Flotation performances of polymorphic pyrrhotite
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2012���1��
�������ߣ������� ������ ��ά�� ����
����ҳ�룺238 - 243
Key words��flotation; pyrrhotite; pulp potential; contact angle
Abstract:
The floatability of different crystalline structures of pyrrhotite (monoclinic and hexagonal) was studied. It is shown that the floatability of monoclinic and hexagonal has obvious difference, and that the flotation recovery of monoclinic pyrrhotite is larger than that of hexagonal pyrrhotite using different collectors. When butyl dithiophosphate is used as the collector, the recovery is larger than that by sodium butyl xanthate and sodium diethyl dithiocarbamate. At the pH values ranging from 6 to 9, monoclinic pyrrhotite can be floated well, and the flotation recovery is higher than 90%. Monoclinic and hexagonal pyrrhotites are more easily activated by Cu2+ in acidic conditions than in alkaline conditions. But Cu2+ cannot activate hexagonal pyrrhotite using sodium diethyldithiocarbamate as the collector. By the measurement of contact angle, it is indicated that monoclinic and hexagonal pyrrhotites float well and are easily activated by Cu2+ when dithiophosphate is used as the collector. Using sodium diethyl dithiocarbamate as a collector, the relationship between potential and pH range for pyrrhotite flotation is established. At pH 5, the optimal potential range for flotation of monoclinic pyrrhotite is about 125-580 mV (vs SHE), with the maximum flotation occurring at about 350 mV (vs SHE); the optimal potential range for flotation of hexagonal pyrrhotite is 200-580 mV (vs SHE), with the maximum flotation occurring at about 300 mV (vs SHE).
J. Cent. South Univ. (2012) 19: 238-243
DOI: 10.1007/s11771-012-0997-2![]()
HE Ming-fei(������), QIN Wen-qing(������), LI Wei-zhong(����), JIAO Fen(����)
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: The floatability of different crystalline structures of pyrrhotite (monoclinic and hexagonal) was studied. It is shown that the floatability of monoclinic and hexagonal has obvious difference, and that the flotation recovery of monoclinic pyrrhotite is larger than that of hexagonal pyrrhotite using different collectors. When butyl dithiophosphate is used as the collector, the recovery is larger than that by sodium butyl xanthate and sodium diethyl dithiocarbamate. At the pH values ranging from 6 to 9, monoclinic pyrrhotite can be floated well, and the flotation recovery is higher than 90%. Monoclinic and hexagonal pyrrhotites are more easily activated by Cu2+ in acidic conditions than in alkaline conditions. But Cu2+ cannot activate hexagonal pyrrhotite using sodium diethyldithiocarbamate as the collector. By the measurement of contact angle, it is indicated that monoclinic and hexagonal pyrrhotites float well and are easily activated by Cu2+ when dithiophosphate is used as the collector. Using sodium diethyl dithiocarbamate as a collector, the relationship between potential and pH range for pyrrhotite flotation is established. At pH 5, the optimal potential range for flotation of monoclinic pyrrhotite is about 125-580 mV (vs SHE), with the maximum flotation occurring at about 350 mV (vs SHE); the optimal potential range for flotation of hexagonal pyrrhotite is 200-580 mV (vs SHE), with the maximum flotation occurring at about 300 mV (vs SHE).
Key words: flotation; pyrrhotite; pulp potential; contact angle
1 Introduction
Pyrrhotite is a common sulphide mineral which has a disordered NiAs structure resulting from a nonstoichiometric composition [1]. Several types of pyrrhotite are known to exist in nature whose general formula is Fe1-xS with x<0.13. The most common formula is monoclinic Fe7S8, while a rare hexagonal stoichiometric pyrrhotite (FeS) is known as troilite. However, pyrrhotite rarely exists as a pure mineral phase and usually consists of a mixture of hexagonal and monoclinic phases [2]. It is found in nature to be commonly associated with pyrite, chalcopyrite, galena and other sulfide minerals. In many flotation plants, pyrrhotite is rejected to the flotation tailings as a waste product. However, a strong interest in pyrrhotite recovery is developed in certain instances. Specifically, the importance of pyrrhotite flotation is evident in the processing of the complex ores from Yunnan Province, China. Mengzi ore-body contains the valuable minerals, such as galena (PbS), marmatite (Fe, ZnS), cassiterite (SnO2), pyrite (FeS), pyrrhotite (Fe1-xS), and precious metals such as silver and indium. The priority of the operations in Mengzi is recovery of lead, zinc, tin and sulfur, while the sulfur should be floated first with the main sulfur concentrate being pyrite and pyrrhotite before the cassiterite gravity concentration in order to get qualified tin concentrate. Consequently, it is important to achieve high pyrrhotite recovery during flotation.
