离子吸附型稀土矿硫酸镁浸取尾矿中钙、镁的淋出行为
来源期刊:中国有色金属学报(英文版)2021年第1期
论文作者:范波 赵龙胜 冯宗玉 刘德鹏 尹伟强 龙志奇 黄小卫
文章页码:288 - 296
关键词:离子吸附型稀土矿;硫酸镁;环境修复;淋出行为;石灰水
Key words:ion-adsorption rare earth ore; magnesium sulfate; environmental remediation; leaching behaviors; lime water
摘 要:研究采用去离子水、氯化钙溶液、石灰水淋洗剂淋洗,离子吸附型稀土矿硫酸镁浸取尾矿中镁、钙的淋出行为。结果表明,浸取尾矿中的镁主要以水溶态形式存在,这部分镁易被淋洗脱除。采用石灰水淋洗,淋洗剂中的绝大部分钙可被静电吸附在淋洗后尾矿中,同时浸取尾矿中的部分水溶态镁因Mg2+在黏土矿物上的反吸附作用而逐渐转化为交换态镁,这与淋洗后尾矿pH值的升高有关。采用石灰水淋洗,当液固比为0.80时,淋洗后尾矿中速效镁、钙含量分别为104.4~207.6和201.7~1426.3 mg/kg,满足植物的生长需求。该研究为离子吸附型稀土矿原地浸矿场的环境修复提供一种有潜力的解决方案。
Abstract: The leaching behaviors of calcium and magnesium in the rare earth tailings leached with magnesium sulfate using deionized water, CaCl2 solution and lime water were investigated. Experimental data indicated that magnesium in the tailings was easy to be leached out since most of the magnesium was in the form of water-soluble phase. Most of calcium in the lime water was electrostatically adsorbed on the clay mineral of the tailings, and the water-soluble magnesium was also gradually converted into exchangeable phase because of back-adsorption of Mg2+ on the clay mineral with increasing the pH values. When the liquid-to-solid ratio was 0.80, the contents of readily-available magnesium and calcium were 104.4-207.6 and 201.7-1426.3 mg/kg, respectively, which could meet the requirements for plants. These results suggest a promising route for the environmental remediation of ion-adsorption rare earth ore after in-situ leaching.
Trans. Nonferrous Met. Soc. China 31(2021) 288-296
Bo FAN1,2, Long-sheng ZHAO1,3, Zong-yu FENG1,3, De-peng LIU1,3, Wei-Qiang YIN1,3, Zhi-qi LONG1,3, Xiao-wei HUANG1,3
1. National Engineering Research Center for Rare Earth Materials,GRINM Group Co., Ltd., Beijing 100088, China;
2. General Research Institute for Nonferrous Metals, Beijing 100088, China;
3. Grirem Advanced Materials Co., Ltd., Beijing 100088, China
Received 13 March 2020; accepted 18 August 2020
Abstract: The leaching behaviors of calcium and magnesium in the rare earth tailings leached with magnesium sulfate using deionized water, CaCl2 solution and lime water were investigated. Experimental data indicated that magnesium in the tailings was easy to be leached out since most of the magnesium was in the form of water-soluble phase. Most of calcium in the lime water was electrostatically adsorbed on the clay mineral of the tailings, and the water-soluble magnesium was also gradually converted into exchangeable phase because of back-adsorption of Mg2+ on the clay mineral with increasing the pH values. When the liquid-to-solid ratio was 0.80, the contents of readily-available magnesium and calcium were 104.4-207.6 and 201.7-1426.3 mg/kg, respectively, which could meet the requirements for plants. These results suggest a promising route for the environmental remediation of ion-adsorption rare earth ore after in-situ leaching.
Key words: ion-adsorption rare earth ore; magnesium sulfate; environmental remediation; leaching behaviors; lime water
1 Introduction
Mid-heavy rare earths are important in many strategic fields, from advanced materials with superior properties to large-scale high-tech devices [1-4]. Ion-adsorption rare earth ore, which is rich in mid-heavy rare earths and is mainly found in seven adjacent provinces of southern China, accounts for over 80% of the world’s total reserves of mid-heavy rare earths [5,6]. At present, acidic ammonium sulfate solution (1%-4%) is usually used as a leaching agent to extract the rare earths during the in-situ leaching of the ion-adsorption rare earth ore [7]. However, acidification of the ore body (pH ~4.0) and serious leaching loss of the nutrients such as calcium and magnesium always occur because of the eluviation of a large amount of acidic ammonium sulfate [8]. More seriously, a large amount of ammonia-nitrogen enters into the ground water and surface water around the ore body. Because of the slow release of the residual ammonium salt during the long-term eluviation of rainwater, and as such, the ammonia-nitrogen (NH3-N) content of the surface water is always as high as 100 mg/L [9]. It is much higher than the limit of classification III in the environmental quality standards for surface water (NH3-N 1.0 mg/L) [10].
