
Diffusion behaviors of helium atoms at two Pd grain boundaries
XIA Ji-xing(夏吉星), HU Wang-yu(胡望宇), YANG Jian-yu(杨剑瑜), AO Bing-yun(敖冰云)
Department of Applied Physics, Hunan University, Changsha 410082, China
Received 10 April 2006; accepted 25 April 2006
Abstract: The diffusion behaviors of helium atoms at two symmetric grain boundaries (Σ5<001>{210} and Σ3<110> {112}) of Pd were investigated using molecular dynamics simulations through an analytical embedded-atom method(MAEAM) model. The simulations demonstrate that the interstitial helium atoms are easily trapped at the grain boundaries and precipitated into clusters. Due to the closed-shell electronic configurations of both helium and palladium, Pd grain boundaries yield strong capability of retaining helium atoms. By calculating the mean square displacements(MSD) of an interstitial helium atom at the grain boundaries, the diffusion coefficients were determined, and the linear fits to Arrhenius relation. The diffusion activation energies of interstitial helium atom at these two Pd grain boundaries were also evaluated.
Key words: helium atoms; diffusion; grain boundary; palladium; molecular dynamics
1 Introduction
Helium atoms could be produced by nuclear reactions of energetic particles, and due to its low solubility in metals, they tended to cluster and precipitated into bubbles evenly. The accumulation of helium atoms may depress the integrity of first-wall materials. At high temperatures, helium could result in degradation of the tensile, creep and fatigue properties, which may even cause the initiation of cracks along grain boundaries (GBs)[1]. It is also generally accepted that GBs provide fast diffusion paths for helium atoms, hence, the research of interactions between helium atoms and GBs is great significant to nuclear and material industry.
BARNES[2] firstly attributed investigated high temperature embitterment of irradiated steels to helium bubbles in the GBs. And two decades later, Baskes et al[3] performed an atomistic study on the trapping of hydrogen and heium atoms at GBs in nickel. Recently, using molecular dynamics(MD) method, GAO et al[4] and Kurtz et al[1] studied the binding effects of GBs to helium atoms and the diffusion behaviors of interstitial helium atoms of GBs in α-Fe. In our previous work, we have performed the simulation of helium behaviors at GBs in nickel with MD method. In the present work, due to the especial closed-shell electronic configuration of palladium, we studied the helium diffusion behaviors at two GBs, one is Σ5<001>{210} θ=53.1?, and the other is Σ3<110>{112} θ=70.53?.
2 Modeling
A slab model was used in this simulation along the direction parallel to the GB planes, periodic boundary conditions were employed to the GB planes, the model were divided into three parts. The inner atoms sandwiched between the outer slabs were free in three dimensions, and the atoms of the outer slabs were frozen in the initial positions. The slabs were allowed to move rigidly and perpendicular to the GB planes. The GBs were created by aligning crystallographic planes paralleling to the desired planes, which followed by an 180? rotation of one grain relative to the other one[5].
The MD simulation was performed in a NPT box, the initial coincident-site-lattice(CSL) GBs were quench to that of 0 K by MD, and the minimum energy configuration for further calculations was obtained. The relaxed structures contain 5 000 and 5 040 atoms respectively, as shown in Fig.1.
In the present work, a modified analytic embedded-
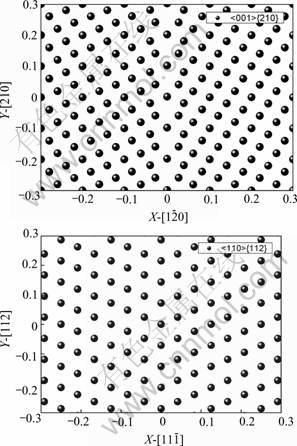
Fig.1 Two bicrystal models for MD simulation
atom-method model (MAEAM) [6] was adopted to describe the interactions between metal atoms. The interaction between a helium atom and a metal atom was described by a Morse potential determined by fitting the free-electron helium-metal pair potentials[7], the form was given by
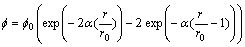 (1)
(1)
The interaction between two helium atoms was described with the Lennard-Jones potential[8], which was given by
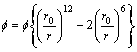 (2)
(2)
The parameters are shown in Table 1.