The mechanisms and kinetics of the oxidation reactions for pyrrhotite are poorly understood, and there is also a scarcity of detailed studies on the effect of different crystal structures of pyrrhotite [3]. Under the traditional flotation conditions, such as at pH 9-9.5, no natural pyrrhotite floatability is observed, pyrrhotite floatability with collector is low, and activators (copper and lead) lose their effectiveness in most cases, precipitating as hydroxides. Significantly improved pyrrhotite flotation can be achieved at pH<5 [4]. It was reported [5] that hexagonal pyrrhotite is more reactive than monoclinic pyrrhotite, and VANYUKOV and RAZUMOVSKAYA [6] noted that oxidation rates tend to increase with an increase in the sulfur to iron ratio in pyrrhotite. In this work, the floatability of different crystal structures of pyrrhotite is studied and the electrochemical flotation behavior of pyrrhotite is investigated.
2 Experimental
2.1 Materials
Natural pyrrhotite was provided by Mengzi Mining & Metallurgy Corporation. In nature, pyrrhotite minerals occur with significant variation in structure and/or composition. Generally, a mixture of hexagonal and monoclinic phases is predominant. ARNOLD [2] made the analysis of 82 terrestrial pyrrhotite samples. Seventy percent was a mixture of h-Po and m-Po, 10% contained hexagonal pyrrhotite only, and 9% contained monoclinic pyrrhotite only. Mengzi ore-body has two lodes, with one mainly containing hexagonal pyrrhotite, and the other mainly containing monoclinic pyrrhotite.
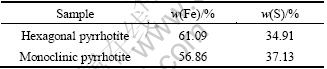
Pyrrhotite specimen was cut and polished for contact angle and X-ray diffraction measurements. Its grains were ground in a ceramic ball mill and stored under nitrogen condition for the flotation. The X-ray diffraction analysis shows that the purity of the two types of pyrrhotite are greater than 94% (Fig. 1 and Fig. 2) and the chemical analysis of each sample is given in Table 1. The reagents used to adjust pulp potential were ammonium persulfate ((NH4)2S2O8) and sodiumdithionite (Na2S2O4). All reagents in the flotation tests were of analytical reagent grade except that the frother butyl aether alcohol was in the industry reagent grade. The dosage of the frother was 10 mg/L.
Fig. 1 X-ray powder diffraction pattern of hexagonal pyrrhotite
Fig. 2 X-ray powder diffraction pattern of monoclinic pyrrhotite
Table 1 Chemical composition of mineral samples
2.2 Flotation tests
The flotation tests of the pure mineral were conducted in common small cells with a volume of 40 mL. A sample of 2.0 g for each experiment was weighed before its surface was cleaned by ultrasonic generator for 5 min to remove the oxidation film. Pulp potential was adjusted in terms of the experiment purpose using reagents. The pulp potential was measured before adding the frother. The electrodes which were used to measure the pulp potential were a saturated calomel electrode (SCE, 0.245 V vs SHE) and a platinum electrode with geometric surface area of 0.28 cm2. The SCE was checked periodically to ensure accurate measurement and the platinum electrode was cleaned first before each experiment. The flotation time was 4 min. The flotation recovery (R) was calculated from R=m1/(m1+m2), where m1 and m2 are floated and unfloated product masses, respectively.
2.3 Contact angle measurements
Contact angle measurements were carried out using the method developed by SUTHERLAND and WARK [7]. Only freshly prepared mineral samples were used without an extensive polishing operation to avoid aging of surfaces. The samples were not allowed to dry or contact air before contact angle tests. The prepared specimen was placed on stable support with the polished surface facing downward in the rectangular glass chamber of the apparatus. The chamber was filled with the desired solution. The sample was then treated in the solution of reagent for 10 min before measurements. A small air bubble was produced at the tip of a specially designed U-shaped needle using a microsyringe. The bubble was then released from the needle tip from a fixed distance (1 cm) below the submerged specimen surface. Released bubble was then captured on the solid surface as a result of buoyant transport and attachment. After the air bubble attached to the solid surface, the angles at each side of the bubble were measured. The measurements were repeated three times for each condition and the mean value was calculated.