In order to eliminate the ammonia-nitrogen pollution and prevent the nutrients loss, many researches on development of new leaching agents (magnesium sulfate [11], aluminum sulfate [12], citrate, acetate [13,14]) and leaching auxiliaries (fulvic acid [15], sesbania gum [16], ascorbic acid [17] and ferrous salt [18]) have been carried out. HUANG et al [19] proposed a new green leaching technology using magnesium sulfate, and the leaching efficiency of rare earths using magnesium sulfate is almost equivalent to that using conventional ammonium sulfate [20]. However, acidification of the ore body and leaching loss of calcium also occur because of the eluviation of acidic magnesium sulfate. It is reported that the suitable pH range of soil for plants is 5.5-8.5 since plants can grow well under neutral, weakly acidic or alkaline conditions [21]. The contents of hydrolyzable nitrogen, readily-available calcium, and readily-available magnesium are expected to be 20-100, 400-4800, and 120-1460 mg/kg, respectively, with the mass ratio of calcium to magnesium of about 1:1-20:1 [22].
Therefore, a remedy for the rare earth tailings leached with magnesium sulfate is of vital importance. But existing researches are highly focused on the remediation of heavy metals contaminated soil using EDTA [23], Ca(H2PO4)2 [24], HPMA and PBTCA [25]. YANG et al [12] used lime water to treat the rare earth tailings leached with aluminum sulfate by column leaching. Most of the aluminum sulfate was transformed to Al(OH)3 sediment and then stably presented in the tailings when the pH of the tailings was higher than 6, and the problems of acidification and landslides were then expected to be solved. As for the rare earth tailings leached with magnesium sulfate, calcium-bearing materials might be promising to solve the above-mentioned ecological problems of the rare earth tailings leached with magnesium sulfate. However, the mechanism for the transportation and transformation of calcium and magnesium in the tailings is not clear yet, which provides the impetus for this work.
In this work, the leaching of the rare earth tailings leached with ammonium sulfate (NH4+- leached tailings) and magnesium sulfate (Mg2+- leached tailings) was investigated, and deionized water, calcium chloride (CaCl2) solution and lime water were used for the leaching of the Mg2+-leached tailings. The leaching behaviors of calcium and magnesium in the Mg2+-leached tailings were studied and discussed in detail. The present work aims to elucidate the mechanism for the transportation and transformation of calcium and magnesium in the Mg2+-leached tailings.
2 Experimental
2.1 Materials
The ion-adsorption rare earth ore was from Longyan, southwestern of Fujian Province, China. The rare earth ore sample was first air-dried, and then treated with conical quartering before it was used in this work. The chemical composition of the rare earth ore sample is listed in Table 1, and the X-ray diffraction pattern is shown in Fig. 1. It can be seen that the rare earth ore sample was composed of quartz, potassium feldspar and kaolinite. Adsorption and cation exchange processes are always largely influenced by clay mineral of kaolinite [26,27]. All chemicals used in this work were of analytical grade.
Table 1 Chemical composition of rare earth ore sample (wt.%)

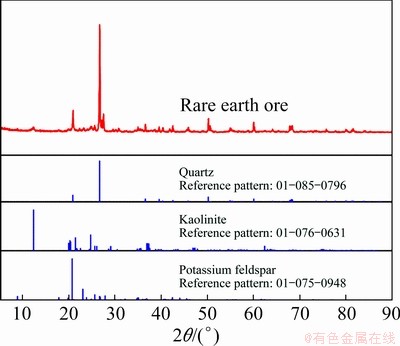
Fig. 1 X-ray diffraction pattern of rare earth ore sample
PVC pipe with an inner diameter of 100 mm was used as a leaching column for the leaching of the ion-adsorption rare earth ore. Ion-adsorption rare earth ore was first packed into the leaching column, and then leached with 0.2 mol/L magnesium sulfate (MgSO4) solution or ammonium sulfate ((NH4)2SO4) solution (pH 2.0) at room temperature. The flow rate of leaching agents was 2 mL/min, and the liquid-to-solid ratio was 2:1. After the leaching was completed, the leaching solutions and tailings were collected for analysis.
