of about 3.5 nm and the thickness of 0.4-0.6 nm. The bright and dark stripes were parallel to the  traces around the ��' phase, which induced obvious lattice distortion, as shown in Fig. 7. When the Ni content was further increased to 2.10 wt.%, the number and size of the ��' phase in the alloy changed slightly; however, a small number of the incoherent �� phase with a size of 6-10 nm formed.
traces around the ��' phase, which induced obvious lattice distortion, as shown in Fig. 7. When the Ni content was further increased to 2.10 wt.%, the number and size of the ��' phase in the alloy changed slightly; however, a small number of the incoherent �� phase with a size of 6-10 nm formed.

Fig. 11 TEM images (a, b, c) and diffraction pattern (d) of Cu-0.4Be-2.10Ni alloy aged at 400 ��C for 60 min
3.4 Influencing mechanism of Ni content on microstructure and properties of Cu-0.4Be alloy
According to the above experimental results, it can be concluded that the phase precipitation behavior of the Cu-Be alloy without Ni was not obvious during aging, and adding Ni element could remarkably promote the phase precipitation of Cu-0.40wt.%Be alloy, where abundant nano-scale dispersed Be-Ni phase formed in the Cu matrix. Taking the alloy with 1.50 wt.% Ni aged at 400 ��C as an example, when the aging time was increased, phase transformation of ��" phase �� ��' phase �� �� phase occurred, and disk-like coherent ��' phase formed. In addition, for the peak-aging, when the Ni content was increased from 0.49 to 1.50 wt.%, the number of the precipitated phase increased apparently but the size of the phase changed slightly. For the alloy with Ni content of 1.50 wt.%, some bright and dark strain contrast stripes formed around the ��' phase. When the Ni content was increased to 2.10 wt.%, the number and size of the ��' phase hardly changed, and a few large incoherent �� phase particles formed.
From the Cu-Be binary phase diagram [22], the solid solubility of Be in Cu was 2.7 wt.% at 866 ��C and 0.16 wt.% at room temperature. The solid solubility of Be decreased obviously with the decrease of temperature. In this work, the Be content of the alloy was about 0.40 wt.% and CuBe phase formed after aging at 300-450 ��C. When the Ni element was added into the Cu-0.40wt.%Be alloy, the solid solubility of Be in ��-Cu solid solution declined [23], which was beneficial to the formation of dispersed precipitated phase in Cu matrix. In addition, the affinity between Ni atom and Be atom was greater than that between Cu atom and Be atom, which was more likely to form Be-Ni phase [19]. Therefore, the addition of Ni element can promote the Be-Ni phase precipitation and facilitated to form a great number of dispersed Be-Ni phase in the Cu-0.40wt.%Be alloy during aging.
Morphology, number and distribution of precipitated phase were the main factors affecting the strength of Cu-Be alloy. In the Cu-Be alloy without Ni element, the precipitated phase forming in the aging process was less, which induced low strengthening effect. Adding Ni element produced a great number of nano-scale dispersed coherent Be-Ni phase in the alloy during aging, and the coherent precipitated phase produced large lattice elastic distortion in the Cu matrix, which impeded the dislocation movement intensively. Furthermore, the pinning effect of dispersive precipitated phase to dislocation movement was strong, which further strengthened the alloy remarkably. According to the principle of precipitation strengthening, the strengthening effect generated by Orowan bypass mechanism can be expressed by [9]
 (1)
(1)
 (2)
(2)
where M is Taylor factor, G is shear modulus of Cu matrix, b is magnitude of Burgers vector, v is Poisson ratio, �� is particle spacing, dp is average diameter of particle and fv is volume fraction of second phase.
Taking the alloy with 1.50 wt.% Ni as an example, in the early stage of aging, the solid solubility of Be and Ni elements in the Cu matrix was relatively large, which had a large driving force of phase precipitation and induced rapid phase precipitation, forming more coherent ��" phase and �á� phase in the alloy. With prolonging aging time, the number of the coherent precipitated phase increased significantly, which induced rapid increase of strength and hardness of the alloy at the initial stage of aging. When the aging time was 60 or 120 min, a great number of dispersed disk-like ��" and ��' phases formed in the alloy, and the number of the precipitated phase increased apparently. The coherent ��' phase contributed to a large elastic strain field in the Cu matrix (bright and dark strain contrast stripes forming around ��' phase, as shown in Fig. 7(c)), which remarkably hindered the dislocation movement and induced the peak strength of the alloy. When the aging time was further increased, the ��' phase grew up and some coherent ��' phase transformed into incoherent �� phase, which decreased the strength of the alloy due to the reduction of obstruction to the dislocation.