3 Results and discussion
3.1 Effect of pH on recovery of pyrrhotites with different collectors
The effects of pulp pH on the flotation recovery of pyrrhotites (hexagonal, monoclinic) are shown in Fig. 3 and Fig. 4. It can be seen that the flotation behaviors of hexagonal pyrrhotite have a remarkable difference from monoclinic pyrrhotite using different collectors (sodium butyl xanthate, sodium diethyl dithiocarbamate and butyl dithiophosphate). The floatability of monoclinic pyrrhotite is larger than that of hexagonal pyrrhotite when pH is from 4 to 12.5. The results demonstrate that the recovery of hexagonal pyrrhotite decreases with increasing pH value using sodium butyl xanthate or sodium diethyl dithiocarbamate as collector, whereas the flotation recovery of hexagonal pyrrhotite increases in the acidic solution at first using butyl dithiophosphate as collector, then it reaches the maximum (87%) at pH 7, but it drops when the pH is larger than 7 (Fig. 3). Butyl dithiophosphate exhibits high collectivity not only for hexagonal pyrrhotite, but also for monoclinic pyrrhotite, as can be seen from Fig. 4. When butyl dithiophosphate is used as the collector, the recovery is larger than that by sodium butyl xanthate and sodium diethyl dithiocarbamate. At the pH values range from 6 to 9, monoclinic pyrrhotite can be floated well, and the flotation recovery is higher than 90%.
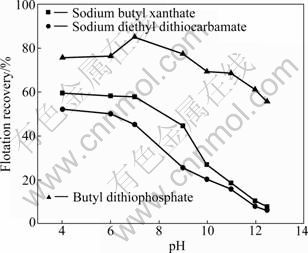
Fig. 3 Effect of pH on recovery of hexagonal pyrrhotite with different collectors (1��10-4 mol/L)
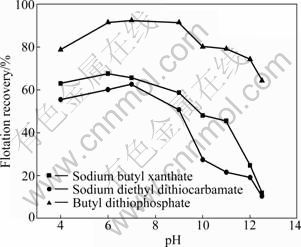
Fig. 4 Effect of pH on recovery of monoclinic pyrrhotite with different collectors (1��10-4 mol/L)
3.2 Activation of pyrrhotite
YU [8] noted that copper sulfate can significantly improve pyrrhotite recovery when pH is smaller than 8. In the experiments, Cu2+ directly exchanges with Fe2+ at the pyrrhotite surface, accounting for the improved flotation of pyrrhotite:
Cu2++FeS=CuS +Fe2+ (1)
But the effectiveness of Cu2+ for the activation of pyrrhotite in alkaline solutions is not fully understood. Generally, activation of pyrrhotite by copper and lead is significant in acidic solutions, but in some cases, it has been found to be negligible in neutral and alkaline solutions [4]. In order to improve recovery in flotation of pyrrhotites with different collectors, using Cu2+ as activator, experiments for activation of pyrrhotites by cupric ions (sulfate) at an initial concentration of 1��10-4 mol/L were carried out and the results are shown in Fig. 5 and Fig. 6. These results show that when the pH is above 7, the presence of cupric ions does not influence the recovery of hexagonal pyrrhotite compared with that in the absence of Cu2+ (Fig. 3). Pyrrhotite particles are likely to be well oxidized and covered with ferric hydroxide/oxide, which may inhibit reaction with the underlying mineral surface in alkaline solutions. In contrast, the recovery of monoclinic pyrrhotite increases greatly. Especially, it is effectively activated using butyl dithiophosphate as collector. For example, the recovery of monoclinic pyrrhotite increases greatly from 68.8% to 92.5% with the addition of Cu2+ at pH 11. When the pH is smaller than 7, copper sulfate significantly improves the recovery of pyrrhotites. The results demonstrate that the recovery of monoclinic pyrrhotite increases from 59.74% to 70.77% for sodium butyl xanthate, from 52.32% to 63.32% for sodium diethyl dithiocarbamate and from 75.92% to 85.62% for butyl dithiophosphate at pH 4. It should be noted that the recovery of monoclinic pyrrhotite is larger than that of hexagonal pyrrhotite with the addition of Cu2+ in acidic solution for the three collectors.