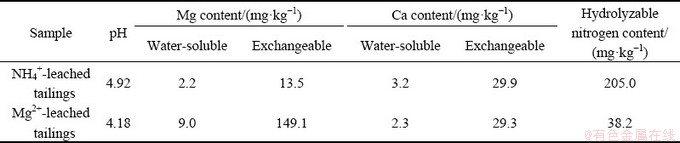
When the concentration of leaching agent was 0.20 mol/L, the leaching efficiency of rare earths in the ion-adsorption rare earth ore using magnesium sulfate solution was 90.3%, which was almost equal to that using ammonium sulfate. This is in good agreement with the results of XIAO et al [20]. The contents of readily-available magnesium and calcium, and hydrolyzable nitrogen in the rare earth ore and the NH4+/Mg2+-leached tailings are shown in Table 2. For the NH4+-leached tailings, the content of hydrolyzable nitrogen was 1540.0 mg/kg, much higher than the suitable content of the soil for plants (20-100 mg/kg), while serious loss of the nutrients of calcium and magnesium occurred. For the Mg2+-leached tailings, the contents of readily-available magnesium and hydrolyzable nitrogen were 1179.7 and 38.3 mg/kg, respectively, which could meet the requirements for the growth of plants. Similar to the NH4+-leached tailings, serious loss of readily-available calcium also occurred. Furthermore, the pH values in the NH4+-leached tailings and the Mg2+-leached tailings were 3.49 and 3.41, respectively, indicating acidification of the rare earth tailings.
2.2 Apparatus and experimental procedure
Perspex column with an inner diameter of 40 mm was used for the leaching of the rare earth tailings, and peristaltic pump (BT100-1F, Baoding Longer Precision Pump Co., Ltd.) was used for the injection of different leaching agents. The experimental set-up for leaching is shown in Fig. 2.
Column with an inner diameter of 40 mm was used for the leaching of the rare earth tailings. Air-dried rare earth tailings were slowly filled into the column, and the leaching agents of deionized water, calcium chloride and lime water were injected using precision pumps with a flow rate of 0.2 mL/min at room temperature of approximately 25 °C. Leaching solutions were collected from the bottom of the column when the liquid-to-solid ratios were higher than 0.3:1 since the saturated water content of the air-dried rare earth tailings is 30%.
2.3 Characterization
The chemical composition was tested by X-ray fluorescence (ZSXPrimus II). The mineral phase was scanned by X-ray diffraction (X'Pert PRO MPD) at 2θ of 5°-90°. The concentrations of metal ions in the aqueous solutions were measured by ICP-OES (Optima 8300, Perkin-Elmer), and the concentration of ammonium ion was determined by acid-base titration. The pH value of the rare earth tailings was measured by a pH meter (LE438, Mettler-Toledo) using the potentiometry method [28]. The hydrolyzable nitrogen was tested using the alkali-diffusion method [29]. The determination of the water-soluble and exchangeable phases in the rare earth tailings were optimized based on the standard methods [30,31].
3 Results and discussion
3.1 Leaching of Mg2+-leached tailings using deionized water
Leaching experiments of the Mg2+-leached tailings using deionized water were conducted in the liquid-to-solid ratio range of 0.30-1.67, and the leaching of the NH4+-leached tailings was also studied for comparison. As seen in Fig. 3, the leaching curves of the readily-available magnesium and calcium, and the hydrolyzable nitrogen in the NH4+-leached tailings were very similar to those in the Mg2+-leached tailings. The concentration of cations decreased almost exponentially with increasing the liquid-to-solid ratio. This is in good agreement with the reported literature [32]. Most of the readily-available magnesium and calcium, and the hydrolyzable nitrogen in the leached tailings were mainly in the form of water-soluble phase, which could be easily leached with deionized water at the initial leaching stage. Therefore, the contents of the readily-available magnesium and calcium, and the hydrolyzable nitrogen in the leached tailings decreased significantly as leaching progressed, and then the exchangeable phases were considered as the main forms in the leaching tailings. The leaching of the exchangeable phases was controlled by the cation exchange reaction. According to Eqs. (1)-(3) [7], the concentrations of calcium, magnesium and ammonium ions are proportional to the concentration of free hydrogen ion. These cation exchange reactions were so difficult to occur via leaching with deionized water that little of calcium, magnesium and ammonium ions were leached out at the final leaching stage.