Table 1 Input parameters of Morse potential and Lennard-Jones potential

3 Results and discussion
3.1 Formation energy of interstitial helium atom in GBs
An interstitial helium atom could easily be diffused into a stable position, in order to decrease the total lattice energy. To obtain the formation energies of one interstitial helium atom at different positions, 0 K MD simulations were performed instead of molecular static simulations with the helium atom placed along an interstitial plane. The formation energies depend strongly on the distance from the GB core, as shown in Fig.2. For <001> {210} GBs, at the GB core, the helium atom yields the lowest formation energy (0.45 eV), while in <110>{112} GBs, the lowest formation energy does not exist, but close to the GB core with an value 1.50 eV. Both the lowest formation energies in these two GBs are lower than 2.38 eV in bulk palladium. An interstitial helium atom should tend to diffuse into the GBs from bulk region to minimize the total energy. The appearance of helium atoms at GBs provides the precursor condition for the nucleation of helium atoms.
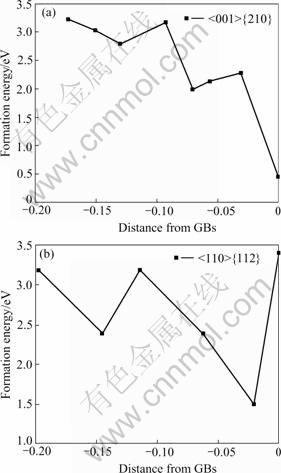
Fig.2 Change curves of formation energies for interstitial helium with distance from GB core
3.2 Binding effect of one helium atom to rest helium cluster at GBs
To study the growth of helium clusters, one supplemental helium atom was added sequently after the new helium cluster was relaxed sufficiently. Hence, the helium cluster size increase by one helium atom each time till to 20 helium atoms. The binding energy of one helium atom to the rest cluster is defined as follows.
EB(He)=Ef(Hen-1)+Ef(He)-Ef(Hen) (3)
where Ef is the corresponding formation energy. The relationship between binding energy and helium cluster size are depicted as shown in Fig.3. Though from Fig.3, the dependencies of helium binding energies on the cluster size were not obvious in our calculation range, the binding energy in both GBs keep positive with the cluster size increasing. It suggests that the supplemental helium atom would be bond to the helium cluster and cause the growth of the helium clusters. The higher binding energies for Σ5 GB may result from the larger excess volume[1]. The cluster configuration with 20 helium atoms is shown in Fig.4(a), which can explain why helium atoms can easily agglomerate into bubbles in GBs.
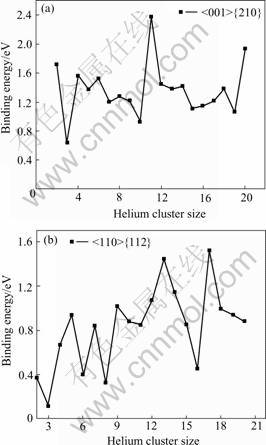
Fig.3 Relationship between binding energy and helium cluster size
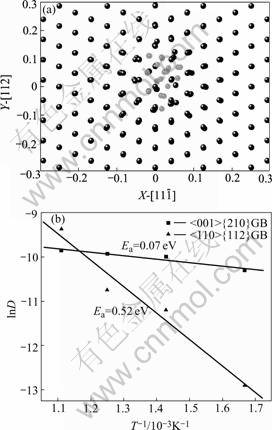
Fig.4 Agglomeration of 20 helium atoms in <110>{112} GBs (a) (Red balls are helium atoms and black ones are palladium atoms; right) and Arrhenius plots of diffusion coefficients of one helium atom in GBs (b)
3.3 Diffusion behaviors of one interstitial helium atoms in GBs
The most stable configuration with one interstitial helium atom was used to perform the diffusion process at different temperatures with MD. To obtain efficient mean square displacements(MSD)[9], the statistical steps should be long enough, and in our simulations, it reached more than one million steps (about more than 2 ns). The diffusion coefficients were derived from linear fitting the obtained MSD curves. The activation energy and diffusion pre-factor of interstitial diffusion could be estimated from Arrhenius relation. The plots of lnD vs 1/T in both GBs are shown in Fig.4(b). The values of Ea and D0 are evaluated to be 0.07 eV and 1.31×10-4 cm2/s for the <001>{210} GB and 0.52 eV and 5.5×10-2 cm2/s for the <110>{112} GB.