In addition, for the peak-aging, as the Ni content increased from 0.49 to 1.50 wt.%, the number of both ��" phase and ��' phase increased, the volume fraction fv of the precipitated phase increased, and the average distance dp between the precipitated phase reduced. According to the Orowan bypass mechanism, the pinning effect of the precipitated phase to the dislocation movement increased, which improved the strength of the alloy. Especially, compared with the alloy with the Ni content of 0.49 and 0.98 wt.%, the number of the coherent ��' phase in the alloy with 1.50 wt.% Ni increased and produced larger lattice distortion in the Cu matrix, which remarkably improved the strength of the alloy. Furthermore, with the increase of the initial Ni content, Ni content in the Cu matrix increased, which induced an increase of the solid solution strengthening effect. With further increasing the Ni content to 2.10 wt.%, the number of the precipitated phase changed slightly, but the ��' phase grew slightly and some ��' phase transformed into incoherent �� phase with a size of 6-10 nm, weakening the impeding effect of the precipitated phase to the dislocation movement, which gave rise to the reduction of strength.
The concentration of Be and Ni atoms in Cu matrix was the main factor affecting electrical conductivity. The higher the concentration of solute atoms, the stronger the electron scattering effect, and the lower the electrical conductivity of the alloy. Before aging, the alloy had supersaturated solid-solution structure, and the concentration of Be and Ni atoms in the alloy was large, which led to a large degree of lattice distortion, and thus the electrical conductivity of the alloy was reduced due to strong scattering effect on electron. At the early stage of aging, due to the rapid precipitation of Be-Ni phase, the concentration of Be and Ni atoms in the alloy decreased rapidly, resulting in the rapid increase of electrical conductivity. With the increase of the aging time, the number of the precipitated phase increased slowly, resulting in the slow increase of electrical conductivity.
Under the experimental conditions in this work, when the aging time exceeded 10 min, the electrical conductivity of Cu-Be alloy was reduced with the increase of Ni content (seen in Fig. 5), which was relevant to electron scattering by the precipitated phase as well as Be and Ni atoms in the Cu matrix. Under the same aging condition, with the increase of the Ni content, the number of Be-Ni precipitated phase in the alloy increased, which reduced the number of Be atom in the Cu matrix and was beneficial to weakening the electron scattering effect of Be atom. However, owing to the limited phase precipitation, the number of Ni atom in Cu matrix increased with the increase of the initial Ni content, which enhanced the electron scattering effect of Ni atom. The electron scattering effect derived from the increase of Ni atom in the Cu matrix was greater than that of Be atom. On the other hand, with the increase of the Ni content, the number of the nano-scale precipitated phase increased, which improved the electron scattering to some extent. Therefore, the electrical conductivity of the alloy decreased with the increase of the Ni content due to the above two aspects.
From the above experimental results and analysis, adding Ni element can remarkably improve the strength of Cu-0.4Be alloy. With the increase of the Ni content, the strength of the alloy initially increased and then decreased. When the Ni content was 1.50 wt.%, Cu-0.4Be-1.5Ni alloy had the maximum strength, and the reasonable aging parameters were aging temperature of 400 ��C and the aging time of 1 h. The tensile strength and yield strength of the alloy were 611 and 565 MPa, respectively, which were 2.8 times and 6.1 times of the tensile strength and yield strength of the alloy without Ni, and the electrical conductivity was 40.3% (IACS). A great number of dispersed nano- scale coherent disk-like ��" phase and ��' phase formed in the alloy, inducing large lattice distortion around the ��" phase and ��' phase, which was mainly responsible for the high strength of the alloy. Moreover, the electrical conductivity of the aged alloy decreased with the increase of the Ni content.
4 Conclusions
(1) Adding Ni element can remarkably improve the strength of Cu-0.4Be alloy. When the Ni content increased from 0 to 2.10 wt.%, the strength of the aged alloy initially increased and then decreased. When the Ni content was 1.50 wt.%, Cu-0.4Be-1.5Ni alloy had the maximum strength. The peak-aging parameters were aging temperature of 400 ��C and the aging time of 60 min. The tensile strength and yield strength were 611 and 565 MPa, respectively, which were 2.8 times and 6.1 times of the tensile strength and yield strength of the alloy without Ni.