Table 2 Contents of readily-available magnesium and calcium, and hydrolyzable nitrogen in rare earth ore and NH4+/Mg2+-leached tailings
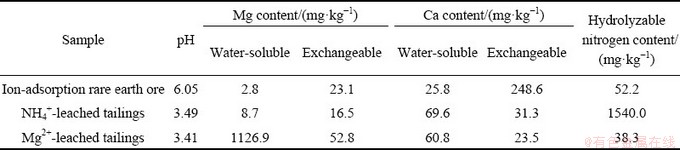
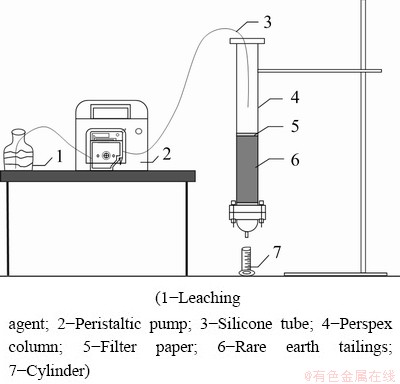
Fig. 2 Experimental set-up for leaching
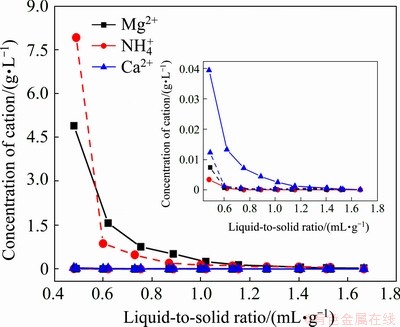
Fig. 3 Leaching curves of Ca2+, Mg2+ and NH4+ in NH4+-leached tailings (dotted line) and Mg2+-leached tailings (solid line)
[Al2Si2O5(OH)4]a・nMg2+(s)+2nH+(aq) [Al2Si2O5(OH)4]a・2nH+(s)+nMg2+(aq) (1)
[Al2Si2O5(OH)4]a・2nH+(s)+nMg2+(aq) (1)
[Al2Si2O5(OH)4]a・nNH4+(s)+nH+(aq) [Al2Si2O5(OH)4]a・nH+(s)+nNH4+(aq) (2)
[Al2Si2O5(OH)4]a・nH+(s)+nNH4+(aq) (2)
[Al2Si2O5(OH)4]a・nCa2+(s)+2nH+(aq) [Al2Si2O5(OH)4]a・2nH+(s)+nCa2+(aq) (3)
[Al2Si2O5(OH)4]a・2nH+(s)+nCa2+(aq) (3)
For the NH4+-leached tailings, the concentration of ammonium ions in the leaching solution at the final leaching stage was 31.6 mg/L, which is still 30 times higher than the limit of Classification III in the environmental quality standards for surface water (1.0 mg/L) [10]. For the Mg2+-leached tailings, the total hardness (calculated as CaCO3) of the leaching solution at the final leaching stage was lower than 254.0 mg/L (Mg 21.2 mg/L), which meets the requirement of the limit of Classification III in the environmental quality standards for ground water (CaCO3 450 mg/L) [33]. Therefore, magnesium sulfate is a green leaching agent for the leaching of ion-adsorption rare earth ore and can reduce the risk of water pollution.
Both of the leached tailings were considered to be completely leached since the concentrations of cations in the leaching solution at the final leaching stage were too low. The contents of readily-available magnesium and calcium, and hydrolyzable nitrogen in the leached tailings are presented in Table 3. The content of the hydrolyzable nitrogen in the NH4+-leached tailings was 205.0 mg/kg, and it is much higher than the suitable content in the soil for plants (20- 100 mg/kg). But the contents of the readily- available magnesium and calcium in the NH4+-leached tailings were as low as 15.7 and 33.1 mg/kg, respectively. But for the Mg2+-leached tailings, both the readily-available magnesium and the hydrolyzable nitrogen could meet the requirements for the growth of plants. Interestingly, the content of the exchangeable magnesium in the Mg2+-leached tailings was increased during the leaching of deionized water, which was most probably caused by the back-adsorption of Mg2+ with increasing the pH values of the Mg2+-leached tailings. Serious loss of the readily-available calcium also occurred in the Mg2+-leached tailings. Furthermore, the pH values of the leached tailings after water leaching were below 5, indicating that deionized water might be difficult to solve the acidification of the leached tailings.
Table 3 Contents of readily-available magnesium and calcium, and hydrolyzable nitrogen in NH4+/Mg2+-leached tailings after water leaching
3.2 Leaching of Mg2+-leached tailings using CaCl2 solution
In order to investigate the effect of the concentration of CaCl2 (0.05-0.25 mol/L) on the leaching of the Mg2+-leached tailings using CaCl2 solution, several tests were performed with the range of liquid-to-solid ratios of 0.30-1.67. As shown in Fig. 4(a), the leaching efficiency of magnesium in the Mg2+-leached tailings was increased rapidly as the liquid-to-solid ratio was increased. According to Eq. (4) [7], higher CaCl2 concentration of the CaCl2 solution is likely to induce the leaching of Mg2+ in the Mg2+-leached tailings. But actually, no obvious differences in the leaching of Mg2+ could be observed because the content of exchangeable magnesium in the Mg2+-leached tailings was only 52.8 mg/kg (see Table 2). Similar to the water leaching process, the concentration of Mg2+ decreased almost exponentially with increasing the liquid-to-solid ratio when leaching using CaCl2 solution (Fig. 4(b)). The leaching curves of calcium indicated that the leaching reaction gradually reached equilibrium as leaching progressed, and the concentrations of Ca2+ and Mg2+ in the leaching solution maintained relatively stable. That is to say, the contents of the readily-available magnesium and calcium in different layers of the Mg2+-leached tailings were almost the same.
