
Influence of freezing rate on microstructure and electrochemical properties of Mg-2%Ga alloys
FENG Yan, WANG Ri-chu, PENG Chao-qun
School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 12 June 2010; accepted 30 September 2010
Abstract: Equilibrium freezing curve of Mg-2%Ga (mass fraction) alloy was calculated by CALPHALD method. Microstructures of the melted Mg-2%Ga alloys solidified by iron and copper moulds, respectively, were investigated using OM and SEM. Electrochemical properties of the Mg-2%Ga alloys with different freezing rates were measured by galvanostatic, potentiodynamic and electrochemical impedance spectroscopy tests. The results show that solidification by copper mould leads to intergranular Mg5Ga2 compounds with small size and large number density. Less adsorbent of Mg+ and oxide corrosion products occur on the surface of the Mg-2%Ga alloys solidified by copper mould, producing lower corrosion current density of 1.8×10-5 mA/cm2. In the galvanostatic tests with 100 mA/cm2 current density, more negative stable potential of -1.604 V exists in the Mg-2%Ga alloys solidified by iron mould due to the lower freezing rate, which leads to smaller inductive and capacity time constants as well as shorter activity time and better electrochemical activity.
Key words: Mg-Ga alloy; electrode materials; electrochemical reaction; microstructure; computer simulation
1 Introduction
Magnesium alloys are widely used in seawater batteries and sacrificial anodes due to many advantages, such as rapid activation, low specific mass, low electrode potential and high current capacity[1-3]. Since 1960s, magnesium alloy batteries have been the subject of comprehensive interest and have been developed for some military and commercial applications, such as torpedo, electromotive and unmanned underwater vehicle power source.
The developed magnesium anode materials with high cell voltage are AP65 (Mg-6%Al-5%Pb), Mg-7%Tl-5%Al[1] and Mg-Hg alloys[4]. The seawater battery using Mg-Hg alloys as anodes can reach a higher specific energy of 150 W・h/kg[5-6], compared with lead acid battery of 30 W・h/kg. However, the good results of the Mg anodes obtained in high power seawater battery still meet with problems such as bad deformability, large self-corrosion velocity and low current efficiency[7-8]. In order to solve these problems, gallium was added into magnesium anodes due to its high hydrogen revolution over-potential and good electrochemical activity[4, 9-11]. For getting fine microstructure, heat treatment of the Mg-Ga anode materials was studied[12-14]. But there are few reports about the influence of the solidifying rates on the electrochemical and corrosion properties of the Mg-Ga alloys. According to researches in Ref.[15], fine distribution of the alloying elements in magnesium anode materials does not imply good anodic properties. The present study will focus on the influence of the solidifying rate on the microstructure and electrochemical properties of the Mg-2%Ga alloys.
2 Experimental
The Mg-2%Ga (mass fraction) alloys were melted in a muffle furnace using 99.9% purity materials and then solidified by iron and copper mould, respectively. The chemical compositions of the specimens were determined by atomic absorption spectrochemical analysis. The deviation of the main composition was under 0.5%.
Microstructure and corroded surface of the specimens were observed using OM and SEM. And SEM observation was carried out using Sirion 200. Potentiodynamic and galvanostatic experiments were performed with a Potentiostat-Galvanostat (Model 263A) in 3.5% (mass fraction) NaCl solution. The scanning rate of potentiodynamic test was 5 mV/s and the anodic current density in the galvanostatic test was 100 mA/cm2 and the testing time was 1 300 s. Electrochemical impedance spectroscopy (EIS) measurements were performed using a Potentiostat-Galvanostat (Model 263A) and a 1255 Solartron frequency response analyzer. The impedance measurements were made over a frequency range of 0.005 Hz to 100 kHz (15 points per decade) with a 10 mV AC signal and interpreted in terms of equivalent circuit (EC) with frequency dependent components. Those specimens were polished with emery paper and buffed to a mirror finish. Each of them was sealed with epoxy resin except for an exposed surface of 10 mm×10 mm submitted to the electrochemical tests in a three-electrode cell. A platinum sheet was used as the auxiliary electrode and a saturated calomel electrode (SCE) was used as the reference electrode.
3 Results and discussion
3.1 Calculation of equilibrium freezing curve of Mg-2%Ga alloy
Using CALPHALD method and the Mg-Ga thermodynamic database[16], the equilibrium freezing curve of the Mg-2%Ga liquid alloys was calculated (Fig.1). Mg with HCP structure was first solidified in Mg-2%Ga liquid alloys. The number of the HCP phase increased with decreasing the temperature. When the temperature decreased to nearly 415 °C, Mg5Ga2 was solidified from the liquid and the temperature did not change until total solidification of liquid. Finally, the Mg-2%Ga alloy contained about 2.8% Mg5Ga2 and 97.2% Mg.