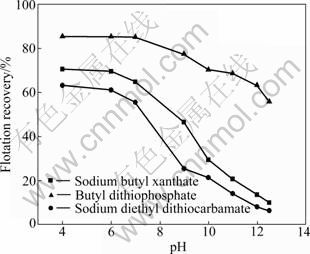
Fig. 5 Effect of pH on recovery of hexagonal pyrrhotite in presence of Cu2+ (1��10-4 mol/L)
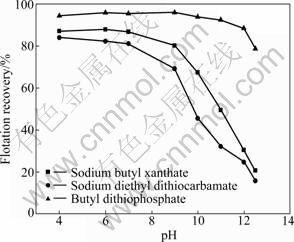
Fig. 6 Effect of pH on recovery of monoclinic pyrrhotite in presence of Cu2+ (1��10-4 mol/L)
3.3 Effect of pulp potential on pyrrhotite flotation in presence of butyl dithiophosphate
The pulp potential values influence significantly the floatability of pyrrhotite either in the absence of a collector or in the presence of a collector. HAMILTON and WOODS [9] pointed out that pyrrhotite-collectorless flotation results from elemental sulfur formation on the mineral surface since sulfur is strongly hydrophobic and may remain stable for a long time, even in alkaline solutions. MILLER et al [4] reported that pyrrhotite is thermodynamically and kinetically unstable and is easily oxidized to ferric hydroxide/oxide, or intermediate compounds, depending on the oxidation potential level and solution pH, and they pointed out that appropriate control of the oxidation potential should be important during flotation process for improved flotation recovery. The relationship between floatability and potential pulp for pyrrhotite using ethyl xanthate as collector in different pH values was investigated by ZHANG [10], displaying potential-pH range for floatable pyrrhotite.
The effect of potential-pH on the flotation recovery of pyrrhotites is given in Fig. 7 and Fig. 8. The results show that the pulp potential decreases with increasing pH value for both types of pyrrhotite. With pH fixed at 5, 9 and 12 by different buffer solutions, respectively, the dependence of pyrrhotite flotation recovery on pulp potential is shown in Fig. 9 and Fig. 10. It can be shown that the flotation of pyrrhotite may be possible only in a suitable range of the pulp potential at the corresponding pH. If the upper and lower potential limits for flotation are defined at 50% flotation recovery above, a relationship between pyrrhotite flotation and upper (��u) and lower (��l) potential limits can be established and is presented in Fig. 11. It can be seen that the flotation of hexagonal pyrrhotite may occur only in a range of pulp potential ��l<��h<��u at different pH values and flotation of monoclinic pyrrhotite may occur in a range of pulp potential ��m,l<��m<��m,u at different pH values. The potential range for flotation recovery decreases with increasing pH. In acidic solution, there is a wider range of pulp potential for flotation. For example, at pH 5, the optimal potential range for flotation of monoclinic pyrrhotite is 125-580 mV with the maximum flotation occurring at about 350 mV, and the optimal potential range for flotation of hexagonal pyrrhotite is 200- 580 mV with the maximum flotation occurring at about 300 mV.
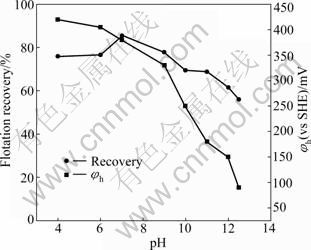
Fig. 7 Flotation recovery of hexagonal pyrrhotite and pulp potential as function of pH
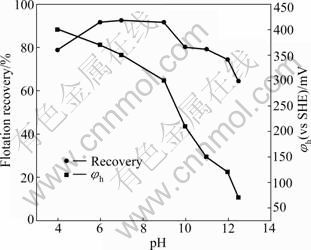
Fig. 8 Flotation recovery of monoclinic pyrrhotite and pulp potential as function of pH
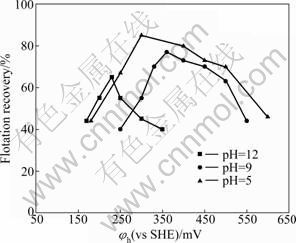
Fig. 9 Dependence of hexagonal pyrrhotite flotation recovery on pulp potential
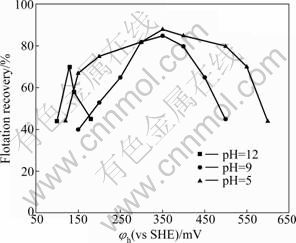
Fig. 10 Dependence of monoclinic pyrrhotite flotation recovery on pulp potential
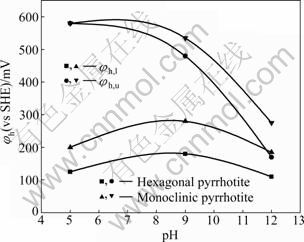
Fig. 11 Flotation potential upper and lower limit of pyrrhotites at different pH values
3.4 Contact angle measurements
The Young��s equation is the thermodynamic equilibrium of condition for an ideal solid-liquid-gas capillary system. The contact angle, ��, is determined by three interfacial tensions, ��sg, ��sl and ��lg as an equilibrium property of the system (��sg, ��sl and ��lg are the solid�Cgas, the solid�Cliquid and the liquid�Cgas interfacial tensions, respectively). However, the validity of Young��s equation requires that the solid surface is smooth, homogenous, inert, non-porous and nondeformable which is usually not met by real surfaces [11-12]. Previous studies showed that the contact angles are usually preferred for flotation systems because these contact angles are supposed to correlate much better with flotation response [13-14]. The natural hydrophobicity of pyrrhotite was examined in air from pH 3.0 to pH 9.2 based on captive bubble contact angle measurements, and the results demonstrated that the pyrrhotite surface has a strong hydrophilic state at pH above 4.5 [4]. Therefore, in order to verify the reaction between the flotation reagents and pyrrhotite, the contact angles were measured.