Both the obtained activation energies were lower than 0.72 eV[10] in perfect bulk. It is consistent with the fact present in the former section that GBs provide fast-diffusion path for helium atoms. Thus, once there exists GBs nearby, the interstitial helium atoms should diffuse easily into GBs and be bonded with each other to form helium clusters or even grow into bubbles. The activation energy in Σ5 GB is much lower than in Σ3 GB, it may result from the lower density of atoms in Σ5 GB.
4 Conclusions
1) An interstitial helium atom tends to diffuse into the GBs to obtain the most stable configurations.
2) The minimum formation energies of an interstitial helium atom are 0.45 eV in Σ5{210} GB and 1.50 eV in Σ3{112} GB, respectively.
3) The binding energies of the supplemental helium atom to the rest helium cluster in the both GBs are positive.
4) The corresponding activation energies for interstitial diffusion are estimated as 0.07 eV and 0.52 eV.
5) The lower activation energies suggest that the GBs provide a fast-diffusion path for helium atoms before they nucleate into bubbles.
References
[1] Kurtz r j, Heinisch h l. The effects of grain boundary structure on binding of He in Fe [J]. J Nucl Mater, 2004, 329-333: 1199-1203.
[2] Barnes r s. Embrittlement of stainless steels and Ni-based alloys at high temperature induced by neutron irradiation [J]. Nature, 1965, 206: 1307-1310.
[3] Baskes m I, Vitek v. Trapping of hydrogen and helium at grain boundaries in nickel: an atomistic study [J]. Met Trans A, 1985, 16: 1625-1631.
[4] Gao f, Krutz r j. The diffusion of He atoms and small He clusters in grain boundaries in Alpha-Fe [J]. JOM, 2004, 56(11): 200.
[5] Sfrensen m r, Mishin y. Diffusion mechanisms in Cu grain boundaries [J]. Phys Rev B, 2000, 62(6): 3658-3673.
[6] Hu w. The application of the analytic embedded atom potentials to face-centered cubic metals [A]. Naka m, Yamane t. New Frontiers of Process Science and Engineering in Advanced Materials [C]. Japan: High Temperature Society, 2004. 7-12.
[7] Baskes m I, Melius c f. Pair potentials for fcc metal [J]. Phys Rev B, 1979, 20: 3197-3204.
[8] Johnson r a. Empirical potentials and their use in the calculation of energies of point defects in metals [J]. J Phs F: Metal Phys, 1973, 3: 295-321.
[9] Kakimoto k, Umehara t, Ozoe h. Molecular dynamics analysis on diffusion of point defects [J]. J Cryst Growth, 2000, 210: 54-59.
[10] XIA Ji-xing, HU Wang-yu, YANG Jian-yu, AO Bing-yun, WANG Xiao-lin. A comparative study of helium atom diffusion via an interstitial mechanism in nickel and palladium [J]. Phys Stat Sol (b), 2006, 243(3): 579-583.
(Edited by LI Yan-hong)
Foundation item: Project(50371026) supported by the National Natural Science Foundation of China; Project supported by the Natural Science Foundation of Hunan Province; Project(20040546) supported by the Science, Technology Foundation of China Academy of Engineering Physics
Corresponding author: HU Wang-yu; Tel: +86-731-8823971; E-mail: wangyuhu2001cn@yahoo.com.cn