(2) After aging at 400 ��C, the electrical conductivity of the alloy with Ni increased with an increase of the aging time, and decreased with an increase of the Ni content. Taking the aging time of 60 min as an example, the electrical conductivity decreased from 47.6% to 39.6% (IACS) when the Ni content was increased from 0.45 to 2.10 wt.%.
(3) After aging at 400 ��C, with the increase of the aging time, the phase transition sequence of ��" phase �� ��' phase �� �� phase occurred in the Cu-0.4Be-1.5Ni alloy. When the aging time was 60 min, numerous dispersed nano-scale coherent ��" phase and ��' phase formed in the alloy, which mainly contributed to the high strength of the alloy.
(4) After the peak-aging, the precipitated phase in the alloy without Ni was much less than that in the alloy with Ni. When the Ni content was increased from 0.49 to 1.50 wt.%, the numbers of the ��" phase and ��' phase in the alloy increased remarkably, which induced strong precipitation strengthening. With increasing the Ni content to 2.10 wt.%, a few incoherent �� phase formed, which reduced precipitation strengthening effect.
Acknowledgments
The authors would like to thank the support from the National Key R&D Programme of China (No. 2016YFB0301404), the National Natural Science Foundation of China (51925401, 92066205), the National Ten Thousand Talents Programme of China and Ningbo ��Science and Technology Innovation 2025�� major project (No. 2019B10087).
References
[1] BEHJATI P, DASTJERDI H V, MAHDAVI R. Influence of ageing process on sound velocity in C17200 copper beryllium alloy [J]. Journal of Alloys and Compounds, 2010, 505: 739-742.
[2] PANG J C, DUAN Q Q, WU S D, LI S X, ZHANG Z F. Fatigue strengths of Cu-Be alloy with high tensile strengths [J]. Scripta Materialia, 2010, 63(11): 1085-1088.
[3] MURTY Y V. Electrical and electronic connectors: Materials and technology [J]. Encyclopedia of Materials: Science and Technology, 2001(2): 2483-2494.
[4] YAGMUR L. Effect of microstructure on internal friction and Young��s modulus of aged Cu-Be alloy [J]. Materials Science and Engineering A, 2009, 523: 65-69.
[5] ARGIBAY N, BARES J A, KEITH J H, BOURNE G R, SAWYER W G. Copper�Cberyllium metal fiber brushes in high current density sliding electrical contacts [J]. Wear, 2010, 268(11-12): 1230-1236.
[6] WANG Wei. Production and application prospect of beryllium-copper alloy [J]. Nonferrous Metals Process, 2014, 43(2): 9-12. (in Chinese)
[7] ZHU D B, LIU C M, HAN T, LIU Y D, XIE H.P. Effects of secondary �� and �� phases on the work function properties of Cu-Be alloys [J]. Journal of Applied Physics, 2015, 120(3): 1023-1026.
[8] YAGMUR L, DUYGULU O, AYDEMIR B. Investigation of metastable �á� precipitate using HRTEM in aged Cu-Be alloy [J]. Materials Science and Engineering A, 2011, 528: 4147-4151.
[9] XIE Guo-liang, WANG Qiang-song, MI Xu-jun., XIONG Bai-qing, PENG Li-jun. The precipitation behavior and strengthening of a Cu-2.0wt.%Be alloy [J]. Materials Science and Engineering A, 2012, 558: 326-330.
[10] MONZEN R, WATANABE C, MINO D, SAIDA S. Initiation and growth of the discontinuous precipitation reaction at [011] symmetric tilt boundaries in Cu-Be alloy bicrystals [J]. Acta Materials, 2005, 53(4): 1253-1261.
[11] ROTEM A, SHECHTMEN D, ROSEN A. Correlation among microstructure, strength, and electrical conductivity of Cu-Ni-Be alloy [J]. Metallurgical Transactions A, 1988, 19(9): 2279-2285.
[12] ZINKLE S J. Evaluation of high strength, high conductivity CuNiBe alloys for fusion energy applications [J]. Journal of Nuclear Materials, 2014, 449(1-3): 277-289.
[13] MONZEN R, HOSODN T, TAKAGAWN Y, WATANABE C. Bend formability and strength of Cu-Be-Co alloys [J]. Journal of Materials Science, 2011, 46(12): 4284-4289.