[Al2Si2O5(OH)4]a・nMg2+(s)+nCa2+(aq) [Al2Si2O5(OH)4]a・nCa2+(s)+nMg2+(aq) (4)
[Al2Si2O5(OH)4]a・nCa2+(s)+nMg2+(aq) (4)

Fig. 4 Leaching behaviors of calcium and magnesium in Mg2+-leached tailings using CaCl2 solutions
The contents of the readily-available magnesium and calcium in the Mg2+-leached tailings after leaching with CaCl2 solution are listed in Table 4, as well as the pH values. According to Eq. (4) [7], the exchangeable acidity in the Mg2+-leached tailings is easier to be leached with increasing the CaCl2 concentration, resulting in an increase in the pH value of the leached tailings. However, the pH value of the Mg2+-leached tailings after leaching with CaCl2 solution was always lower than that after water leaching (pH=4.18) at the CaCl2 concentration of <0.20 mol/L. It has been reported that the pH value of the red soil containing calcium chloride was decreased by 0.2-0.45 probably because of an increase of the ionic strength and the H+ activity of the leaching solution in the presence of calcium salt during the pH determination process [34]. The pH value of the Mg2+-leached tailings after leaching with 0.25 mol/L CaCl2 solution was 4.32, indicating that CaCl2 solution is difficult to solve the acidification of the leached tailings.
Table 4 Contents of readily-available magnesium and calcium in Mg2+-leached tailings after leaching with CaCl2 solution

As the CaCl2 concentration was increased from 0.05 to 0.25 mol/L, the content of the readily- available calcium rapidly increased from 650.1 to 2333.0 mg/kg, which could meet the requirements for the growth of plants (readily-available calcium 400-4800 mg/kg). The content of readily-available calcium increased rapidly most probably because of the multiple-layer adsorption of water-soluble calcium on the surface of the clay minerals via van der Waals force. When CaCl2 solution was used for the leaching of the Mg2+-leached tailings, the problem of acidification was not solved, and part of electrostatic adsorption sites on the clay surfaces might still be occupied by hydrogen ions [26,35]. That is to say, the readily-available calcium in the leached tailings was mainly in form of water-soluble. Besides, the CaCl2 solution strengthened the process of cation exchange reaction, promoting further conversion of exchangeable magnesium into water-soluble phase. The serious loss of magnesium occurred since the content of the readily-available magnesium was lower than 30 mg/kg, and as such, the mass ratio of calcium to magnesium was much higher than 20. Therefore, calcium chloride is more likely to be used for preliminary adjustment of the readily- available calcium content in the Mg2+-leached tailings.
3.3 Leaching of Mg2+-leached tailings using lime water
The leaching experiments of the Mg2+-leached tailings were carried out using 0.02 mol/L lime water, and the range of liquid-to-solid ratios was 0.30-1.67. As shown in Fig. 5, the concentration of Mg2+ in the leaching solution decreased almost exponentially with increasing the liquid-to-solid ratio, while the concentration of Ca2+ was relatively stable and was lower than 48.3 mg/L. This is because most of Ca2+ is electrostatically adsorbed on the leached tailings during the neutralization of water-soluble and exchangeable H+ by OH- (Eq. (5)) [26,35].