The measured contact angle values at the hexagonal and monoclinic pyrrhotite surfaces in deionised distilled water are shown in Fig. 12 and Fig. 13. It can be observed that an increase in pH can lead to a great decrease in the contact angle both on the hexagonal and monoclinic pyrrhotite surface in the absence of flotation reagents and that during the whole pH range, the contact angle of hexagonal pyrrhotite is smaller than that of monoclinic pyrrhotite. When the pH is below 7, the natural hydrophobicity of both types of pyrrhotite increases with decreasing pH value, and the results demonstrate that the hexagonal pyrrhotite surface has a strong hydrophilic state at pH values above 9 (contact angle 0��). When the pH value is greater than 9, the contact angle of monoclinic pyrrhotite is about 25��.
The floatability of pyrrhotite has been investigated from contact angle measurements using butyl dithiophosphate as collector for solution pH from 5 to 12.5. Figures 12 and 13 suggest that the addition of collector improves the hydrophobicity compared with that in the absence of collector. In this work, it is demonstrated that butyl dithiophosphate is adsorbed on pyrrhotite surface. At pH 9, the contact angle increases greatly from 0�� to 50�� with the addition of collector (1��10-4 mol/L) for hexagonal pyrrhotite, and the contact angle increases from 25�� to 75�� for monoclinic pyrrhotite.
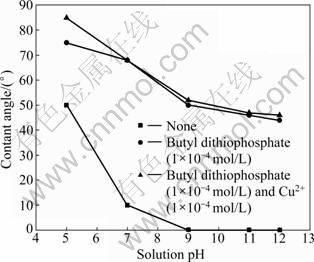
Fig. 12 Contact angle on hexagonal pyrrhotite surface with and without flotation reagents as function of pH
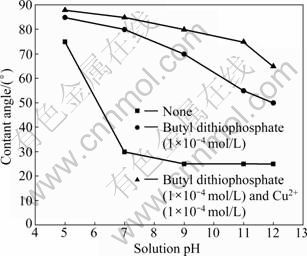
Fig. 13 Contact angle on monoclinic pyrrhotite surface with and without flotation reagents as function of pH
The experiments for activation of pyrrhotite by cupric ions (sulfate) at an initial concentration of 1��10-4 mol/L butyl dithiophosphate have been carried out with contact angle measurements, as shown in Fig. 12 and Fig. 13. These results show that when the pH is greater than 7, the presence of cupric ions does not influence the contact angle on the hexagonal pyrrhotite surface compared with the contact angle measured only in the presence of butyl dithiophosphate. Similar research [15-16] demonstrated that copper activation is not possible since copper is essentially insoluble above pH 8 and thus not available for reaction at the pyrrhotite surface. In contrast, the contact angle of monoclinic pyrrhotite increases greatly in the alkaline solutions. When pH<7, cupric ions increase the contact angle, from 50�� to 85�� for hexagonal pyrrhotite and from 85�� to 88�� for monoclinic pyrrhotite, indicating that pyrrhotites are effectively activated in acidic solutions. It should be noted that the hydrophobicity of monoclinic pyrrhotite is larger than that of hexagonal pyrrhotite obtained with cupric ions activated in the whole pH range.