[14] TANG Yan-chuan, ZHU G M, KANG Yong-lin, YUE Li-juan, JIAO Xiao-liang. Effect of microstructure on the fatigue crack growth behavior of Cu-Be-Co-Ni alloy [J]. Journal of Alloys and Compounds, 2016, 663: 784-795.
[15] TANG Yan-chuan, KANG Yong-lin, YUE Li-juan, JIAO Xiao-liang. Mechanical properties optimization of a Cu-Be-Co-Ni alloy by precipitation design [J]. Journal of Alloys and Compounds, 2017, 695: 613-625.
[16] WATANABE C, MONZEN R. Precipitation process in a Cu-Ni-Be alloy [J]. Solid State Phenomena, 2011, 172-174: 432-436.
[17] PENG Li-jun, XIONG Bai-qing, XIE Guo-liang, WANG Qiang-song, HONG Song-bai. Precipitation process and its effects on properties of aging Cu-Ni-Be alloy [J]. Rare Metals, 2013, 32(4): 332-337.
[18] HE Shuang-jiang, JIANG Yan-bin, XIE Jian-xin, LI Yong-hua, YUE Li-jian. Effects of Ni content on cast and solid-solution microstructures of Cu-0.4Be alloys [J]. International Journal of Minerals Metallurgy and Materials, 2018, 25(6): 641-651.
[19] ZHANG Xiao-zhong. Electronic micro-analysis [M]. Beijing: Tsinghua University Press, 2006. (in Chinese)
[20] RYOICHI M, TSUTOMU S, TOSHIRO S, CHIHIRO W. Precipitation processes in Cu-0.9mass%Be single crystal [J]. Materials Transactions, 2006, 47(12): 2925-2934.
[21] ZHOU Y J, SONG K X, XING J D, ZHANG Y M. Precipitation behavior and properties of aged Cu-0.23Be-0.84Co alloy [J]. Journal of Alloys and Compounds, 2016, 658: 920-930.
[22] LIU Chu-ming , LI Hui-zhong, HAN Tan. Phase diagram of copper alloy [M]. Changsha: Central South University Press, 2011. (in Chinese)
[23] SPAIC S, MARKOLI B. Microstructural characterization of alloys of the quasibinary Cu-Ni-Be system [J]. Zeitschrift Fur Metallkunde, 2003, 94(8): 876-879.
�����1,2����ͬͬ2���� ��2����˫��2�����»�2,3��л����2,3
1. ���ϴ�ѧ ���Ͽ�ѧ�빤��ѧԺ����ɳ 410083��
2. �����Ƽ���ѧ �����Ƚ��Ʊ������������ص�ʵ���ң����� 100083��
3. �����Ƽ���ѧ �������ϻ��̸߾��ⴴ�����ģ����� 100083
ժ Ҫ���о�Ni����(0��2.10%����������)��Cu-0.4Be(����������%)�Ͻ�ʱЧ�����ࡢǿ�Ⱥ͵����ʵ�Ӱ�죬������Ӱ��������������������NiԪ�ؿɴٽ�ǿ������������������ʱЧǿ�����á���Ni������0����2.10%ʱ���Ͻ�ʱЧ���ǿ�ȳ���������С�ı仯���ƣ���Ni����Ϊ1.50%ʱ�ﵽ���1.50%Ni�Ͻ�ķ�ֵʱЧ����Ϊ400 ��C����60 min��ʱЧ��Ͻ�Ŀ���ǿ�Ⱥ�����ǿ�ȷֱ�Ϊ611 MPa��565 MPa���ֱ��Dz���Ni�Ͻ��2.8����6.1������Ni�Ͻ�ĵ���������ʱЧʱ����ӳ������ӣ�������Ni������������½�����400 ��C������ʱЧʱ����ӳ���Cu-0.4Be-1.5Ni�Ͻ����á�����á��������ת�䣻��ʱЧʱ��Ϊ60 minʱ���Ͻ��г��ִ�����ɢ�ֲ�����������á���ͦá��࣬����ǿ�������������ǺϽ���нϸ�ǿ�ȵ���Ҫԭ��
�ؼ��ʣ�Cu-Be �Ͻ𣻺Ͻ���䣻����֯����ѧ���ܣ�������
(Edited by Bing YANG)
Corresponding author: Xin-hua LIU, E-mail: liuxinhua18@163.com; Jian-xin XIE, E-mail: jxxie@mater.ustb.edu.cn
DOI: 10.1016/S1003-6326(21)65529-2
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press