[Al2Si2O5(OH)4]a・2nH+(s)+nCa2+(aq)+2nOH-(aq) [Al2Si2O5(OH)4]a・nCa2+(s)+nH2O(aq) (5)
[Al2Si2O5(OH)4]a・nCa2+(s)+nH2O(aq) (5)
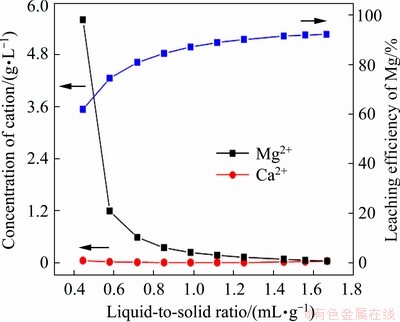
Fig. 5 Leaching curves of Ca2+ and Mg2+ during leaching of Mg2+-leached tailings using lime water
The liquid-to-solid ratio of lime water is an important factor affecting the contents of readily-available magnesium and calcium in the Mg2+-leached tailings, and the results are presented in Table 5. The content of the readily-available calcium and the pH value were gradually increased, while those of the readily-available magnesium were decreased with increasing the liquid-to-solid ratio from 0.40 to 0.93. Also, obvious differences in the contents of the readily-available magnesium and calcium were observed in different layers of the Mg2+-leached tailings. This could be attributed to the flow direction of lime water during the column leaching. When lime water was used for the leaching of the Mg2+-leached tailings, the pH value of the clay mineral of kaolinite was increased rapidly, forming negatively charged surfaces of the kaolinite. Therefore, most of Ca2+ in lime water was electrostatically adsorbed on the leached tailings and exchangeable phase became the main form of the readily-available calcium, which follows the linear relationship of the pH value of clay and the cation exchange capacity [26,35]. Moreover, the content of the exchangeable magnesium was increased compared with that in the Mg2+-leached tailings because of the back-adsorption of Mg2+ with higher pH values. When the liquid-to-solid ratio of lime water was 0.80, the contents of the readily-available magnesium and calcium were 104.4-207.6 and 201.7-1426.3 mg/kg, respectively, and the mass ratios of calcium to magnesium were 1.0-13.7. Furthermore, the pH range of the Mg2+-leached tailings was 5.50-7.75, thus the problem of acidification was solved.
Table 5 Contents of readily-available magnesium and calcium in Mg2+-leached tailing after leaching with lime water

4 Conclusions
(1) Deionized water was used for the leaching of the Mg2+-leached tailings, and the concentrations of the Mg2+ and Ca2+ in the leaching solutions decreased almost exponentially. The contents of readily-available magnesium and hydrolyzable nitrogen were 158.1 and 38.1 mg/kg, respectively, which meets the requirements for plants. But serious loss of the readily-available calcium also occurred.
(2) When CaCl2 solution was used for the leaching of the Mg2+-leached tailings, the contents of the readily-available calcium were 650.1- 2333.0 mg/kg with the CaCl2 concentration of 0.05-0.25 mol/L, and most of them was in the form of water-soluble phase. Serious loss of magnesium occurred owing to the enhanced ion-exchange of calcium salt solutions, and the mass ratios of calcium to magnesium were much higher than 20.
(3) When lime water was used for the leaching of the Mg2+-leached tailings, little Ca2+ was detected in the leaching solution probably because most of Ca2+ is electrostatically adsorbed on the tailings. The content of the exchangeable magnesium was increased, which might be attributed to back-adsorption of Mg2+ with increasing the pH values. When the liquid-to-solid ratio was 0.80, the contents of readily-available magnesium and calcium were 104.4-207.6 and 201.7-1426.3 mg/kg, respectively, and the mass ratio of calcium to magnesium was 1.0-13.7. Therefore, lime water has great potential for the environmental remediation of ion-adsorption rare earth ore after in-situ leaching using magnesium sulfate.
Acknowledgments
The authors are grateful for the financial supports from the National Key Research and Development Program of China (2018YFC1801803) and the Major Research Plan of the National Natural Science Foundation of China (91962211).
References
[1] ABREU R D, MORAIS C A. Study on separation of heavy rare earth elements by solvent extraction with organophosphorus acids and amine reagents [J]. Minerals Engineering, 2014, 61: 82-87.
[2] BINNEMANS K, JONES P T, BLANPAIN B, van GERVEN T, YANG Y X, WALTON A, BUCHERT M. Recycling of rare earths: A critical review [J]. Journal of Cleaner Production, 2013, 51: 1-22.
[3] JORDENS A, CHENG Yin-ping, WATERS K E. A review of the beneficiation of rare earth element bearing minerals [J]. Minerals Engineering, 2013, 41: 97-114.
[4] STEGEN K S. Heavy rare earths, permanent magnets, and renewable energies: An imminent crisis [J]. Energy Policy, 2015, 79: 1-8.
[5] YANG Xiao-Jin, LIN Ai-jun, LI Xiao-liang, WU Yi-ding, ZHOU Wen-bin, CHEN Zhan-heng. China’s ion-adsorption rare earth resources, mining consequences and preservation [J]. Environmental Development, 2013, 8: 131-136.
[6] LIU Ying-hui, CHEN Ji, LI De-qian. Application and perspective of ionic liquids on rare earths green separation [J]. Separation Science and Technology, 2011, 47(2): 223-232.