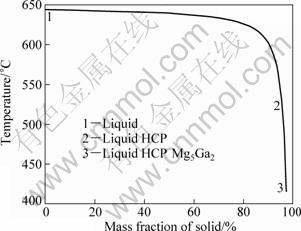
Fig.1 Equilibrium freezing curve of Mg-Ga alloy
Figure 2 shows the composition of the Mg-2%Ga liquid alloy during equilibrium freezing. It can be seen that the content of Mg in the Mg-2%Ga liquid alloy decreases with decreasing the temperature, while the content of Ga increases. This leads to the solidification of Mg5Ga2 compounds at the last stage.
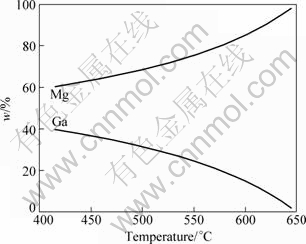
Fig.2 Composition of Mg-Ga liquid alloy during equilibrium freezing
3.2 Influence of freezing rate on microstructure of Mg-2%Ga alloy
Figure 3 shows the microscopic structures of the Mg-2%Ga alloy with different freezing rates. According to the calculated freezing curve, the intergranular and transgranular compounds are Mg5Ga2 in the Mg-2%Ga alloys. From Fig.3 it can be observed that the Mg-2%Ga alloy solidified by copper mould has smaller grain size and larger number density of Mg5Ga2 compounds than the alloy solidified by iron mould. It may be due to the fast freezing rate.

Fig.3 Microscopic structures of Mg-2%Ga alloys with different freezing rates solidified by copper mould (a) and iron mould (b)
3.3 Influence of freezing rate on electrochemical corrosion behavior
3.3.1 DC measurements
To obtain information about the influence of freezing rate on the electrochemical corrosion behavior of the Mg-2%Ga alloys, galvanostatic and potentiodynamic curves were conducted in a 3.5 mol/L NaCl solution (Fig.4). The activation of the surface is confirmed by the trend of the curve φ―t. The stable potential was calculated from the curves (Table 1). From Fig.4(a) and Table 1, it can be observed that polarization exists in the Mg-2%Ga alloy solidified by copper mould but stable potential of -1.604 V was obtained in the alloy solidified by iron mould, which demonstrates better electrochemical activity.
During potentiostatic polarizations, a steady state current was attained for each applied potential step. Near the rest potential the anodic dissolution rate increases with the increase of the potential, and steeper anodic polarization curves exist in the alloy solidified by iron mould. Pitting potential of about -1.60 V occurs in the Mg-2%Ga alloys with different freezing rates. From the galvanostatic measurements, it can be seen that the stable potential of the alloy solidified by iron mould is close to -1.6 V. The oxide film in this alloy destroyed quickly in the measurement, which leads to dissolution of the Mg matrix. The stable potential of the alloy solidified by copper mould is close to -1.3 V, which is more positive than pitting potential. So the oxide film on the surface is hard to destroy, which impedes the electrochemical activation reaction and leads to polarization in the galvanostatic measurements.
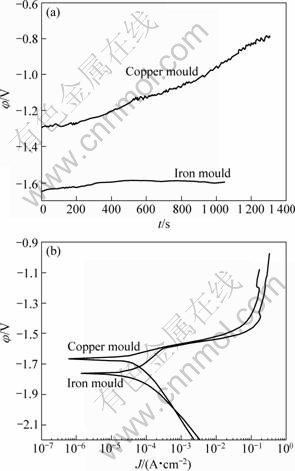
Fig.4 Galvanostatic and potentiodynamic curves of Mg-2%Ga alloys: (a) Galvanostatic curves; (b) Potentiodynamic curves
Table 1 Electrochemical corrosion properties of Mg-2%Ga alloys

The corrosion potentials and corrosion current densities, calculated by Tafel linear extrapolation, are listed in Table 1. It can be seen that the corrosion current density of the alloy solidified by iron mould is 3.5 times the one of the alloy solidified by copper mould. It may be due to the damage of the oxide film on the surface. Figure 5 shows the corrosion surface of the alloys after potentiodynamic measurements. It can be observed that a lot of loose corrosion products exist in the alloy solidified by iron mould while compact oxide film exists in the alloy solidified by copper mould. The corrosion resistance depends on the properties of two electric poles in galvanic couple corrosion. According to the microscopic structures of the alloys in Fig.3(a), the closer grain and larger number density of second phases in the alloy solidified by copper mould lead to more homogeneous structure and smaller corrosion driving force.