4 Conclusions
1) The flotation behavior of hexagonal pyrrhotite has a remarkable difference compared with that of monoclinic pyrrhotite with collectors (sodium butyl xanthate, sodium diethyl dithiocarbamate and butyl dithiophosphate). It is shown that the floatability of hexagonal pyrrhotite is larger than that of monoclinic pyrrhotite. The effect of pH value is very important from the results for pyrrhotites collector flotation. The floatability of both types of pyrrhotite in alkaline solutions is not as good as that in acidic solutions.
2) Activation of pyrrhotite by copper is significant in acidic solutions for both types of pyrrhotites, but the effect is found to be negligible for hexagonal pyrrhotite using butyl dithiophosphate as collector in alkaline solutions. In contrast, monoclinic pyrrhotite shows better floatability in alkaline solutions when activated by copper ion.
3) The flotation of pyrrhotites is dependent on pulp pH and potential, thus the pulp potential-pH correlation for pyrrhotites flotation is established. Appropriate control of the oxidation potential is important during flotation process for improved flotation recovery.
References
[1] WARD J C. The structure and properties of some iron sulphides [J]. Rev Pure and Appl Chem, 1970, 20: 175-206.
[2] ARNOLD R G. Range in composition and structure of 82 natural terrestrial pyrrhotites [J]. Can Miner, 1967, 9: 31-50.
[3] JANZEN M P, NICHOLSON R V, SCHARER J M. Pyrrhotite reaction kinetics: Reaction rates for oxidation by oxygen, ferric iron, and for nonoxidative dissolution [J]. Geochimica et Cosmochimica Acta, 1999, 64(9): 1511-1522.
[4] MILLER J D, LI A J, DAVIDTZ J C, VOS F. A review of pyrrhotite flotation chemistry in the processing of PGM ores [J]. Minerals Engineering, 2005, 18: 855-865.
[5] ORLOVA T A, STUPNIKOV V M, KRESTAN A L. Mechanism of oxidative dissolution of sulphides [J]. Zhurnal Prikladnoi Khimii, 1988, 61: 2172-2177.
[6] VANYUKOV A V, RAZUMOVSKAYA N N. Hydrothermal oxidation of pyrrhotites [J]. Izv Vyssh Uchelon Zaved, Tsvetn, Metall, 1979, 6: 605-610.
[7] SUTHERLAND K L, WARK I W. The principles of flotation [M]. Australasian Institute of Mining and Metallurgy, 1955: 25-29.
[8] YU Run-lan. Study on the basic theory of flotation electrochemistry of Pb-Sb-Fe-Zn sulfide minerals [D]. Changsha: University of Central South University, 2004: 78-87. (in Chinese)
[9] HAMILTON I C, WOODS R. An investigation of surface oxidation of pyrite and pyrrhotite by linear potential sweep voltammetry [J]. J Electrochem, 1981, 118: 327-343.
[10] ZHANG Qin. The study of electrochemistry flotation behavior and surface adsorption of lead-antimony-zinc-iron sulfides [D]. Changsha: University of Central South University, 2004: 62-72. (in Chinese)
[11] PRESTIDGE C A, RALSTON J. Contact angle studies of particulate sulfide minerals [J]. Minerals Engineering, 1996, 9: 85-102.
[12] PRESTIDGE C A, RALSTON J. Contact angle studies of galena particles [J]. Journal of Colloid and Interface Science, 1995, 172: 302-310.
[13] CRAWFORD R, RALSTON J. The influence of particle size and contact angle in mineral flotation [J]. International Journal of Mineral Processing, 1988, 23: 1-24.
[14] DIGGINS D, FOKKINK L K J, RALSTON J J. The wetting of angular quartz particles: Capillary pressure and contact angles [J]. Colloids and Surfaces, 1990, 44: 299-313.
[15] BUSWELL A M, NICOL M J. Some aspects of the electrochemistry of the flotation of pyrrhotite [J]. Journal of Applied Science, 2002, 32: 1321-1329.
[16] CHEN Jian-hua, LI Yu-qiong, CHEN Ye. Cu-S flotation separation via the combination of sodium humate and lime in a low pH medium [J]. Minerals Engineering, 2011, 24: 58-63.
(Edited by YANG Bing)
Foundation item: Project(50774094) supported by the National Natural Science Foundation of China
Received date: 2011-01-07; Accepted date: 2011-03-25
Corresponding author: HE Ming-fei; Tel: +86-15887733998; E-mail: hemingfei1@126.com