[7] CHI Ru-an, TIAN Jun. Chemical and metallurgy process of weathered crust elution-deposited rare earth ore [M]. Beijing: Science Press, 2006. (in Chinese)
[8] SHI Xiao-yan, CHENG Hong-wen. Contamination and restoration of abandoned pool and heap leaching sites of rare earth mine [J]. Journal of Rare Earths, 2019, 37(4): 409-417. (in Chinese)
[9] LIU Zu-wen, ZHANG Jun. Mechanism of nitrogenous compounds pollution in the soil of ion-adsorption rare earth mining area [M]. Beijing: Metallurgical Industry Press, 2018. (in Chinese)
[10] China’s Ecological and Environmental Protection Mascot, General Administration of Quality Supervision, Inspection and Quarantine of China. GB 3838―2002 Environmental quality standards for surface water [S]. Beijing: China Environment Publishing Group, 2002. (in Chinese)
[11] XIAO Yan-fei, CHEN Ying-ying, FENG Zong-yu, HUANG Xiao-wei, HUANG Li, LONG Zhi-qi, CUI Da-li. Leaching characteristics of ion-adsorption type rare earths ore with magnesium sulfate [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(11): 3784-3790.
[12] YANG Li-feng, LI Cui-cui, WANG Da-shan, LI Feng-yang, LIU Yan-zhu, ZHOU Xue-zhen, LIU Ming-biao, WANG Xiu-feng, LI Yong-xiu. Leaching ion-adsorption rare earth by aluminum sulfate for increasing efficiency and lowering the environmental impact [J]. Journal of Rare Earths, 2019, 37(4): 429-436.
[13] LI Qiong, HE Zheng-yan, ZHANG Zhen-yue, ZHANG Ting-ting, ZHONG Cheng-bin, CHI Ru-an. Studies on coordination leaching of weathered crust elution-deposited rare earth ore with citrate [J]. Chinese Rare Earths, 2015(1): 18-22. (in Chinese)
[14] HE Zheng-yan, ZHANG Zhen-yue, YU Jun-xia, ZHOU Fang, XU Yuan-lai, XU Zhi-gao, CHEN Zhe, CHI Ru-an. Kinetics of column leaching of rare earth and aluminum from weathered crust elution-deposited rare earth ore with ammonium salt solutions [J]. Hydrometallurgy, 2016, 163: 33-39.
[15] LUO Xian-ping, FENG Bo, WANG Peng-cheng, ZHOU He-peng, CHEN Xiao-ming. The effect of fulvic acid on the leaching of a weathered rare earth ore [J]. Metallurgical and Materials Transactions B, 2015, 46: 2405-2407.
[16] TIAN Jun, TANG Xue-kun, YIN Jing-qun, CHEN Ji, LUO Xian-ping, RAO Guo-hua. Enhanced leachability of a lean weathered crust elution-deposited rare-earth ore: effects of sesbania gum filter-aid reagent [J]. Metallurgical and Materials Transactions B, 2013, 44: 1070-1077.
[17] LAI Fu-guo, GAO Guo-hua, HUANG Li, XIAO Yan-fei, YANG Run, LI Kai-zhong. Compound leaching of rare earth from the ion-adsorption type rare earth ore with magnesium sulfate and ascorbic acid [J]. Hydrometallurgy, 2018, 179: 25-35.
[18] XIAO Yan-fei, FENG Zong-yu, HU Gu-hua, HUANG Li, HUANG Xiao-wei, CHENG Ying-ying, LONG Zhi-qi. Reduction leaching of rare earth from ion-adsorption type rare earths ore with ferrous sulfate [J]. Journal of Rare Earths, 2016, 34(9): 917-923.
[19] HUANG Xiao-wei, YU Ying, FENG Zong-yu, ZHAO Na. A method for the recovery of rare earths from ion-adsorption rare earth ore: Chinese Patent, CN201010128302.9 [P]. 2011-09-21. (in Chinese)
[20] XIAO Yan-fei, FENG Zong-yu, HUANG Xiao-wei, HUANG Li, CHEN Ying-ying, WANG Liang-shi, LONG Zhi-qi. Recovery of rare earths from weathered crust elution-deposited rare earth ore without ammonia-nitrogen pollution: (I) Leaching with magnesium sulfate [J]. Hydrometallurgy, 2015, 153: 58-65.
[21] HUANG Chang-yong, XU Jian-ming. Soil science [M]. Beijing: Chinese Agricultural Press, 1983. (in Chinese)
[22] JIN Ji-yun, BAI You-lu, YANG Li-ping. Efficient technologies and equipments for soil testing [M]. Beijing: Chinese Agricultural Press, 2006. (in Chinese)
[23] DERMONT G, BERGERON M, MERCIER G, RICHER- LAFLECHE M. Soil washing for metal removal: A review of physical/chemical technologies and field applications [J]. Journal of Hazardous Materials, 2008, 152(1): 1-31.