Fig.5 Corrosion surface of Mg-2%Ga alloys after potentio- dynamic measurements: (a) Solidified by copper mould; (b) Solidified by iron mould
3.3.2 AC measurements
The EIS results are shown in Fig.6. It can be observed that capacitive impedance appears in high frequency. Inductive reactance appears in low frequency and two time constants occurs in the bode diagrams. The EC can be attained, as shown in Fig.7, where R0 is the electrolyte resistance; Cdlt and Rct refer to the capacitance of the double layer and the charge transfer resistance in the Faradic reaction; L and Ru refer to the inductive reactance and resistance of adsorbent of Mg+ and oxide on the surface. A method of parameter adjustment based on the equivalent circuit in Fig.6 was applied and the results are shown in Table 2.
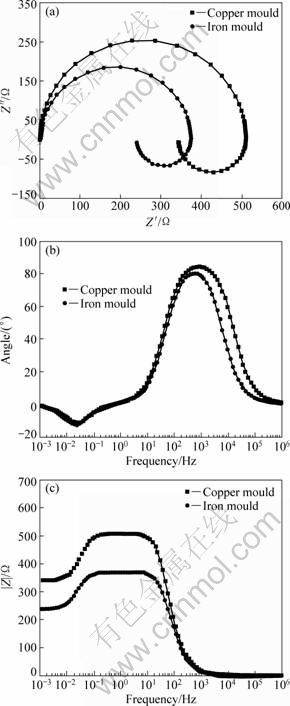
Fig.6 Nyquist and Bode diagrams of Mg-2%Ga alloys in 3.5%NaCl solution
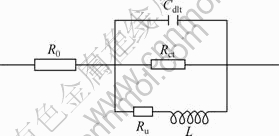
Fig.7 Equivalent circuits of Mg-2%Ga alloys/3.5% NaCl interfaces
Table 2 Electrochemical parameters obtained by fitting analysis of Nyquist diagrams of Mg-2%Ga alloys

The equation of τ=L/Ru is the time constant of the inductive reactance, which presents the stable time of the inductive reactance. The expression of RctCdlt presents the stable time of the electric double layer. From Table 2, the values of τ in the Mg-2%Ga alloys solidified by iron and copper mould can be calculated to be 6.52 and 7.14, respectively. The values of RctCdlt in the Mg-2%Ga alloys solidified by iron and copper moulds can be calculated to be 3 444.2 and 4 060.8, respectively. According to Ref.[17], smaller time constants of the capacitive impedance and inductive reactance lead to shorter activity time and better electrochemical activity. So the Mg-2%Ga alloy solidified by iron mould has more negative stable potential than the Mg-2%Ga alloy solidified by copper mould, as seen in Fig.4(a).
Considering that Ru refers to the resistance of adsorbent of Mg+ and oxide on the surface, it can be seen that smaller value of Ru in the Mg-2%Ga alloy solidified by copper mould leads to smaller number density of the adsorbent. It produces smaller moving resistance of the reactant H+, resultant Mg(OH)2 and reaction promoter Cl- in the corrosion reaction. Reversely, the larger value of Ru in the Mg-2%Ga alloy solidified by iron mould leads to larger number density of the adsorbent of Mg+ and oxide on the surface. This increases moving resistance of the reactant H+, resultant Mg(OH)2 and reaction promoter Cl- in the corrosion reaction and decreases the velocity of the corrosion reaction. The Mg-2%Ga alloy solidified by iron mould has larger corrosion current density than the alloy solidified by copper mould, as seen in Table 1.
4 Conclusions
1) Using CALPHALD method the equilibrium freezing curve of the Mg-Ga liquid alloys was calculated. Through thermodynamic calculation the Mg-2%Ga alloy after equilibrium freezing contains 2.8% Mg5Ga2 and 97.2% Mg.
2) High freezing rate exists in the Mg-2%Ga alloys solidified by copper mould and leads to smaller size of grain and larger number density of intergranular Mg5Ga2 compounds. Due to less adsorbent of Mg+ and oxide corrosion products on the surface, lower corrosion current density of 1.8×10-5 mA/cm2 occurs in the Mg-2%Ga alloy solidified by copper mould.
3) In the galvanostatic measurements with current density of 100 mA/cm2, more negative stable potential of -1.604 V occurs in the Mg-2%Ga alloy solidified by iron mould due to the lower freezing rate. This is due to small inductive and capacity time constants as well as short activity time and better electrochemical activity.
References
[1] AURBACH D, SURESH G S, LEVI E, MITELMAN A, MIZRAHI O, CHUSID O, BRUNELLI M. Progress in rechargeable magnesium battery technology [J]. Advanced Materials, 2007, 19(23): 4260-4267.
[2] GIRAUDET J, CLAVES D, GUERIN K, DUBOIS M, MASIN F, HAMWI A. Magnesium batteries: Towards a first use of graphite fluorides [J]. Journal of Power Sources, 2007, 173(1): 592-598.