[24] LI Zhong-wu, HUANG Bin, HUANG Jin-quan, CHEN Gui-qiu, XIONG Wei-ping, NIE Xiao-dong, MA Wen-ming, ZENG Guang-ming. Influence of different phosphates on adsorption and leaching of Cu and Zn in red soil [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(2): 536-543.
[25] CAO Ya-ru, ZHANG Shi-rong, WANG Gui-yin, LI Ting, XU Xiao-xun, DENG Ou-peng, ZHANG Yan-zong, PU Yu-lin. Enhancing the soil heavy metals removal efficiency by adding HPMA and PBTCA along with plant washing agents [J]. Journal of Hazardous Materials, 2017, 339: 33-42.
[26] ZHANG Zhen-yue, HE Zheng-yan, ZHOU Fang, ZHONG Cheng-bin, SUN Ning-jie, CHI Ru-an. Swelling of clay minerals in ammonium leaching of weathered crust elution- deposited rare earth ores [J]. Rare Metals, 2018, 37(1): 72-78.
[27] JING Qing-xiu, CHAI Li-yuan, HUANG Xiao-dong, TANG Chong-jian, GUO Huan, WANG Wei. Behavior of ammonium adsorption by clay mineral halloysite [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(7): 1627-1635.
[28] Ministry of Agriculture of the People’s Republic of China. NY/T 1121.2―2006 Soil testing (Part 2): Method for determination of soil pH [S]. Beijing: Chinese Agricultural Press, 2006. (in Chinese)
[29] National Forestry Administration. LY/T 1228―2015 Nitrogen determination methods of forest soils [S]. Beijing: Standards Press of China, 2015. (in Chinese)
[30] Ministry of Agriculture of the People’s Republic of China. NY/T 1121.13―2006 Soil testing (Part 13): Method for determination of soil exchangeable calcium and magnesium [S]. Beijing: Chinese Agricultural Press, 2006. (in Chinese)
[31] Ministry of Agriculture of the People’s Republic of China. NY/T 1121.16―2006 Soil testing (Part 16): Method for determination of total water-soluble salt [S]. Beijing: Chinese Agricultural Press, 2006. (in Chinese)
[32] TANTON T W, RYCROFT D W, HASHIMI M. Leaching of salt from a heavy clay subsoil under simulated rainfall conditions [J]. Agricultural Water Management, 1995, 27(3-4): 321-329.
[33] General Administration of Quality Supervision, Inspection and Quarantine of the People's Republic of China, Standardization Administration of China. GB/T 14848―2017 Standard for groundwater quality [S]. Beijing: Standards Press of China, 2017. (in Chinese)
[34] WANG Ai-hua, DUAN Zeng-qiang, DONG Jin-long, ZHAO Yu, TANG Ying, LI Xun, SHEN Wen-zhong. Effect of soluble salts on determination of soil pH and its elimination [J]. Acta Pedologica Sinica, 2014, 51(6): 1298-1308. (in Chinese)
[35] MA C, EGGLETON R A. Cation exchange capacity of kaolinite [J]. Clays and Clay Minerals, 1999, 47(2): 174-180.
范 波1,2,赵龙胜1,3,冯宗玉1,3,刘德鹏1,3,尹伟强1,3,龙志奇1,3,黄小卫1,3
1. 有研科技集团有限公司 稀土材料国家工程研究中心,北京 100088;
2. 北京有色金属研究总院,北京 100088;
3. 有研稀土新材料股份有限公司,北京 100088
摘 要:研究采用去离子水、氯化钙溶液、石灰水淋洗剂淋洗,离子吸附型稀土矿硫酸镁浸取尾矿中镁、钙的淋出行为。结果表明,浸取尾矿中的镁主要以水溶态形式存在,这部分镁易被淋洗脱除。采用石灰水淋洗,淋洗剂中的绝大部分钙可被静电吸附在淋洗后尾矿中,同时浸取尾矿中的部分水溶态镁因Mg2+在黏土矿物上的反吸附作用而逐渐转化为交换态镁,这与淋洗后尾矿pH值的升高有关。采用石灰水淋洗,当液固比为0.80时,淋洗后尾矿中速效镁、钙含量分别为104.4~207.6和201.7~1426.3 mg/kg,满足植物的生长需求。该研究为离子吸附型稀土矿原地浸矿场的环境修复提供一种有潜力的解决方案。
关键词:离子吸附型稀土矿;硫酸镁;环境修复;淋出行为;石灰水
(Edited by Wei-ping CHEN)
Corresponding author: Zong-yu FENG; Tel: +86-10-82241180; E-mail: fengzongyu1120@163.com
DOI: 10.1016/S1003-6326(21)65495-X
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier B.V. & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier B.V. & Science Press