[3] ZHAO H Y, BIAN P, JU D Y. Electrochemical performance of magnesium alloy and its application on the sea water battery [J]. Journal of Environmental Sciences, 2009, 21(1): S88-S91.
[4] FENG Y, WANG R C, YU K, PENG C Q, ZHANG J P, ZHANG C. Activation of Mg-Hg anodes by Ga in NaCl solution [J]. Journal of Alloys and Compounds, 2009, 473(1-2): 215-219.
[5] BAGSHAW N E, WARRELL S. Anodic dissolution of magnesium alloys related to lead chloride-magnesium seawater batteries [C]//THOMPSON J. Power Sources: Research and Development in Non-Mechanical Electrical Power Sources. London: E-Publishing Inc, 1981: 117-139.
[6] FIRA S S, KIBL L, LIW L W. Water-activated disposable and long shelf-life microbatteries [J]. Sensors and Actuators A, 2004, 111(1): 79-86.
[7] VENKATESARA R K. Performance evaluation of Mg-AgCl batteries for under water propulsion [J]. Denfence Science Journal, 2001, 5(2): 161-170.
[8] GUANG L S, ANDREJ A. Corrosion mechanisms of magnesium alloys [J]. Advanced Engineering Materials, 1999, 1(1): 11-33.
[9] FLAMINI D O, SAIDMAN S B, BESSONE J B. Aluminium activation produced by gallium [J]. Corrosion Science, 2006, 48(6): 1413-1425.
[10] FENG Y, WANG R C, YU K, PENG C Q, LI W X. Influence of Ga content on electrochemical behavior of Mg-5at%Hg anode materials [J]. Materials Transaction, JIM, 2008, 49(5): 1077-1080.
[11] PINSSCO M R, ANGELINI E, CORDANO E, PRSALBINO F. Structural characterization and corrosion resistance of Ga-precious metal alloys formed by liquid-solid reaction at room temperature [J]. Journal of Alloys and Compounds, 2001, 317-318(12): 411-418.
[12] FENG Yan, WANG Ri-chu, YU Kun, LI Wen-xian. Influence of heat treatment on electrochemical behavior of the Mg anode materials [J]. Journal of Central South University of Technology, 2007, 14(2): 12-15.
[13] XI B H, XIA T. Survey of power battery for torpedo propulsion [J]. Torpedo Technology, 2005, 13(2): 7-12.
[14] FONT S, DESCROIX J P, SARRE G. Advanced reserve batteries for torpedoes propulsion [C]//CHERRY H. Proceedings of the 31st Power Sources Symposium. Penniton: Electrochemical Soc, 1984: 362-368.
[15] LIU N, WANG J L, WANG L D, WU Y M, WANG L M. Electrochemical corrosion behavior of Mg-5Al-0.4Mn-xNd in NaCl solution [J]. Corrosion Science, 2009, 51(6): 1328-1333.
[16] FENG Yan, WANG Ri-chu, LIU Hua-shan, JIN Zhan-peng. Thermodynamic reassessment of the magnesium-gallium system [J]. Journal of Alloys and Compounds, 2009, 486: 581-585.
[17] JIA Zheng, DAI Chang-song, CHEN Lin. Electrochemical measurements [M]. Beijing: Chemical Industry Publisher, 2006. (in Chinese)
凝固速度对Mg-2%Ga合金显微组织和电化学性能的影响
冯 艳,王日初,彭超群
中南大学 材料科学与工程学院,长沙 410083
摘 要:采用CALPHALD方法计算Mg-2%Ga合金的平衡凝固曲线。分别采用铁模和铜模冷却,用熔炼铸造法制备该合金,并用金相OM和扫描电镜SEM方法分析其显微组织,用恒电流法、动电位扫描和交流阻抗法测量不同冷速对Mg-2%Ga合金电化学性能的影响。结果表明:铜模冷却的Mg-2%Ga合金晶界存在尺寸小、数量多的Mg5Ga2化合物,其合金表面吸附了较少的Mg+和氧化腐蚀产物,因此产生较低的腐蚀电流密度1.8×10-5 mA/cm2。铁模冷却的Mg-2%Ga合金在恒流100 mA/cm2检测时具有更负的稳定电位-1.604 V,这是由于铁模冷却产生较低冷却速度导致双电层具有较小的感抗和容抗时间常数,因此产生较短的活化时间和更好的电化学活性。
关键词:Mg-Ga合金;电极材料;电化学反应;显微组织;计算机模拟
(Edited by YANG Hua)
Foundation item: Project (JPPT-115-4-1682) supported by the National Defense Science and Technology Industry Committee of China
Corresponding author: FENG Yan; Tel: +86-731-88836638; E-mail: fengyanmse@yahoo.com.cn
DOI: 10.1016/S1003-6326(11)60820-0